Abstract
Lipophilicity is an important parameter for any potential drug candidate. Accurate and efficient lipophilicity measurements facilitate the development of high-quality predictive in silico models that support the design of future drugs. Lipophilicity estimates derived from the traditional 1-octanol/water shake flask techniques have been the most widely employed and are therefore the best understood. This technique can be considered to give a good measure of a compound’s lipophilicity, albeit slower and more labor intensive to run compared with some other methodologies. Herein is described and validated an efficient 1-octanol/water shake flask technique that has sufficient capacity to be run as a primary screen within the drug discovery process. This is achieved by the simultaneous measurement of the distribution coefficients of mixtures of up to 10 compounds using high-performance liquid chromatography and tandem mass spectrometry. Concerns regarding ion pair partitioning that could result in erroneous results due to interactions between compounds within a mixture are discussed.
Introduction
I
It was recently argued that the accuracy of in silico lipophilicity predictions that are widely available from globally trained algorithms is unacceptably poor, with an average error in excess of 1 log unit. 3 It was further argued that such inaccuracy can mislead the design of new analogues and that available experimental lipophilicity data from a particular chemical series can be used to develop high-quality local models that dramatically improve predictivity. This reflects our own experience and is an important driver for the development of accurate and efficient experimental methods.
The traditional shake flask method is considered the gold-standard technique for determining log
The shake flask method has been miniaturized and automated by transferring it to a 96-well microplate format, with direct sampling of the two phases prior to the use of liquid chromatography (LC) and selected ion recording (SIR) mass spectrometry (MS) quantification.
5
A similar technique, which makes use of a water plug aspiration and injection method, has also been described.
6
The determination of log
The method that has been developed is a miniaturized and automated shake flask log
Materials And Methods
Materials
Columns for LC were obtained from Waters Ltd. (Elstree, Hertfordshire, UK). 1-Octanol was obtained from Sigma Aldrich (St. Louis, MO), and all other consumables where obtained from Thermo Fisher Scientific (Loughborough, UK). The pH 7.4, 0.02 M, phosphate buffer was prepared by dissolving NaH2PO4.2H2O (0.62 g) and Na2HPO4.2H2O (2.85 g) in water (1000 mL). The 1-octanol and pH 7.4, 0.02 M, phosphate buffer were shaken together for 18 h and the phases allowed to separate prior to use.
Instrumentation and analysis
Pre- and postdata handling procedures were carried out using custom-built Microsoft Excel workbook add-ins. Automated liquid sample handling was performed using a Tecan Freedon Evo 150 (Tecan, Männedorf, Switzerland) robot fitted with an eight-disposable tips configuration and controlled by EVOware software (Tecan). The use of disposable tips eliminated contamination issues relating to sample carryover. Centrifugations were carried out using a Beckmann Allegra R6 (Beckmann Coulter, Palo Alto, CA). Shaking of samples was carried out using an IKA KS 260 (IKA, Wilmington, NC) orbital shaker set at 450 rpm. Experiments were carried out in a constant-temperature laboratory held at 20 °C. All LC analyses were carried out using a Waters 2690 separations module and Waters 2777 autosampler fitted with a Peltier cooler set to 20 °C. A Waters 996 diode array detector was used for UV peak confirmation. MS detection was carried out using a Waters Quattro Premier XE atmospheric pressure tandem quadrupole mass spectrometer (Premier-MS) operating in the multiple-reaction monitoring (MRM) mode. 16 The Premier-MS was controlled using MassLynx software (Waters; refer to operating guides available from Waters Ltd. at www.waters.com). Waters QuanOptimise application manager was used to automatically generate MRM methods for compounds. Waters QuanLynx software was used to automatically process all the MRM chromatograms. Waters Symmetry C8 3.5 µm, 4.6 × 50-mm columns were used for LC along with a gradient of 1% acetonitrile/99% 0.1% aqueous formic acid to 99% acetonitrile/1% 0.1% aqueous formic acid at a flow rate of 2 mL min−1 over 5.0 min. All pH measurements were conducted using a Fisher Scientific AR20 pH/Conductivity meter (Thermo Fisher Scientific). A Milli-Q water purification system (Millipore, Billerica, MA) was used to supply water.
General procedure for log D
7.4 measurements
Compounds (1 mg) were dispensed into individual 1-mL polypropylene vials, held within a 96-well plate along with 1-octanol (700 µL), and presaturated with 0.02 M phosphate buffer (pH 7.4). The plate was then shaken overnight followed by centrifugation (800
Results
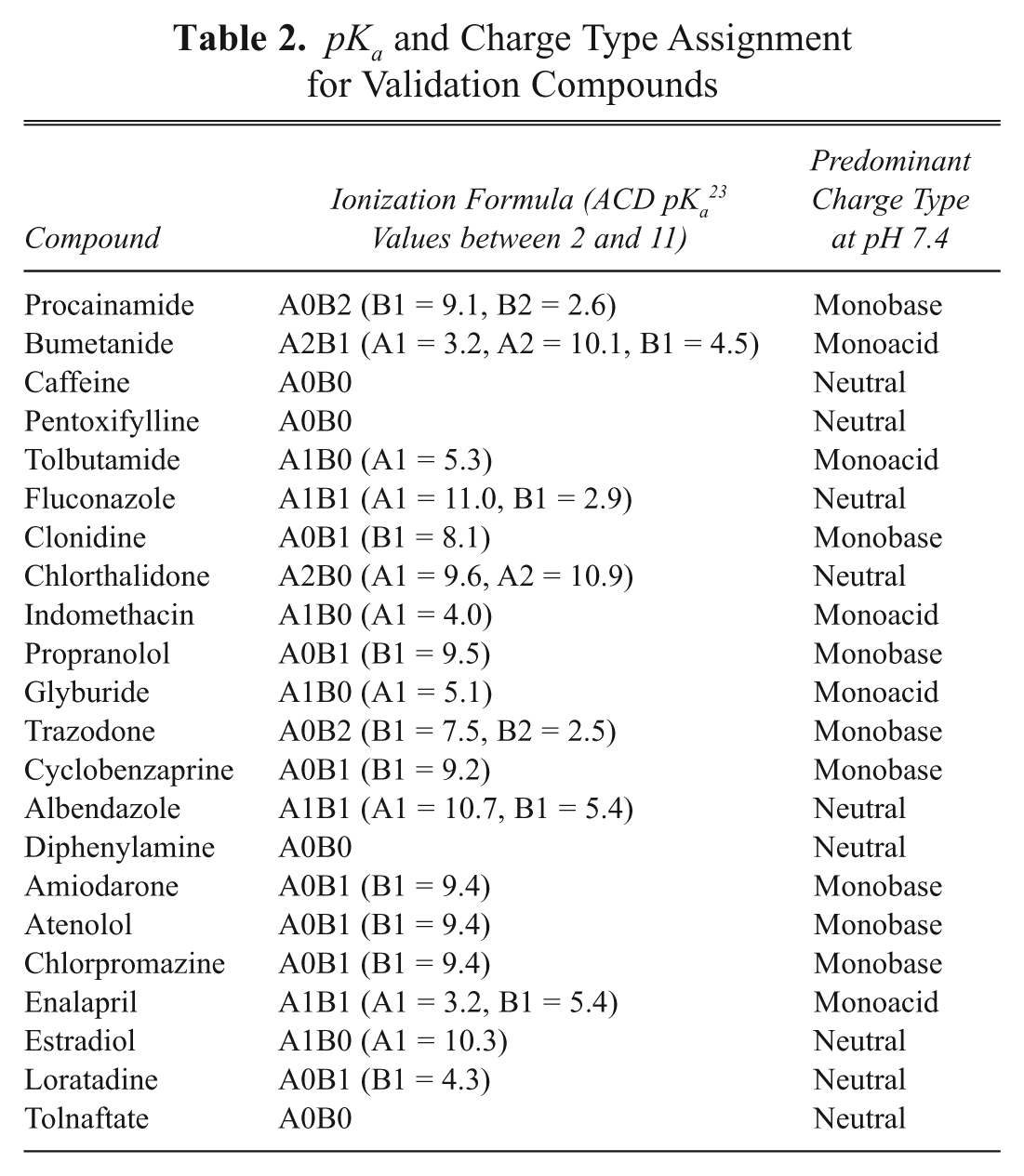
A set of 22 commercially available compounds exhibiting a wide range of lipophilicity and ionization class were selected for validation of the experimental method. To validate the use of mixtures of compounds, the method was first applied to the log
Log
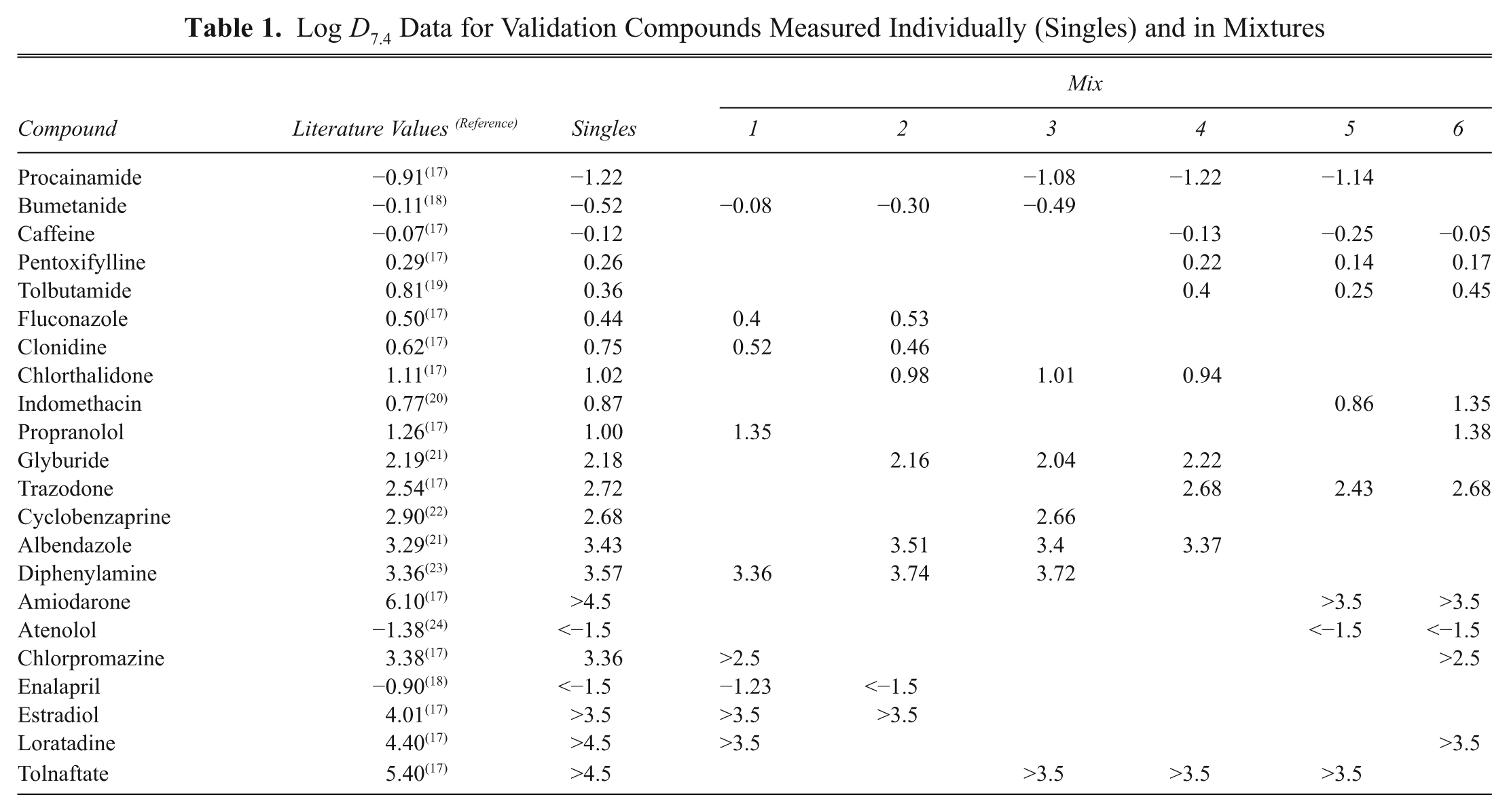
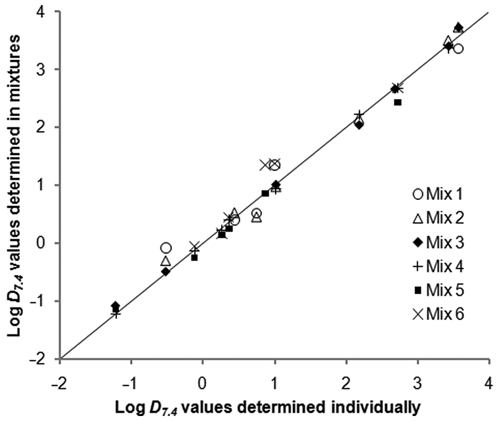
Comparison of log
To test the assay-to-assay precision of the mixture-based shake flask technique, the log
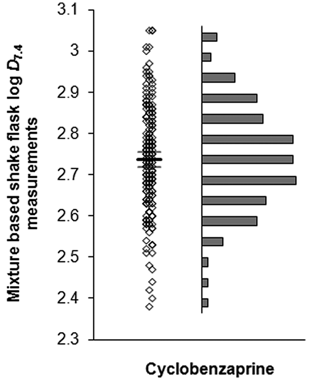
Plot of the spread in the 200 repeat log
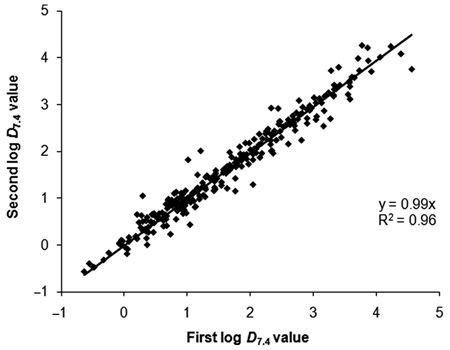
Repeat log
Discussion
Experimental complexity
The experimental methodology extensively uses standard robotic liquid handling techniques and automated MS data processing to vastly reduce the manual effort needed to run a traditional log
Two aspects of running a log
Dynamic range
The experimental procedure leads to an effective log
Technical information regarding MRM MS
The procedure uses mixture-based MRM MS methods containing the individual MRM MS method for each compound in a mixture. During the LC/MS analysis stage, the Premier-MS applies the approximate mixture-based MRM MS method and acquires the data by cycling through the individual MRM MS methods for each compound in a mixture. Each individual MRM MS method is tailored so that the Premier-MS only collects data for the major daughter ion of the parent ion, making this detection method highly sensitive. The Premier-MS is capable of collecting enough MS scans for each daughter ion in a mixture of 10 compounds for a typical peak width (refer to operating guides available from Waters Ltd. at www.waters.com). This is illustrated in Figure 4 . The top chromatogram shows the MS response peak versus the number of scans when the Premier-MS is set to just collect data for bumetamide. Similarly, the bottom chromatogram shows the data for bumetamide where it has been collected along with data for nine other compounds. The only difference in the methods used to generate these two chromatograms is the dwell time: 0.2 s (top) and 0.035 s (bottom)—the former resulting in 35 scans across the peak compared to 30 for the latter, with both satisfying the minimum quantification requirement of 15 scans to define a peak. However, the bottom chromatogram shows a 7-fold reduction in peak area. Such reductions are tolerable because of the increased sensitivity provided by the Premier-MS. In our experience, the simultaneous analysis of mixtures of up to 10 compounds does not compromise quantification. Mixture-based MRM MS methods of more than 10 compounds are possible, but this will affect the number of MS scans that are collected, and proper quantification of peaks may be compromised. Conversely, compounds with poor MS response or that show chemical instability in aqueous or 1-octanol media may need to be run in mixtures of less than 10 to maximize the collection of MS data. Care should be taken to avoid the pooling of compounds that may chemically react with one another. The risk of this happening is low as lead optimization projects tend to avoid synthesizing drug-like compounds that are chemically reactive because of their potential to cause toxicity.
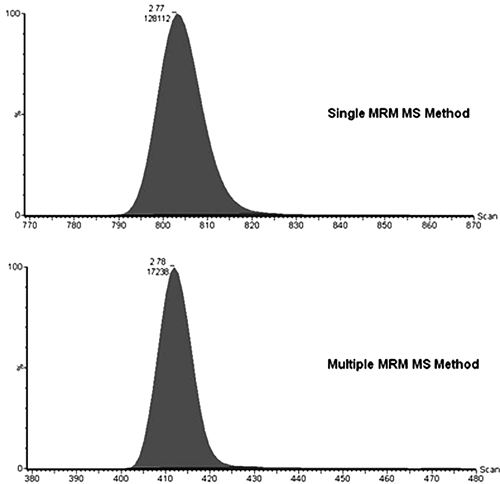
Response chromatographs showing the number of mass spectrometry (MS) scans collected when using a single multiple-reaction monitoring (MRM) MS method for detecting bumetamide alone, compared to a multiple MRM MS method where bumetamide is detected along with nine other MRM transitions. Parent ion (m/z) = 365.03, daughter ion (m/z) = 240.13, cone voltage = 24 v, collision energy = 16 eV.
The Premier-MS is a very high-end capability MS, and the applicability of the described log
Using the aqueous phase dilutions to generate a calibration curve
The situation can arise when the concentration–response calibration curve generated from the 1-octanol phase dilutions is poor or it is inappropriate to be used. This tends to happen for very hydrophilic compounds that have poor MRM MS response, where only one or two of the most concentrated 1-octanol phase dilutions can be quantified. A log
Ion pair partitioning: consideration of erroneous results due to compound–compound interactions
Highly ionized compounds can be extracted as an ion pair, with a buffer counter ion, into the 1-octanol phase, which will cause the observed log
Austin et al.
26
used equation (1) to describe the observed 1-octanol/water distribution coefficient,
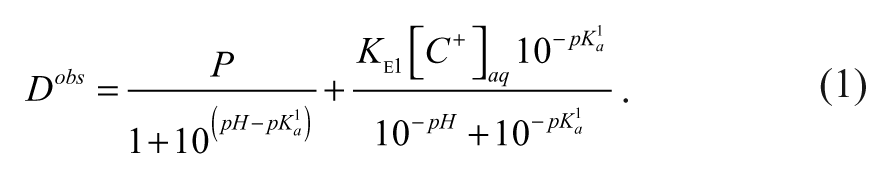
In equation (1),
Scherrer and Donovan
27
have developed a potentiometric titration method in KCl/water-saturated 1-octanol that enables the ion pair partitioning coefficient,
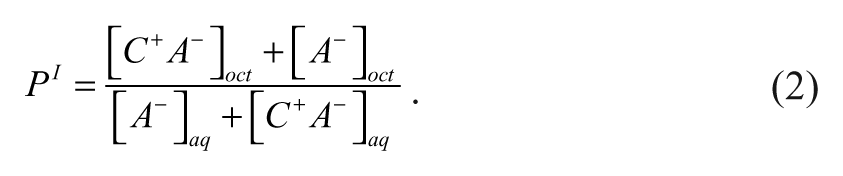
Equation (2) can be simplified if it is assumed that the ionized compound predominantly exists as the ion pair in the 1-octanol phase and as the dissociated ion in the aqueous phase such that [

Equation (3) enables the estimation of
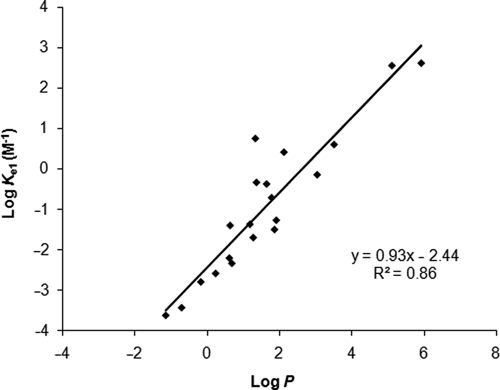
Plot of extraction constants (log
It can be predicted from the correlation in
Figure 5
that
When log
With respect to monobasic compounds, the effect of ion pairing on
This mixture-based shake flask log
