Abstract
Multicellular tumor spheroids (MCTS) are routinely employed as three-dimensional in vitro models to study tumor biology. Cultivation of MCTS in spinner flasks provides better growing conditions, especially with regard to the availability of nutrients and oxygen, when compared with microtiter plates. The main endpoint of drug response experiments is spheroid size. It is common practice to analyze spheroid size manually with a microscope and an ocular micrometer. This requires removal of some spheroids from the flask, which entails major limitations such as loss of MCTS and the risk of contamination. With this new approach, the authors present an efficient and highly reproducible method to analyze the size of complete MCTS populations in culture containers with transparent, flat bottoms. MCTS sediments are digitally scanned and spheroid volumes are calculated by computerized image analysis. The equipment includes regular office hardware (personal computer, flatbed scanner) and software (Adobe Photoshop, Microsoft Excel, ImageJ). The accuracy and precision of the method were tested using industrial precision steel beads with known diameter. In summary, in comparison with other methods, this approach provides benefits in terms of semiautomation, noninvasiveness, and low costs.
Introduction
There are a variety of culture techniques available for obtaining MCTS.2,3,9 Our previously described approach combines the two major culturing methods. 10 First, a defined volume of cell suspension is given into the wells of a 96-well plate coated with agarose. One spheroid is formed in each well. Spheroids obtained with this method are of nearly identical size, establishing the basis for drug response experiments. Second, a defined number of spheroids is transferred into a spinner flask. In these specially shaped flasks equipped with a stirring device, the spheroids are kept under constant motion. Thus, a standardized supply with gases and nutrients is guaranteed. Consequently, spheroids can be cultured over a longer experimental period and reach much larger sizes than in multititer plates. One major advantage of stationary culture techniques such as MCTS growth in multiwell plates is the requirement of only minimum amounts of media and active pharmaceutical ingredients. But with the use of mini spinner flasks (20 mL volume of cell suspension vs 100–300 mL in conventional spinner flasks), the consumption of agents is clearly reduced.
In the clinical setting, the efficacy of any treatment is proven by reduced tumor growth and, eventually, reduced tumor mass. Analogously, spheroid volume can be used as a measure of efficacy in preclinical therapy studies. The more accurately this parameter is registered, the less the investigator needs to interfere with the cultures and the greater is the informative value of such investigations. Using the method presented here, the growth of a complete spheroid population is analyzed directly in culture containers with transparent and flat bottoms. For measurement, the bottom of an appropriate culture container with the sedimented MCTS is scanned digitally, and the diameter and volume of every MCTS are determined by analyzing the imaged spheroid areas. A conventional personal computer and a flatbed scanner are used. Common software applications Microsoft Excel and Adobe Photoshop are employed for the analysis. The actual digital image processing is performed using the open source software ImageJ of the National Institutes of Health.
Materials and Methods
Cultivation of MCTS
The method was established using MCTS cultivated in stirred culture medium. For this purpose, spheroids are placed in special spinner flasks (Wheaton, Millville, NJ) with a 20 mL culture medium capacity. Through an opening at the top, a glass bar is inserted into the flask. At one end stands a self-established polyether ether ketone–coated dark-colored stirring device. The magnet of the stirring device is rotated by means of an induction stirring plate (VWR, Darmstadt, Germany) on which the spinner flask is positioned. As the culture medium is constantly stirred, the spheroids remain in continuous motion. The entire assembly is placed in an incubator at 37 °C and 5% CO2 in humidified air.
Hardware
For volume growth analysis, a standard office flatbed scanner is used (Epson Perfection 2400 PHOTO). However, any flatbed scanner with a resolution that fulfills the required standard may be used. The scanning region is set manually to the synthetic mask (
Software
The standard computer programs Adobe Photoshop, Microsoft Excel, and the open-source software ImageJ were used. All programs are available for Microsoft Windows as well as Mac OS X.
Adobe Photoshop
The protocol is run in Photoshop via the menu item “Actions.” The version used here was Photoshop CS3. However, as the action protocols are upward compatible, they can also be executed in Photoshop CS4 or CS5.
Adobe Photoshop Action Protocol

ImageJ
ImageJ is a platform-independent open-source program of the National Institutes of Health (http://rsb.info.nih.gov/ij/, version 1.44). The image is processed and analyzed using several existing and automatic plug-ins that require no intervention on the part of the user. From the large number of options provided by ImageJ, three principal functions are used for our purposes: the creation of a 2-bit image by the threshold procedure, watershed transformation, and calculation of area by the “analyze particles” function. The individual work steps can be recorded as a macro in ImageJ and need not be executed manually (
ImageJ Plug-in

Microsoft Excel
The pixel values analyzed by ImageJ are calculated in Excel. First, the data from the image stack of the “results” window is separated for each “slice” by implementing an inquiry for the slice number. In addition, only pixel areas that fit into a defined area range—thus excluding small artifacts or spheroid aggregates from the analysis—can be selected for further calculation. By using the known scan resolution, the pixel counts are transformed into millimeters. In the prepared Excel file, the mean diameter (mm), mean volume (mm3), and respective standard deviation or the standard error of mean are calculated. Likewise, the total number of spheroids and the minimum and maximum values are given.
Results and Discussion
Measurement is fast and noninvasive
For measurement, the spinner flasks are taken from the incubator and placed on the scanning field of the flatbed scanner (
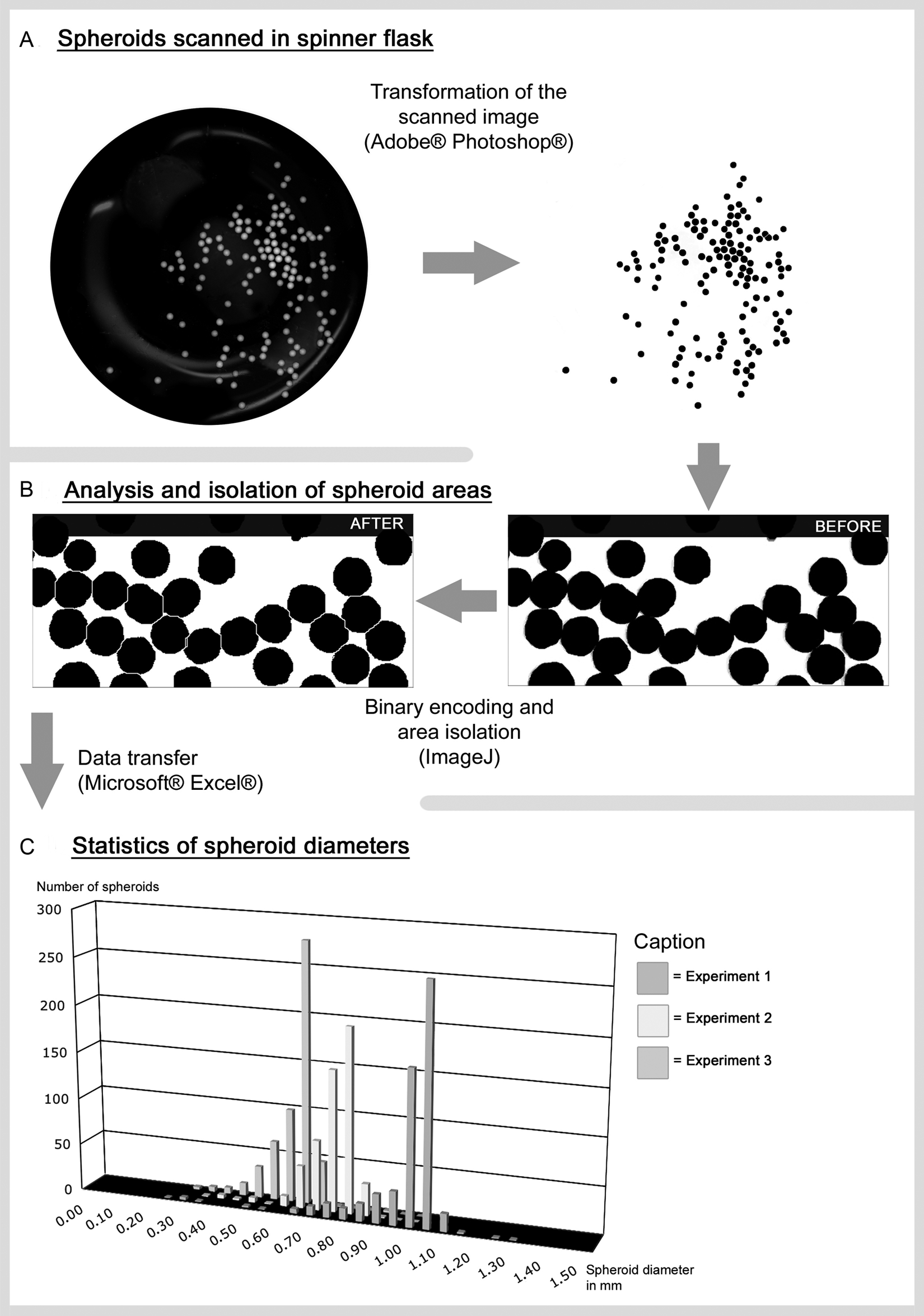
Working steps of spheroid analysis. The steps from the scanned image to the data diagram involve several semiautomatic steps (
Image processing by Photoshop is mainly automated
From the color image thus obtained, a gray-scale image is created via several steps, using the Photoshop software (
ImageJ generates spheroid sizes in the form of pixels
The gray-scale image is first transformed into a binary image through a threshold procedure. Here the spheroid areas are colored black and the background is white (
Microsoft Excel is used to calculate spheroid volume
The raw data (pixel values) obtained from ImageJ are converted into calibrated diameters (mm) and volumes (mm
3
) using the spreadsheet program MS Excel (
Steel beads demonstrate good precision and accuracy
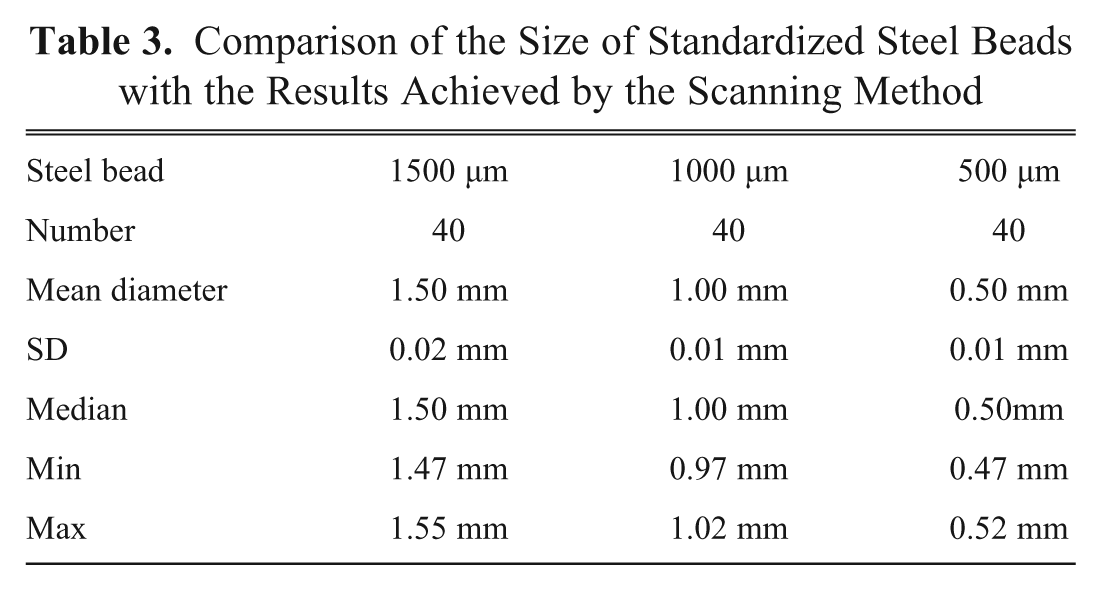
The accuracy and precision of the method were tested by the use of steel beads. Industrial precision beads made of steel according to ISO 3290, and diameters of 500, 1000, and 1500 µm were used (JSK°Nanoball GmbH, Wermelskirchen, Germany, www.nanoball.com). The beads were filled into the spinner flasks and then subjected to the above-mentioned procedure. However, in contrast to scanning MCTS, the specular reflections of the steel had to be removed manually on Photoshop. Forty steel beads of every size were analyzed (
Comparison of the Size of Standardized Steel Beads with the Results Achieved by the Scanning Method
Advantages of the method
Automation-related benefits
By fully scanning the bottom of the spinner flask, practically the entire spheroid population can be analyzed in a single step. The data thus obtained permit a more valid statement about alterations in the growth behavior of spheroids under experimental conditions when compared with the extrapolation of values concerning the size of individual MCTS obtained by microscopic investigation. The test measurements on high-precision steel beads (500, 1000, and 1500 µm) yielded the same average diameters as the corresponding reference values (difference <10%). The standard deviations were only between 10 and 20 µm.
In contrast to the measurement of random samples, this analytical procedure permits the investigator to perform measurements at user-defined intervals. Over a period of several days, precise data can thus be obtained about volume growth under treatment or control conditions. For instance, the investigator will be able to determine whether spheroid growth is restricted only at the start of treatment or persists during the entire period of investigation.
The results of semiautomatic spheroid evaluation can be registered in numbers as well as images and can also be recalled and checked as files. This uncouples data assessment and analysis. As standard graphic software (Adobe Photoshop) is used, there is no difficulty with regard to the compatibility of image files; such difficulties may well be encountered when using exclusive software solutions for special equipment. Thus, the investigator can share the images with coworkers at any personal computer equipped with Photoshop. Furthermore, the files can also be converted later—at any time—into any available file type or processed for the purpose of publication.
As the procedure is automated, spheroid analysis is executed much more rapidly than by the previously used microscope. Individual operators are much less likely to influence the results.
Noninvasiveness
This new method does not require an opening of the flask. This greatly simplifies handling and reduces the risk of contamination by bacteria and fungi. Furthermore, by placing a lay-on mask on the scanner, several spinner flasks can be scanned simultaneously. This step further reduces flask manipulation time outside the incubator and with it the effects on the cultures.
As spheroid size is registered only in the form of images, the investigator need not remove tumor cells for performing the measurements. The number of MCTS used at the beginning does not change. Independent of this fact, however, a change in the number of spheroids due to necrosis of cells or new growth of MCTS may still occur.
When using the conventional measuring procedure, individual spheroids are taken with a pipette from the spinner flasks. In addition to the previously mentioned loss of tumor cells, a small quantity of the medium is also removed by this procedure. Depending on the experimental approach, this involves a shift in concentration ratios at every measurement, such as the ratio between tumor cell count and active substance. This can be avoided by the noninvasive method of spheroid assessment.
Low costs
In contrast to many other laboratory devices, semiautomatic growth analysis of three-dimensional cell cultures involves no major financial investment. For the analysis protocol, one needs only a flatbed scanner, a computer, and three programs: Adobe Photoshop, Microsoft Excel, and the open-source software ImageJ.
Disadvantages of the method
Spinner flask cultures do not allow for a follow-up of individual spheroids, which is also true for the current method. Although miniaturized, the current spinner flasks still require relatively large amounts of culture media and tested drugs. Cleaning and sterilizing of a significant number of glassware represent further disadvantages of the method. Finally, prerequisite to the spheroid scanning procedure is a high optical quality of the bottom of the spinner flask. This may limit the applicability of the technique.
Comparison with other analytical procedures
As spheroid cultures have been used for several decades now in tumor research as a three-dimensional model, a large variety of analytical techniques has been applied during this time to determine spheroid size. A common approach is to measure the MCTS with an inverse microscope using a calibrated ocular micrometer.11–15 To enhance contrast, a phase contrast microscope may be used. 6 The disadvantages of this method of evaluation are (1) the fact that one needs to interfere with the culture, which hinders its use as an instrument to monitor growth, and (2) manual analysis of large spheroid numbers is tedious and laborious, whereas this evaluation can be performed automatically by the developed method. Similar problems are encountered when photographing spheroids with a microscope and a Polaroid MicroCam. 16 Compared to analysis with the ocular micrometer, here one has the option of saving the images and evaluating them later. A further approach is the method of flow cytometry, which was modified and established for spheroid analysis by J. P. Freyer et al. 17 This procedure permits automatic analysis of very large numbers of spheroids but requires modification of the measuring device. 6 Besides, at the time of its establishment, the measurable spheroid size was limited to about 500 µm. 18 Although this limit can be increased to 1500 µm by the use of new devices for flow cytometry (Union Biometrica COPAS), the method can be used only outside the suspension environment. 19 Impedance spectroscopy is subject to similar limitations. 20 By this method, which was submitted for a patent in the year 2000, a large number of MCTS can be analyzed, but the spheroid size is limited to about 400 µm. 21 Characterization of MCTS is performed by individual measurements—as is done in flow cytometry—and is unsuitable for the observation of spheroids in spinner flasks. It may be summarized that the method of semiautomatic growth analysis of three-dimensional cell cultures is a very good alternative to conventional analytical procedures or commercial solutions for the determination of spheroid size. This should encourage other investigators to devote more attention to spinner flask usage for MCTS drug response experiments due to the better correlation with in vivo conditions. The next step in maximizing the attractiveness of the culture method would be the implementation of even smaller spinner flasks to reduce the costs of culture media and test drugs. Another improvement would be the commercial availability of smaller disposable flasks. Maximizing ease of use and minimizing the negative effects on the culture could be achieved by the development of devices that combine stirring and scanning capabilities.
In summary, the method of semiautomatic growth analysis of three-dimensional cell cultures here described is a good alternative to conventional analytical procedures or commercial solutions for the determination of spheroid size. The method allows for a time-efficient, reproducible, and standardized method of size measurement of multicellular tumor spheroids. This is of great interest in preclinical pharmaceutical testing.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
