Abstract
The gp41 subunit of the human immunodeficiency virus type 1 (HIV-1) envelope glycoprotein mediates the fusion of viral and host cell membranes. As the HIV-1 enters the host cells, the 2 helical regions, HR1 and HR2, in the ectodomain of gp41 can form a 6-helix bundle, which brings the viral and target cell membranes to close proximity and serves as an attractive target for developing HIV-1 fusion inhibitors. Now, there are several cell- and molecule-based assays to identify potential HIV-1 fusion inhibitors targeting gp41. However, these assays cannot be used universally because they are time-consuming, inconvenient, and expensive. In the present study, the authors expressed and purified GST-HR121 and C43-30a proteins that were derived from the HIV-1 gp41 ectodomain region. GST-HR121 has a function similar to the HR1 peptide of gp41, whereas C43-30a is an HR2-derived peptide that added 50 amino acid residues (aa) in the N-terminal of C43. Further research found they could interact with each other, and a potential HIV-1 fusion inhibitor could inhibit this interaction. On the basis of this fact, a novel, rapid, and economic enzyme-linked immunosorbent assay was established, which can be developed for high-throughput screening of HIV-1 fusion inhibitors.
Introduction
T
Nuclear magnetic resonance and crystallographic analyses demonstrated that the core structure of gp41 consists of 3 HR1 helices as an interior, parallel coiled-coil trimer, whereas 3 HR2 helices are packed in an oblique, antiparallel manner into the highly conserved, deep hydrophobic cavity on the surface of the HR1 helical trimer, which forms a 6-helix bundle (6-HB) of gp41. 4-6 When HIV-1 and host cell membrane fusion occurs, Gp120 binds to CD4 and a coreceptor (e.g., CCR5 or CXCR4), and then gp41 changes its conformation to a prefusion (intermediate) state by inserting its fusion peptide into the target cell membrane. Consequently, the gp41 HR1 associates with the HR2 region to form 6-HB, bringing the viral and target cell membranes to close proximity and eventually fusion of HIV-1 with the target cells.
The “intermediate state” of gp41 provides a window in terms of time and space for some synthetic peptides and small molecular compounds to block the formation of the fusion-active core, and they inhibit fusion. Synthetic peptides mimic the N-peptides and C-peptides to form a heterogeneous 6-HB with HR1 or HR2, and small molecules bind to the deep, well-defined pocket on the surface of the HR1 helical trimer, thereby disrupting the gp41 six-helix bundle formation and inhibiting the gp41-mediated membrane fusion. Understanding of the function of the fusion inhibitors has opened a new direction for designing some new methods to identify more antiviral peptides and small molecular compounds against HIV-1 gp41.
In a previous study, the authors had designed an HIV-1 fusion inhibitory protein, HR121, formed by linking HR1 (N34) to the C-terminus of HR1 (N34)–HR2 (C34). This protein has been expressed and purified in Escherichia coli as soluble proteins. As a consequence, 3 molecules of the proteins can form a stable 6-helix bundle with 3 free heptad repeats of N34 exposed. 7 The conformation of these 3 N34 is similar to that of 3 HR1 in the gp41 prehairpin intermediate. It is also found that HR121 binds to the HR2-derived peptide in the viral gp41, which is similar to the function of HR1 in the gp41 prehairpin intermediate. However, HR121 easily aggregates in aqueous solutions. In the present study, the authors expressed and purified the recombinant GST-HR121, which was found to be more soluble and could be more easily purified than HR121 with no detectable inhibitory activity of glutathione S-transferase (GST). The authors also expressed and purified an HR2-derived peptide: C43-30a. Further research demonstrated that these 2 proteins could interact with each other. Therefore, it was hypothesized that this interaction could be inhibited by a potential HIV-1 fusion inhibitor. On the basis of this hypothesis, a novel enzyme-linked immunosorbent assay (ELISA) was established that could effectively detect HIV-1 fusion inhibitors targeting 6-HB of HIV-1 gp41.
Materials and Methods
Reagents and chemicals
Glutathione-Sepharose 4B was purchased from Amersham Pharmacia Biotech (Piscataway, NJ). Ni-NTA His Bind Resins were purchased from Novagen (Madison, WI). SureLINK HRP Conjugation kit was purchased from Kirkegaard & Perry Laboratories (KPL; Gaithersburg, MD). BCA protein determination assay kit was purchased from Pierce Biochemicals (Rockford, IL). Sifuvirtide was synthesized by FusoGen Pharmaceuticals (Tianjin, China), and enfuvirtide (T20) was purchased from Roche Molecular Biochemicals (Indianapolis, IN). C34 and N36 were kindly donated by Prof. Yu-Xian He of the Institute of Pathogen Biology, Chinese Academy of Medical Sciences, and Peking Union Medical College. The peptides were dissolved in phosphate-buffered saline (PBS), sterilized by 0.22-µm filtration, and stored in aliquots at −20°C. AGB (4′-acetamidophenyl-4′-guanidinobenzoate hydrochloride; molecular weight [MW] = 348.8) was purchased from Sigma (St. Louis, MO). Zidovudine (AZT, 3′-azido-3′-deoxythymidine) was purchased from Sigma. Indinavir (IDV) was purchased from Glaxo Wellcome (Brentford, UK).
Plasmids, cell lines, and viruses
The expressed vectors pET-30a/HR2, pGEX-6p-1/HR121, and pGEX-6p-1/HR212 have been described previously. 7 Chronically infected H9 cells (H9/HIV-1IIIB) and C8166 cells were kindly donated by the Medical Research Council, AIDS Research Project (London, UK).
Protein expression and purification
GST-HR121 was expressed in E. coli strain BL21 (DE3) and purified by the Glutathione-Sepharose 4B affinity column according to the method described earlier. 6 Briefly, E. coli strain BL21 (DE3) transformed with the recombinant pGEX-6p-1/HR121 plasmid was grown at 37°C in Luria-Bertani (LB) medium to an optical density of 0.8 to 1.0 (OD 600 nm) before induction with 0.5 mM isopropyl-l-thio-3-D-galactoside (IPTG) for 6 h. Bacterial cells were harvested and lysed by sonication in PBS (10 mM sodium phosphate, pH 7.4; 150 mM NaCl). The lysate with a final concentration of 1% Triton X-100 was incubated for 30 min at 4°C and subsequently clarified by centrifugation at 12,000 g for 30 min at 4°C. The clarified supernatants were passed over the Glutathione-Sepharose 4B column (equalized by PBS). The GST fusion protein-bound column was washed by PBS with more than 10 column volumes and eluted with reduced glutathione (10 mM) for 3 column volumes. The resultant protein was concentrated to a proper concentration by ultrafiltration and stored at −70°C for further analysis. Proteins were analyzed on 15% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS–PAGE). Protein concentration was determined by the BCA protein determination assay. GST-HR212 was expressed and purified in the same way as GST-HR121 and used as a negative control. 7,8
C43-30a were expressed in E. coli strain BL21 (DE3) and purified by a Ni-chelated Sepharose affinity column as described. 9 Briefly, the recombinant plasmids of pET-30a/HR2 transformed into E. coil strain BL21 (DE3). A single colony from the respective transformation was grown at 37°C in the LB medium to an optical density of 0.8 to 1.0 (OD 600 nm) and then induced with 1 mM IPTG at 25°C for 5 h. Bacterial cells were harvested and lysed by sonication in PBS. Then, the lysate with a final concentration of 1% Triton X-100 was incubated for 30 min on ice and subsequently clarified by centrifugation at 12,000 g for 15 min at 4°C. The clarified supernatants were applied on the Ni-NTA His Bind Resin affinity column. The column was washed with 5 column volumes of PBS, then with 5 column volumes of imidazole (20 mM), and eluted with imidazole (100 mM) for 3 column volumes. The fusion protein was named C43-30a.
GST pull-down assay
First, 50 µL of the Glutathione-Sepharose 4B gel containing GST-HR212 or GST-HR121 was mixed with 50 µL of 1 mg/mL C43-30a. The mixtures were incubated for 30 min at room temperature with rotation of the microcentrifuge tubes. Then, 1 mL of PBS was added into the microcentrifuge tube and centrifuged at 500 g for 5 min to pellet the glutathione-agarose beads. After washing with PBS 4 times, the beads were analyzed by 15% SDS-PAGE.
ELISA for screening HIV-1 fusion inhibitors targeting gp41 6-HB
The ELISA was established based on the principle that GST-HR121 could bind to C43-30a. The binding activity of HR121 was analogous to the monomer HR1; therefore, it could form a 6-helix bundle with a horseradish peroxidase (HRP)–labeled C43-30a. If a testing compound or a peptide interacts with HR1 or HR2, the 6-HB formation would decrease and the percentage of inhibition could be detected by a microplate spectrophotometer. Therefore, the percentage inhibition of the compound or peptide on the 6-HB formation could be calculated based on the levels of reduction of optical density.
First, C43-30a was labeled with HRP according to the instructions of the manufacturer. Then, 96-well polystyrene plates were coated with 1 µg/mL GST-HR121 in coating buffer (15 mM Na2CO3, 35 mM NaHCO3, pH 9.6) overnight at 4°C or at 37°C for 2 h. After removing the coating buffer and washing with PBS containing 0.05% Tween-20 (PBST) 5 times, the plate was blocked with PBST containing 5% milk (PBST-M) at 37°C for 2 h. Then, the plate was washed 5 times. Meanwhile, C34-30a-HRP (100 ng/mL) and a compound at gradient concentration in PBST-M were preincubated for 30 min and then added to each well of the plate for 1 h at 37°C. The plate was washed and the OPD substrate was added into the wells. The optical density of the plates was read on a Bio-Tek ELx800 ELISA reader (BioTek, Winooski, VT) at 490 nm/630 nm. The inhibition percentage of 6-HB was calculated by the following formula: % of inhibition = [1 – (E – N)/(P – N)] × 100, where E represents the optical density in the presence of a compound, and P represents the optical density in the absence of a compound. N corresponds to the wells where neither the compound nor C43-30a-HRP was added. The concentration of reducing 6-HB formation by 50% (IC50) was determined using the Reed-Muench method.
Cell-to-cell fusion assay
A cell-based assay was used for the detection of HIV-1-mediated cell fusion as previously described. 10 Briefly, in the presence or absence of 100 µL of gradient concentrations of compound, 6 × 104 of C8166 cells (50 µL) were cocultured with 2 × 104 of H9/HIV-1IIIB (50 µL) at 37°C in a humidified atmosphere of 5% CO2. After a 6-h incubation, the number of syncytia was scored under an inverted microscope. The 50% inhibitory concentration to blocking syncytia formation (IC50) was calculated.
Quality evaluation of the ELISA
To assess the statistical confidence of the ELISA, a simple statistical parameter, Z′ factor (Z′), was determined. The Z′ factor is a statistic value designed to reflect the dynamic range of the assay as well as the variation associated with the signal measurements. Z′ factor was calculated by the following formula: Z′ = 1 − 3 × (SDmax + SDmin)/(Mmax – Mmin), where Mmax and Mmin represent the mean optical density values of positive and negative controls, respectively; SDmax and SDmin are the respective standard deviations for those signals. The larger the Z′ factor value of an assay, the higher the quality of the assay. For example, 0.5 > Z′ > 0 suggests that it is an acceptable assay, whereas 1 > Z′ > 0.5 indicates that it is an excellent assay.
Assay performance was also analyzed by measurements of the coefficient of variation (CV), the signal to background (S/B), and the signal to noise (S/N) as follows: CV = SD/M; S/B = Mmax/Mmin; S/N = (Mmax – Mmin)/[(SDmax)2 + (SDmin)2]0.5. The smaller the values of CV, the larger the values of S/B and S/N, and the better the quality of the assay.
Results
Detection of interaction between GST-HR121 and C43-30a
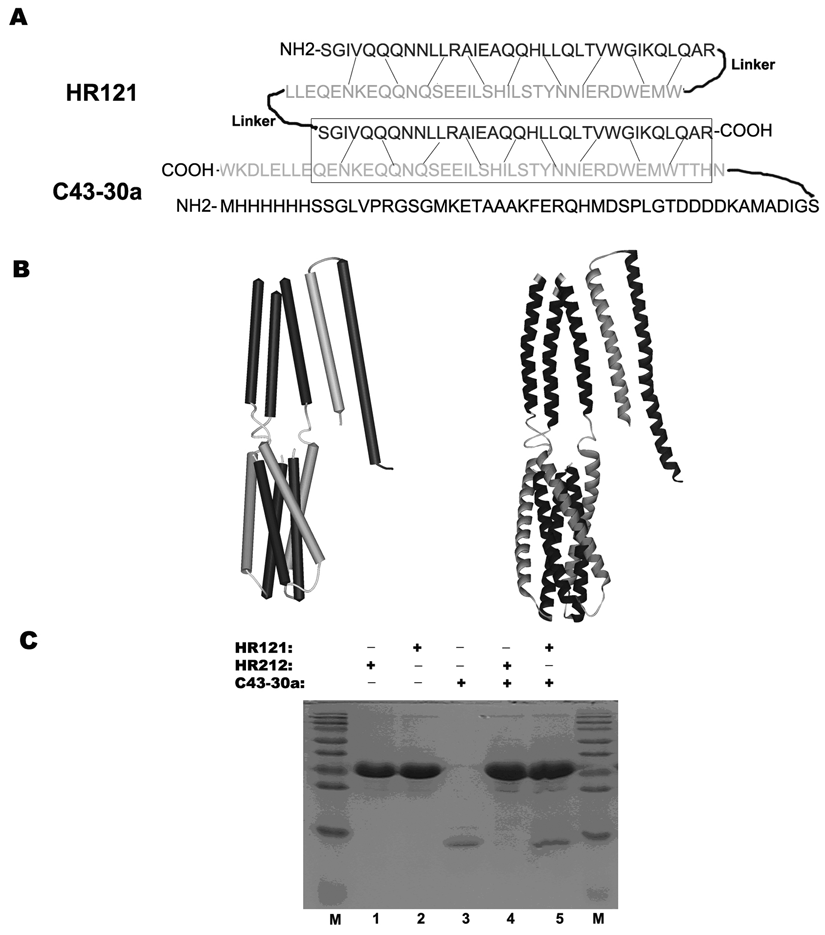
The fusion proteins GST-HR121 and C43-30a were purified by the Glutathione-Sepharose 4B and the Ni-NTA affinity columns. Based on the structure of HR121 and C43-30a, it could be predicted that HR121 could bind to an HR2-derived peptide C43-30a ( Fig. 1A , B ). When a fusion protein GST was added into the N-terminal of HR121 and 50 aa was derived from the vector pET-30a into the C-terminal of C43, the interaction between these 2 proteins was not changed from SDS-PAGE analysis ( Fig. 1C ). The control protein GST-HR212, which has 3 free HR2, could not bind to C43-30a. These results suggest that GST and redundant peptide 50 aa did not disturb the binding activity of HR121 and C43, and the function of HR121 was analogous to the monomer HR1 ( Fig. 1C ).
Sequence analysis, computer modeling, and glutathione S-transferase (GST) pull-down assay demonstrated that HR121 can interact with C43-30a. (
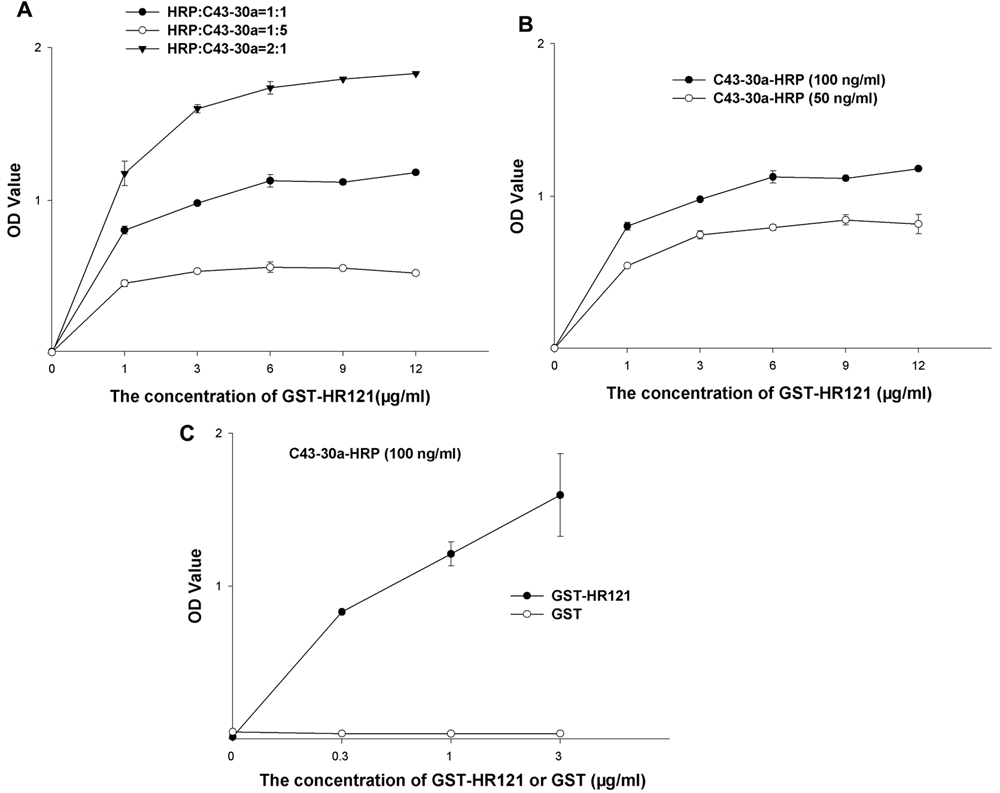
Determination of appropriate ratio of GST-HR121 and C43-30a-HRP in an ELISA-based assay
To optimize the molar ratio of HRP and C43-30a in the process of labeling C43-30a, 3 different groups were set. It was found when the molar ratio of HRP:C43-30a was 2:1, the value of optical density was the highest, and the signal to background was the same as the other 2 groups ( Fig. 2A ).
Crisscross serial dilution assays to optimize (
To obtain the optimal binding concentrations of GST-HR121 and C43-30a-HRP, the 2 proteins with gradient concentrations were tested by crisscross serial dilution assay. As the concentrations of GST-HR121 increased, the values of the optical density of the 1000-fold diluted C43-30a-HRP (the final concentration was 100 ng/mL) were always higher than those of the 2000-fold diluted C43-30a-HRP ( Fig. 2B ). Therefore, the 1000-fold diluted C43-30a-HRP was picked in the ELISA screening assay. To determine the best ratio of GST-HR121, a series of concentrations of GST-HR121 (GST as the control) ranging from 0 to 3 µg/mL was also tested, and it was shown that GST-HR121 at 1 µg/mL could obtain a proper value of optical density at 1.21. However, GST could not bind to C43-30a-HRP with a very low value of optical density ( Fig. 2C ).
Specificity and stability of ELISA-based assay for screening HIV-1 fusion inhibitors targeting gp41 6-HB
To determine the specificity of the ELISA, some anti-HIV peptides or drugs with known targets were tested. C34, N36, HR212, enfuvirtide (T20), and sifuvirtide, all targeting at the gp41 prehairpin intermediate, were used as positive controls. Among them, C34, N36, HR212, and sifuvirtide could firmly inhibit gp41 6-HB formation. 8,10,11 However, in some previous reports, T20 only weakly hindered the 6-HB formation. 11-13 In addition, IDV (HIV-1 protease inhibitor) and AZT (HIV-1 reverse transcriptase inhibitor) were included as negative controls. Besides these known peptides or drugs, an active compound, AGB, acting as an HIV-1 fusion inhibitor confirmed by our laboratory, 14 was tested to determine the specificity of the ELISA-based screening assay.
As shown in Figure 3 , C34 and N36 could compete with C43-30a to form 6-HB by IC50 values of 0.023 ± 0.005 µg/mL and 1.33 ± 0.013 µg/mL, respectively ( Fig. 3A , B ), and HR212 and sifuvirtide also could inhibit 6-HB formation with IC50 values of 0.072 ± 0.017 µg/mL and 0.030 ± 0.005 µg/mL, respectively ( Fig. 3D , E ). However, T20 showed low inhibiting activity to block 6-HB formation in this ELISA, with a high IC50 value of 3.77 ± 0.08 µg/mL ( Fig. 3C ). Besides these peptides, an active compound AGB also could weakly block 6-HB formation with IC50 values of 186.81 ± 11.73 µg/mL ( Fig. 3F ). On the other hand, IDV and AZT did not show any inhibitory effect on 6-HB formation (data not shown). Therefore, these data suggested that this ELISA was specific for the fusion inhibitors that target at the gp41 prehairpin intermediate.
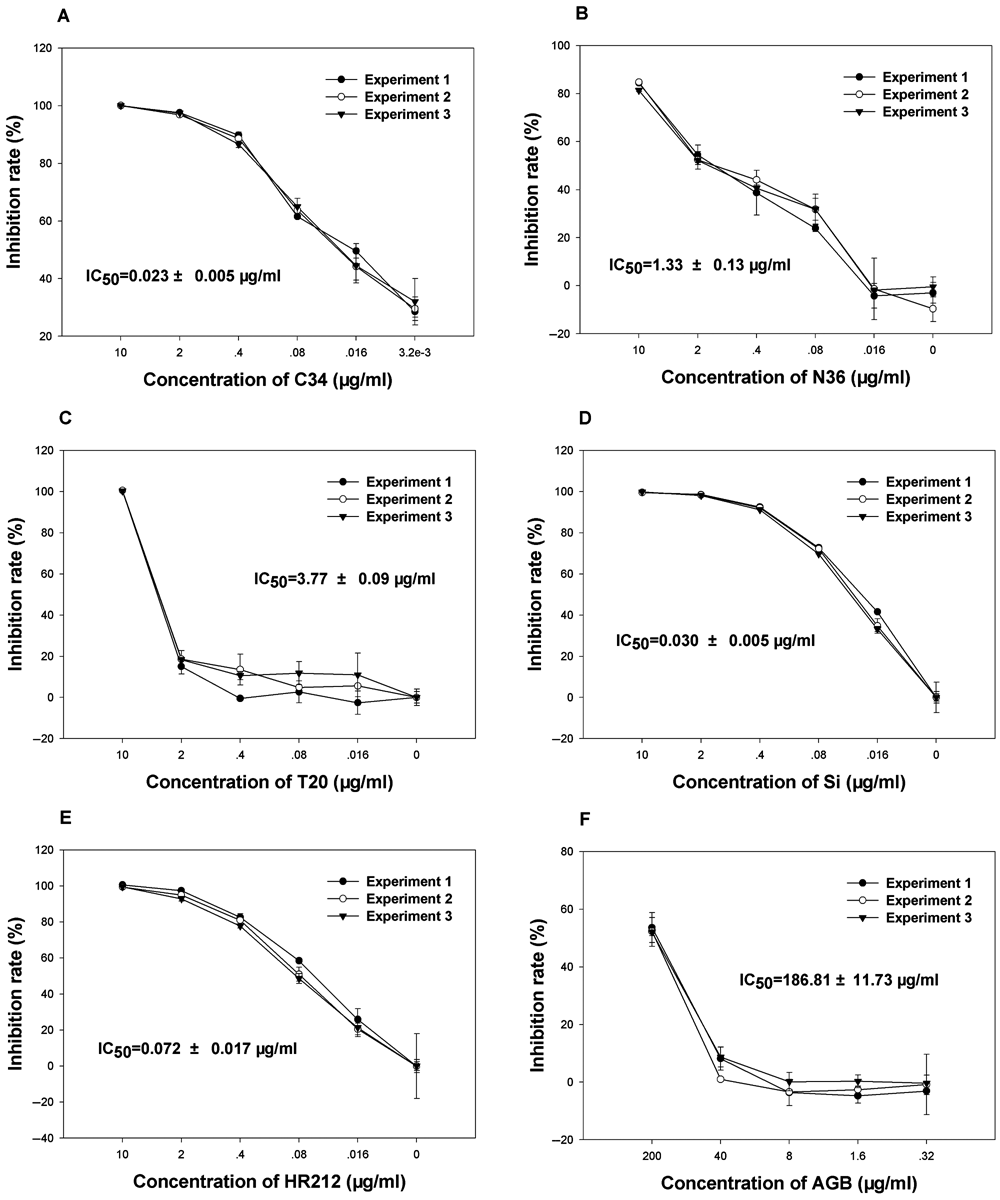
Specificity and sensitivity of enzyme-linked immunosorbent assay (ELISA)–based screening assay. Inhibitory activities of (
To test the stability of the ELISA, each compound was measured in triplicate, and the curve of the inhibiting rate in each experiment was similar. This indicates that this ELISA was not easy to fluctuate in repeats ( Fig. 3 ). In addition, the well-to-well variations in 96-well plates also were evaluated by the measurement of the standard statistical parameters Z′, S/B, and S/N. As shown in Figure 4 , the coefficients of variation were 4.32% for the positive controls and 9.11% for the negative controls. The signal-to-background ratio was 31.26, signal-to-noise ratio was 25.01, and the Z′ factor was 0.86. This demonstrates that the ELISA was of excellent quality and was suitable to be developed for high-throughput screening (HTS). This assay is used in the authors’ laboratory to screen chemical libraries.
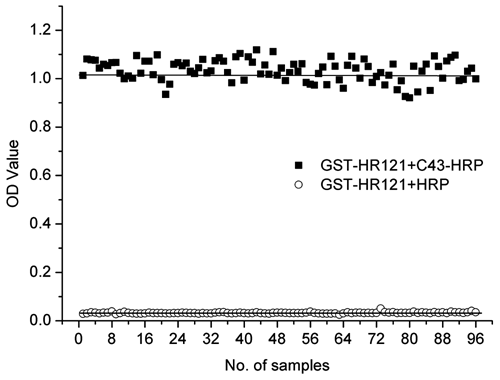
Well-to-well variation for determination of Z′ factor in the enzyme-linked immunosorbent assay. The procedure used was the following: GST-HR121 was coated on a 96-well polystyrene plate, and C43-30a-HRP or horseradish peroxidase (HRP) was added as positive or negative control. Then, OPD substrate was added. The optical density was read on a microplate reader at 490 nm/630 nm and the coefficient of variation, signal to background, signal to noise, and Z′ factor were calculated.
Sensitivity of ELISA-based screening assay
The cell-to-cell fusion assay is widely used in evaluating anti-HIV efficiencies of HIV fusion inhibitors. To determine the sensitivity of the ELISA, the IC50 values of each testing compound in ELISAs and cell-to-cell fusion assays were compared and listed ( Table 1 ). One can deduce that there was little difference between the inhibiting activities N36 and HR212. To achieve effects similar to those in the cell-to-cell fusion assay, the concentrations of C34, sifuvirtide, and AGB needed to be about 5 to 7 times higher in the ELISA, whereas T20 needed to reach about 500-fold concentration by ELISA detection.
Summary of Anti-HIV Activities of Anti-HIV Reagents in Novel ELISA and in Cell-to-Cell Fusion Assay
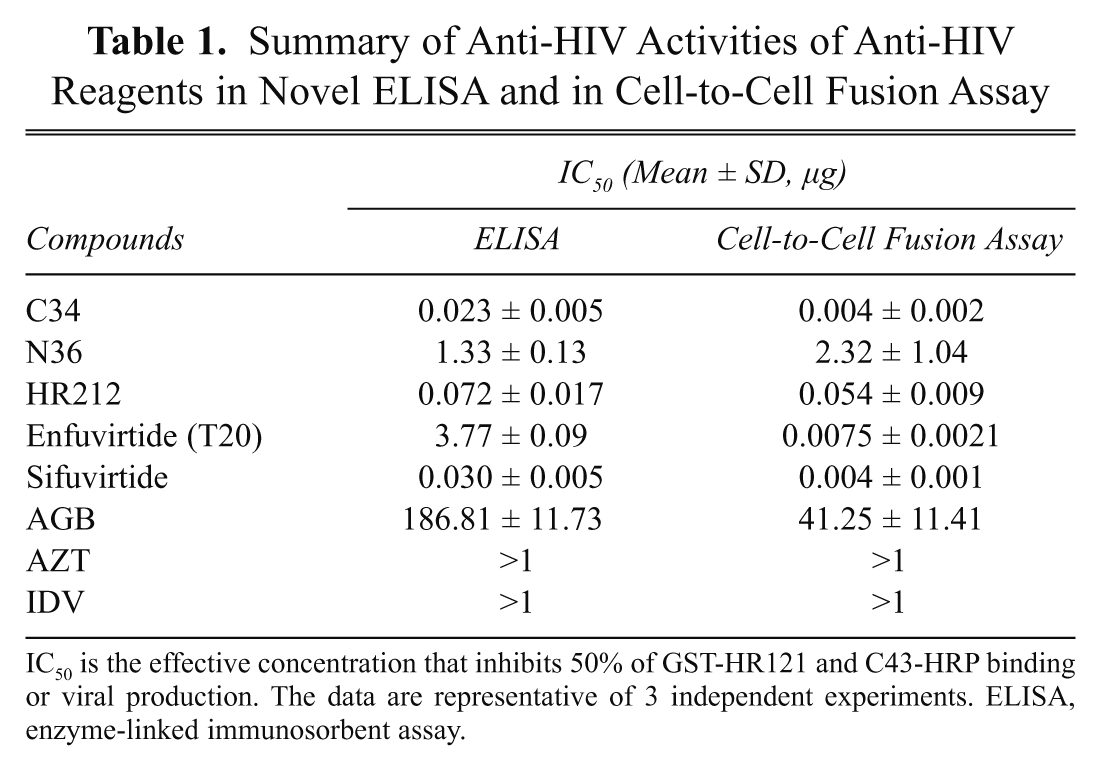
IC50 is the effective concentration that inhibits 50% of GST-HR121 and C43-HRP binding or viral production. The data are representative of 3 independent experiments. ELISA, enzyme-linked immunosorbent assay.
Discussion
Up to now, the assays for screening compounds that block gp41 6-HB formation and inhibit fusion between HIV and host cells could be classified into 2 main systems: assays based on cellular level and those on molecular level. Cellular-level assays are established on the principle that cells expressing the HIV-1 envelope glycoprotein gp160 could fuse with cells expressing receptor CD4, and a potential HIV-1 fusion inhibitor could block this Env-mediated fusion in a dose-dependent manner. 15-21 These assays can serve as determinants to the identification of fusion inhibitors; however, they are difficult to be adapted for HTS of HIV-1 fusion inhibitors because of some limitations. First, it is time-consuming, requiring about 2 to 3 days to culture cells for a single screening cycle. Second, it is laborious because of the number of syncytia needed to be carefully scored. Third, it is inconvenient because these assays are to be done in biosafety laboratories. Therefore, they are time-consuming and laborious.
One of the solutions to overcome these shortcomings is to establish some molecular-level screening methods. In the late 1990s, it was found that when a potential HIV-1 fusion inhibitor was added into the N36 and C34 mixture, it could not form 6-HB. On the basis of this principle, Jiang et al. 22 developed an ELISA for screening organic compounds for HIV-1 fusion inhibitors. This assay was then developed and improved for HTS. 22-25
However, N36 is relatively insoluble in aqueous solution. Therefore, it was replaced by some soluble trimeric coiled coil molecules, IQN17 26 or IQN36. 27 Using IQN36 and C34-Eu, Dams et al. 27 developed a time-resolved fluorescence assay to screen small-molecule HIV-1 fusion inhibitors.
In 2006, Frey et al. 28 expressed a soluble, single-chain protein construct called gp41-5, which contains a free HR1 segment and can bind firmly to C34-FITC. On the basis of this, they established a fluorescence enzyme-linked immunosorbent assay for screening HIV-1 fusion inhibitors.
The present research predicts that HR121 can bind to an HR2-derived peptide C43-30a and has developed a novel ELISA to screen potential HIV-1 fusion inhibitors that target the gp41 prefusion intermediate. The principal steps in this assay are as follows: GST-HR121 was coated onto the wells of a 96-well polystyrene plate; meanwhile, C43-HRP and a compound were mixed and then added to the washed wells. The compound that could interfere with the interaction of GST-HR121 and C43-HRP might decrease the optical absorption, thus having a potential inhibitory activity against HIV-1 fusion ( Fig. 5 ).
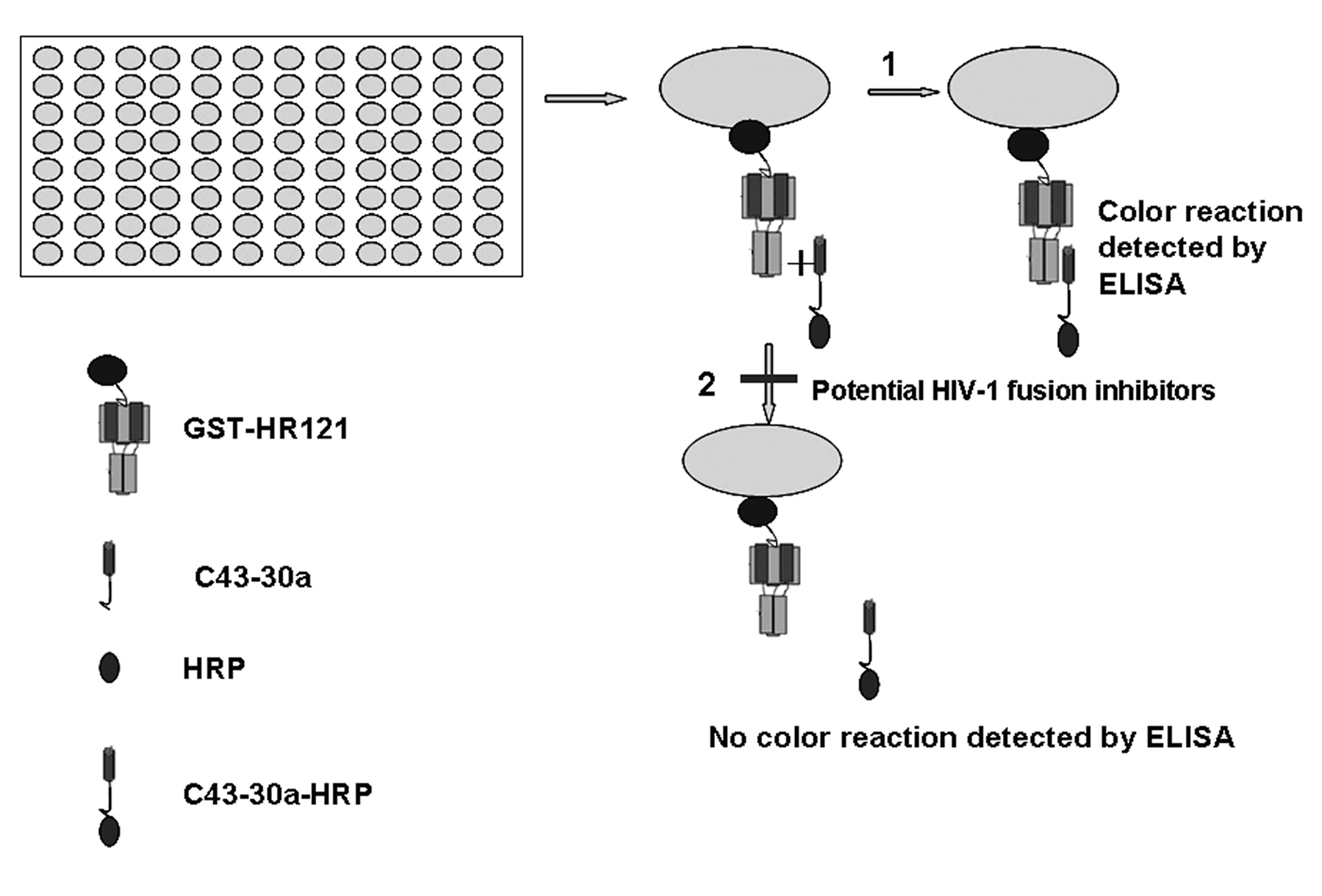
The procedure of a novel enzyme-linked immunosorbent assay (ELISA)–based screening assay. First, GST-HR121 and C43-30a were purified; C43-30a was labeled with horseradish peroxidase (HRP). Second, GST-HR121 was coated on a 96-well polystyrene plate, and C43-30a-HRP was added. Finally, in the absence of a potential inhibitor, C43-30a-HRP bound to GST-HR121 and the optical density could be detected, whereas in the presence of a potential inhibitor, the interaction was blocked and the optical density reduced.
Up to now, 3 main molecular-level screening systems have been widely used in HTS of HIV-1 fusion inhibitors, as mentioned earlier. Compared with the other 3 screening systems, the present ELISA-based assay has 2 main improvements: (1) much more economic (synthesizing the peptides was not needed) and (2) much more convenient—it is read on an ELISA reader and could be widely used in a normal laboratory, whereas the other 3 screening systems need a fluorescence microplate reader that is a bit expensive for some laboratories.
In the present study, a cell-to-cell fusion assay was performed to evaluate the anti-HIV efficiencies of the compounds used in the ELISA. Compared with the cell-based assays, the ELISA has several advantages: (1) it is much more rapid (the time for 1 screening cycle was shortened to about 7 h); (2) it has the potential to develop HTS, with which the authors could screen about 100 compounds per day; (3) it is sensitive—all tested anti-HIV-1 reagents showed similar anti-HIV-1 activities with the virus-mediated cell fusion assays; and (4) it is much easier—the experiment procedure was reduced by adding several reagents, no cellular culture, and syncytia calculation.
Notably, in the studies of anti-HIV-1 activity of T20, there was quite a difference in the IC50 values between ELISA and the cell-cell fusion assay ( Table 1 ). This result is consistent with some recent reports. Researchers believed that T20 targets multiple sites in gp41 and gp120, and its anti-HIV-1 activity may be a combinatory effect mediated by different mechanisms of action. They have demonstrated that T20 does not form stable 6-HB with N36 like C34 in fluorescence native polyacrylamide gel electrophoresis, ELISA, and circular dichroism spectroscopy. 11-13 Also, in other fluorescence-linked immunosorbent assays developed to screen HIV-1 fusion inhibitors, T20 failed to inhibit 6-HB formation at concentrations at least up to 100 µM. 25,28 Therefore, these reports confirmed ELISA would, in particular, identify the compound that targets the 6-HB formation of the HIV-1 gp41. For this reason, competitive inhibition of the binding of GST-HR121 and C43-HRP should be a more stringent criterion for fusion inhibitor screening than that of cell-cell or virus-cell fusion assays.
In conclusion, the novel ELISA-based assay is specific, sensitive and stable, and adaptable to an HTS format for identification of potential HIV-1 fusion inhibitors that target the 6-HB core structure of the HIV-1 gp41.
Footnotes
Acknowledgements
The authors thank Professor Yu-Xian He for providing peptides C34 and N36 and acknowledge the MRC AIDS Research Project for providing cell lines and viruses. This work was supported in part by grants from the Chinese Academy of Sciences (KSCX1-YW-R-24, KSCX2-YW-R-185), Eleventh Five-Year Key Scientific and Technological Program of China (2009ZX09501-029, 2008ZX10001-002, 2008ZX10001-015, 2008ZX10005-005) and Yunnan province (2007BC006, 2009CD109), National Basic Research Program of China (2009CB5223006), China Postdoctoral Science Foundation (20090451433), and the National Natural Science Foundation of China (U0832601).
