Abstract
Chitin and its derivatives are widely used as biomedical materials because of their versatility and biocompatibility. Chitinases are enzymes that produce chito-oligosaccharides from chitin. The assay of chitinase activity is difficult because few appropriate substrates are available. In this study, the authors developed an efficient and low-cost chitinase assay using colloidal chitin azure. The assay feasibility is evaluated and compared with traditional assays employing colloidal chitin and chitin azure. The authors found that the optimum pH for determining chitinase activity using colloid chitin azure was pH 5 or 8. The method was sensitive, and the assay was complete within 30 min. When the assay was used to measure chitinase activities produced by 2 strains of chitinolytic bacteria, BCTS (an Escherichia coli BL21 [DE3] expressing a secretory recombinant chitinase) and AS1 (a chitinolytic bacterium with low levels of chitinase), it was shown that cultivation in Bushnell-Haas selection medium caused AS1 to secrete a higher level of chitinase than was secreted when the bacterium grew in other media. In summary, colloid chitin azure is a sensitive, feasible, reproducible, and low-cost substrate for the assay of chitinase activity.
Introduction
C
Chitinases can be isolated from some viruses, bacteria, higher plants, and animals. 2,6 Preparation costs are influenced by the ease with which chitinase activity can be measured. This, in turn, depends on the availability of appropriate enzyme substrates. Insoluble substrates include chitin, colloidal chitin, chitin covalently bonded to dyes, or tritium-labeled chitin. Chemically modified chitins, such as glycol-chitin, carboxymethyl chitin, and 6-O-hydroxypropyl-chitin, are soluble substrates. Using these substrates, chitinase activity can be determined by spectrophotometry or with a radiometer. 7,8 Typically, chitinase activity is evaluated by measuring the production of reducing sugars (e.g., chito-oligosaccharides) using colloidal chitin (CC) as substrate. Reducing sugars react with a color reagent such as 0.05% (w/v) potassium ferricyanide, in 0.5 M sodium bicarbonate. Products of this reaction can be measured by absorbance at 420 nm. The chitinase assay is often affected by the presence of other reducing sugars in the medium, 9 however, and this imposes a limit on assay sensitivity. Although a sensitive fluorometric technique has been developed recently, this assay seems to be very expensive and time-consuming. 10
Chitin azure (CA), chitin covalently coupled with the Remazol brilliant violet 5R dye, was introduced in 1964 for the measurement of chitinase activity. 11,12 The dye, a product of CA digested by chitinase, can be measured at 560 nm in a colorimeter, and the assay is not affected by reducing sugar of the medium. The assay is also very inexpensive. The sensitivity is low, however, because CA is poorly soluble.
In this study, we rendered the CA colloidal to increase dispersion in solvent by chemical techniques and characterized the resulting colloidal CA (CCA); in particular, we investigated its utility in a chitinase assay. We successfully identified an optimum growth medium for the bacterium AS1, a chitinolytic strain that produces low levels of chitinase. Therefore, CCA is a good substrate for a sensitive, fast, inexpensive chitinase assay.
Materials and Methods
Chemicals
Chitin was purchased from local markets and had been imported from Vietnam. CA, chitinases from Streptomyces griseus as standard, and other chemicals were purchased from Sigma Chemical Co. (St. Louis, MO).
Preparation of CC and CCA
The chitin, or CA, powder of 4 g was suspended in 37% (v/v) HCl (40 mL) for 50 min. Then, 1 L of distilled water was slowly added. After centrifugation at 8000 g for 20 min, the pellet was collected and washed with distilled water after resuspension. The CC or CCA was sterilized by autoclaving after the pH values rose, upon washing, to pH 3.5.
Measurements of chitinase activity using a reducing sugar assay
Chitinase was incubated with 3 mg/mL CC in 0.2 M potassium phosphate buffer at indicated pH and temperature. After incubation, enzyme activity was terminated by boiling for 5 min. The reaction mixture was centrifuged at 13,000 g for 5 min. The supernatant (750 µL) was incubated with the color reagent, 0.05% (w/v) potassium ferricyanide, in 0.5 M sodium bicarbonate, in water for 15 min. After boiling, absorbance was measured at 420 nm. The phosphate buffer was used as a negative control, and each reported value is the mean of triplicate samples.
Measurements of chitinase activity using CA or CCA
Chitinase was incubated with 3 mg/mL CA or CCA in 0.2 M potassium phosphate buffer at indicated temperature and pH. After incubation, enzyme activity was terminated by boiling for 5 min. The reaction mixture was centrifuged at 13,000 g for 5 min. Then, absorbance was measured at 560 nm. Enzyme-free phosphate buffer was used as a negative control, and each reported value is the mean of triplicate samples.
Bacterial culture medium
Luria-Bertani medium (LB) contained 10 g/L tryptone, 5 g/L yeast extract, and 10 g/L NaCl. Terrific broth (TB) was composed of 12 g/L tryptone, 24 g/L yeast extract, 12.5 g/L potassium biphosphate, 2.3 g/L potassium dihydrogen phosphate, and 4 mL/L glycerol. The YMP medium had 3 g/L yeast extract, 3 g/L malt extract, 5 g/L peptone, and 2 g/L magnesium sulfate. The composition of Bushnell-Haas selection medium (BHS) was 0.4 g/L magnesium sulfate, 1 g/L potassium dihydrogen phosphate, 1 g/L ammonium biphosphate, 1 g/L potassium nitrate, 0.2 g/L calcium chloride, and 0.5 g/L ferric chloride.
Bacterial culture and protein measurements
To compare various substrates in chitinase activity assays, a wild-type chitinolytic bacterial strain of commercial interest was inoculated into 50 mL medium with 2% (w/v) chitin powder and grown at 28°C for 3 days. Also, strain BCTS, which is Escherichia coli BL21 (DE3) expressing a secretory recombinant chitinase, was grown at 25°C in medium with ampicillin and chitinase expression induced with isopropyl-β-D-thiogalactopyranoside (IPTG). 13 All growth media were harvested for chitinase activity assays as well as total protein concentration, which was determined by measuring absorbance at 280 nm and a Pierce BCA kit (Rockford, IL) using bovine serum albumin as a standard. 14 The standard curve for quantifying chitinase activity was fitted with the absorbance measured at 560 nm from 0 to 2 U of commercial S. griseus chitinase reacting with 3 mg/mL of CCA as substrate.
Results and Discussion
Comparison of 3 different substrates in assays of chitinase activity
Chitinases are the specific enzymes that are able to degrade chitin, a polymer of GlcNAc, by hydrolyzing β-glycosidic bonds. Here, 0.2 U of the commercial chitinase from S. griseus was applied to examine 3 substrates—CC, CA, and CCA—for assaying chitinase activity. Figure 1 summarizes the results by measuring the absorbance at 10-min intervals for 50 min. It appeared that the colloidal substrates were better and efficiently digested due to the greatly elevated absorbance values, parallel with the previous findings that colloidal substrate molecules were more accessible to the enzyme than powder substrates. 15 Moreover, it can be seen that the optimum incubation time was 30 min. To more accurately investigate differences between substrates, we performed dose-dependent experiments. It is shown in Figure 2 that, with CC, the formation of product reached the steady state when more than 3 U of enzyme was added. In contrast, with CCA as substrate, the steady state was reached if the enzyme level was more than 4 U. With CA, however, the steady state was not attained even when the enzyme was added to the level of 5 U. These results indicate that colloidal substrates are most sensitive for analyzing chitinase activity. Accordingly, less than 1 U of chitinases from S. griseus was used for the following chitinases activity assay, and CCA was chosen as the preferable substrate.
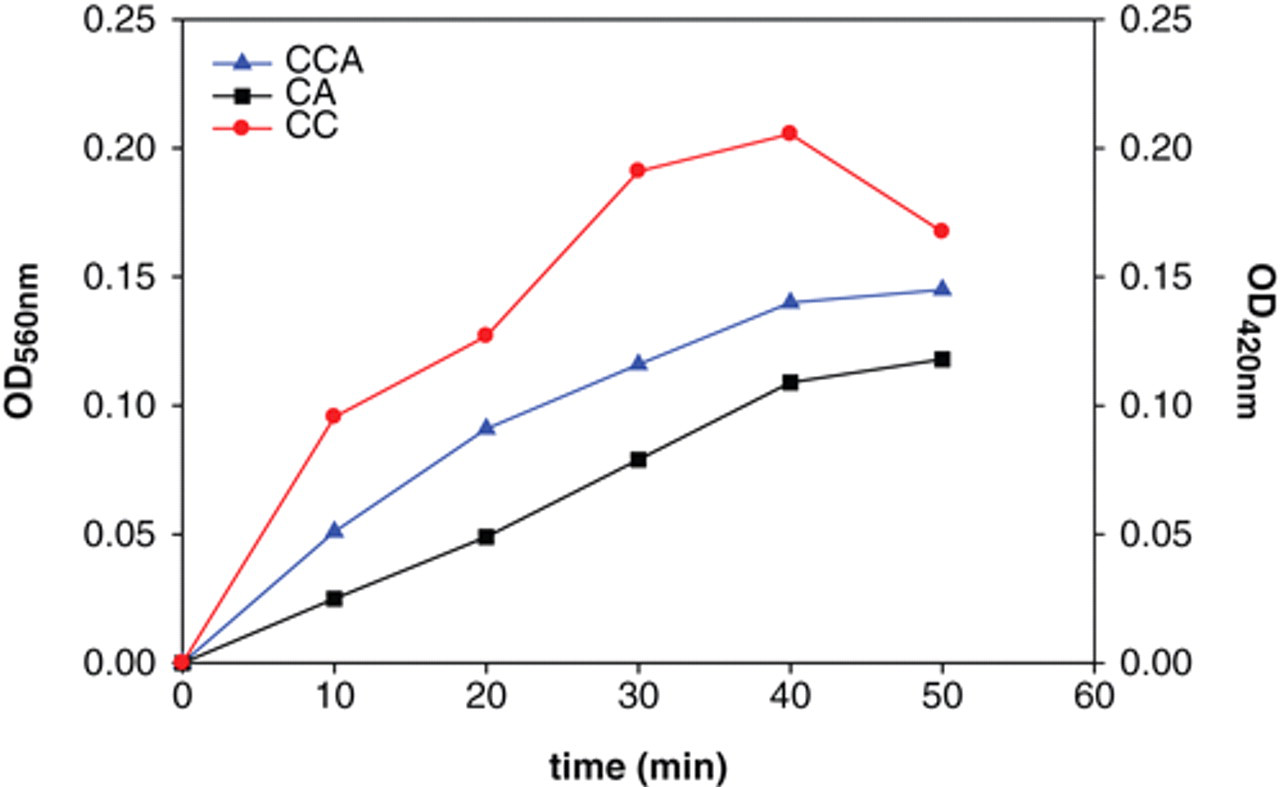
Kinetics of Streptomyces griseus chitinase acting on different substrates: colloidal chitin (CC), chitin azure (CA), or colloidal CA (CCA). S. griseus chitinase (0.2 U) was incubated with 3 substrates—CC, CA, or CCA—in potassium phosphate buffer, pH 6, at 25°C. The absorbance of the reactions (OD560 for CA and CCA, OD420 for CC) was detected at 10-min intervals for 50 min.
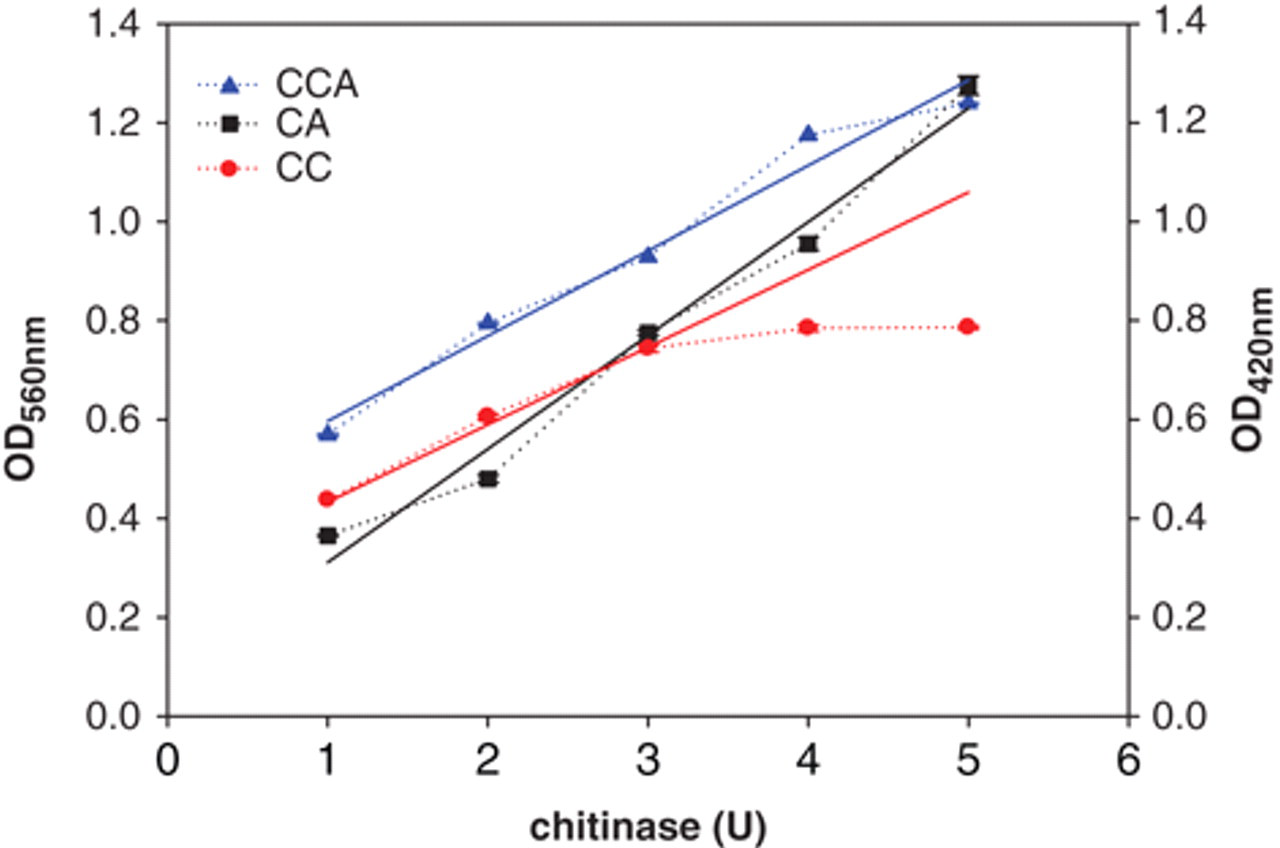
The chitinase activity of Streptomyces griseus chitinase assaying with colloidal chitin (CC), chitin azure (CA), or colloidal CA (CCA) as substrates. Different doses of S. griseus chitinase were assayed for their activities in potassium phosphate buffer, pH 6, at 25°C for 30 min. Three different substrates—CC, CA, or CCA—were used. The absorbance (OD560 for CA and CCA, OD420 for CC) was then determined.
The colloid substrates CC and CCA are somewhat granular gels with 94.8% and 94.5% (both w/v) water, respectively. CA is composed of amorphous, rough, semitransparent, solid particles of various sizes. CC is white in color, CA dark blue, and CCA blue-violet. The ferric iron in CC would be reduced to ferrous iron in the enzymatic reaction of chitinase. The optimum absorbance of ferrous iron is at 420 nm. Azure, a soluble violet substance with λmax at 560 nm, was released from CA or CCA when reacting with chitinases.
Next, we attempted to identify the effects of temperature (range, 20-37°C) or pH (5-9) on assaying chitinase activity using the above-described substrates. It is shown in Figure 3 that the linearity of absorbance can be observed in the reaction with CCA as substrate at a temperature lower than 25°C but lower than 30°C with CC or CA as substrate. Figure 4 demonstrates that pH 5 or 8 is the optimum pH value in the use of CC or CCA, respectively, and pH 7 for CA. Changes in pH apparently affected the enzymatic reactions with colloid substrates but not with the powder form. Such findings indicate that the solid substrate is more difficult to be hydrolyzed, and it might result from the solid form having a closed molecular organization in the quasi-native crystalline structure. 15
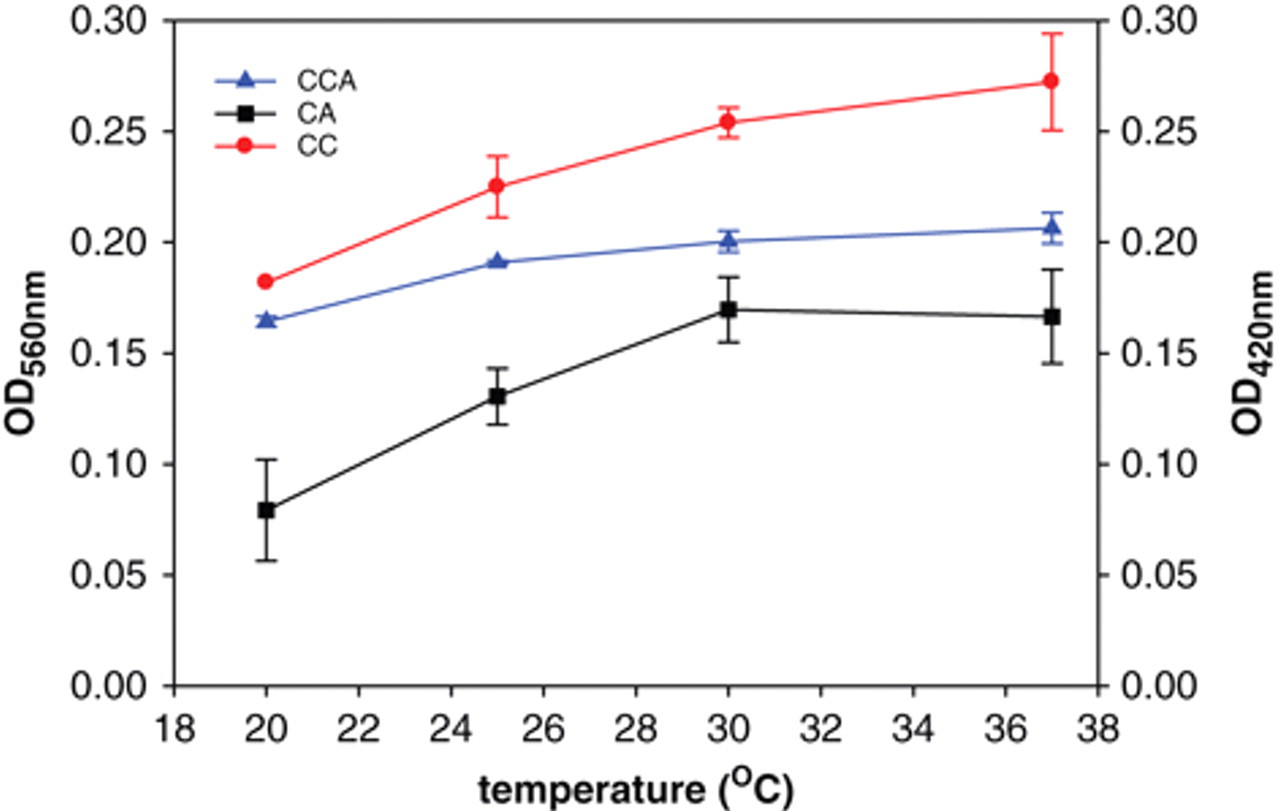
Effects of temperature on the activity of Streptomyces griseus chitinase, using chitin (CC), chitin azure (CA), or colloidal CA (CCA) as substrates. The reaction mixtures, containing 0.2 U S. griseus chitinase, were in potassium phosphate buffer, pH 6, at the temperature indicated. The chitinase activities can be obtained from the absorbance (OD560 for CA and CCA, OD420 for CC).
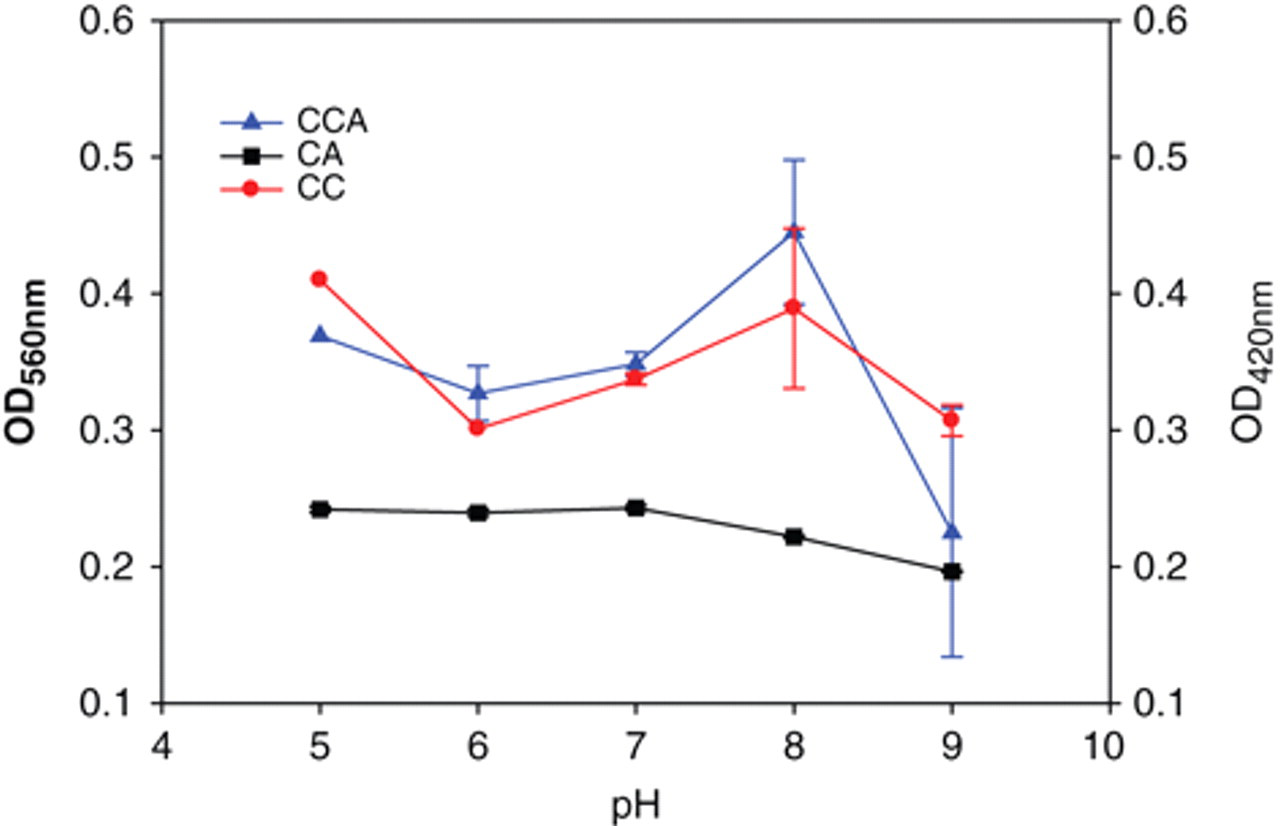
Effects of pH on the activity of Streptomyces griseus chitinase, using chitin (CC), chitin azure (CA), or colloidal CA (CCA) as substrates. The reaction mixtures, containing 1 U S. griseus chitinase, were prepared in the working solution at pH 5 to 9. The chitinase activities were determined and presented in the values of the absorbance (OD560 for CA and CCA, OD420 for CC).
Determination of the thermostabilities of CC, CA, and CCA
To determine the interference, if any, of boiling treatment for stopping the chitinase reaction, we performed thermostability tests. The reaction mixtures containing CC, CA, or CCA were prepared but without adding chitinases and then treated by boiling for 5 min. The untreated samples served as controls. It was found that only CCA appeared to be unstable when receiving the boiling treatment. The absorbance was increased by 0.154 units at 560 nm. Presumably, some azure dye was released from CCA when CCA was heated. To overcome this problem, the supernatants of chitinase activity assays using CCA could be collected by centrifugation before terminating reactions.
Kinetic studies
The Km, kcat, Vmax, and specificity constant (kcat/Km) were determined for the substrates CC, CA, and CCA and are shown in Table 1 . The lowest Km value, 20.42 mg/mL, and the highest Vmax value, 1.25 S−1, were found for CC. The results suggested that the enzyme might have evolved to maximize rates against its original substrate, chitin. However, the assay with chitin or CC as substrate is interfered with reducing sugar. 9
Kinetic Parameters
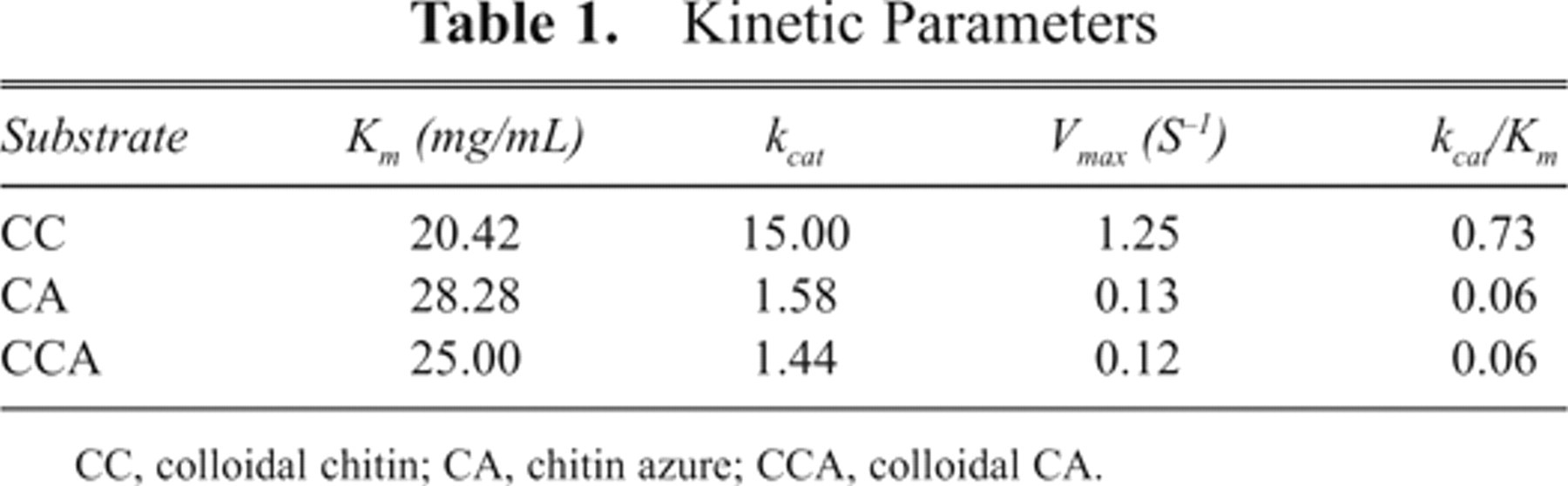
CC, colloidal chitin; CA, chitin azure; CCA, colloidal CA.
The kinetics study also revealed that CCA was more accessible and degradable than CA due to the lower Km and higher Vmax of CCA. It may be argued that the difference of Km between CA and CCA was only 3.72 mg/mL. However, it cannot be ignored that chitin is a polymer with the molecular weight of a million Dalton. Therefore, the Km of CA and CCA in mM would differ by 7 orders of magnitude.
Application of CCA to the evaluation of chitinolytic microorganisms
Typically, chitinolytic bacteria are detected either by the production of clearing zones on culture plates containing chitin or by fluorescence when exposed to a fluorogenic substrate analog of chitin. 3 Both methods have drawbacks. 16 Chitinases of microorganisms that must be exported and diffuse into the surrounding medium can meet the requirement of the screening method with the selective culture plates, but the capacity to degrade small oligomers may be simply detected by hydrolysis of a chitin analog. 16,17 Usually, only 10% of culturable bacteria that degrade chitin can be detected using selective plates. 18-20
In this study, therefore, we applied the new substrate, CCA, to identify chitinolytic microorganisms with relatively low levels of chitinase activity. First, strain BCTS, E. coli BL21 (DE3) expressing a secretory recombinant chitinase, was used to analyze its chitinase activity by CCA as the substrate. Enzyme activities of culture medium and cell lysates, 1 to 12 h postinduction, were assayed. The longer the incubation time, the more chitinase was produced at 4 h after induction (
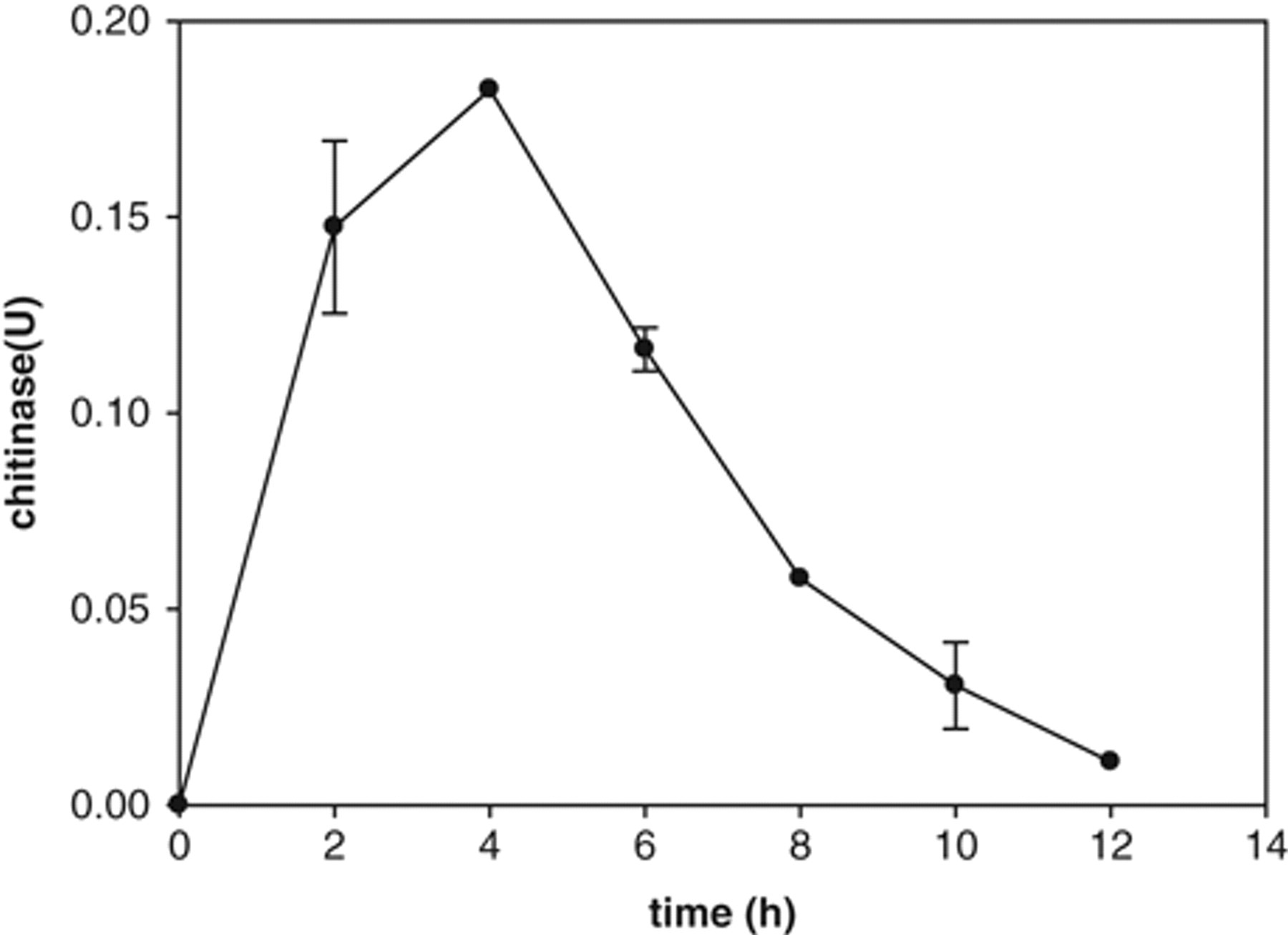
The activity of BCTS chitinase, using colloidal chitin azure (CCA), as substrate. The cell lysates of BCTS were harvested after the isopropyl-β-D-thiogalactopyranoside (IPTG) induction for 12 h. The chitinase activities were detected using CCA as substrate at 2-h intervals and are shown in the values of the absorbance (OD560).
Absorbance of Chitinase Activity from AS1 Cultures in Different Media (OD560 for CA and CCA, OD420 for CC)

CC, colloidal chitin; CA, chitin azure; CCA, colloidal CA; YMP, yeast extract/malt extract/peptone; LB, Luria-Bertani medium; TB, terrific broth; BHS, Bushnell-Haas selection medium; UD, underdetected.
These results indicate that the CCA substrate is much more sensitive than is CA. The low absorbance seen using CA is presumably due to a high detection threshold. To calculate chitinase levels, we constructed a standard curve, using data obtained when a chitinase from S. griseus acted on CCA. Chitinase concentrations in the range of 10−5 to 2 U/mL were used. Total protein levels were also measured ( Table 3 ). The specific activity of chitinase was highest in BHS medium. In future, AS1 may be commercially fermented in BHS medium with chitin.
Comparison of Growth Media Used For Chitinase Production from Strain AS1
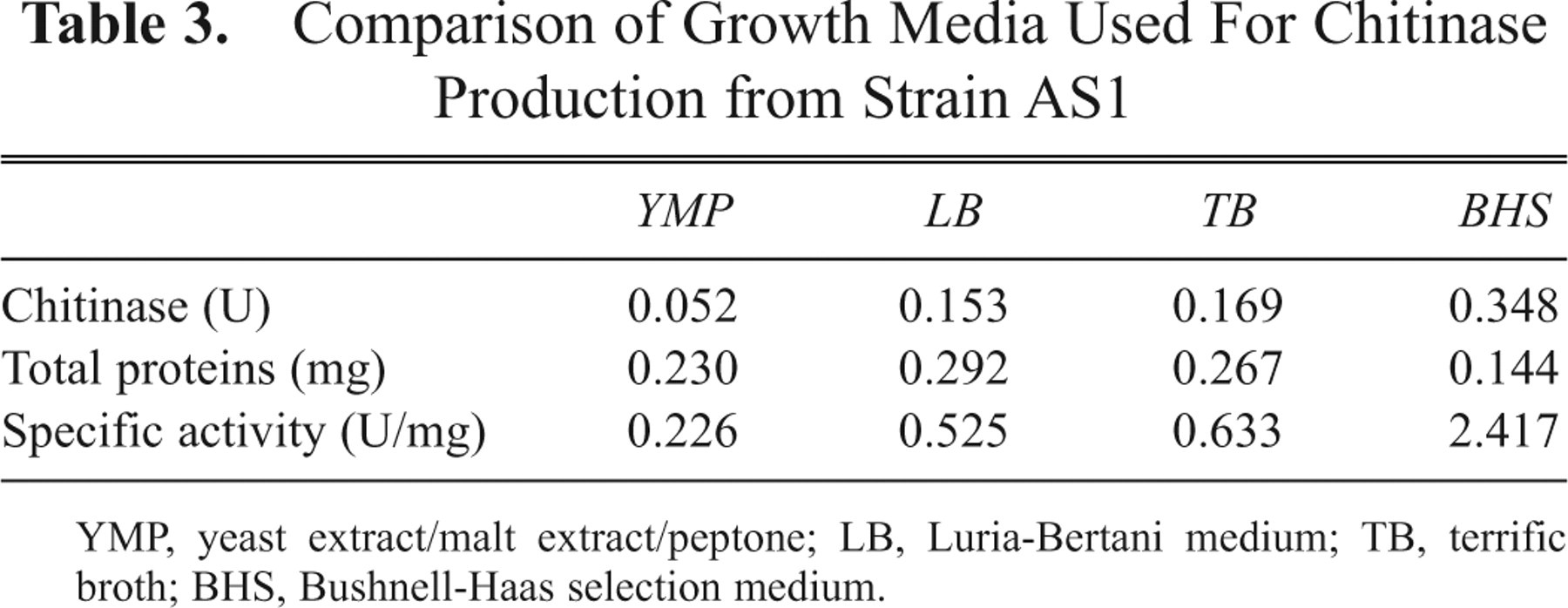
YMP, yeast extract/malt extract/peptone; LB, Luria-Bertani medium; TB, terrific broth; BHS, Bushnell-Haas selection medium.
Conclusion
In conclusion, we have developed an efficient and low-cost chitinase assay using CCA. This assay can not only detect relatively low levels of chitinase but also takes only 30 min. The assay can be applied to identify chitinolytic bacteria and appropriate culture media. The CCA substrate is a sensitive, practicable, feasible, reproducible, and low-cost material for assay of chitinase and has industrial applications.
Footnotes
Acknowledgements
We thank Dr. Pei-Tzu Wu for paper editing. Also, English Manager Science Editing is also acknowledged for the careful reading of the manuscript.
This work was financially supported by National Science Council grant 94-2214-E-131-001 to C. L. Liu, as well as 98-2320-B-182-014-MY3 and the Chang Gung Memorial Hospital Grant CMRPD150382 to C. R. Shen.
