Abstract
Natural products are an inexhaustible source for drug discovery. However, the validation and selection of primary screening assays are vital to guarantee a selection of extracts or molecules with relevant pharmacological action and worthy of following up. The assay must be rapid, simple, easy to implement, and produce quick results and preferably at a low cost. In this work, we developed and validated a colorimetric microtiter assay using the resazurin viability dye. The parameters of the resazurin method for high-throughput screening (HTS) using natural extracts against Aspergillus fumigatus were optimized and set up. The extracts plus RPMI-1640 modified medium containing the spores and 0.002% resazurin were added per well. The fluorescence was read after 24 to 30 h of incubation. The resazurin proved to be as suitable as Alamar Blue for determining the minimal inhibitory concentration of different antifungals against A. fumigatus and effective to analyze fungicidal and fungistatic compounds. An HTS of 12 000 microbial extracts was carried out against two A. fumigatus strains, and 2.7% of the extracts displayed antifungal activity. Our group has been the first to use this methodology for screening a collection of natural extracts to identify compounds with antifungal activity against the medically important human pathogen A. fumigatus.
Keywords
Introduction
The opportunistic human pathogen Aspergillus fumigatus is a widely spread airborne fungus. This fungus causes various diseases in humans, of which invasive aspergillosis (IA) deserves a very special clinical consideration. In the past decade, the incidence of IA has increased threefold. Consequently, IA is now considered as one of the major causes of death in immunocompromised patients. 1
Despite intrinsic difficulties due to the characteristic filamentous growth of Aspergillus, a few methods have been reported to determine by in vitro assays in liquid medium the susceptibility of Aspergillus clinical isolates to antifungal drugs. 2 However, the main drawback of these methods, including the standard broth microdilution susceptibility method developed by the Clinical Laboratory Standard Institute (CLSI), is the low reproducibility to determine the endpoint of antifungal compounds against Aspergillus species. 3
Since 1956, several tests have been used for minimal inhibitory concentration (MIC) determination in Aspergillus. 4 One of the most frequently used assays to determine the MICs of antifungal compounds against A. fumigatus is the M38-A method, developed by the CLSI, with the most commonly employed medium in this assay being the RPMI-1640.
In addition to this information, an extra consideration must be taken into account when an antifungal susceptibility test is developed for filamentous fungi. The assay will require strict pH control of the medium (i.e., using a buffered medium), as acidic conditions disturb the activity of most antifungal compounds used in clinical tests, such as polyenes or azoles. 5 Accordingly, RPMI-1640 (containing 0.3 g/L of glutamine) with 165 mM MOPS buffer (pH 7.1) is the standard medium recommended by both the CLSI and the European Committee on Antimicrobial Susceptibility Testing (EUCAST) to determine MICs of Aspergillus.
In this work, we have developed a novel and highly accurate MIC determination method to identify potential antifungals against A. fumigatus in microplate high-throughput screenings (HTS). This novel method is based on the previously described Alamar Blue assay 6 for testing antifungal susceptibility of Aspergillus spp against voriconazole. Mania et al. in 2010 7 described a related methodology for Aspergillus nidulans based on resazurin. Our method, which also employs resazurin, permits one to perform HTS assays with a huge collection of microbial natural extracts using the economically and clinically important fungus, A. fumigatus. Accordingly, using this method, we were able to identify, by a simple and accurate detection technique, positive extracts exhibiting antifungal activity against A. fumigatus from a collection of 12 000 different natural extracts.
In addition, the optimal settings of these method parameters for HTS using 96- or 384-well microplates are also described. By means of these experimental conditions, we identified 261 positive microbial extracts with antifungal activity against A. fumigatus.
Materials and Methods
Strains, HTS Medium, Antifungals, and Reagents
Three A. fumigatus strains were used in this study: (1) A. fumigatus American Type Culture Collection ATCC 46645 (wild-type strain), (2) A. fumigatus CBS 144.89 (wild-type), and (3) A. fumigatus ΔakuBKU80. 8 The ΔakuBKU80 strain lacks the nonhomologous end-joining pathway and is commonly used in many laboratories for genetic manipulation of A. fumigatus.
A modification of the RPMI-1640 standard medium (CLSI and EUCAST) was used in HTS experiments. The RPMI-1640 modified medium 9 contains 10.4 g/L of RPMI-1640 medium (R8755; Sigma, St. Louis, MO), 6.7 g/L of Yeast Nitrogen Base (YNB; Becton, Dickinson and Company, Franklin Lakes, NJ), 1.8% (w/v) glucose, and 40 mM HEPES (pH = 7.1). Cultures were incubated at 37 °C for 24 h.
Inoculum
All manipulations were carried out in a laminar flow hood using aseptic techniques. Single-spore isolates were cultivated in potato dextrose agar (PDA) plates for 2 to 3 d. A stock spore suspension was prepared from a PDA culture by adding 15 mL of Tween saline (0.025% [v/v] Tween 80, Merck, 8 g/L NaCl). A sterile rubber spatula was used to gently scrape the surface of the agar, and the resulting suspension was filtered through four layers of sterile chiffon. The concentration of this spore stock solution was determined by counting the conidia in a Neubauer chamber.
Optimization studies to determine the optimal inoculum for HTS analyses were performed as described in the Results section. The optimal inoculum per well for a 96-well microplate was 2.5 × 104 fresh conidia/mL (150 µL/well) and for a well of the 384-well microplate, half of this number.
Two incubation times were tested: 24 h and 48 h. The incubation time of 24 to 30 h was chosen in order to not lose any potential positive extract because with longest incubation time, we obtain less positive antifungal extracts due to the loss of sensibility with extended incubation.
Resazurin Solution
Resazurin sodium salt (C12H6NNaO4; R7017, Sigma, St. Louis, MO) stock solution of 0.02 g in 100 mL of sterile distilled water was sterilized by filtration. Two final concentrations of the dye resazurin were tested: 0.001% (w/v) and 0.002% (w/v). The best results were achieved with the final concentration of 0.002% of the dye. Consequently, all the experiments were performed using 0.002% (w/v) as final concentration of the dye.
HTS Assay Conditions
HTS were performed in 96- and 384-well plates. A 96-well plate was loaded as follows: 7 µL of aqueous extracts from the Fundación MEDINA’s collection were added per well using an automated Aquarius Multi-channel Pipettor (Tecan). Then, a volume of 150 µL of the RPMI-1640 modified medium containing the spore inoculum (described above) and 0.002% (w/v) resazurin were added using Multidrop Combi (Thermo). In the case of 384-well plates, we used half of the volumes added to each well of the 96-well plates. In these assays, the volume of natural extract (3.5 µL) was added by using the automated Biomek FX workstation (Beckman Coulter).
Each microplate contained a set of control wells located in the left and right columns. The control wells of a 96-well microplate were established as follows: (1) the four top wells of the left column without inoculum (medium + resazurin) were the negative controls, (2) the four bottom wells of this column contained an amphotericin B (A-4888 Sigma) dose-response curve (1, 0.5, 0.25, and 0.125 µg/mL) as positive control, (3) the four top wells of the right column without testing sample were the growth controls, and (5) the four bottom wells of the right column contained a replica of the amphotericin B dose-response curve earlier described.
For experiments performed in the 384-well plates, the four times number of the same set of controls was established. Culture plates were incubated at 37 °C for 24 h. The variation of the color of resazurin was visually observed. After 24 h of incubation, the data were quantified by measuring fluorescence (excitation 570 nm, emission 615 nm) using a VICTOR multilabel counter (Perkin Elmer, Waltham, MA). All of the experiments were performed in triplicate.
Fundación MEDINA’s Natural Extracts Collection
Six thousand actinomycetes and 6000 fungal strains were cultivated in different production media from 7 to 28 d at temperatures ranging from 22 to 28 °C, according to the microbial strain requirements. 9 Secondary metabolites produced in the cultivation broths were collected and extracted with acetone. Extracts were prepared by mixing 10 mL of culture broth with 10 mL of pure acetone. After 1 h of shaking in orbital Kuhner, the acetone/water extracts were transferred to 16 mm glass tubes and evaporated to half of their volume after centrifugation at 1500 g for 15 min. The final aqueous extract was approximately 1× whole broth equivalent. Four 500 µL aliquots from each well were transferred to two 800 µL well AB-gene AB-0765 storage blocks. After a long time of storage at –20 °C, an aliquot of 500 µL was thaw stored at 4 °C and mixed briefly before testing.
Evaluation of Antifungal Activity
To standardize the assay conditions, the efficacy of two commercial fungicides with different modes of action was evaluated by the resazurin assay. The antifungals tested were (1) amphotericin B and (2) itraconazole (I16657 Sigma). Initially, the effective concentration range for each commercial antifungal was determined. Each chemical was prepared at 1 mg/mL in DMSO (Merck, Darmstadt, Germany) and serially diluted in 20% DMSO (v/v), to reach a final DMSO concentration lower than 0.1% (v/v). All antifungal dose-response curves started at 16 µg/mL of compound per well in 1:2 serial dilutions. The inoculum was dispensed in the wells using 150 µL for 96-well plates and 75 µL for 384-well plates. A range of spore density from 103 to 106 was tested in both types of plates using the resazurin methodology. Finally, a concentration of 2.5 × 104 spore/mL was selected as the inoculum size giving the most suitable and consistent results. Also, the tests were performed with RPMI-1640 standard medium by CLSI and EUCAST and with the RPMI-1640 modified medium. The latter was selected as the medium providing the clearest results. The experiment results were compared with previous information available for these antifungals to select the parameters to be used in the HTS with natural products against A. fumigatus using resazurin as an indicator of cell growth. 10
Fluorometric Measurement, Visual Inspection, and Statistical Analyses
Fluorescence data were expressed as the percentage of resazurin reduced depending on incubation time. After 24 h, the percentage of resazurin reduction was determined fluorometrically by measuring fluorescence at the wavelengths 570 nm (excitation) and 615 nm (emission). Fluorescence was determined with a microplate reader VICTOR multilabel counter (Perkin Elmer). The mean fluorescence values were used in subsequent calculations. The percentage of resazurin reduction and growth inhibition was calculated using the following normalization: % reduction = 100 × (fluorescent intensity of test agent – fluorescent intensity of untreated control/fluorescent intensity of reduced resazurin – fluorescent intensity of untreated control) and % inhibition = 100 – % reduction. The color of the wells was also visually recorded. A blue color was interpreted as absence of metabolic activity (no spore germination). A fluorescent pink color was interpreted as presence of metabolic activity (spore germination; Fig. 1A ). A purple color in the well was interpreted as a trailing result, where some metabolic activity was present but a longer incubation time allowed the purple color to change to pink. Statistical data analyses for the resazurin assay were obtained with the Genedata Screener program (Genedata AG, Switzerland). An extract was considered to have activity when its percentage of inhibition was superior to 60%. The Z′ factor predicts the robustness of an assay by taking into account the mean and standard deviation of both positive and negative controls. 11 The robust Z′ factor (RZ′ factor) is based on the Z′ factor, but standard deviations and means are replaced by the robust standard deviations and medians, respectively. In all experiments performed in this work, the RZ′ factor obtained was between 0.85 and 0.95.
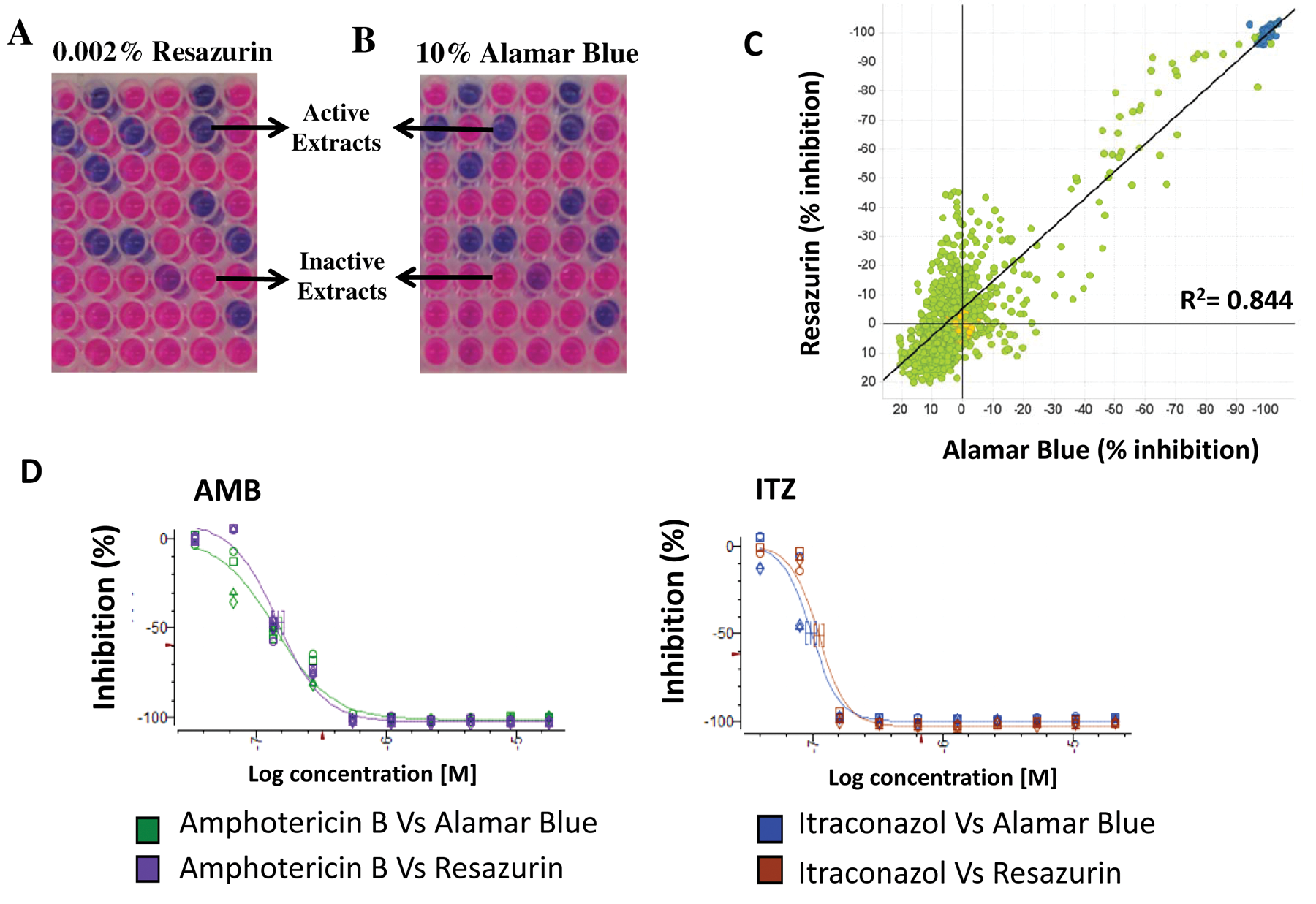
Resazurin dye used to identify microbial extracts with antifungal activity against A. fumigatus. (
Results and Discussion
The main objective of this study was the development of an easy and inexpensive high-throughput colorimetric assay to detect the viability of A. fumigatus enabling the identification of extracts with antifungal activity against this species from a collection of microbial natural products extracts enriched in secondary metabolites.
Resazurin Is a Valuable Indicator to Determine A. fumigatus Viability in HTS of Microbial Natural Extracts
Filamentous fungi form hyphae and mycelia by tip extension, and the mycelia are usually dispersed in the liquid medium, which makes it difficult to measure the turbidity. Resazurin is an oxidation-reduction indicator used for determining the viability of eukaryotic cells. This nonfluorescent blue dye is converted by reduction into the pink-colored highly fluorescent resorufin. Resazurin was initially commercialized under the Alamar Blue trademark (Trek Diagnostic Systems, Cleveland, OH), and now it is also available under other names such as AB assay, Vybrant (Molecular Probes, Carlsbad, CA) and UptiBlue (Interchim, San Pedro, CA). Yamaguchi et al. 6 described the use of the commercial reagent Alamar Blue to determine the MIC of several antifungals against Aspergillus species. The high price of this commercially available substrate hampered similar analyses by HTS to evaluate the activity of thousands of microbial extracts. This limitation led us to test the suitability of resazurin sodium salt (C12H6NNaO4) powder, a more economical reagent, to detect antifungal activity against A. fumigatus from a collection of microbial natural extracts of Fundación MEDINA (Granada, Spain). Accordingly, we tested whether the low-cost compound resazurin, the active substrate of the Alamar Blue trademark also used by Mania et al. 7 for assays with A. nidulans, was a suitable alternative for HTS assays with microbial natural product extracts against A. fumigatus.
First, we performed studies to ascertain the suitability of resazurin to determine A. fumigatus viability and compared these results with those obtained with the commercial reagent Alamar Blue. 12 Our data demonstrated that Alamar Blue (DAL1100 Invitrogen) is able to identify natural extracts exhibiting antifungal activity against A. fumigatus using our laboratory experimental conditions ( Fig. 1B ). More importantly, 0.002% resazurin was chosen because it presents the same effectiveness as the Alamar Blue working solution (10% Alamar Blue; Invitrogen) in identifying the MICs of known antifungals (amphotericin B and itraconazole; Fig. 1D ) as well as positive natural extracts from our collection ( Fig. 1C ), using both the 96- and the 384-well microplates. In contrast, the 0.001% resazurin solution presented a weaker fluorescence emission sign (data not shown). These results enabled us to afford further screenings of more than 12 000 microbial natural extracts to identify potential novel antifungals against A. fumigatus.
Optimization of the Resazurin Method for HTS
Resazurin assay is an excellent alternative to reference viability assays as XTT/MTT and tritiated thymidine-based techniques. Previous studies show excellent correlation among the resazurin assay and these reference viability assays while being much easier and safer to use. 13 Resazurin is a nonradioactive and nontoxic reagent and perfectly soluble in water; XTT has limited solubility in aqueous buffers (and the stock solutions proved to be not easy to prepare) in addition to its limited stability as a reagent. 14
Once the suitability of resazurin to determine A. fumigatus viability in HTS was demonstrated, we optimized all parameters for further screenings. These parameters included the inoculum size, the temperature and time of incubation, the optimal liquid medium including its pH control, the viability detection system, and the analysis of the data. Such studies were performed in both 96- and 384-well microplates.
Concerning the inoculum size, for the 96-well microplate system, we tested a range of conidia concentrations between 104 to 106CFU/mL. The concentration range of conidia recommended by the NCCLS for the M38-A assay with filamentous fungi is 0.4 × 104 to 5 × 104 CFU/mL. Our studies showed that the optimal concentration of conidia with our method using resazurin as the indicator of A. fumigatus growth is 2.5 × 104 CFU/mL. A similar inoculum was found to be optimal for the 384-well microplate system (data not shown). This concentration was chosen as optimal because concentrations higher than 2.5 × 104 CFU/mL ensure an overgrowth at the surface of the well determining false- positive results when it partially covers the well and blocks the fluorescence emission.
Regarding the medium, we employed several liquid media to select the medium that gives the clearest results in terms of sensitivity and reproducibility (data not shown). Results obtained showed that the optimal medium for this assay, in both microplates, was a modified version of the RPMI-1640 medium. 9 This RPMI-1640–modified medium contains 10.4 g/L of RPMI-1640 medium, 6.7 g/L of YNB (Becton, Dickinson and Company), 1.8% glucose, and 40 mM HEPES (pH = 7.1). This medium was chosen because it allows the easy identification of microbial extracts presenting total inhibition and extracts with presumably antifungal activity (between 80% and 60% of inhibition) against A. fumigatus. By contrast, using other media such as the complete medium or RPMI-1640 standard liquid medium, the antifungal activity was hardly distinguished because of the difficult differentiation between total and partial inhibition.
Once the medium conditions were established, we performed several experiments to select the optimal incubation time and temperature for this assay in both microplates. Three different temperatures were chosen: 30 °C, previously used by Yamaguchi et al. 6 for MIC determination in Aspergillus, 34 °C, and 37 °C, the optimal temperature for the A. fumigatus growth. 12 We selected 37 °C, as this temperature significantly reduced the incubation time for the assay (24–30 h), especially in comparison with results obtained at 30 °C.
In parallel to the optimization of these parameters, the reproducibility and sensitivity of our method were evaluated by performing susceptibility dose curves with two different antifungals: amphotericin B and itraconazole. The methodology proved to be effective for analyzing these antifungal and fungistatic compounds. The MICs for amphotericin B were between 0.5 and 1 µg/mL, whereas for itraconazole, it was in the range of 0.125 to 0.25 µg/mL ( Fig. 2 ). These data agree with the MICs reported by other authors using different methods including the standardized M38-A method and the EUCAST. 12 The results obtained show that the resazurin methodology is perfectly suitable to assay pure compounds. In addition, this methodology can be applied to assay both fungicidal and fungistatic antifungals such as amphotericin B and itraconazole. 15
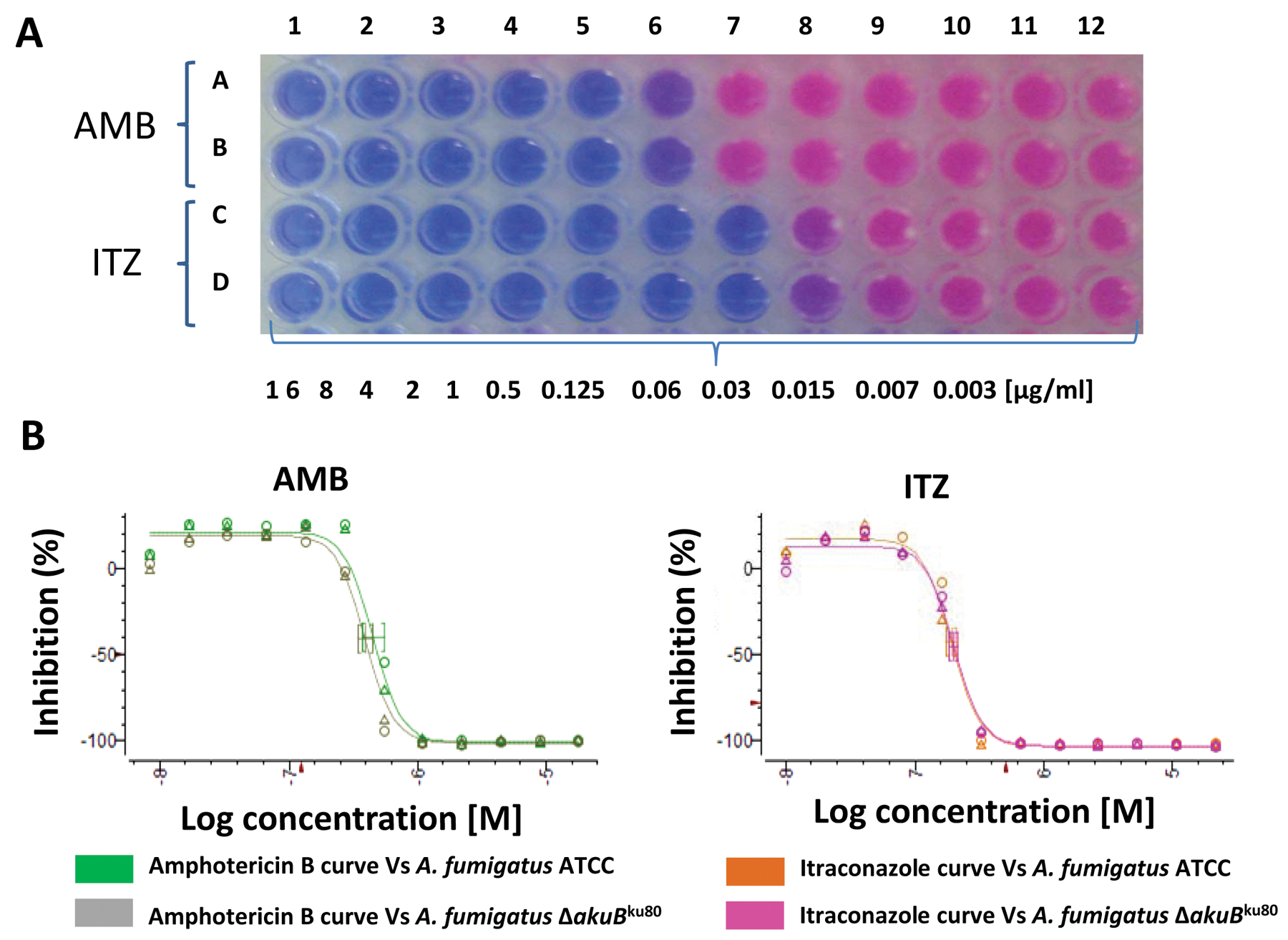
(
Screening Campaigns
The MEDINA’s natural extract collection, employed in these assays, contains secondary metabolites from actinomycetes and fungal cultures. Microbial natural products have been one of the major sources of novel drugs, and the inexhaustible chemical diversity of these extracts suggests that novel molecules with potential therapeutic applications are still waiting to be discovered from these natural sources. Despite this, research in antibiotics from natural products has declined during the past decade as a consequence of diverse factors, among them the lack of new approaches to improve the efficiency of the discovery process.
Once all parameters were optimized, we carried out HTS of 12 000 different microbial extracts described in the Materials and Methods section. Two A. fumigatus strains were selected for a primary screening campaign; A. fumigatus ATCC 4664 (wild-type strain) and A. fumigatus ΔakuBku80,8 a mutant strain that lacks the nonhomologous end-joining pathway, a condition that dramatically increases the homologous recombination process in this fungus. This mutant strain was selected as it has become an A. fumigatus strain routinely used in molecular biology experiments focused on the discovery of novel therapeutic targets of this pathogenic fungus. Accordingly, many mutants deficient in specific pathways related to cell-wall or ergosterol synthesis, the main targets of clinical antifungal drugs, were established in this genetic background, and some of these mutants are likely to be part of further screening assays.
Of the 12 000 microbial extracts, 6000 derived from actinomycetes and 6000 extracts from fungi. Both 96- and 384-well microplate systems were employed in these analyses ( Fig. 3 ). Results obtained are summarized in Table 1 . A high number of natural extracts (3.9%) displayed antifungal activity against only one of the A. fumigatus strains tested. Active natural products extracts were found more frequently in actinomycetes cultures (73% of 791 total positive extracts) than in fungal culture extracts (27%; Table 1 ). Interestingly, 2.7% of extracts analyzed (327 samples) displayed antifungal activity against both A. fumigatus strains tested (inhibition cutoff ≥60%). In addition, whereas 1% of extracts showed antifungal activity against only the A. fumigatus ATCC46645 strain, 2.9% of extracts showed activity against only the A. fumigatus ΔakuBku80strain. All the experiments were performed in triplicate and the standard deviation (SD) of the experiments were between ± 3 and ± 5. In addition, all the assay plates had an amphotericin B curve as a control in order to evaluate the sensitivity of the strain. It is noteworthy to mention that an important number of microbial natural extracts inhibited growth of the A. fumigatus ΔakuBku80mutant strain but were inactive against the A. fumigatus ATCC 46645 (wild-type) strain. Because the A. fumigatus ΔakuBku80strain shows defects in the nonhomologous end-joining pathway, it is conceivable that these extracts contain compounds acting on the DNA and induce extensive repair activity.
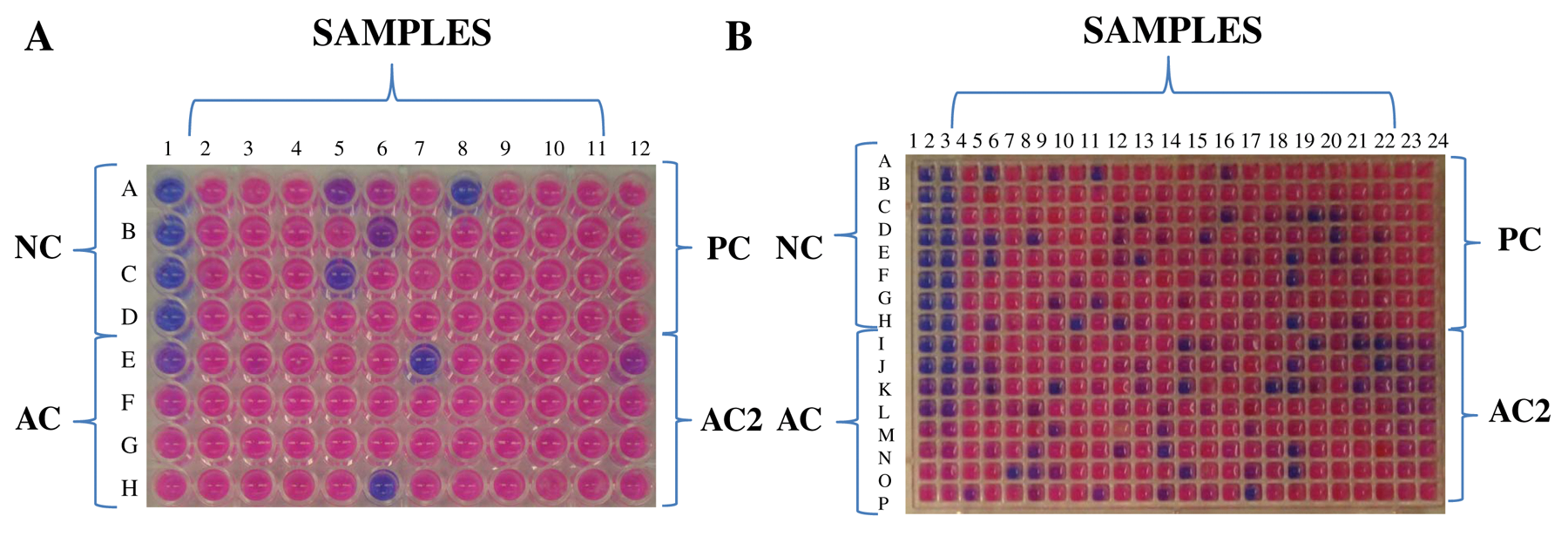
A new method to identify natural extracts with antifungal activity against A. fumigatus using resazurin. (
Primary Screening Campaign Results a
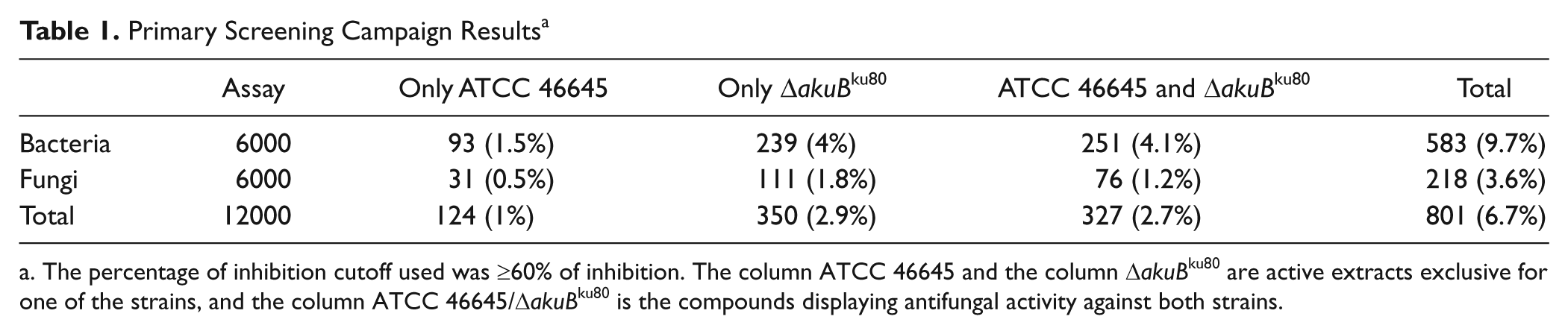
The percentage of inhibition cutoff used was ≥60% of inhibition. The column ATCC 46645 and the column ΔakuBku80 are active extracts exclusive for one of the strains, and the column ATCC 46645/ΔakuBku80 is the compounds displaying antifungal activity against both strains.
A “cherry-pick” campaign was carried out to confirm the activity and the reproducibility of the natural extracts showing antifungal effect against both strains tested. These extracts were called PEFH (positive extracts for both strains in first HTS). In this cherry-pick campaign, besides the employ of the tow strains used in the primary screening, a third strain was added, the A. fumigatus CBS 144.89 isolate, another wild-type strain of A. fumigatus. The use of this third A. fumigatus strain in the cherry-pick assay would help confirm the specific activity against A. fumigatus of the PEFH samples. From the 327 PEFH samples identified, 262 extracts (80% of PEFH samples) displayed antifungal activity against all three A. fumigatus strains assayed. From these extracts, 201 samples derivate from bacterial cultures and 61 from fungal cultures.
The methodology described in this work is an advance in the field of the research with natural products allowing a rapid and simple screening of extracts; in addition, it is easily implementable and produces quick results and at very low cost. The same methodology can be used for the further steps of the screening as the analysis of the extract fractions until a pure active compound is obtained, as well as to identify putative novel antifungal compounds inhibiting the growth of fungal pathogens such as A. fumigatus and other related fungi. In addition, this method can be scaled up to HTS assays of a large collection of microbial natural products extracts. Lastly, this methodology can be applied to different types of assays as the analysis of synergistic, antagonistic, or potentiating effects of microbial extracts with known antifungals.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the project ERA-NET Pathogenomics (7th FP). ANTIFUN “The Cell Wall as a Target to Improve Antifungal Therapy against Aspergillosis,” BFU2008-04709-E/BMC and Fundación MEDINA, a public-private partnership of Merck Sharp & Dohme de España S.A./Universidad de Granada/Junta de Andalucía. We would also like to thank all the technicians from Fundación MEDINA for their contribution to this project.
