Abstract
Cell migration plays a key role in both normal physiological and pathological conditions. The study of cell migration and its underlying mechanisms is of great significance in various fields of research, including basic biology and pharmaceutical development. The cell migration or scratch wounding assay is an easy and economical in vitro method that allows researchers to assess a large number of testing compounds. Even though this simple assay has been used for decades, researchers are still trying to modify such experimental protocols and wounding devices. In this study, an 8-channel mechanical “wounder” was designed for performing a cell migration assay, particularly in a 96-well culture plate format. With special designs of a guiding bar and adjustable pins for use with disposable pipette tips, this wounder confined the scratch area within the center of each well to ensure a perfect contact between the pins and the well surface. As a result, this mechanical wounder produces a uniform denudation of a cell monolayer in a 96-well plate with a wound size of around 600 µm. Using this improved wounding device, the effects of epidermal growth factor and DL-α-difluoromethylornithine on the reepithelialization of rat intestinal epithelial cells (IEC-6) and serum on the wound recovery of human umbilical vein endothelial cells were demonstrated. This wounder facilitates cell migration study and can be applicable for multiple sample analysis.
Introduction
C
Cell migration is a complex process requiring the coordination of numerous inter- and intracellular events, such as cytoskeleton reorganization, matrix remodeling, cell-cell adhesion modulation, and induction of chemoattractants. 2 The Boyden chamber and phagokinetic assay have been used for a long time in the in vitro study of cell migration. 3 However, in many instances, the study of cell migration usually involves the response of confluent monolayer cells to mechanical injury (mechanical wound). These wounds are usually created by the removal of a proportion of confluent cells grown on an individual coverslip or in a multiwell plate using pipette tip, razor blade, syringe needle, mechanical scraper, or spatula. 4,5 The closure of the denuded area can then be observed, measured, and quantified over a time course using microscopy or a computer imaging system.
In general, cell migration/wounding assay is a commonly used method to study cell migration and its underlying biology such as cytoskeletal remodeling or cell polarization. 6 Furthermore, its versatility allows the study of angiogenesis, metastasis, immune response, and other physiological or pathological processes. 7,8 Previously, we have demonstrated the effect of total polysaccharides from Si-Jun-Zi decoction in the reepithelialization of wounded rat intestinal epithelial cells (IEC-6) using a cell migration assay. 9
In fact, most of the cell migration assays require individual treatment of each sample well. Even though tools and assays are commercially available for handling a large number of samples, improvement is still necessary for some parameters such as cost of experiment and the problem of cross-contamination. Therefore, an improved wounding device or method is needed. In this study, we report the development of an 8-channel mechanical wounder especially designed for performing a cell migration assay in a 96-well plate format. It is a user-friendly, contamination-free, easy-to-handle device for analysis of cell migration in a simple and reproducible manner.
Materials
Reagents
Dulbecco’s modified Eagle’s medium (DMEM), fetal bovine serum (FBS), phosphate-buffered saline (PBS), penicillin-streptomycin (PS), trypsin, gentamicin sulfate, insulin, and sodium pyruvate were obtained from Invitrogen (Carlsbad, CA). M199 medium, endothelial cell growth supplements (ECGS), heparin, amphotericin-B, and epidermal growth factor (EGF) were obtained from Sigma (St. Louis, MO). Matrigel was from BD Bioscience (Palo Alto, CA). DL-α-difluoromethylornithine (DFMO) was purchased from Calbiochem (San Diego, CA).
Cell lines and culture condition
The rat intestinal epithelial cell line (IEC-6 cells; ATCC CRL 1592) was purchased from American Type Culture Collection (Rockville, MD). The stock culture was maintained in a 75-cm2 flask in DMEM supplemented with 10% FBS, 10 µg/mL insulin, and 50 µg/mL gentamicin sulphate. The cells were incubated at 37°C in a humidified CO2 incubator (5% CO2). Cells at passages 14 to 20 were used in this study.
The human umbilical vein endothelial cells (HUVECs) were purchased from Clonetics (TCS Biologicals, Buckingham, UK). HUVECs were cultured in M199 medium supplemented with 20 µg/mL ECGS, 20% heat-inactivated FBS, 1% PS, 50 ng/mL amphotericin-B, and 90 µg/mL heparin in 0.1% gelatin-coated culture flasks. The cells were grown at 37°C in a humidified CO2 incubator (5% CO2). All experiments were conducted with HUVECs at passages 2 to 7.
Wounding assay
IEC-6 cells were seeded at a density of 4 × 104 cells per well into the 96-well flat-bottom microplates (Iwaki, code: 3860-096), which were precoated overnight with PBS-diluted Matrigel (1:7, v/v). After incubation in 10% FBS-supplemented DMEM for 24 h, the cells were starved in serum-free medium for another 24 h. For wounding of HUVECs, cells were seeded at a density of 3 × 104 cells per well in gelatin (0.1%) precoated 96-well flat-bottom microplates.
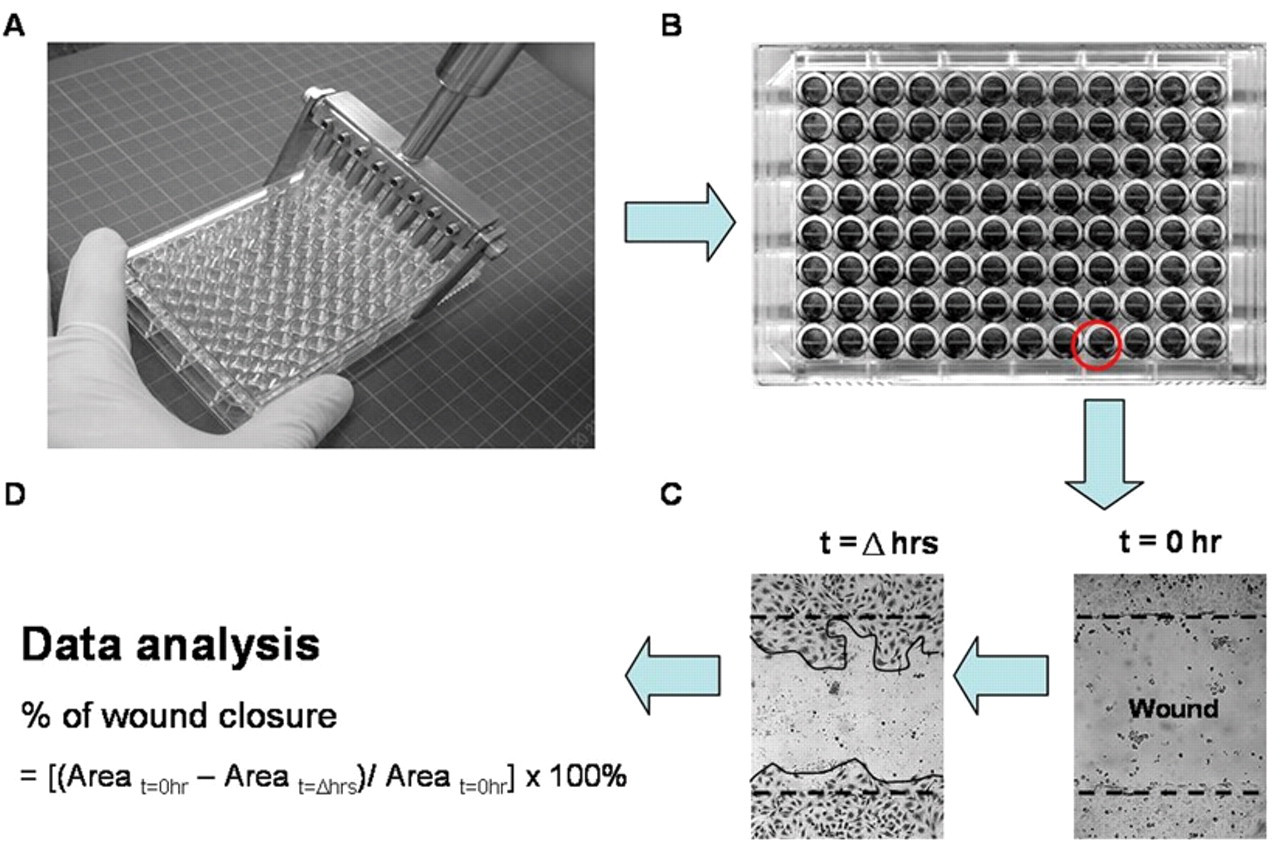
Before wounding, the pins of the wounder were assembled with sterilized p-10 pipette tips (Axygen T-300), and the level of each tip was calibrated using a hex wrench (see Fig. 2 , steps 1-4). In performing the wounding process, the wounder fitted with pipette tips was engaged perpendicularly at the leftmost edge of each well in the same column and moved laterally to the rightmost edge. The same procedure was repeated for all columns (see Fig. 3 ).
Then the medium was replaced with fresh serum-free medium for control wells or with medium containing 20 ng/mL EGF or 5 mM DFMO for IEC-6 cells or with different percentages of FBS for HUVECs.
Image capture and data analysis
Images at time zero (t = 0 h) were captured to record the initial area of the wounds, and the recovery of the wounded monolayers due to cell migration toward the denuded area was evaluated at 16 and 24 h (t = Δ h). The images were captured using an inverted phase-contract microscope (TMS Nikon; 4× objective) equipped with a Motic digital camera (Motic image plus 2.0 software, Motic Instruments, Inc., Richmond, Canada). The area of wound was quantified by Java’s Image J software (http://rsb.info.nih.gov) using the polygon selection mode. The migration of cells toward the wounds was expressed as percentage of wound closure:
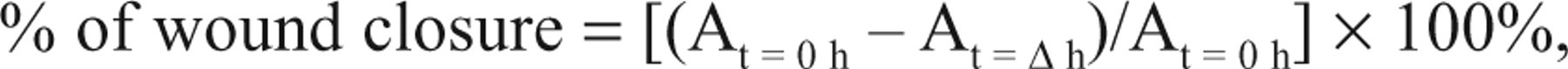
where, At=0h is the area of wound measured immediately after scratching, and At=Δh is the area of wound measured 16 or 24 h after scratching.
Statistical analysis
Each experiment was performed at least 3 times, and each treatment was conducted in 8 replicates. The data were expressed as mean ± standard deviation. The Student t-test was employed for statistical analysis. Statistical significance was established at p < 0.05.
Results and Discussion
We have constructed an 8-channel mechanical wounder ( Fig. 1A ) for the purpose of scratching the cell monolayer in performing the cell migration assay. As shown in Figures 2 and 3 , this wounder is a user-friendly device that researchers can use to perform cell scratching in 96-well plates for less than a minute per plate, and no special training is needed. Using this wounder, a series of artificial wounds of uniform width (600 ± 36 µm) with sharp edges were created in each well of the 96-well plate containing a confluent cell monolayer. In comparison with conventional scratching tools or methods, this wounder has several advantages ( Table 1 ). As this wounder is tailor-made for a 96-well plate format, the adjustable guiding-bar design guarantees that the scratch area is at the central line of each well and also ensures the wounder is applicable for different brands of culture plates with different geometry ( Fig. 1B ). The “central-positioned wound” feature facilitates image capturing using a digital camera–equipped inverted microscope. Even though the photos obtained at different time points are found to have a certain degree of variation, this does not encumber the data analysis of in vitro migration or wound healing in the 96-well plate format because more than 90% of the wound can be visualized under a low-magnification objective (4×). For the scratching pins, they are the typical pipette tips that are commercially available; the protruded length can be adjusted by releasing the screw, and the evenness of pins can be calibrated within a minute ( Fig. 2 ). This design ensures perfect contact between the tips and the bottom of wells (i.e., cell monolayer). Furthermore, the plastic pipette tips have many advantages: (1) they provide sufficient elasticity to ensure perfect contact between the tips and the well surface, (2) they cause no serious mechanical damages on the well surface (i.e., smoothness), (3) they are inexpensive and commercially available, and (4) they can be sterilized (i.e., contamination free) and disposable for every single use. Therefore, those defects such as scrape, wound irregularity, or cross-contamination caused by razor blade, rubber policemen, or a multichannel pipetter can all be overcome by this newly designed wounder.

Diagram of the 8-channel mechanical wounder. (

Schematic diagrams showing the calibration of pins before the wounding process. Step 1: hold the wounder (fitted with pipette tips) vertically on a flat surface (e.g., 96-well plate, Petri dish) and then release the hex screw with wrench. Step 2: simply tap the wounder or the pins until all the tips touch the flat surface. Step 3: lock the hex screw again. Step 4: check the evenness of each tip by pushing the wounder on a flat surface.
Schematic diagrams showing the wounding process. (
Comparison of 8-Channel Mechanical Wounder with Other Conventional or Commercial Wounders

Plastic traditional wounder, including cell scraper, rubber policemen, and so on.
Metallic traditional wounder, including razor blade, forceps, and so on.
Oris™ Cell Migration Assay by Platypus Technologies (Madison, WI).
Wounding replicators by V&P Scientific, Inc. (San Diego, CA).
High-throughput wounder by Yarrow et al. 10
Mechanical wounder by Lauder et al. 11
Figure 3B exemplifies the scratched 96-well plate using our wounder. We noticed that the consistency of the wound width (area) being created in each well is critical for the development of such a device. Every well of the culture plate was captured after wounding, and the scratched area (wound size) was quantitated by Java’s Image J software. Analysis showed that the average width is about 600 µm, with a standard deviation of 6% throughout the 96-well plate, and is reproducible under different trials.
Using this wounding device, we have been able to observe cell migration of normal and cancer cell lines, such as human endothelial cells (e.g., HUVEC), dermal fibroblasts and adipocytes (e.g., 3T3-L1), rat epithelial cells (e.g., IEC-6), and vascular smooth muscle cells and cancer cells (e.g., HepG2, NPC HK-1). In this report, reepithelialization and reendothelialization of wounded IEC-6 cells and HUVECs were chosen as examples. Restitution of epithelial damage in the intestine was modeled using IEC-6 cells, the cell monolayer was removed using our wounder, and the recovery of the denuded area due to cell migration was observed at 16 and 24 h. The applicability of this new wounder was further evaluated by introducing substances that can modulate the migration property of IEC-6 cells. DFMO, a well-known inhibitor of cell migration, inhibits ornithine decarboxylase and decreases the synthesis of polyamines. 12 Because polyamines are required for the early phase of restitution, deficiency in polyamine would affect cell migration. 13 Besides, EGF was chosen to be the inducer of cell migration. 14 In our system, the IEC-6 cells responded typically to the actions of DFMO and EGF ( Fig. 4A , B ). The percentage of wound closure at 16 h after wounding was about 60% in control cells, whereas in EGF-treated cells, the percentage was more than 90%, and in DFMO-treated cells, the percentage was reduced to only 33%. At the 24-h time point, the recovery was recorded as 76%, 100% and 50%, respectively. These results were consistent with a previous wounding method using a razor blade. 15 On the other hand, HUVECs are commonly used in studying angiogenesis and wound healing. 16 Migration of cells was observed at 16 and 24 h after wounding ( Fig. 5A ). The cells treated with different percentages of serum (0%, 1%, and 20%) showed different migration rates in a serum-dependent manner ( Fig. 5B ). At 24 h, cells cultured in plain medium migrated relatively less, with wound closure less than 40%, whereas complete wound closure was found in the presence of 20% FBS. Under low serum conditions, cells could only attain 60% and 72% of wound closure at 16 and 24 h, respectively.
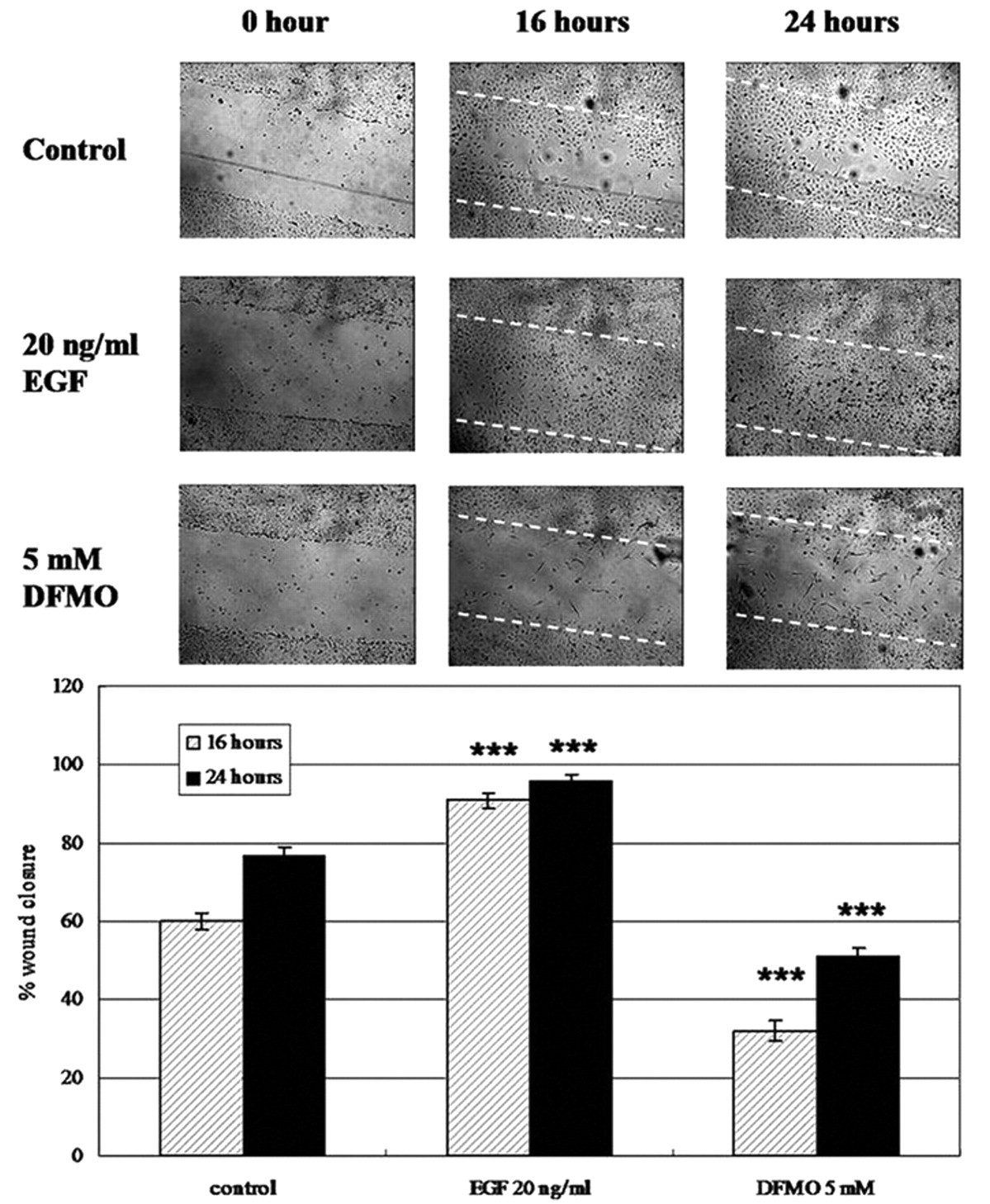
Migration of IEC-6 cells after wounding. (
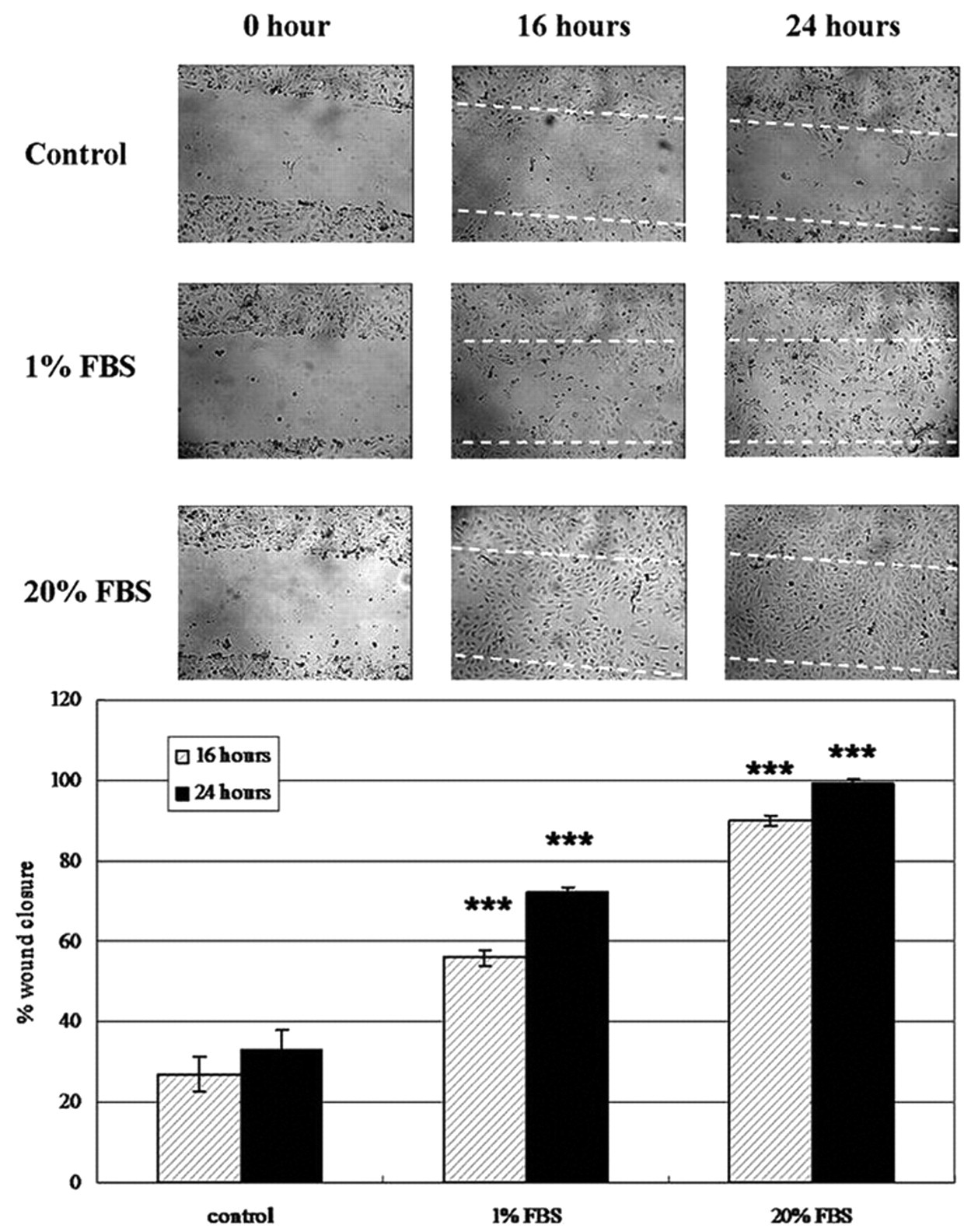
Migration of human umbilical vein endothelial cells (HUVECs) after wounding. (
These results suggest that the present wounder is especially well suited for a cell migration assay in a 96-well plate format. We noted that cell migration study is a very simple and basic assay. It is routinely performed in a wide range of laboratories because of its versatility; it allows analysis not only for a cell motility study but also for cell-matrix and cell-cell interaction levels such as tissue reorganization, cell polarization, or matrix remodeling. It can also be used for screening of compounds in a visually based manner. In the past, researchers have modified such experimental protocols and wounding devices to adapt to a high-throughput manner. 10 However, such high-content image-based screening always requires automated fluorescence microscopy, image and data analysis algorithms, or even an automatic liquid handling workstation. In fact, it is not feasible for most research laboratories to have such capital investment for a relatively simple assay. Besides, screening for hundreds of compounds would not be a common practice in most laboratory settings. As a consequence, a simple device as reported in this study for handling samples in a 96-well plate format is highly desirable. There is a possibility that this device will be marketed to make it available to the scientific community.
Conclusions
We have designed an 8-channel mechanical wounder that is a useful tool for performing a cell migration/wound-healing assay for multiple-sample analysis by providing consistent denudation of a monolayer of cells for a 96-well plate format. This easy-to-assemble and user-friendly device can greatly enhance experimental efficiency and accuracy.
Footnotes
Acknowledgements
The authors are extremely grateful to Mr. Tam Po Leung and Mr. Wong Chi Kin and the technical staff of the Science Faculty Workshop, Hong Kong Baptist University, for their technical skills and advice in the making of this wounder.
This work was supported in part by a research grant (HKBU 1/06C) of the Research Grant Committee, Hong Kong SAR Government. This work is under patent pending (publication no. 101339104).
The authors declare that they have no competing financial interests.
