Abstract
Circulating immune complexes formed by tumor antigens and immunoglobulin M (IgM) represent a novel class of biomarkers with diagnostic value for early cancer detection. The quantitative analysis of these immune complexes is achieved by enzyme-linked immunosorbent assay (ELISA) methods using a purified calibrator from samples of patients with cancer. These complexes obtained from samples of human origin are not suitable for cost-effective production processes with high safety standards. Given the ill-defined biomarker/IgM ratio in these complexes, semisynthesis with retention of functional properties is difficult to achieve and may vary widely according to the batch-to-batch heterogeneity of starting biological preparations. Here the authors describe the development of a combinatorial method for defining the optimal reaction conditions for the reproducible semisynthesis of biomarker-IgM complexes by exploiting the biotin-avidin technology. The method relies on screening by ELISA the 3D composition space defined by the combinatorial variation of biotinylated-biomarker, biotinylated-IgM, and avidin concentrations aiming to select those conditions leading to biomarker-IgM complexes with the highest immunoreactivity. The method allows the reproducible synthesis of species with immunoreactivity comparable to that of natural immune complexes and endowed with sufficient stability to be used as calibrators in ELISA.
Introduction
A
The development of effective processes for the profitable large-scale preparation of synthetic substitutes of the immune complexes to be used for calibration purposes in ELISA procedures represents an urgent industrial need. Such a methodology could allow significant cost reductions and enhance the safety standards associated with the production of diagnostic tools aimed to detect the onset of neoplastic diseases. Here we report a cost-effective semisynthetic methodology circumventing the synthetic issues correlated to the limited knowledge of the immune complexes’ structure. The method exploits a combinatorial bioconjugation approach, allowing the tethering of tumor antigens to human naive IgM after a quick and reproducible screening of a large number of reaction conditions. The conjugation chemistry exploited is based on the biotin-avidin technology because of the mild conditions it requires, although giving fast reactions and quantitative yields. 7-11 The selection of the best synthetic conditions is headed by a maximization process considering the immunoreactivity and solubility of the constructs. This combinatorial method represents a novel approach to the synthesis of protein conjugates, preserving the immunoreactivity of each partner involved. This issue is not a primary concern in the usual methodologies exploited for the protein-protein conjugation, 12,13 and to the best of our knowledge, it represents the first successful attempt of combinatorial optimization of conjugation conditions applied to the development of calibrators in ELISA procedures. The approach was developed and validated selecting as a model molecular target the immune complex formed by the squamous cell carcinoma antigen (SCCA) and IgM. This immune complex proved to be a useful marker in the detection of hepatocellular carcinoma (HCC). 1 The system studied comprises recombinant SCCA, polyclonal human IgM, and avidin, where the latter species was used to assemble the biotinylated SCCA and IgM molecules. The immunometric methodology of combinatorial screening consists of a sandwich ELISA assay aimed to assess the existence of chemical connectivity between SCCA and IgM in the construct.
Materials and Methods
Materials
Bovine serum albumin (BSA) and inorganic and organic materials used for buffer preparation were obtained from standard suppliers, and the reagent grade water was obtained by using the Elix 3 and the milli-Q Synthesis A10 systems (Millipore, Billerica, MA). All the buffers used were freshly prepared and filtered though 0.45-micron Nalgene filters (Millipore) prior to use. Protein purifications by affinity chromatography were run on the ÄKTA Purifier system (Amersham Biosciences, Piscataway, NJ), analytical gel filtration chromatography was run using either a biosep-SEC-S 4000 column (Phenomenex, Torrance, CA) or Superdex™ 200 (Amersham Biosciences) packed on a 50-cm glass column assembled on a Waters 600 high-performance liquid chromatography (HPLC) system equipped with an in-line degasser and dual-wavelength detector. Centrifugal filtrations were performed using either the Amicon® Ultra 10000 MWCO or the microcon filtration devices (Millipore). ELISA tests were run using high-binding 96-wells plates (Greiner Bio-One, Monroe, NC). The washing procedures required in ELISA were automatically performed by using the Biotrak II Plate Washer (Amersham Biosciences). The ELISA required the use of the chromogenic substrate ABTS 2,2′-azino-bis(3-ethylbenzthiazoline-6-sulphonic acid) (Sigma-Aldrich, St. Louis, MO), and color development was monitored at 405 nm using the Viktor3™ 1420 Multilabel Counter plate reader (PerkinElmer, Waltham, MA) or the Biotrak II Plate Reader (Amersham Biosciences). Rabbit oligoclonal anti-SCCA antibody is a product of Xeptagen SpA (Marghera, Italy), and goat polyclonal antihuman IgM conjugated to horseradish peroxidase was obtained from Sigma-Aldrich. The total protein assay was performed in a microwell format according to the Bradford method against BSA or human IgG used as standards, and optical densities of the solutions were read at 620 nm using the Biotrak II Plate Reader (Amersham Biosciences).
Methods
Purification of IgM from human serum
Purification of human IgM from human serum was accomplished by affinity chromatography on goat-antihuman IgM (µ-chain specific) agarose (Sigma-Aldrich A9935). The affinity matrix was loaded onto a glass column (0.5 cm ID, 2.0 mL bed volume) and packed at a flow rate of 1.5 mL/min using phosphate-buffered saline (PBS) as mobile phase. The column was conditioned at a flow rate of 0.5 mL/min with PBS using at least 5 column volumes of buffer prior to sample loading. A sample of diluted human serum obtained diluting 3.0 mL of serum to 12 mL with PBS was loaded onto the column at a flow rate of 0.5 mL/min; the chromatographic run was monitored at 220 and 280 nm. The unretained material (unbound fraction) was collected, and the column was washed with reagent grade water at 1.5 mL/min flow rate until the conductivity of the flowtrough dropped to zero. The bound fraction was then eluted using glycine/HCl buffer 0.1 M at pH 2.5, and the flow rate during elution was set to 1.5 mL/min. The collected solution was neutralized, adding a saturated solution of NaHCO3, and 100 µL of saturated bicarbonate solution was used for each milliliter of collected solution. In a typical experiment, the bound fractions from 3 different chromatographic runs were pooled together, and the buffer was exchanged to PBS by centrifugal filtration by using the Amicon® Ultra 10000 MWCO centrifugal filtration devices (Millipore). The volume of the concentrated solution was then reduced to about 0.5 mL by centrifugal filtration; the protein concentration was determined by the Bradford method. The solution was divided into 200-µL aliquots and stored at −20°C until further use.
Human IgM biotinylation
The concentration of the IgM solution resulting from the purification process was adjusted to 1260 µg/mL adding PBS; a solution of bicarbonate buffer 0.1 M (pH 8.5) containing NaCl 0.5 M was added to the solution in a 1:1.5 volume ratio. The mixture was treated with 25 µL of a freshly prepared solution of 6-(biotinamidocaproylamido)caproic acid N-hydroxysuccinimide ester (Sigma B3295-50MG) prepared at the concentration of 1.0 mg/mL in dimethyl sulfoxide (DMSO). The solution containing IgM and activated biotin was shaken for 4 h and afterwards dialyzed overnight against 1 L of PBS. The dialysis process was repeated 2 more times against 1 L of PBS. The protein solution was collected and assayed for total protein content.
Biotinylation of SCCA
Squamous cell carcinoma antigen was obtained from cell lysate as previously described. 14 Then, 500 µL of SCCA solution in MES buffer 0.1 M (pH 6.5) was subjected to centrifugal filtration to change the milieu to PBS using the microcon centrifugal filtration devices as recommended by the manufacturer. The resulting solution was diluted to 600 µL with PBS. An equal volume of bicarbonate buffer 0.1 M (pH 8.5) containing NaCl 0.5 M was added. To the solution, 60 µL of 6-(biotinamidocaproylamido)caproic acid N-hydroxysuccinimide ester (Sigma B3295-50MG) 1.0 mg/mL in DMSO was added, and the mixture was shaken for 4 h and afterwards dialyzed overnight against 1 L of PBS. The protein solution was collected and assayed for total protein content.
ELISA for the estimation of the conjugate reactivity
Ninety-six-well microplates were coated with 100 µL/well of polyclonal rabbit anti-SCCA solution at the concentration of 10 µg/mL in PBS. The coating procedure was performed overnight at 4°C in a humid closed chamber. The solution was removed and the plates washed with PBS containing 0.05% Tween-20 (3 × 300 µL per well). The wells were filled with 300 µL of PBS containing 3% bovine serum albumin to saturate the residual reactive sites of the plastic plate, and the plate was allowed to stand at room temperature for 2 h, washed with PBS containing 0.05% Tween-20 (3 × 300 µL per well), and tapped dry. The samples were loaded on the plate and incubated for 1 h at room temperature. The solutions were removed by suction, and the plate was washed with PBS containing 0.05% Tween-20 (6 × 300 µL per well). A solution of goat-antihuman IgM conjugated to horseradish peroxidase (HRP) at the 1:1000 dilution (100 µL/well) was added to the wells and incubated for 1 h at room temperature. The solution of secondary antibody was removed by suction, and the plate was washed with PBS containing 0.05% Tween-20 (6 × 300 µL). A solution containing ABTS 0.2 mg/mL and 5 mM hydrogen peroxide in phosphate-citrate buffer (pH 5.0) was added to each well (150 µL/well), and the enzymatic reaction was allowed to proceed for 20 min at 37°C in the dark; absorbance was read at 405 nm.
Screening of reaction conditions
The combinatorial screening of reaction conditions for the optimization of the bioconjugates’ immunoreactivity was carried out using a microplate for ELISA determinations as a reaction vessels array. The procedure required the inactivation of a medium binding plate (Greiner Bio-One) with a 5% solution of nonfat dried milk reconstituted in PBS to prevent any nonspecific adsorption of proteins on the plastic surface. The plate containing the reconstituted milk solution (300 µL per well) was allowed to stand overnight at 4°C in a humid closed chamber. The plate was washed with a solution of PBS containing 0.05% Tween-20 (6 × 300 µL per well) and tapped dry. The proper volumes of protein solutions were delivered to the wells and mixed thoroughly. Each well was loaded with a constant amount of biotinylated IgM, and the concentration of biotinylated SCCA was continuously varied across the vertical direction of the plate, whereas the avidin concentration was varied according to the horizontal direction. The total volume of each solution was brought to 150 µL by adding PBS (pH 7.2). A single screening allows the analysis of 96 different reaction conditions in which the antigen and avidin concentrations are continuously varied by doubling concentrations. After addition of all the protein solutions, the plate was allowed to stand at room temperature for 1 h in a humid closed chamber. An aliquot (100 µL) of each conjugate solution was withdrawn from each well and tested for its reactivity.
Results and Discussion
Immune complexes are inherently multivalent species; in fact, a single antigenic molecule, displaying different antigenic epitopes, may, in principle, be complexed by different cognate antibodies. In the design of an effective immune complex mimetic conjugate, this multivalency should be taken into account. A minimalist approach to mimic the immunoreactivity of SCCA-IgM immune complexes could be to contrive the formation of oligomeric species where few SCCA molecules are tethered to a large number of IgM. However, the synthesis of such an oligomeric construct poses a series of challenges mainly related to its solubility properties and to the preservation of the immunoreactivity of each single component. The latter point deserves special attention because in this framework, immunoreactivity means capacity to effectively interact with the antibodies used in the ELISA tests as the natural immune complex does. Clearly, in an oligomeric species, steric hindrance may severely impair the recognition events by specific antibodies, resulting in an unpredictable behavior of the construct. All these issues can be simultaneously addressed by designing a screening method of synthetic conditions capable of selecting those preparations of the conjugate producing soluble materials and displaying a sufficient availability to binding, by specific antibodies, of both SCCA and IgM molecules. This can be achieved by using an anti-SCCA antibody to capture the antigen part of the conjugate and, after removal of the unbound material, using an anti-IgM antibody conjugated to a suitable enzyme to detect the presence of IgM. Although various tethering chemistries could be used to connect SCCA and IgM, ideally they should consist of mild, quick, and quantitative reactions, enabling the formation of robust species. As a proof of principle, we exploited the biotin-avidin interaction to bring together the SCCA and IgM molecules previously biotinylated, SCCAB and IgMB, respectively. The quest for the system composition (concentration of biotinylated SCCA, biotinylated IgM, and avidin) attaining the highest immunoreactivity signal requires devising a method capable of handling a large number of reaction conditions. In fact, screening the system composition by maximizing the immunoreactivity signal obtained corresponds with an exploration of an available space of dimension 3 (concentration of 3 species). Our approach consisted of using 96- or 384-well microplates as reaction vessels array where different mixtures of biotinylated species could be prepared. In a typical experiment, the SCCAB concentration was varied along the vertical direction, whereas the avidin concentration was varied according to the horizontal direction of the plate. The IgMB concentration was kept constant in each experiment and varied from one experiment to another. After 1 h of incubation at room temperature, the reactivity of the solution contained in each well was assayed.
The recorded optical density values due to the different conjugate solutions were indexed according to the amount of SCCAB and avidin used for their synthesis, thus providing an unequivocal addressing method. The optical density values and the amounts of proteins used for each synthesis may be considered a set of coordinates to represent the outcome of the experiment as a 3D surface. In closer detail, with reference to Figure 1 , the optical density represents the z coordinate, whereas the amount of SCCAB and avidin used in the conjugate preparations is represented as the y and x coordinates, respectively.
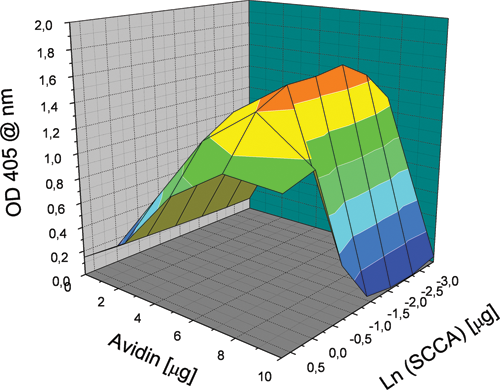
Representation as a 3D surface of the outcomes of a screening experiment. Coordinates x and y represent the system composition in terms of amounts of protein used for each different condition. The amount of IgM used was 10 µg per preparation. The z coordinate indicates the optical density signal recorded at 405 nm at the end of the enzyme-linked immunosorbent assay screening.
Because the IgMB concentration was kept constant during the experiment, the graph in Figure 1 can be conveniently considered a 2D section of the 3D available “space.” An analysis of the profile reported in Figure 1 infers that the highest conjugate reactivity is obtained at the intermediate SCCAB concentration, and the dependence of the measured reactivity from the SCCAB concentration is bell shaped and reaches its maximum at a concentration corresponding to an amount of protein close to 0.5 µg/well (3.3 µg/mL). The dependence of the observed reactivity from the concentration of avidin is also bell shaped; the highest reactivity is obtained for those preparations that required an amount of avidin corresponding to 7 µg/well (33.3 µg/mL). These dependences give the 3D surface the shape of an oblong bell.
The shape of the graph can be explained easily considering that at low SCCA concentrations, the number of reactive sites will be too low for an efficient binding to the anti-SCCA capturing antibody, whereas at high concentrations, the content of the SCCAB molecule might be too high with respect to IgMB, preventing an efficient binding by the revealing antibody (anti-IgM). On the other hand, an excess of unreacted SCCAB may compete with the conjugate for the anti-SCCA antibody used as the capturing phase. The shape of the graph, by looking at the reactivity profile as a function of the avidin content, can be explained easily considering that at low avidin content, the conjugate cannot form, whereas at high avidin content, the excess of protein may react with nearly all the biotin moieties available, leading to steric hindrance, preventing the binding by either the capturing antibody or the revealing one. This method allows the identification of the synthetic conditions corresponding to the most reactive conjugate, thus unequivocally defining its composition. Remarkably, this process allowed the identification of a tiny region of the composition space in which the conjugate reactivity is sufficient for application as a calibrator in ELISA; similar limited regions could be out of reach when exploiting synthetic methodologies that are not guided by effective screening procedures. The reproducibility of the optical density signals obtained in this screening for reaction conditions was assessed by repeating the experiment in a narrower region, using 10 µg/well of IgMB and 5 µg/well of avidin, and varying the amount of SCCAB used from 1 to 0.016 µg/well (6.7-0.1 µg/mL), thus 7 different conditions were explored. In this reproducibility assessment, the proteins were taken from the same protein stock; the experimental results are reported in Figure 2 . The data support a fairly good reproducibility of the screening method, with the differences in measured optical densities within the experimental uncertainty of ±10%.
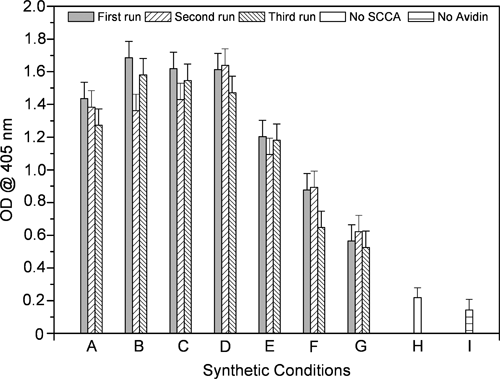
Graphical representation of the results obtained in the experiment aimed to assess the reproducibility of the screening of synthetic conditions. Letters on the x-axis describe the different synthetic conditions: (A-G) serial dilutions of SCCA from 13.3 to 0.0208 µg/mL; (H) no SCCA used; (I) no avidin used.
The preparative-scale synthesis of the conjugate could be performed by repeating the reactions a large number of times using the conditions identified in the screening. To this solution, BSA was added to the final concentration of 20 mg/mL to prevent adsorption of the newly synthesised conjugate to the plastic surfaces.
The immunoreactivity of the newly formed conjugate was compared, by ELISA, to the reactivity of a standard preparation of natural immune complex obtained from human serum by gel filtration methods as previously described. 1 To this aim, the optical densities obtained for a serial dilution of 2 different lots of the artificial conjugate were compared to the optical densities of a 250-AU/mL natural SCCA-IgM immune complex preparation; Figure 3 displays the obtained experimental data. The comparison with the natural reference allowed us to determine the reactivity of the artificial conjugates to be 200 AU/mL in the first case and 230 AU/mL in the second. The intra- and interassay coefficients of variation on the standard curves were, on average, less than 10%, as determined in quadruplicate analysis.
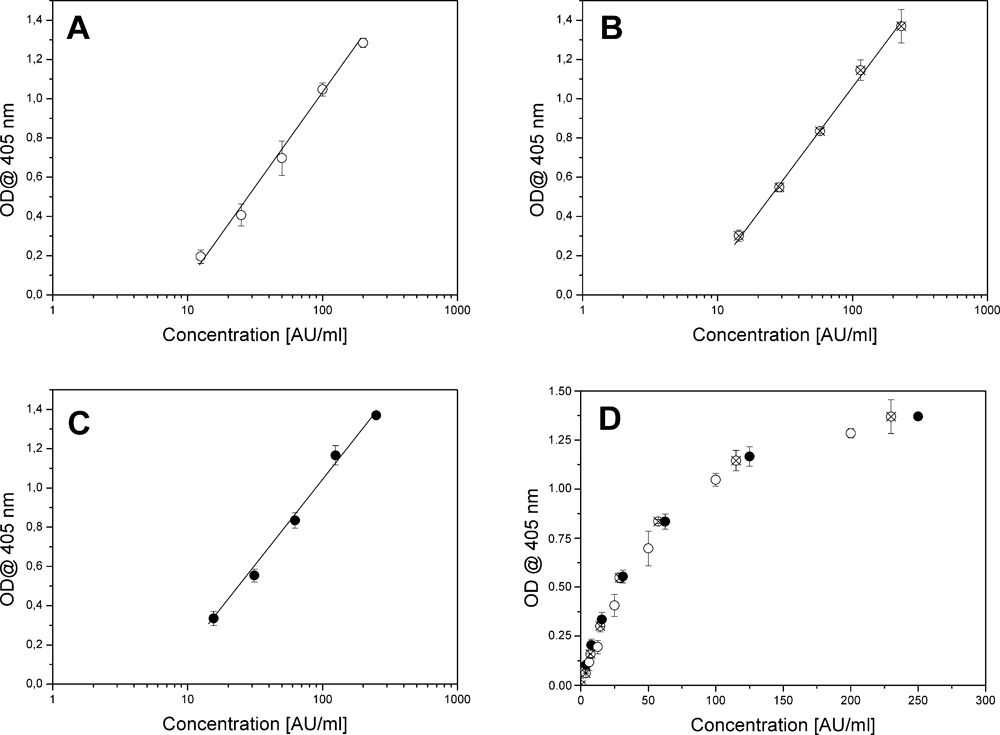
Comparison of the calibration curves for the natural calibrator and different preparations of the conjugate; error bars represent signals uncertainty as ±2 SD. (
In a further analysis, the screening method was applied to the optimization of the reactivity of the SCCA-IgM mimetic conjugate, starting with different stock solutions. In nearly all of the cases explored, the method allowed us to obtain conjugates with very similar reactivities, even though the exact compositions of the reacting mixtures were not the same.
Gel filtration chromatography
A preliminary mass analysis of the oligomeric conjugate was carried out by gel filtration, using a column (1.0 cm ID, 50 mL bed) packed with Superdex™ 200; the column was calibrated by using a set of proteins of known molecular weight, spanning the range of 900 to 17 KDa (human IgM, human IgG, BSA, horse myoglobin). A total volume of 1.0 mL of conjugate solution (150 µg/mL) was injected at a 1.0-mL/min flow rate using PBS (pH 7.2) as the mobile phase. The obtained chromatogram ( Fig. 4 ) displays a single peak eluting with the void volume of the column, confirming the oligomeric nature of the construct. The column effluent was collected in 500-µL fractions, and their immunoreactivity was assayed as previously described. More than 90% of the total immunoreactivity was recovered in the peak eluting with the void volume, thus confirming the nature of the peak. No other peaks corresponding to proteins with masses in the range of 900 to 17 KDa could be detected in the chromatographic trace, confirming the quantitative nature of the conjugation approach exploited.
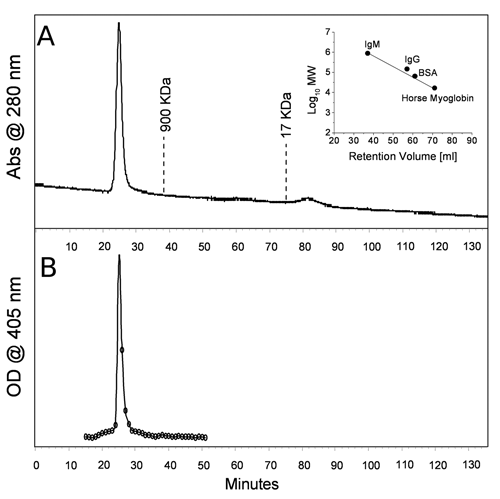
(
Specificity of binding
To assess the binding specificity of the conjugate, a binding competition experiment was performed by mixing the conjugate at the concentration of 50 AU/mL with increasing amounts of free (nonbiotinylated) SCCA and monitoring by ELISA the extent of conjugate binding to the anti-SCCA antibody used for the plate coating. A series of different mixtures of conjugate and SCCA were prepared by mixing 50 µL of 100 AU/mL conjugate solution with 50 µL of SCCA solutions at different concentrations; the total volume of each solution was 100 µL. In the mixtures, the concentration of free SCCA ranged from 20 µM to 0.5 nM; an aliquot of 50 µL of these solutions was loaded on the plate sensitized with the anti-SCCA antibody, and the ELISA test was then run. The percentage of residual reactivity is reported as a function of the free SCCA concentration in Figure 5 . The inflection point of the competition curve was found at a concentration of free antigen of 0.5 µM.
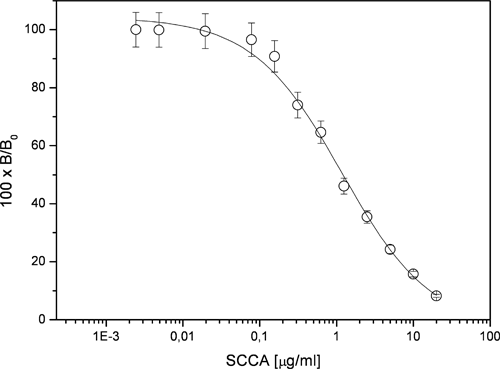
Inhibition of bioconjugate binding by increasing concentrations of free SCCA. The extent of binding decreases in a dose-dependant manner with increasing SCCA concentration. The inflection point of the curve (50% inhibition) is obtained at a concentration of SCCA corresponding to 0.5 µg/mL.
The total amount of SCCAB involved in the formation of the conjugate investigated was 0.023 µg, corresponding to a formal concentration of 0.23 µg/mL. Because only a small fraction of it would be exposed on the conjugate surface and thus available for binding, a fairly large excess of free SCCA was necessary to completely inhibit the conjugate binding; this finding could suggest a multipoint binding of the conjugate to the capturing phase. The data obtained in this experiment clearly infer the existence of a specific binding event in the recognition of the conjugate by the anti-SCCA antibody used in the capture phase of the assay. As a negative control, the presence of an excess of an unrelated protein such as BSA did not cause any reduction in the measured reactivity; in fact, the excess BSA used for the conjugate storage was never found to be a source of diminished reactivity.
The specificity of the conjugate reactivity was also analyzed to assess to what extent the binding could be affected by nonspecific interactions. This is particularly important because weak nonspecific interactions could be exacerbated in a large oligomeric species. Thus, to specifically address this point, we carried out a series of experiments aimed to determine the intensity of the recorded signal due to the possible nonspecific interactions. In closer detail, we prepared a series of solutions containing a constant amount of IgM (5 µg) and avidin (5 µg) but without using SCCA in the mixture; in this case, the conjugate did not contain the species responsible for the binding to the antibody adsorbed on the plastic surfaces of the microplate used in the assay. In these conditions, the signal corresponded to the 10% of a typical conjugate reactivity ( Fig. 2 ). These results altogether infer that the nonspecific binding of the conjugate to the solid phase of the assay is negligible, if any.
Conclusion
In this report, we presented a combinatorial methodology for the semisynthesis of soluble oligomeric species with immunoreactivity mimetic to that of natural SCCA-IgM immune complexes. The synthetic construct consists of biotinylated IgM and SCCA molecules held together by exploiting the biotin-avidin interaction. The best synthetic conditions were determined by screening the immunoreactivity of the oligomeric construct by varying the concentrations of its components. This selection method required the analysis of a 3D space consisting of 3 “concentration coordinates” and an optical density signal proportional to the reactivity of the conjugate. Despite its complex nature, this analysis could easily be implemented in an ELISA platform. The developed method allowed the reproducible synthesis of conjugates, even starting from different batches of protein preparations, and the screening also allowed the identification of those synthetic conditions in which a postsynthetic purification step was not required, and the yield was essentially quantitative. The newly synthesized SCCA-IgM conjugate proved to be specifically recognized by anti-SCCA and anti-IgM antibodies, displaying immunoreactivity very similar to that of the natural compound. Altogether, the properties of this mimetic species allowed its use as an artificial calibrator in ELISA tests aimed to quantitatively determine SCCA-IgM immune complexes. The development of the methodology described in this study allowed us to overcome relevant industrial limitations in the large-scale preparation of diagnostic tools for cancer early diagnosis.
