Abstract
Background:
Boron neutron capture therapy (BNCT) is a precision binary radiotherapy. In this modality, thermal neutrons combine with 10B to induce a nuclear reaction that kills tumor cells. Its therapeutic efficacy depends on the targeted accumulation of boron delivery agents. BNCT has demonstrated clinical efficacy in treating head and neck cancers and recurrent gliomas. However, there is limited evidence regarding its application in hepatocellular carcinoma (HCC). This review systematically examines recent advances in novel boron carriers. It also assesses the potential of BNCT for treating HCC and aims to provide a new therapeutic option for HCC.
Methods:
We used a systematic research approach to investigate the latest advances in novel boron carrier development. We also analyzed existing clinical data related to BNCT treatment for HCC. We aimed to systematically assess the feasibility and potential of applying BNCT to HCC.
Results:
Newly designed boron carriers show significantly enhanced targeted aggregation within tumor cells and reduced systemic toxicity compared to traditional carriers. Preliminary clinical studies have confirmed the potential efficacy of BNCT in inhibiting HCC growth. Notably, BNCT possesses the unique advantage of precise tumor targeting, which shows promising potential in minimizing damage to surrounding normal liver tissue.
Conclusion:
The progress made in the development of boron carriers has built a solid foundation for improving the efficacy and safety of BNCT treatment for HCC. By addressing current limitations in boron delivery and clinical evidence, BNCT has the potential to complement existing treatment modalities, improve outcomes for HCC patients, and provide new directions for the clinical treatment of HCC.
Boron neutron capture therapy (BNCT) is a dual target-guided particle radiotherapy technique at the cellular level. By selectively generating a high-dose gradient between tumor and normal tissue, it achieves significant clinical efficacy after single or split treatments. Its efficacy depends on the specific accumulation of boron carriers in the tumor tissue. Early low-molecular weight boron compounds (first-generation boron carriers), reported by Albert Soloway in 1964, 1 faced challenges in clinical translation due to insufficient tumor-targeting efficiency. The second generation of boron carriers, represented by 4-dihydroxyborylphenylalanine (BPA) and sodium borocaptate (BSH), has significantly enhanced tumor specificity and has been successfully applied in the treatment of recurrent head and neck cancers and gliomas with remarkable clinical efficacy. Due to its high linear energy transfer (LET) characteristics, bio-targeting advantages, and potential application at complex anatomical sites, BNCT has recently expanded its applicability to solid tumors such as hepatocellular carcinoma (HCC). As a structural analog of phenylalanine, BPA utilizes the high-affinity active transport mechanism of L-type amino acid transporter 1 (LAT1) to achieve selective accumulation in LAT1 overexpressing HCC. This is closely related to the abnormal affective amino acid metabolism of HCC that drives the upregulation of LAT1. 2 Given the positive correlation between intratumoral boron concentration and BNCT efficacy, the development of novel boron carriers has become a focus of preclinical research. In this article, the authors systematically review the recent advances in boron carriers for BNCT and provide an in-depth discussion of their potential application in the precision treatment of HCC.
Principles of BNCT
BNCT is a biologically targeted binary radiation therapy modality whose therapeutic advantages derive from the following core features: precise spatial selectivity, dual-targeting mechanism (chemo-targeting + radio targeting), single-session treatment regimen, and radioprotective effect on normal tissues. 3 The mechanism of BNCT involves two sequential phases as follows: first, tumor-specific accumulation of the 10B by boron carriers, followed by thermal neutron irradiation to trigger a nuclear reaction (10B + 1n → 7Li + 4He [α] + 2.79 MeV), generating LET α particles and lithium recoil nuclei. These secondary particles show a limited killing range (5–10 μm) in biological tissues, similar to the diameter of a single cell 4 (Fig. 1). Tumor cell DNA double-strand breaks can be caused while maximizing the protection of adjacent normal tissues.
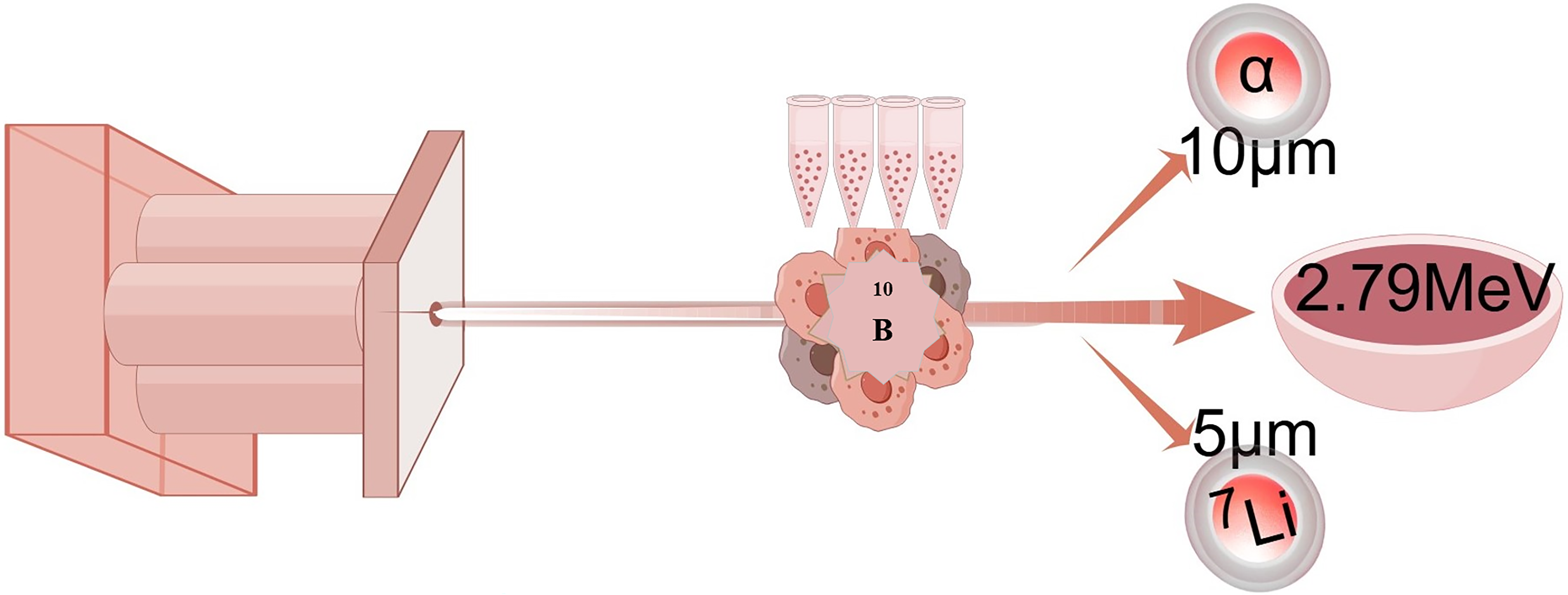
Boron delivery agents accumulate in the tumor tissue. After the tumor is irradiated with a neutron beam, a nuclear reaction occurs between the neutrons and the boron agent.
Research Progress in Boron Delivery Carriers
First-generation boron carriers
Early boron delivery agents primarily included sodium tetraborate, boric acid, and their derivatives. These agents exhibited poor tumor-targeting and short intracellular retention times, resulting in limited clinical adoption. Currently, BPA and BSH (Fig. 2) are the main agents used clinically. However, BPA faces challenges such as poor water solubility and low boron loading. BSH is another clinically approved BNCT agent that contains multiple boron atoms in its structure and exists as a sodium salt, which improves water solubility. However, due to the lack of tumor-targeting functional molecules, the tumor accumulation efficiency of BSH is not satisfactory. 5 Therefore, there is an urgent need to develop new boron carriers with higher tumor specificity, lower systemic toxicity, and higher boron-loading capacity for BNCT applications.
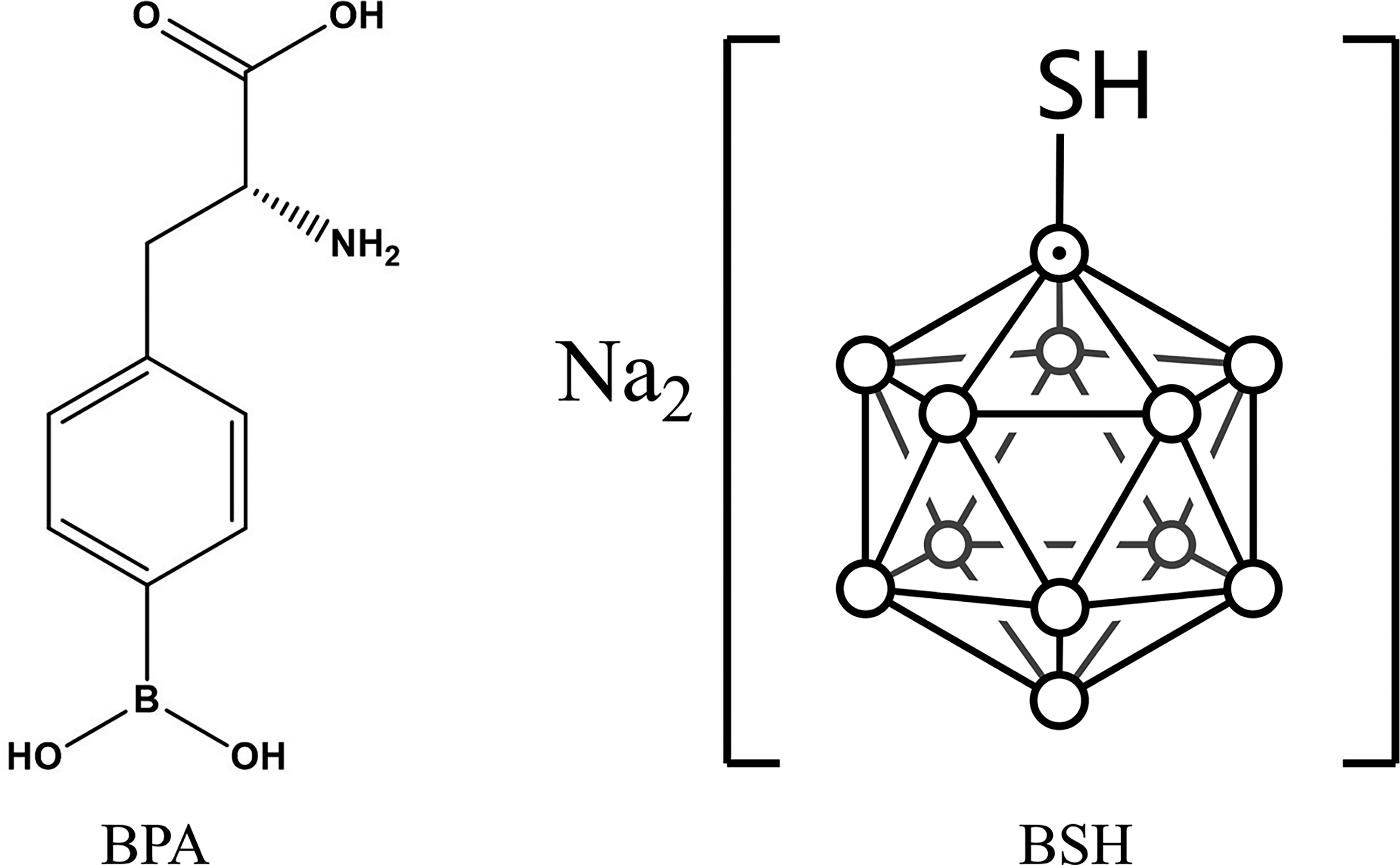
The molecular structures of BPA and BSH. BPA, 4-dihydroxyborylphenylalanine; BSH, sodium borocaptate.
BPA/BSH derivatives
BPA-fructose complex resolves the water solubility limitation of BPA through sugar coordination. On this basis, Takahashi et al. 6 developed a fructose-modified block copolymer-BPA conjugate system. Its cationic nature not only accelerates renal excretion (reducing systemic toxicity) but also enhances tumor-targeted accumulation through charge-mediated endocytosis. To overcome the boron-loading limitations of conventional BSH, carbaborane scaffolded with polycyclic boranes has high boron density (>10 boron atoms/molecule) and adjustable hydrophobicity. 7 Researchers integrate aggregation-induced emission motifs into “diagnostic-integrated” boron carriers, 8 which enable boron dose monitoring and tumor-specific delivery. Kawasaki et al. 9 further designed a carbon–borane–extracellular vesicle hybrid system (CB@EVs) through the host–guest interaction between hydrophobic carbon–borane complexes and extracellular vesicles. In a colorectal cancer model, CB@EVs showed a higher tumor-to-normal tissue uptake ratio (T/N = 420) and tumor-to-blood ratio (T/B = 63), and it was 3-fold more efficacious than the BPA-fructose complex for BNCT, highlighting the effectiveness of the synergistic delivery strategy.
Amino acid-based boron carriers
Boronated amino acid derivatives, as novel small-molecule boron carriers, enable tumor-targeted delivery through the amino acid metabolic pathway. Although the natural amino acid derivative BPA has clinical utility, it is rapidly cleared from the body, limiting therapeutic effectiveness. Raitano et al.
10
developed the unnatural amino acid 3-boron-based-
Boron-containing DNA-binding molecules
Boronated nucleotides are highly promising targeted boron delivery systems by virtue of their molecular recognition properties with genetic material. These compounds are converted to 5′-monophosphate derivatives catalyzed by phosphorylase enzymes, and their negatively charged phosphate groups are selectively retained across tumor cell membrane potential gradients. 16 Wang et al. 17 synthesized boronated nucleotide esters through thermally induced esterification reaction, which significantly enhanced their water solubility and tumor selectivity. Różycka’s team 18 designed acridine polymers functionalized with carbon boranes that induce S-phase blockade and activate reactive oxygen species storms in HeLa cells, exerting a dual cytotoxicity mechanism by changing the cellular DNA topology. To address the toxicity limitations of acridine derivatives, Druzina et al. 19 developed low-toxicity 9-methoxyacridine-N3CH2CH2NH2 conjugates by combining a DNA insertion agent (to enhance nuclear localization) with the high boron density of borane. In vitro, mixing it with calf thymus DNA demonstrated its DNA-binding ability and optimized the “localization-loading” efficiency. Notably, due to the fact that nucleosides are affected by multiple carrier protein transporters and deoxyribonuclease degradation pathways in vivo, the uptake of delivery agents exhibits uncertainty and differences. 20
Boron-containing porphyrin delivery agents
Boron-containing porphyrin compounds have emerged as ideal candidates for diagnostic-integrated boron carriers due to their tumor-selective accumulation characteristics and inherent fluorescence imaging capabilities. Although the early development of borated derivatives of tetraphenylporphyrin showed targeting potential, they had problems with systemic toxicity, such as the risk of hemolysis. Shi’s team 21 addressed these limitations by constructing a carbaborane-tetraboronated porphyrin micellar system. This system enables tumor-targeted delivery through the enhanced permeation retention effect. It significantly increased drug loading while reducing hematological toxicity to clinical safety thresholds. Hiramatsu et al. 22 reported a carbon-boronated chlorin derivative with excellent tumor affinity, low cytotoxicity under dark conditions, and dual functionality of 650 nm near-infrared fluorescence imaging capability and single-linear-state oxygen quantum yield, which enables intraoperative fluorescence navigation and temporal and spatial synergies between BNCT and photodynamic therapy. Notably, metal-chelated porphyrins such as ZnTCPH and CuTCBr incorporate diagnostic isotopes (64Cu/76Br).23,24 Their half-life was extended to 12.7 and 16.2 h, respectively, overcoming the limitations of the short half-life of 18F (insufficient for monitoring drug metabolism), while retaining the biodistribution characteristics of the host compound. Current challenges in preclinical studies focus on porphyrin-associated skin phototoxicity responses and mitochondrial dysfunction, 21 which need to be either optimized by rational drug design or subcellular organelle-targeting strategies.
Nanoparticle-based targeted delivery systems
Nanoparticles (NPs) provide a revolutionary solution for BNCT delivery systems due to their high drug-carrying capacity and diverse surface modification capabilities. Tang’s team 25 developed SP94 peptide-targeted boronic acid-functionalized mesoporous silica NPs, achieving HCC targeting by the 78 kDa glucose-regulated protein receptor. In tumor-bearing mice, the tumor boron concentration reached 40.18 ± 5.41 mg/kg at 4 h after injection, which was 8-fold higher than that in the BPA group. Wang et al. 26 designed boronated metal-organic frameworks, which were verified by Monte Carlo simulations to have enhanced neutron absorbed dose with a relative biological effectiveness (RBE) of 6.78, which is a 4.1-fold improvement compared with boric acid. To address the challenge of cyclic stability of NPs, Feine et al. 27 innovated a pretargeting strategy. Tumor-targeting antibodies modified with trans-cyclooctene were first injected, followed by precise targeting of tetrazine-functionalized NPs mediated by bio-orthogonal click chemistry, achieving reduced off-target rates.
Liposomal nanosystems exhibit unique advantages due to their bionic membrane structure. The thermosensitive liposomes developed by the Luderer team 28 remain stable at physiological temperatures (37°C), but trigger drug release in the tumor microenvironment (41°C–43°C), enabling selective enrichment of boron by localized thermal therapy. The Kawasaki team 29 constructed HER-2-targeted immunoliposomes by the host–guest exchange technique that showed 14-fold higher BNCT activity than BPA in SK-OV3 cells. Li et al. 30 developed carbaborane–phospholipid hybrid liposomes that enhanced membrane stability through hydrophobic interactions and increased drug retention time up to 40 h. Shirakawa et al. 31 significantly enhanced tumor boron enrichment by covalently anchoring BSH to the surface of polyethylene glycolated liposomes using surface engineering techniques. Despite these advances, NP delivery systems still face serious physicochemical stability challenges as follows: the risk of dissociation of metal-organic framework NPs in acidic microenvironments, the stability of antibody-coupled NPs, and the difficulty of maintaining the membrane integrity of liposomes under cyclic shear stress. 32
In Vivo Distribution Monitoring of Boron Delivery Agents
In BNCT, accurate monitoring of boron delivery agents is critical for optimizing treatment time windows, dose calculations, and efficacy assessments. In terms of fluorescence-based tracer technology, Fukuda et al. 33 developed an aryl boronic acid-specific fluorescent labeling technique by Suzuki coupling reaction. The palladium-catalyzed cross-coupling was used to generate cyanodiphenyl derivatives that enabled real-time detection of BPA in fingertip blood samples. Kondo’s team 34 designed a small-molecule fluorescent probe, which reacts specifically with BPA in a borate esterification reaction to achieve quantitative detection in less than 1 min by fluorescence bursting and emits a distinctive red fluorescence at 631 nm. In terms of imaging monitoring, although 18F-BPA PET (4-borono-2-[18F]fluoro-L-phenylalanine, positron emission tomography) remains the clinical gold standard, its spatial definition limits the accuracy of brain tumor target area outlining. After Grunewald et al. 35 demonstrated that 18F-BPA is bioequivalent to its host compound, the Lo team 36 innovatively applied zero-echo time magnetic resonance imaging (MRI) sequences for decay correction to reduce quantitative errors in standardized uptake measurements of gliomas by PET-MRI. To satisfy multimodal imaging needs, Okada et al. 37 developed a gadolinium–boron bimodal albumin coupling, whose MRI signal intensity was strongly and positively correlated with boron concentration measured by inductively coupled plasma photoemission spectroscopy. In terms of breakthroughs in real-time monitoring technology, Silarski et al. 38 designed a transient γ analysis system based on lanthanum bromide scintillators, achieving a boron detection sensitivity of 10 ppm. The Murata team 39 has elevated single photon emission computed tomography technology to clinical standards, achieving 5% quantitative accuracy and 5 mm spatial resolution. Current technological barriers include in vivo metabolic interference with fluorescent probes, spatiotemporal alignment errors in multimodal imaging, and clinical translational validation of real-time monitoring systems.
Optimization of Boron Delivery Agent Administration Modes
In BNCT, it is crucial to optimize the route of administration of the boron delivery system to overcome efficacy limitations. To address the low T/N ratio (<4:1) and systemic toxicity of conventional intravenous (i.v.) delivery, 40 researchers have developed innovative strategies from multiple dimensions. In terms of pharmacodynamic synergy, Fukumura et al. 41 found in HGG13 glioblastoma-bearing mice that pretreatment with 5-aminolevulinic acid administered orally 24 h before BPA administration and neutron irradiation significantly increased intratumor boron concentrations, T/B ratios, and improved survival compared with BPA-BNCT alone. In terms of physically assisted drug delivery, Yura’s team 42 pioneered ultrasound-responsive microbubble targeted cavitation technology. Triggered by high-intensity ultrasound, the BPA-containing lipid microbubbles produce instantaneous vascular permeability through the cavitation effect, resulting in 2- to 3-fold uptake of tumor boron compared with i.v. drug delivery. Olaiz et al. 43 applied electroporation to deliver decahydrodecaborate, which enhanced tumor delivery by improving the electrical conductivity of tumor cell membranes in a hamster oral cancer model. In terms of precise local delivery, Barth et al. 44 utilized an intra-arterial catheterization technique to create a blood boron concentration gradient in gliomas, prolonging median survival to 30 days. The Tsujino team 45 used convection-enhanced delivery technology for continuous intracranial boron infusion, bypassing the blood–brain barrier and maintaining tumor boron concentrations for more than 24 h. Kusaka et al. 46 developed an intrathecal delivery method that achieved a peak T/N ratio of 4.4:1. Current technological challenges are focused on the following areas: ensure long-term biocompatibility of the local delivery system, overcome the limited penetration depth of physically assisted modalities in deep tissues, and enable time-synchronized control of multimodal coadministration. Future developments will require the exploitation of tumor microenvironment-responsive smart drug release platforms to achieve four-dimensional (spatial and temporal) precision in boron delivery to optimize therapeutic efficacy while minimizing off-target effects.
Therapeutic Potential of BNCT in Liver Cancer
Liver cancer is the sixth most common malignant tumor worldwide (annual incidence >900,000 cases) and the third leading cause of cancer-related deaths, causing major challenges in clinical treatment. From a pathological point of view, primary liver cancer consists mainly of HCC (75%–85%), intrahepatic cholangiocarcinoma (10%–15%), and rare subtypes such as combined hepatocellular–biliary carcinoma. 47 Due to the lack of specific biomarkers for early detection, approximately 60% of patients are diagnosed at an advanced stage (BCLC stage C) and cannot receive radical treatment. Currently, systemic therapy for advanced liver cancer is based on immune checkpoint inhibitors (Programmed Cell Death Protein 1 Inhibitor) combined with tyrosine kinase inhibitors (lenvatinib), but the objective reductions rate of these regimens is only 30%−40%, and the median progression-free survival is 6.2–7.4 months. 48 From the radiobiological point of view, HCC exhibits moderate radiosensitivity (α/β ratio ≈10 Gy), higher than pancreatic cancer (α/β ≈ 3 Gy), but requires ≥60 Gy biologically effective dose for radical treatment. However, the risk of radiological liver disease (RILD; incidence >15%) in the livers of cirrhotic patients severely limits the use of conventional radiotherapy. BNCT has a stronger DNA double-strand break effect (several folds higher than photons) and dual-targeting precision (boron localization and neutron activation), which can kill tumor cells while protecting normal tissues. This selective radiobiological advantage provides an opportunity for patients with Child–Pugh class B cirrhosis or multifocal HCC (populations contraindicated to conventional radiotherapy). Currently, BNCT has shown significant efficacy for all types of HCC (Table 1). In the future, BNCT combined with surgery or chemotherapy will likely further improve patient prognosis (Fig. 3).
Conversion therapy for patients with liver cancer.
Boron Neutron Capture Therapy Has Shown Significant Efficacy for All Types of Hepatocellular Carcinoma

BPA, 4-dihydroxyborylphenylalanine; BSH, sodium borocaptate; i.p., intraperitoneal administration; i.v., intravenous administration.
Locally advanced HCC
High LET radiation, with its superior RBE >2.8, effectively overcomes tumor radioresistance mechanisms. Huang et al. 54 established a radioresistant HCC cell model (HepG2-R) by γ-radiation gradient induction and demonstrated that the survival fraction of BNCT-treated cells was significantly lower compared with the conventional radiotherapy group. The expression of γ-H2AX, a biomarker of DNA double-strand breaks, was 9-fold higher in BNCT-treated cells than in γ-irradiated control cells. The Suzuki team 49 reported the first clinical case of BNCT for multifocal HCC in a 60-year-old man (Child–Pugh class B) who received transarterial chemoembolization for the left liver tumor and BNCT for the right liver tumor. After 3 months, computed tomography assessment showed complete remission in the BNCT-irradiated area and no grade ≥3 treatment-related adverse events. Although the patient ultimately suffered from liver failure due to the development of a new tumor (overall survival 10 months), this case validates the feasibility and efficacy of BNCT for locally advanced HCC.
Locally recurrent HCC
For recurrent HCC with RILD risk limitations, BNCT shows a unique dose-related advantage. The spatially selective secondary radiation generated by the neutron capture reaction enables precise dose deposition through the tumor/normal tissue boron concentration gradient, effectively controlling the radiation absorption in healthy liver tissue. Yanagie et al. 50 reported a typical case of a patient with right liver resection combined with Child–Pugh class B cirrhosis who developed new metastases after eight cycles of chemotherapy. BNCT was given on day 5 after infusion of 10B iodinated emulsion through a super selective arterial catheter intervention. At 3-month follow-up after treatment, CT showed stabilization of the target lesion and no worsening of liver function.
HCC metastatic lesions
In the field of oligo-metastatic liver cancer treatment, BNCT shows unique advantages due to its spatial dose precision. To address the challenge of hepatic target displacement caused by respiratory motion (with an average amplitude of 15–20 mm), its “boron-mediated, neutron-triggered” dual-targeting mechanism enables dynamic dose conformality. Zonta teams 51 pioneered the use of in vitro liver perfusion neutron irradiation technology to accomplish metastatic irradiation while maintaining liver activity in an out-of-body circulatory system (with boron concentrations up to 30 μg/g), with fluctuating and stable postoperative liver function indices (alanine/aspartate aminotransferase). In a mouse model of HCC metastases, Pavanetti et al. 55 showed that polyethylene glycol (PEG)-modified boron-containing liposomes achieve therapeutic boron accumulation (>30 μg/g) in liver metastases and that PEGylated liposomes promote metastatic recession by enhancing tumor-specific boron delivery. Garabalino and Pozzi teams52,53 conducted dosimetric validation in a rat model of colon cancer (DHD/K12/TRb) liver metastases, confirming that multiple routes of administration delivered BNCT therapeutic doses to the tumor through differential dose calculations for normal liver tissue and metastases. Significant reduction in metastatic tumor size without liver toxicity was observed. Cardoso et al. 56 in a model of partial liver resection-induced liver regeneration found that BNCT irradiation did not affect regeneration, function, or proliferation of the normal liver. The distant effect of BNCT provides a new direction for the treatment of liver cancer metastases, and Trivillin’s team 57 observed a significant reduction in the size of tumors in the left hind limb following BNCT treatment of right hind limb tumors in a rat colon cancer xenograft model, suggesting that BNCT activates a systemic antitumor immune response.
Challenges of BNCT in the Treatment of HCC
Although BNCT has shown promise in preclinical studies for the treatment of liver cancer, its clinical translation still faces three core challenges. (1) Limited availability of neutron sources: Nuclear reactors, the main neutron source for BNCT, are being phased out by many research centers due to operational constraints and uncontrollable lifetimes. While new emerging accelerator neutron sources (cyclotrons, radiofrequency quadrupole accelerators, and electrostatic accelerators)58,59 have advantages in controllability and ease of operation, clinical applications still require the development of a high-throughput compact accelerator. (2) Safety and pharmacokinetic drawbacks of boron delivery agents: Currently used agents (BPA, BSH) have insufficient tumor uptake in HCC (T/N ratios are typically <4:1), requiring higher doses but potentially increasing the risk of liver toxicity. The development of targeted boron delivery agents for HCC biomarkers is urgent. (3) Lack of standardized segmented regimens: There is a lack of standardized segmented BNCT regimens to reduce liver radiation damage. Although the current clinical data show that BNCT is well tolerated and has low acute and chronic toxicity, the reported adverse effects (skin toxicity and liver fibrosis) 60 suggest the need for further clarification of the mechanisms of liver cancer-specific toxicity and for the establishment of a multimodal toxicity monitoring framework to enhance treatment safety.
Conclusions
Driven by technological improvements in accelerator-based neutron sources, the clinical translation of BNCT has overcome the limitations of conventional reactors in terms of radiation protection and facility accessibility, enabling patient precision radiotherapy. Novel targeted boron delivery agents (small molecules, large molecules, and NPs) based on BPA and BSH expand the clinical indications for BNCT. Research on boron visualization and multiple routes of delivery is aimed at diagnostic–therapeutic integration and optimization of treatment efficacy. For patients with locally advanced or recurrent liver cancer who are unable to undergo surgical resection due to liver insufficiency or comorbidities, BNCT offers a promising local–regional treatment option that may improve survival outcomes and quality of life. Although current evidence supports the feasibility and safety of BNCT in this population, further studies are needed to maximize clinical efficacy and to assess whether BNCT combination therapy improves survival.
Authors’ Contributions
Conceptualization: T.Z., H.Z., and J.R.; formal analysis: Z.Z. and J.R.; investigation: T.Z., Z.Z., and J.R.; methodology: P.Z. and J.R.; project administration: J.R.; resources: P.Z. and J.R.; supervision: H.Z. and J.R.; writing—original draft: T.Z., H.Z., and Z.Z.; and writing—review and editing: P.Z. and J.R.
Footnotes
Funding Information
This work was supported by the National Natural Science Foundation of China (82360583); Gansu Provincial Science and Natural Science Foundation of Gansu Province of China (23JRRA0952); Lanzhou University Medical Research Innovation Capacity Enhancement Program (lzuyxcx-2022-159); Gansu Province Major Special Project (ldyyyn2023-6); and Gansu Provincial Science and Technology Major Program (25JRRA1244).
Disclosure Statement
There are no existing financial conflicts.
