Abstract
Angiogenesis is the process of generating new blood vessels from preexisting vessels and is considered essential in many pathological conditions. The purpose of the present study is to evaluate the effect of aspartame on angiogenesis in vivo chick chorioallantoic membrane (CAM) and wound-healing models as well as in vitro 2,3-bis-2H-tetrazolium-5-carboxanilide (XTT) and tube formation assays. In CAM assay, aspartame increased angiogenesis in a concentration-dependent manner. Compared with the control group, aspartame has significantly increased vessel proliferation (p < 0.001). In addition, in vivo rat model of skin wound-healing study showed that aspartame group had better healing than control group, and this was statistically significant at p < 0.05. There was a slight proliferative effect of aspartame on human umbilical vein endothelial cells on XTT assay in vitro, but it was not statistically significant; and there was no antiangiogenic effect of aspartame on tube formation assay in vitro. These results provide evidence that aspartame induces angiogenesis in vitro and in vivo; so regular use may have undesirable effect on susceptible cases.
Introduction
Aspartame is a food additive marketed under a few brand names, and it has subsequently been approved for use in a number of food products including multivitamins, fruit juices, stick-type confections, breath mints, and iced tea. 1 Aspartame is a methyl ester of an aspartic acid and phenylalanine dipeptide, used as a synthetic nonnutritive sweetener in over 6000 products across 90 countries worldwide. 2 Although aspartame is used in many products for nearly 30 years, it is not clearly established whether it has an undesirable effect. Recent study suggests that useable aspartame extracts have angiogenic activity in vitro. 3 Angiogenesis is thought to be important in some pathological processes such as chronic inflammatory diseases, psoriasis, diabetic retinopathy, and some cancers. 4,5 If aspartame has a possible inducing effect on angiogenesis, it may have unfavorable long-term results on these patients. Therefore, we aimed to examine the effects of aspartame on angiogenesis in vivo chorioallantoic membrane (CAM) model and rat model of skin wound healing; in addition, in vitro 2,3-bis-2H-tetrazolium-5-carboxanilide (XTT) assay and endothelial cell tube formation assay were also studied.
Materials and methods
Aspartame solution
Aspartame was purchased from Bilim Pharmaceuticals Corp. (Istanbul, Turkey). For XTT assay, various aspartame solutions were prepared with serial dilutions from a stock solution of 1 mM, which was prepared by dissolving 6 mg aspartame in a final volume of 20 mL solution. For other models, a stock solution of 100 mM aspartame was prepared and various concentrations were used with dilutions.
Cell viability assay
Cell viability was evaluated using the XTT method. Human umbilical vein endothelial cell (HUVEC; 100 µL) suspensions were plated in 96-well flat-bottom tissue culture plates at a density of 4 × 104 cells/well. The cells were allowed to attach to the well overnight and then treated with 100 µL of aspartame solution at various concentrations (20, 40, 60, 80, and 100 µM) for 24–48 h. Then, 50 µL of XTT reagent solution (Biological Industries, Israel) was added to each well, and then the plate was incubated in a humidified carbon dioxide incubator at 37°C for 2–4 h. Control wells were the wells containing the cells and the blank background. Absorbance was read at 450 nm in an enzyme-linked immunosorbent assay plate reader (model ELx 800 bioelisa microplate reader; Biokit, Spain), and the results were expressed as percentage variation with respect to controls. The cell viability was repeated in triplicate with three independent experiments.
In vitro endothelial cell tube formation assay
BD Matrigel (BD cat. no.: 354234, USA; 50 µL) matrix was plated in 96-well flat-bottom cell culture cluster plates. After incubating for 30 min, 50 µL HUVEC (10,000 cells/well) and 20 µL of 20 mM aspartame solution were placed on matrigel. Control group was prepared by following the same procedure as mentioned previously except aspartame solution. The plate was incubated at 37°C for 16–18 h. 6 Following incubation, the plated wells were photographed, and the results were evaluated.
CAM assay
Angiogenesis was evaluated in vivo using the CAM assay. Atak-S fertilized chicken eggs were obtained from Poultry Institution (Ankara, Turkey) and incubated at 37°C, and 85–90% of relative humidity were maintained throughout the experiment.
Aspartame with 6, 15, 30, and 60 mM concentrations were prepared, and 50 µL of each solution was used on CAMs.
Experiments were performed to determine the mode of action of aspartame on vascular development in the sixth day and seventh day on CAM. On the sixth day, between 10 a.m. and 12 p.m., a window was opened on each egg to allow subsequent access to the CAM and 50 µL of test solution was placed on the surface of each CAM. 7 Some control CAMs received phosphate-buffered saline (PBS), and then all the eggs were sealed with a transparent tape. On day 7, CAMs were screened and taken pictures using a computer-aided stereomicroscope (model S6D; Leica Microsystems, Heerbrugg, Switzerland). The captured images were processed with the software Leica Application Suite V4 Automated Image Analysis (Leica Microsystems). Finally, the increase in vessel formation was scored. 8
Rat model of skin wound healing
Male Sprague Dawley rats, aged 8 weeks and weighing 200–250 g, were used for this model. They were anesthetized by intraperitoneal injection of ketamine (90 mg/kg) and xylazine (10 mg/kg). Before excision, the surgical area, which is the skin over the dorsal area, was shaved off and disinfected with 70% ethanol. With the help of a biopsy punch and dissecting scissors, two round sections of 5 mm diameter full thickness skin were excised from each rat. 9 Each rat had two groups of wounds: control wound (treated with PBS) and aspartame-treated wound. Aspartame-treated wounds received 50 µL of 50 mM aspartame solution. The same treatment protocol was applied for 7 days. On day 8, the surface area of each wound was calculated and the wounds were harvested. All specimens were fixed in 10% formalin. After getting thin perpendicular sections, embedding in paraffin and deparaffinization procedures were carried out, and the specimens were stained with hematoxylin and eosin in Department of Pathology of Gülhane Military Medical Academy. Histopathological examination for all slides was carried out under a light microscope by a pathologist blinded to the groups. The wound-healing process was scored according to the following parameters: re-epithelization, granulation tissue formation, collagen deposition, inflammatory cells, and angiogenesis. 10,11 Also, the wound areas were statistically compared. The procedures in this study were approved by the Local Animal Ethics Committee.
Statistics
The statistical analysis was carried out using Statistical Package for the Social Sciences software for Windows, Version 15.00 (SPSS Inc., Chicago, Illinois, USA). The value of p ≤ 0.05 was considered statistically significant.
Results
Cell viability assay of aspartame
HUVECs were incubated with increasing doses of aspartame for 24–48 h, and cell viability was observed with the XTT assay. There is a slight proliferative effect on HUVECs in the presence of aspartame for 24 h, but it was not statistically significant (Figure 1).
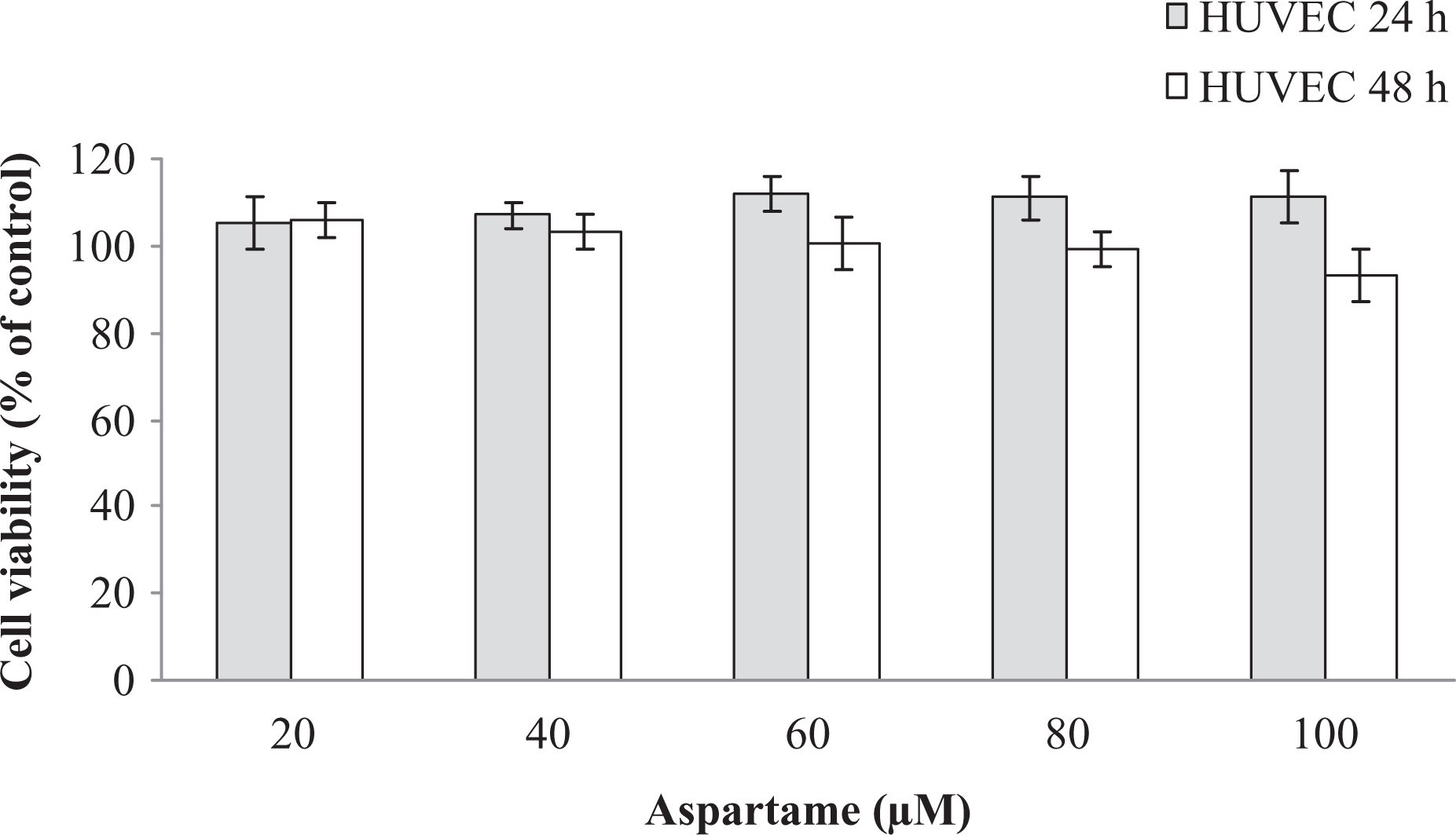
Cell proliferation of HUVECs under aspartame incubation (cell viability was indicated as percentage of control). HUVECs: human umbilical vein endothelial cells.
Effect of aspartame on tube formation
In the tube formation assay, on the 18th hour of incubation, the results were evaluated. Comparing the tube length/area ratio values of control and aspartame groups, there was no significant statistical difference. So, it was found that aspartame had no antiangiogenic effect (Figures 2 and 3).

Ratio of tube length to area under aspartame incubation and control.
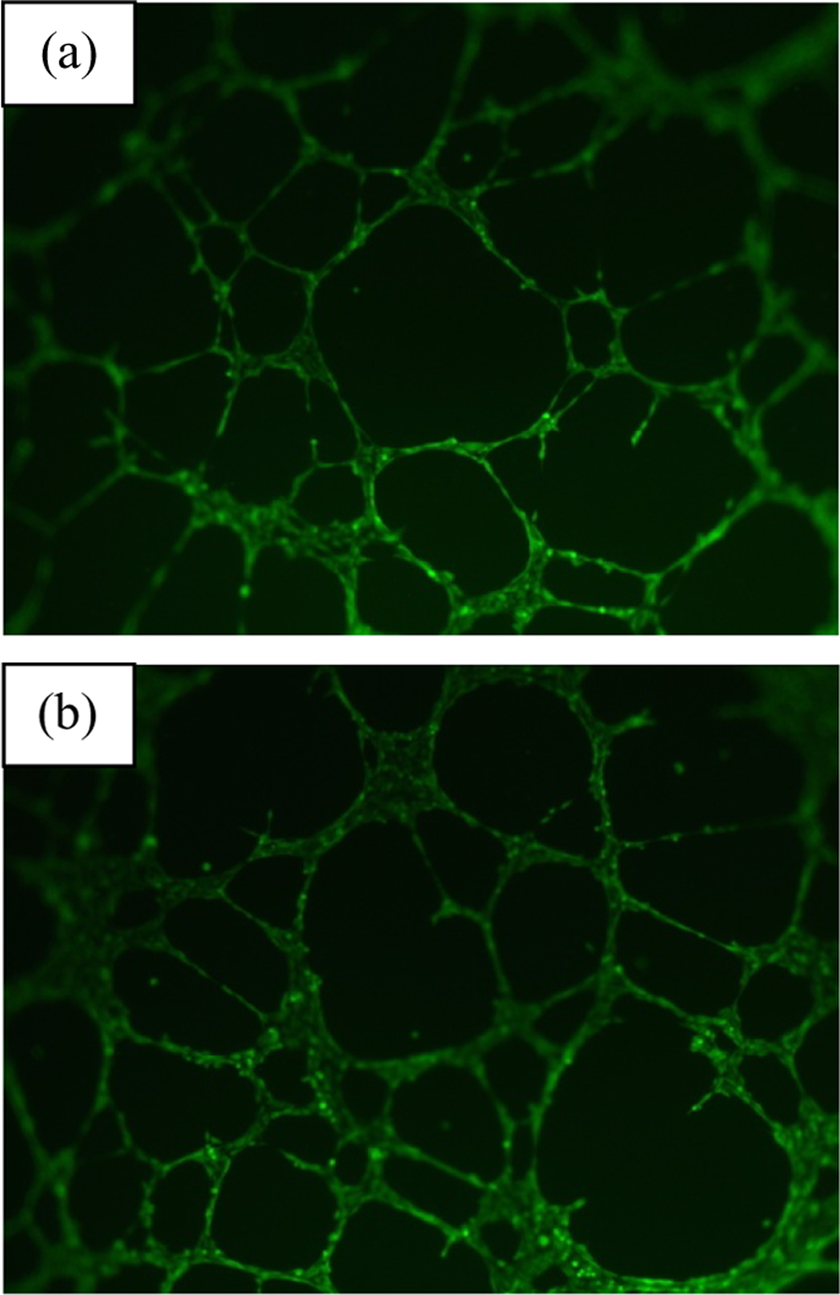
Tube formations of control group (a) and under aspartame incubation (b).
Aspartame induces angiogenesis on CAM
To determine the mode of action and dose dependency of drug during days 6 and 7 on CAMs, we applied various concentrations of aspartame solution to CAMs on day 6 and evaluated their effects on CAM vessels growth, angiogenesis on day 7. On day 7, the treatment of CAMs with aspartame solutions caused a significant dose-dependent increase in CAM vascular area. The increase in vessel formation was scored. 8 While physiological angiogenesis was observed in the form of some allantoic vessels in the control group (score 0), a significant increase in CAM area of eggs treated with aspartame solution was appreciable macroscopically (score +2). Significant growth was seen for 60 mM aspartame solution.
In affected CAMs, the vessels were thicker and showed more branching (Figure 4). Efficacy was not enough for low doses; while there was a significant increase for high doses in comparison with the control group. Aspartame caused a significant increase in blood vessels when compared with the control group. However, the lower doses of drug did not show discernible effect on blood vessel pattern.

Effect of aspartame solution on CAM before (a) and after 24 h (c). In images (b) and (d), the same vessels with (a) and (c) were highlighted with yellow to be seen clearly. CAM: chorioallantoic membrane.
The efficacies at different doses were compared using χ2 test, and there was a statistically significant difference (Yates correction χ2 = 10.60, p = 0.001). The efficacy of increasing dose was evaluated with Spearman’s correlation test and a strong relation was found (r = 0.704, p = 0.001). Macroscopically evaluated results of CAMs are shown in Table 1.
Macroscopic evaluation of the effect of aspartame treatment on CAM.

CAM: chorioallantoic membrane.
Aspartame may have a healing effect on wounds by angiogenesis
Aspartame-treated wounds were found to heal better than the control group. For control group, mean for wound surface area was found as 32.40 ± 5.58 mm2, and for aspartame group, the mean was 21.25 ± 2.59 mm2. According to the results of independent samples t test analyses, the two groups were found to be significantly different in wound healing (p < 0.05), and aspartame had a better healing effect.
Histopathological examination showed that aspartame-treated wounds had better healing (Figure 5). The thickness of granulation tissue and collagen deposition were close to each other comparing the control and aspartame groups. All the specimens indicated a complete re-epithelization. Therefore, aspartame-treated wounds showed enhanced cellularity and increased angiogenesis compared with the control group.

Aspartame-treated wounds (a) showed better angiogenesis and thus better healing compared with the control group (b). Vessel formation seems to be much more in (a) than that of (b).
Discussion
Artificial sweeteners are added to a wide variety of foods, drinks, drugs, and hygienic products. 12 Since the introduction of aspartame into the American food supply in 1981, it has grown to become the most widely used and accepted artificial sweetener. Aspartame has been the predominant artificial sweetener in the United States for the past 25 years and is currently found in over 6000 food and beverage products. 13 Although many scientific papers have reported contradictory data about aspartame’s safety, there are few studies explaining the relation between aspartame and angiogenesis. 3 The strength of this study is the way it shows the effects of aspartame on both in vivo and in vitro models.
In this study, we aimed to explain clearly the relation between aspartame and angiogenesis using both in vivo and in vitro models because aspartame may have a role in many diseases via angiogenesis.
According to the in vitro results of this study, it was clearly seen that aspartame had a mild proliferative effect on HUVECs in cell viability assay, during short exposure times, similar to the results of Alleva et al. 3 However, these results were not statistically significant.
CAM assay is a widely used in vivo method to analyze the effects of many different molecules on angiogenesis at varied concentrations. In this study, we examined the effects of aspartame at different concentrations (6–60 mM) on CAM assay. We found that aspartame had an angiogenic effect especially at high concentrations compared with the control group and this was statistically significant. Also, we showed the in vivo effects of aspartame on rat model of skin wound healing and aspartame-treated wounds obviously showed better healing at a concentration of 50 mM, and the results were statistically significant. By inducing angiogenic response, aspartame might have a proangiogenic effect on CAM and wound healing model in vivo. Thus, it is believed that aspartame had similar effects on different species and increased angiogenesis. US Food and Drug Administration has set the acceptable daily intake (ADI) of aspartame at 50 mg/kg of body weight. The European Food Safety Authority has recommended a slightly lower ADI of aspartame at 40 mg/kg. Aspartame concentrations used in our in vivo models were much lower than these recommended limits. In addition, our results showed that increasing concentrations of aspartame had stronger angiogenic effects. Past studies claimed that aspartame induces regenerative cytokine production leading to the activation of mitogen-activated protein kinases and resulting in the formation of new blood vessels. 3 In our opinion, a change on angiogenic switch with aspartame might play an important role in neovascularization.
There are many contradictory studies about aspartame and its negative effects on health. 14,15 Many diseases such as Alzheimer’s, phenylketonuria, congenital defects, some types of cancer, lupus, and multiple sclerosis are thought to have possible relation with aspartame. 16 The results of this study, which aims to indicate the relation between aspartame and angiogenesis, showed that aspartame might have an inducing effect on angiogenesis in vitro and in vivo models. Long-term aspartame intake may have unfavorable results on patients with diseases; those pathologic bases are related to angiogenesis. Therefore, other detailed studies, such as tumor models, are needed for a final decision about worldwide usage of aspartame.
Footnotes
Acknowledgment
The authors would like to give special thanks to Ozlem Bitisik from Bilim Pharmaceuticals Corp. (Istanbul, Turkey) for providing aspartame.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This study was supported by Gülhane Military Medical Academy (AR no.: 2012-37).
