Abstract
The authors designed a chemical genomics screen with the aim of understanding genes and pathways that modulate neural stem/precursor cell differentiation. Multipotent mouse neural precursor cells isolated from cortices of embryonic day 12 (E12) embryos were subjected to spontaneous differentiation triggered by growth factor withdrawal. A quantitative whole-well immunofluorescence assay was set up to screen tool compound sets to identify small molecules with potent, dose-dependent, and reproducible effects on increasing neural stem cell differentiation toward neuronal lineage. Among the pro-neuronal compounds, kinase inhibitors were shown to exert pro-neuronal effect via a signaling pathway associated with the kinase. The global effect of hit compounds on modulating neuronal differentiation was confirmed by an in vivo mouse study and human neural stem cells culture. This study demonstrates that a phenotypic assay using cell type–specific antibody markers can be used for a large-scale compound screen to discover targets and pathways with impacts on differentiation of lineage-restricted precursor cells toward specific lineages.
Introduction
Neurons differentiated from neural stemcells/precursor cells (NSCs/NPCs) hold great promises in applications such as drug discovery and regenerative medicine for neurodegenerative diseases.1–3 However, under the current knowledge and method of in vitro culture conditions, NPCs, whether from mouse embryonic/adult brains or from human embryonic stem cells/inducible pluripotent stem cells (hESCs/iPSCs), can only be differentiated inefficiently into a mixed population of three neural lineages: neurons, oligodendrocytes, and astrocytes.4–6 Researches have been focused on identifying the genes and regulators involved in the lineage-restricted differentiation process so that purer neuronal population for drug discovery can be achieved.7–9 However, very little is known of small molecules that can modulate neuronal differentiation through targeting these genes.
Despite the potential usefulness of compounds in scaling up production of neurons from NPCs, only a few are able to perform screens using these stem/progenitor cells, partly due to (1) technical difficulties in maintaining a homogeneous population of NPCs and (2) only a few suitable assays to quantify the readout.10–17 Using mouse embryonic carcinoma cells, Ding et al. 18 introduced the luciferase reporter construct carrying promoter of specific neuronal gene Tα1 tubulin to identify compounds capable of inducing neural fate from embryonic carcinoma P19 cells. However, screening with P19 cells leads to identification of the modulators of embryonic cells to neural progenitor fate, instead of molecules that modulate lineage-restricted progenitors to terminally differentiate to neurons. Using lineage-restricted progenitors from mouse embryonic brain, Saxe et al. 14 screened for molecules capable of promoting differentiation of neurons via TuJ1 immunoreactivity measured by chemiluminescence. This approach of screening with a cell type–specific antibody allows phenotypic screens to be performed where stable clones are not available, as is the case for most lineage-restricted progenitors. However, by measuring the total amount of TuJ1 immunoreactivity instead of counting the number of cells positive for TuJ1 staining, researchers have had to perform secondary assays to make sure the hits were not simply increasing TuJ1 protein expression levels. Thus, an assay capable of directly quantifying the number of specific cell type via antibody would be more straightforward and reduce false-positive rates.
We designed our screen as a chemical genomics exercise to search for tool compounds with significant effects on NPC differentiation toward neurons. Using multipotent NPC from mouse embryonic brain, a morphology-based phenotypic assay was developed using neuron-specific antibody TuJ1 and an imaging device capable of unbiased quantification of every signal within the entire well. By taking advantage of tool compound collections with known target associations, we screened for chemical modulators and identified genes and pathways potentially critical for neuronal differentiation and validated their pro-neuronal effect in vivo and also on human NSCs. Our study shows that lineage-restricted progenitors can be used to identify chemical compounds capable of modulating differentiation of progenitor cells, elucidate the genes and pathways involved, and find potential tools for regenerative therapy.
Materials and Methods
Culture and spontaneous differentiation of primary neural precursor cells
Embryonic cortices from E12 ICR mice were dissected out and dissociated into single cells. Primary cortical NPCs were adhered to a poly-L-ornithine (PO)–coated (15 µg/mL; Sigma, St. Louis, MO) and fibronectin (FN) (10 µg/mL; GIBCO, Carlsbad, CA)–coated 10-cm dish with 10 mL Dulbecco’s modified Eagle’s medium (DMEM)/F12 1:1 (GIBCO) containing 2% B27-supplement (GIBCO) and 10 ng/mL basic fibroblast growth factor (bFGF). The cells were passaged with TrypLE (GIBCO) upon confluence every 5 to 6 days and stocked frozen at passage 2. All assays were carried out using passage 3 NPC. Twenty thousand or 5000 undifferentiated passage 3 NPCs were plated onto PO/FN-coated 96-well or 384-well plates, respectively, in DMEM/F12/B27 media without bFGF. Cells were allowed to differentiate spontaneously for 5 days.
Compound collections and assay plates preparation
The Library of Pharmacologically Active Compounds (LOPAC) collection (cat. #LO3300; Sigma) and GlaxoSmithKline’s Cell & Pathway (C&P) compound collection (company proprietary compound library with known pathways and targets, 8438 compounds) were dissolved in DMSO at 10 mM in 384-well stock plates. Assay plates were prepared by dispension of compounds in stock plates onto PO/FN-coated 96- or 384-well plates by the Echo Liquid Handling System (Labcyte, Sunnyvale, CA). Using the MultiDrop Combi Reagent Dispenser (Thermo Scientific, Waltham, MA), dissociated cells were plated onto compound containing assay plates. The same amount of DMSO was added to wells that did not contain compounds to serve as proper controls.
Antibodies, immunofluorescence, and quantification
Cells were fixed by 4% paraformaldehyde (PFA) for 20 min and washed three times with phosphate-buffered saline (PBS) by a BioTek xL405 (BioTek, Winooski, VT). Antibodies against TuJ1 (1:1000; Covance, Princeton, NJ), GFAP (1:1000; Sigma), Nestin (1:1000; Millipore, Billerica, MA), Sox2 (1:200; Santa Cruz Biotechnology, Santa Cruz, CA), and CNPase (1:500; Millipore) were diluted in PBS containing 3% normal donkey serum (Jackson ImmunoResearch, West Grove, PA). Appropriate Alexa480 secondary antibodies (Invitrogen, Carlsbad, CA) were used. All liquid additions were carried out using MultiDrop (Thermo Scientific).
Screen design
In the primary screen, NPCs plated on 384-well assay plates were allowed to differentiate for 5 days in medium described above without bFGF, with each well containing 10 µM of a single compound dissolved in DMSO or DMSO only. The sides of the 384-well plate were not used due to a possible edge effect; thus, one 384-well plate could screen approximately 280 compounds plus 28 DMSO controls. After 5 days of culture, cells were immunostained for TuJ1 and counterstained with DAPI. Fluorescently labeled plates were analyzed with Acumen Explorer X3 (TTP LabTech, Royston, UK). We worked out a semi-automatic way to perform the immunostaining washing steps using the MultiDrop Combi Reagent Dispenser (Thermo Scientific); the assay throughput was about 10 plates per day. Potential hit compounds that increased the percentage of the TuJ1-positive neurons but not the total DAPI (cell) numbers were tested further in triplicate (1, 3, and 10 µM) and 10-point full dose-response studies.
Gene expression analysis by quantitative real-time PCR
Total cellular RNA was extracted using the RNeasy Mini Kit (74104; QIAGEN, Valencia, CA), and real-time (RT) reactions were performed with PrimeScript RT reagent Kit (DRR037A; Takara, Shiga, Japan) according to the manufacturer’s protocol. Real-time PCR analyses were performed with the Roche 480 (Roche, Basel, Switzerland), quantified using the 2ΔΔCt method. For primer sequences, please refer to
siRNA knock-down experiment
GSK3β siRNA (sc-35525), β-catenin siRNA (sc-29210), and control siRNA (sc-37007) were purchased from Santa Cruz Biotechnology. siRNAs were transfected with Amaxa Nucleofector II (Lonza, Basel, Switzerland) according to the manufacturer’s instructions.
Western blot
The following antibodies were used: anti-TuJ1 (MMS-435P; Covance), anti-GSK3β (sc-9166; Santa Cruz Biotechnology), anti-β-catenin (AB19022; Millipore), anti-active-β-catenin (05-655; Millipore), anti-actin (ab3280; Abcam, Cambridge, UK), and anti-GAPDH (G8795; Sigma).
In vivo injection, immunofluorescence, and quantification
Male C57BL/6 mice were obtained from the Shanghai Laboratory Animal Center. Bromodeoxyuridine (BrdU) (Sigma; 100 mg/kg body weight) was injected intraperitoneally at 4-hour intervals on day –1 and then repeated on day 0. GSK3β inhibitor (SB-627772) diluted in PBS (10 mg/mL) was injected intracerebroventricularly (2 µL/mouse) on day 0 immediately following final BrdU injection. GSK3β inhibitor–injected group and vehicle control group were sacrificed 3 or 6 days after compound injection. Each time point consisted of four animals in the GSK3β inhibitor group and two to three mice in the control group. The 10-µm coronal cryostat brain sections were stained with anti-doublecortin (DCX) (1:200; Santa Cruz) and anti-BrdU (1:100; Sigma) antibodies. Digital images of immunofluorescent staining on defined regions of interest within the subventricular zone (SVZ) areas were captured on a Zeiss (Oberkochen, Germany) LSM 710 META multiphoton confocal system. Data are presented as number of BrdU+ and DCX+ cells per square millimeter (mean ± SEM), and two-tailed Student t test was used to determine significance at a threshold of p < 0.05. All mouse protocols were approved by the GlaxoSmithKline Institutional Animal Care and Use Committee.
Human NSC and differentiation
Culture and spontaneous differentiation of GIBCO Human Neural Stem Cell Kits (H9-Derived) from Invitrogen (cat. N7800-200) was performed according to the manufacturer’s protocols.
Statistical analysis
All values except for in vivo data were processed and expressed as mean ± SD using the GraphPad Prism 5 (GraphPad Software, San Diego, CA). Differences between means were analyzed using a two-tailed Student t test or one-way analysis of variance (ANOVA), with p < 0.05 considered statistically significant. Dose-responsive curves were generated from XLfit using the dose-response one-site formula as follows: fit = (A + (B/(1 + ((x/C)^D)))); inv = ((((B/(y – A)) – 1)^(1/D))*C); res = (y-fit).
Results
Quantitative whole-well immunofluorescence assay for neural stem cell differentiation
To identify tool compounds that modulate differentiation of neurons from NPCs, we established a quantitative differentiation assay. Primary NPCs isolated from telencephalon of E12 ICR mice were expanded and differentiated at passage 3 for 5 days in the absence of bFGF. Nestin+/Sox2+ NPC gave rise to all three differentiated neural cell types of the CNS: neurons, astrocytes, and oligodendrocytes ( Fig. 1A ). Using a laser-scanning cytometer Acumen eX3 (TTP LabTech), which is capable of capturing and quantifying whole-well immunofluorescence, the relative percentages of cells labeled with each cell type–specific marker were measured ( Fig. 1B ). 19 Antibodies against TuJ1, GFAP, and CNPase were used to measure relative percentages of neurons, astrocytes, and oligodendrocytes, respectively, and DAPI was used to measure total cell number in the well ( Fig. 1C ; DAPI data not shown). During 5 days of differentiation, cells expressing neural stem cell marker Nestin steadily decreased from 90% to 15% ( Fig. 1D ), whereas cells expressing differentiation markers TuJ1, GFAP, or CNPase increased to 35%, 15%, and 8% of total population, respectively ( Fig. 1D ). A considerable amount of assay development work was spent on automating the multiple washing steps required for the immunofluorescence assay to achieve the coefficient of variation of 10%.
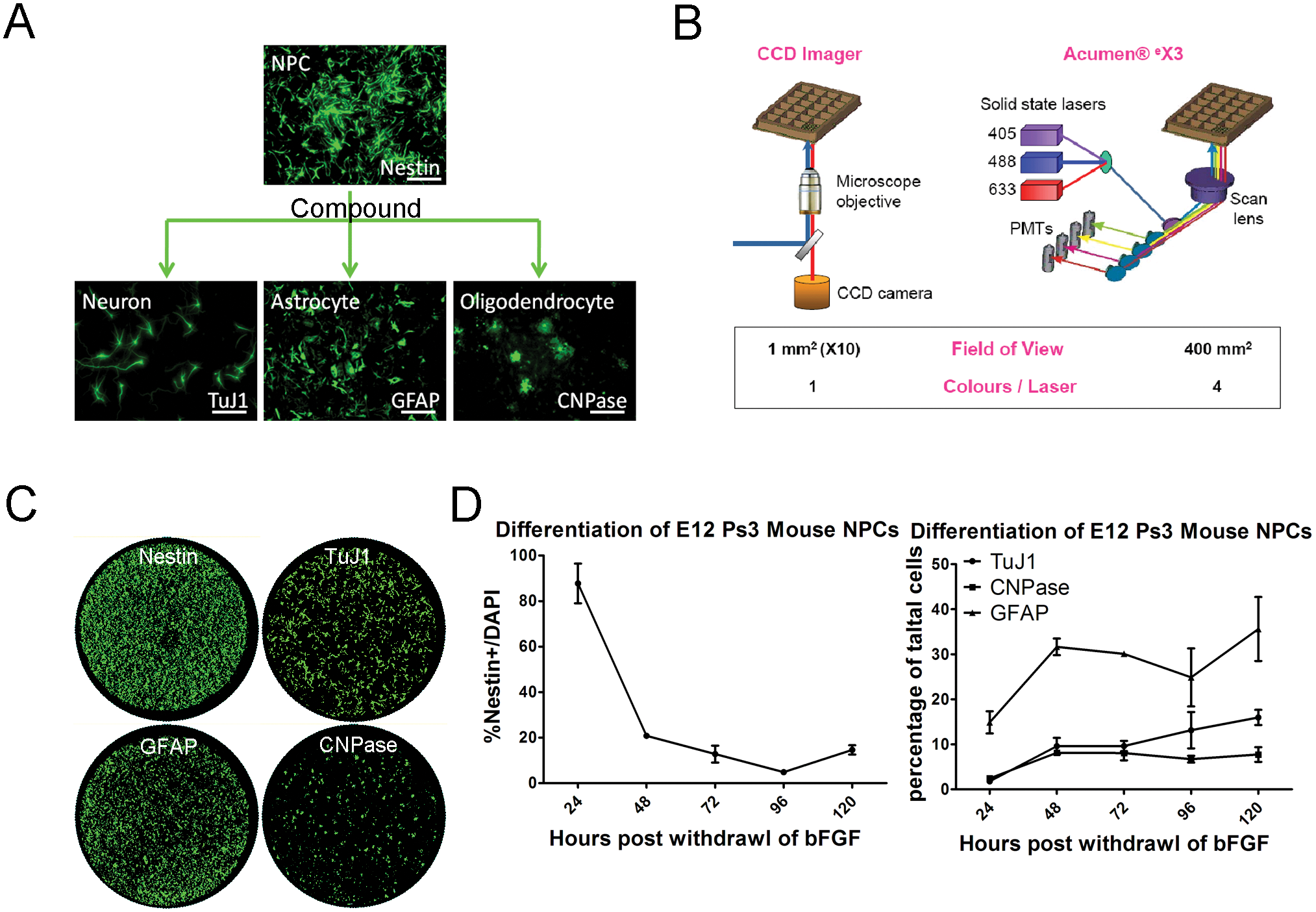
High-content image-based screening of mouse neural precursor cell (NPC) differentiation. (
Screening of LOPAC and C&P compound set
LOPAC and C&P library compounds were screened at 10 µM for their effects on NPC differentiation toward neurons. Compounds were added on the day of bFGF removal to detect a direct effect on differentiation. Compounds that increased percentages of TuJ1+ cells without significantly changing the total number of cells were chosen for confirmation in replicates and in dose–response studies. Hits were chosen at a stringent criterion of three times the standard deviation above the average of the DMSO control. Images of hits were analyzed to eliminate false positives with high background fluorescence from the compounds. Of 1258 LOPAC and 8438 C&P compounds, 9 and 33 compounds increased the percentage of TuJ1-positive neurons in a reproducible and dose-dependent manner, respectively. The signal (reference compound) to background (DMSO) ratio was about 4; Z′ factor = 0.4; coefficient of variation (CV) = ~10%.
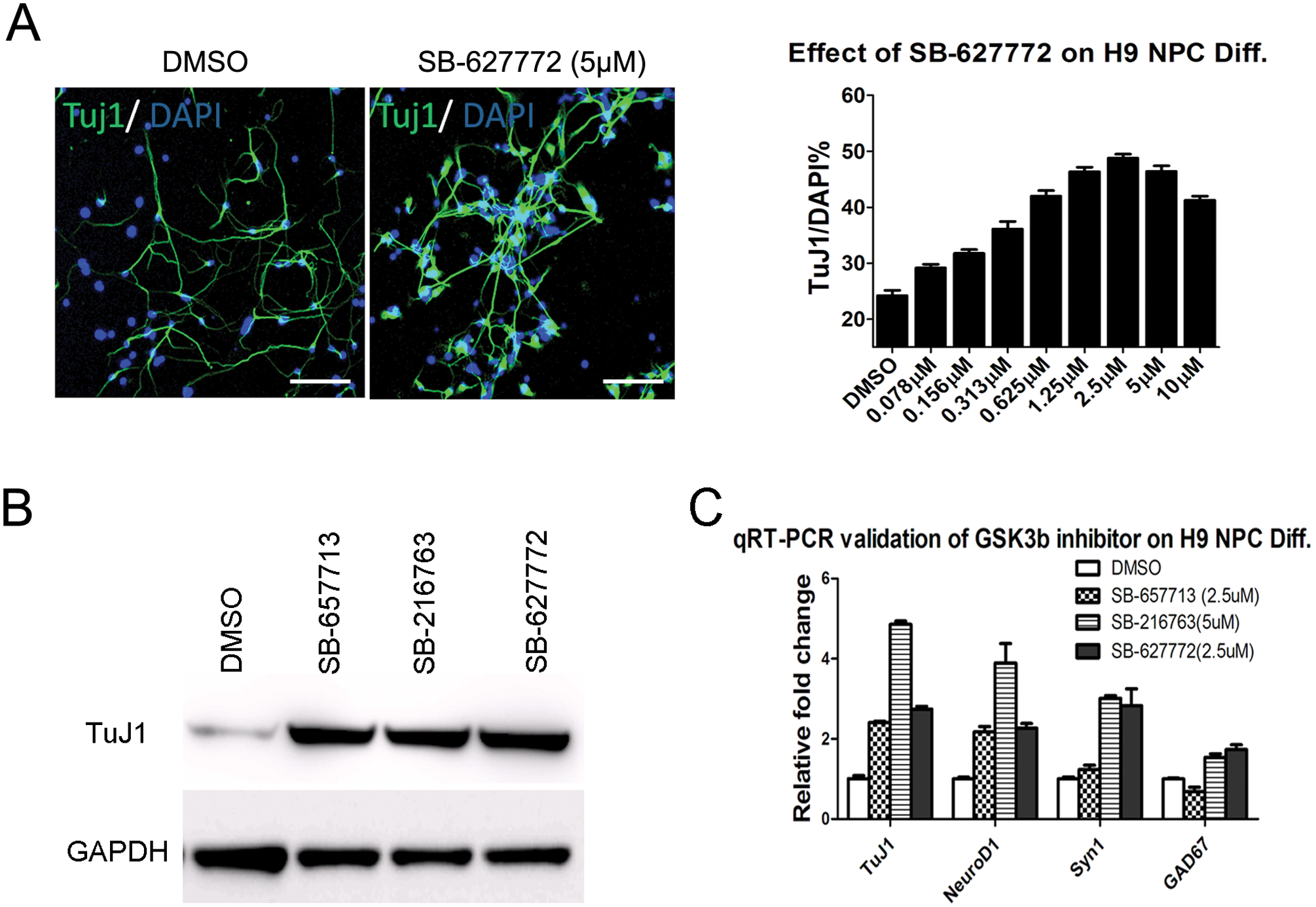
Interestingly, of 42 pro-neuronal compounds from two libraries, 27 compounds are known GSK3β inhibitors. Given its known role in Wnt signaling pathway and in stem cell differentiation, it is not surprising that GSK3β inhibitors were identified through the NPC differentiation screen. 20 Thus, we decided to validate our screening by confirming the effect of GSK3β hit compounds. Whole-well immunofluorescence analyses and calculated percentages of three representative GSK3β inhibitor hit compounds (SB-216763 from the LOPAC library and SB-657713 and SB-627772 from the C&P library) are shown in Figure 2A . These GSK3β inhibitors increased the percentage of TuJ1+ cells from a baseline of 20% (DMSO) to about 50% to 60% (hit compound) in 384-well plates. By manually inspecting whole-well images captured by the Acumen eX3, we noted a significant increase in TuJ1+ cells in GSK3β inhibitor hit compound-treated wells. Via dose–response study, the EC50 (~1 µM) of three hit compounds was found ( Fig. 2B ). The Western blot analysis of compound-treated, 5-day differentiated cells showed an increase in the total amount of TuJ1 protein expression when compared to the DMSO control ( Fig. 2C ). In addition to TuJ1 staining and Western blot, the expressions of additional cell type–specific genes were evaluated at mRNA levels to confirm an increase in neuronal phenotype after treatment of the hit compounds. Expressions of neuronal markers, such as NeuroD1, TuJ1, and synapsin1 (Syn1), increased, whereas expressions of glial markers, such as CNPase, MBP, and GFAP, decreased ( Fig. 2D ).
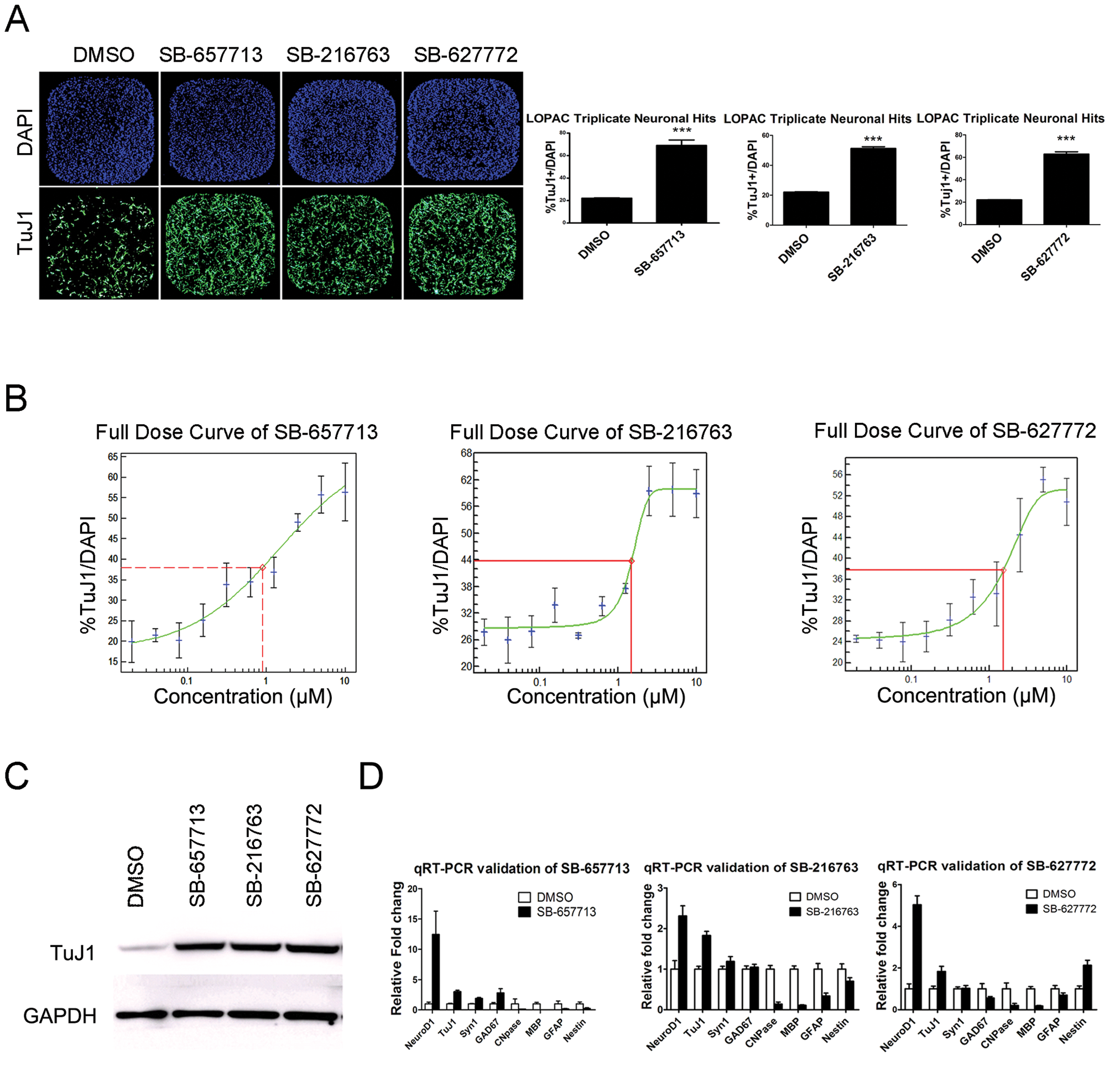
Compound screening result and validation of the pro-neuronal differentiation compounds. (
GSK3β inhibition enhances neural stem cell differentiation toward neurons
Because it is common for a kinase inhibitor to target multiple kinases, we profiled 15 hit compounds that have been shown to target GSK3β, including the aforementioned SB-216763, SB-657713, and SB-627772 (data not shown). SB-216763 is reported to inhibit multiple other kinases, including ERK8, SRPK1, HIPK2, and CDK2. 21 According to our profiling, GSK3β inhibitors also inhibited multiple kinases besides GSK3β, but CDK2 was the only other target shared among them. To figure out whether the pro-neuronal effects of compounds SB-657713, SB-216763, and SB-627772 are indeed through inhibiting GSK3β, we performed the differentiation assay using a highly selective GSK3β inhibitor, CT99021, which only inhibits GSK3β and CDK2, and two CDK2-specific inhibitors, roscovitine and purvalanol, which do not inhibit kinase activity of GSK3β. 21 CT99021 increased TuJ1+ neuronal percentage in a dose-dependent manner ( Fig. 3A ), whereas CDK2 inhibitors had no effect ( Fig. 3B ), suggesting that the pro-neuronal effect of hit compounds is not mediated by inhibition of CDK2. In addition to increasing neuronal percentage, total TuJ1 protein expression of CT99021-treated cells was higher than DMSO- or roscovitine-treated cells ( Fig. 3C ). This suggests that the pro-neuronal effect of hit compounds is not mediated by inhibition of CDK2.
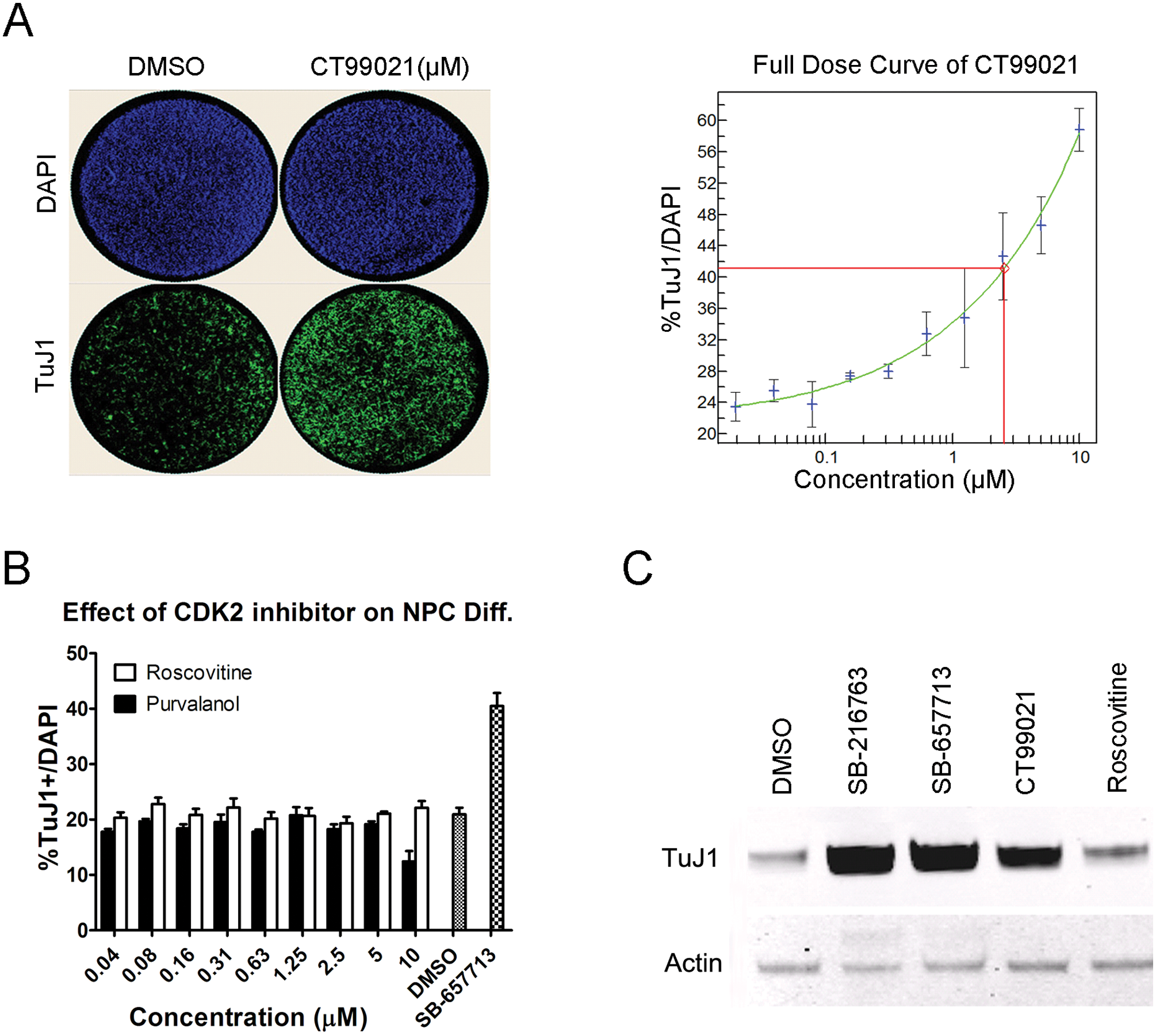
Specific effect of GSK3β inhibitor on promoting neurogenesis. (
As a well-known negative regulator of the Wnt signaling pathway, GSK3β phosphorylates β-catenin, leading to eventual degradation of β-catenin. Thus, unphosphorylated (activated) β-catenin level is an indicator of GSK3β activity. As expected from the inhibition of GSK3β, expression of activated β-catenin was upregulated in NPCs treated with pro-neuronal GSK3β inhibitors SB-216763, SB-727772, and SB-657713 for only 2 h ( Fig. 4A ). In addition, after 16 h of compound treatment, gene expression of β-catenin target Lef1 was upregulated as well as the pro-neural basic helix-loop-helix (bHLH) transcriptional factor NeuroD1 ( Fig. 4B ).
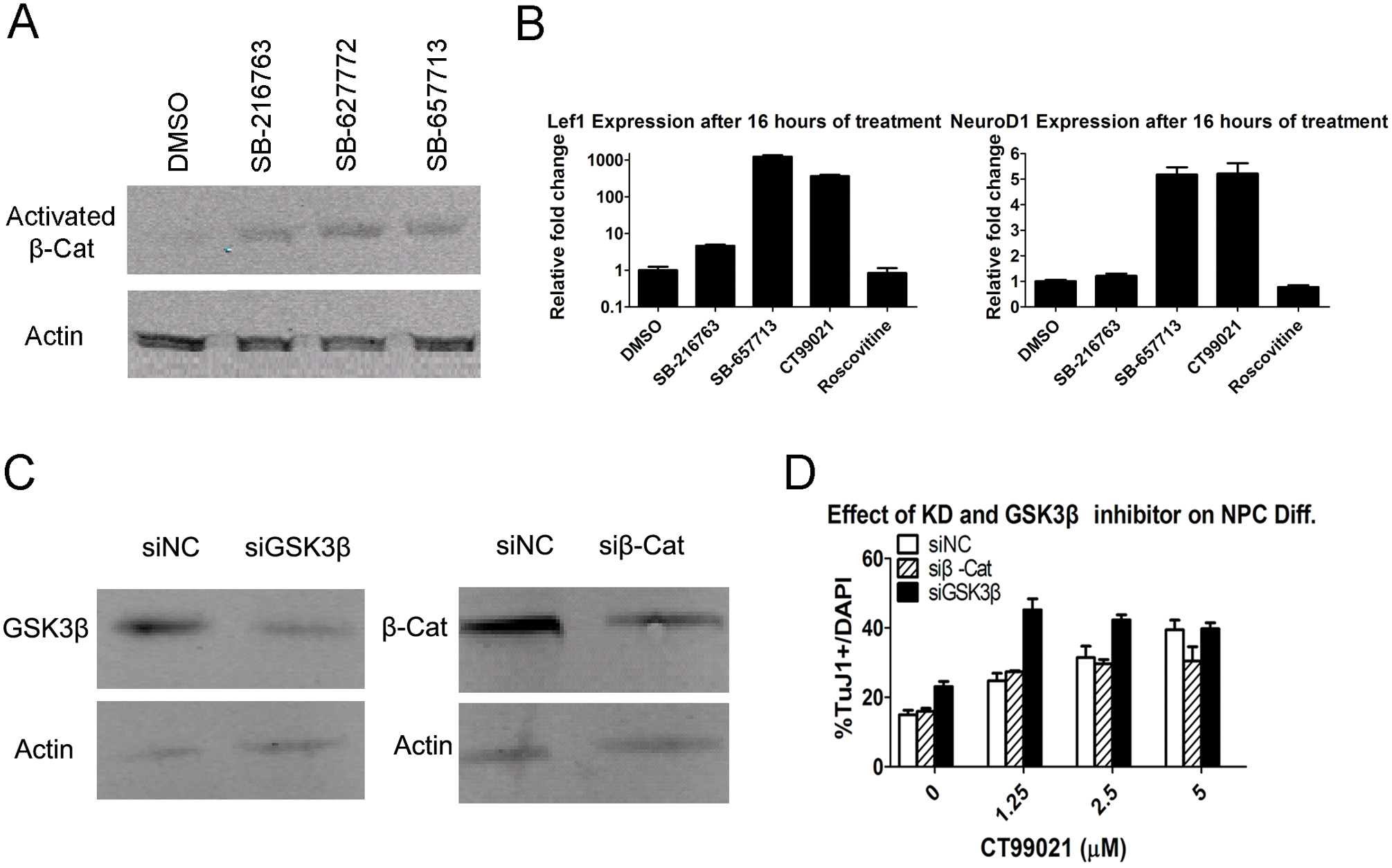
Validation of GSK3β as a target of hit compounds. (
To fully validate GSK3β as a target of these pro-neuronal hit compounds, we designed siRNAs to knock down expression of GSK3β and β-catenin ( Fig. 4C ). The knockdown of GSK3β, but not β-catenin, increased TuJ1-positive neuronal percentages ( Fig. 4D ). In addition, GSK3β knockdown cells treated with GSK3β inhibitor CT99021 created a synergistic effect and caused knockdown cells to reach maximum neuronal differentiation at a lower concentration than control cells (1.25 µM vs. 5 µM). Taken all together, GSK3β is a potent and reproducible target of pro-neuronal hits found through our screening.
GSK3β inhibitors promote neurogenesis in the adult SVZ in vivo
The SVZ of the lateral ventricle (LV) is considered one of two neurogenic regions of the adult brain, in addition to the subgranular zone (SGZ) of the hippocampus.22,23 In the normal rodent brain, NPCs generated from SVZ migrate through the rostral migratory stream (RMS) and differentiate into olfactory bulb interneurons. 24 Moreover, reports suggest that under damage conditions, SVZ NPCs will differentiate into neurons and migrate to the injury site.2,25,26 Thus, it is of interest to see whether GSK3β inhibitors can enhance endogenous adult neurogenesis in the mouse brain, specifically in the SVZ. We examined the generation of newly generated neurons in the SVZ of the adult mouse brain at different time points after GSK3β inhibitor (SB-627772) treatment. SB-627772 was selected because it was easily dissolved in PBS at a high concentration. BrdU and DCX antibody were used to label newborn neurons in the SVZ. Three days after SB-627772 administration into the LV, we did not observe any significant differences between the control and SB-627772-treated groups ( Fig. 5A ). However, 6 days after SB-627772 administration, we observed a significant increase in DCX and BrdU double-labeled neurons in the SVZ in the SB-627772-treated group versus the control group (80 ± 13 vs. 21 ± 6 double-positive cells/mm2; Fig. 5A , B , representative immunofluorescence image of SVZ). Our GSK3β inhibitor (SB-627772) identified from an in vitro phonotypic screen can significantly promote endogenous neurogenesis in the SVZ region in vivo.
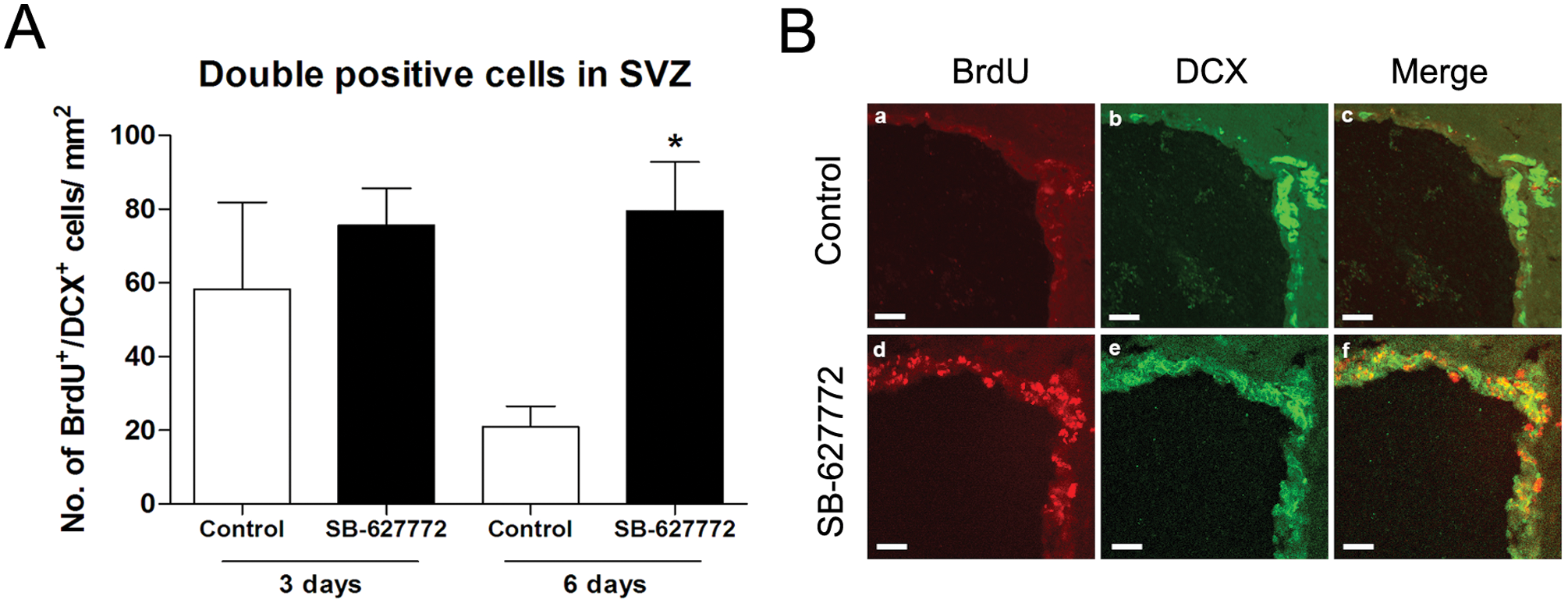
GSK3β inhibitor promotes neurogenesis in the subventricular zone (SVZ) of mouse brain. (
GSK3β inhibitors promote neurogenesis in human ES-derived NSCs
Although mouse NPCs are a great system for phenotypic screen assays, to be translated into human biology, they need to be tested on human cells. Nestin+ and Sox2+ GIBCO human NSCs derived from a National Institutes of Health (NIH)–approved H9 ESC line were obtained for validation of the effect of pro-neuronal GSK3β inhibitors. A dose-dependent increase in neurogenesis was observed in samples treated with GSK3β inhibitors measured by the number of TuJ1+ cells (
Fig. 6A
and
Validation of pro-neuronal GSK3β inhibitors in human H9 neural precursor cells (NPCs). (
Discussion
We describe a phenotypic screen for compounds with the ability to enhance neural stem cell differentiation toward neurons. By combining methods readily used in academia and industry, we demonstrate the possibility of using lineage-specific stem cells to identify small molecules and genes involved in stem cell differentiation. Applying cell type–specific marker and whole-well imaging analysis of primary or progenitor cells, additional screens can be devised in the future.
Following the establishment of human embryonic stem cells in 1998, there has been tremendous interest in using human cells differentiated from stem cells to model the disease process for screening, as they would represent a scalable source of human cells with a consistent genetic background and reproducible quality.27,28 Yet, most current protocols to induce differentiation of a specific cell type are not efficient or cost-effective. Desbordes et al. 29 used hESCs and screened for compounds capable of altering the expression of pluripotency marker OCT4. The authors identified compounds capable of inducing expression of endoderm lineage marker Sox17 or neuroectoderm marker Pax6. Replacing growth factors with small molecules in the differentiation protocol will likely reduce the cost of large-scale production and, in some cases, will likely improve the yield of the process, as demonstrated by our study.
Through our proof-of-principle phenotypic screen using mouse NPCs for small-molecule modulators of neurogenesis, we identified a group of GSK3β inhibitors as potent inducers of neuronal differentiation. We further validated their function in mouse adult SVZ and also human NSCs. Inhibition of GSK3β activity will lead to activation of Wnt signaling, which has been shown to direct neuronal differentiation of cortical neural precursors. 20 Thus, it is highly likely that our GSK3β inhibitors promote neurogenesis through activating Wnt/β-catenin function, at least in part, as suggested by our siRNA knockdown β-catenin experiment ( Fig. 4D ). GSK3β has long been implicated in major neurological disorders such as Alzheimer disease, schizophrenia, and bipolar disorders.31,32,33 It will be of interest to test GSK3β inhibitors in a hippocampus lesion model and examine whether it can induce neurogenesis and have functional behavioral recovery.
A more intriguing possibility of these compounds is the therapeutic use of small molecules targeting endogenous neural stem cells. Although SVZ and SGZ are widely recognized as brain regions harboring neural stem cells capable of differentiating into neurons, other regions of the brain, such as the midbrain, also have been suggested to be neurogenic. 33 The robust evidence of neurogenesis in these regions and of their integration into proper neural circuits to support neural repair is an area of debate, but if the repair mechanism is hampered in the disease state, enhancing the endogenous progenitor cells’ ability using chemical compounds can be explored.
Footnotes
Acknowledgements
We thank Dr. Yi E. Sun (UCLA) for setting up the mouse neural precursor cell differentiation system at GlaxoSmithKline(China)R&D Co., Ltd. and initial conceptual contribution to this work.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
