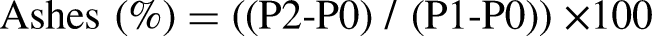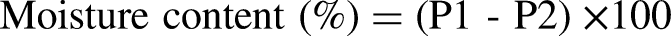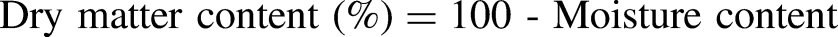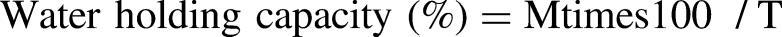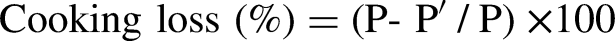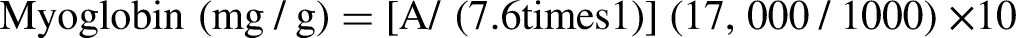Abstract
The effect of lairage time on animal stress and its impact on meat quality were evaluated on 14 male dromedaries of the Sahraoui population, raised in the El-Oued region, aged from 12 to 18 months. The animals were divided into two groups according to the lairage time: from 1 to 8 h (G01) and for 48 to 96 h (G02). At the end of the holding period, physiological stress parameters were measured (heart rate, rectal temperature, cortisol, creatine phosphokinase, and glucose levels). Longissimus Lumborum muscle taken at 24 h postmortem was used to assess meat quality parameters (pH, moisture, dry matter and mineral content, water-holding capacity, cooking loss, myoglobin content, myofibrillar fragmentation index and the electrophoretic profiles of myofibrillar and sarcoplasmic proteins). Only cortisol level and pH showed a significant difference between the two groups. Negative correlations were observed between cortisol and both pH and cooking loss. Principal component analysis showed that animals subjected to short-time lairage were characterized by high cortisol and plasma glucose levels and meat with low pH, mineral content, and moisture. The data suggest that controlling the lairage period in terms of duration and conditions is essential for ensuring consistent and high-quality dromedary meat.
INTRODUCTION
The camel enjoys an exceptional status, considered a valuable resource, an asset, or even capital for breeders and their families, revealing significant prestige, while its possession provides the ability to survive due to its resilience and great mobility. Indeed, the extraordinary range of products and services provided by this animal, including meat, milk, and wool for weaving, skin and dung for processing, makes it unique in the industry (Senoussi et al., 2024).
The dromedary species present in Algeria is the Camelus dromedarius, with an estimated population of 416,519 head in 2019, ranking Algeria 14th in the world (Smili et al., 2022a). This species has been described for centuries as having a high productive potential, used by populations (especially nomads) in arid and hot regions as versatile animals for physical labor, transport, milk, meat, wool, hair and skin production, as well as for racing and tourism (Faye et al., 2022).
Compared with other red meats, camel meat stands out for its low fat and cholesterol content, which makes it a beneficial choice for human health and has led researchers to take a much greater interest in improving its quality parameters (Tabite et al., 2018). Indeed, the quality of camel meat depends on factors both intrinsic and extrinsic to the animal. These include breed, sex, age, rearing system, diet, preslaughter handling and postslaughter treatment, all of which affect the chemical composition and quality attributes of this meat (Kadim et al., 2008; Smili et al., 2022a, 2022b; Suliman et al., 2019).
Livestock intended for meat production are often subjected to stressful conditions prior to slaughter. These conditions include stocking densities, handling, loading and unloading, transport time and distance, passage through livestock markets, fasting and lairage, all of which affect the animal welfare and meat quality (Barka et al., 2016). These events trigger physical, physiological, and behavioral reactions in the animals, which are used as biomarkers to assess their stress states through numerous indicators considered important and reliable using physiological biomarkers (cardiac and respiratory rhythms, rectal temperature), hematological (hemolysis, neutrophil/lymphocyte ratio), biochemical and hormonal analyses (enzymes, cortisol, thyroid hormones, glycolytic potential, proteins, oxidative stress, lipid peroxidation, and tissue damage) (Broom, 2014; Kumar et al., 2023; García-Torres et al., 2021). Transport and handling stress, as well as waiting at the slaughterhouse, induce an increase in cortisol levels, as well as creatine kinase and lactate in muscle (Kannan et al., 2000).
Preslaughter stress with high cortisol levels deteriorates meat quality and consumer acceptance due to increased pH, changes in tenderness and color (Terlouw and Bourguet, 2022). A low muscle glycogen content at slaughter results in a high final pH of the meat due to a lack of sufficient glycogen to produce enough lactic acid/net H + ions during postmortem anaerobic glycolysis in the absence of blood circulation (Matarneh et al., 2023). A slight deviation from the normal situation in postmortem pH drop and temperature could have a negative effect on the various physicochemical parameters (such as water holding capacity, cooking yield, cooking loss, exudation loss) and organoleptic attributes of processed meat products. Time spent in lairage at the slaughterhouse is the resting time that allows animals to recover from transport stress and restore their energy reserves (Kumar et al., 2023).
The aim of the study was to explore the effect of lairage time after transport on dromedary camel physiological parameters and meat quality.
MATERIALS AND METHODS
Animals and sampling
Our study covered 14 male dromedaries “Camelus dromedarius” from the Sahraoui population, aged from 12 to 18 months, with an average live weight of 125 ± 7 kg, clinically healthy. The animals were raised under the same conditions on a farm in the Hassi Khalifa commune, 30 km from the abattoir. Rearing was semi-intensive, based on roughage (hay and straw) and concentrated feed (crushed barley, bran, and date waste).
The slaughter took place between October and November at the municipal abattoir of Oued Souf in south-eastern Algeria (33°22’ 00"N; 6°53'23"E). On arrival at the abattoir, the animals were guided to an open-air stabling area, isolated from noise and human activity. Two groups (G) were formed according to lairage time. The first group (G01) was stabled between 1 and 8 h (5h17 min ± 2.8; n = 6) and had no access to feed. However, the second group (G02) was held in lairage for 48 to 96 h (66 h ± 21.27; n = 8) and had access to water and feed until the day before slaughter. This sampling is a part of the real-life practices, since the dromedaries were destined to be sold in the local market and they were submitted to the usual preslaughter practices.
To assess the effect of lairage time on physiological parameters and dromedary meat quality, blood, and muscle samples were taken.
While the animal was held in lairage, before being loaded for slaughter, blood samples were taken by puncturing the jugular vein. The blood was transferred to dry EDTA tubes, stored in a cooler and sent immediately to the laboratory for analysis.
The slaughtering operation was carried out according to the Halal procedure in compliance with Algerian legislation. At 24 h postmortem, around 450 g of Longissimus Lumborum muscle was removed from the last lumbar vertebra of each carcass. Samples were transported to the laboratory in a cooler. After removal of fat and connective tissue, samples were packed in plastic bags and stored at 4 °C for 24 h, then transferred to −20 °C until analysis.
Physiological parameters measured
Physiological parameters measured on the animal include heart rate (HR) and body rectal temperature (TR), and on the blood sample were blood glucose (Glu), cortisol (COR), and creatine phosphokinase (CPK).
Plasma glucose measurement
Plasma glucose levels were measured using a Vital Check glucometer (Vital Check ®—MM 1200, Korea) with disposable strips.
Measurement of serum cortisol
Serum cortisol levels were quantitatively determined on the VITROS 5600 system using Intellicheck technology with the aid of a reagent cartridge and VITROS Cortisol calibration samples on the VITROS ECi/ECIQ and 3600 immunodiagnostic systems. The assay is based on an immunometric technique involving competition between cortisol present in the sample and a cortisol conjugate labeled with horseradish peroxidase (HRP) for a limited number of binding sites on a biotinylated antibody (sheep anticortisol antibody).
Measurement of creatine phosphokinase
Creatine phosphokinase was measured by the plate assay method, involving VITROS CPK plates and VITROS Chemistry products calibrator kit 3 calibration samples on the VITROS 250/350/950/5.1 FS and 4600 clinical chemistry systems, and on the VITROS 5600 integrated system.
Heart rate measurement
After restraint, HR per minute was measured by auscultation of the heart region using a stethoscope.
Body temperature measurement
Body temperature was determined rectally, using a Boso digital clinical thermometer (Bosotherm basic-D-72417, Germany). The thermometer was gently inserted into the animal's rectum until a reading was obtained.
Meat quality parameters
pH measurement
pH was determined according to the protocol described by McGeehin et al. (2001). One gram of muscle was sampled and homogenized on the polytron (Polytron ® PT- MR 2100, Kinematica AG, Switzerland) for 15 s with 10 mL of sodium iodoacetate (5 mM) and potassium chloride (150 mM) buffer adjusted to pH 7.0. The resulting homogenate was measured using a portable pH meter, model 616.01.001 microprocessor-based water proof pH/mV/Temp tester (ISOLAB, laborgerӓte GmbH, Germany).
Determination of ashes
Ashes were determined according to the protocol of Marra et al. (1999) and Komprda et al. (2012). One gram of ground muscle was incinerated in the muffle furnace (LINN-HightThern) at 550 °C for 6 h. Crucibles were transferred to a desiccator and weighed using a KERN analytical balance (ALS 160, Germany).
Moisture content and dry matter
Muscle moisture was analyzed according to the standard methods of the Association of Official Analytical Chemists (AOAC, 2000). Two grams of muscle were weighed on a precision balance, then placed in petri dishes and placed in an oven set at 105 °C for 24 h. After 24 h, the petri dishes were placed in a desiccator for at least half an hour to cool in a dry atmosphere. Finally, the petri dishes were weighed.
Determination of water holding capacity
Water holding capacity was assessed by the Pressure Method on filter paper according to the protocol of Grau and Hamm (1953) modified by Hafid et al. (2016). In this method, 300 ± 5 mg of intact meat was weighed and then placed on 11 cm diameter Wathmann n°1 filter paper which had been previously dried and weighed. The samples were then placed between two glass plates, to which a weight of 2.25 kg was applied for 5 min. Circles representing the meat (M) and relegated juice (T) were then carefully traced on a transparent plastic sheet. The surface areas of the meat (M) and released liquid (T) spots were measured using Image J 1.48 software.
Determination of cooking loss
The protocol of Pascual and Pla (2007) was used to determine the percentage of water lost during cooking. To do this, 10 g of each muscle was taken (P), packed in plastic cooking bags, then frozen at −20 °C. Before analysis, the muscles were thawed in the refrigerator for 24 h, then placed in a water bath thermostated at 80 °C for 1 h. After cooking, the muscles were wiped dry with absorbent paper, then reweighed (Pʹ). Cooking water losses, expressed as a percentage (%), were calculated according to the formula:
Myoglobin content
The protocol adopted by Canto et al. (2015) was used to determine myoglobin content. One gram of muscle was homogenized in 10 mL of sodium phosphate buffer (pH = 6.8). The resulting homogenate was filtered with Wattman paper in the dark to avoid oxidation. The absorbance of the filtrate was measured at a wavelength of 525 nm with a spectrophotometer, using sodium phosphate buffer as a blank. Myoglobin concentration was calculated using the following formula:
Myofibrillar fragmentation index
The degree of myofibrillar fragmentation was estimated according to the protocol of Culler et al. (1978) modified by Li et al. (2012). Two grams muscle sample was taken, weighed, then homogenized for 25 s using a polytron (Polytron ® PT- MR 2100, Kinematica AG, Switzerland) in 10 mL extraction buffer (RB) (75 mM KCl, 10 mM KH2PO4, 2 mM MgCl2, 2 mM EGTA, 1 mM NaN3, at pH 7.0). Next, two consecutive centrifugations (Centrifuge NF 400R, NUVE-Turkey) were performed at 2000 rpm for 15 min at 4 °C. During the first centrifugation, the supernatant liquid was removed and the precipitate recovered, then solubilized in 5 to 10 mL of RB. For the second centrifugation, the precipitate was solubilized in 5 to 10 mL of RB, then filtered through filter paper to remove fat and connective tissue. Finally, the myofibrillar fragmentation index, expressed as a percentage, was determined by measuring the absorbance of the myofibrillar protein suspension at 540 nm spectrophotometrically (Spectrophotometer 7305, Jenway-Bibby scientefic Ltd -UK) and multiplying this value by 200.
Electrophoretic analysis of proteins (SDS-PAGE)
A 0.5 g of muscle sample was added to 10 mL extraction buffer (RB) containing 75 mM KCl, 10 mM KH2PO4, 2 mM MgCl2, 2 mM EGTA, 1 mM NaN3 at pH 7.0, then homogenized for 15 s on the polytron. The homogenate was filtered through gauze, then centrifuged at 4000 rpm for 15 min at 4 °C. The supernatant containing sarcoplasmic proteins (SP) was collected in Eppendorf tubes. The pellet containing myofibrillar proteins (MP) was solubilized with 2.5 mL RB, filtered through filter paper and stored in Eppendorf tubes. The Eppendorf tubes containing the protein solutions were stored at −20 °C until used for protein assay using the method of Bradford (1976) and electrophoretic analysis (Joo et al., 1999).
SDS-PAGE electrophoresis, was run according to Laemmli (1970) using 12% separation and 5% concentration gels. Samples were mixed at a ratio of 1:1 with denaturation buffer (containing Tris/HCl 312.5 mM, EDTA 0.4 mM, SDS 7.5%, glycerol 25%, DTT 150 mM and bromophenol blue 0.05%, pH 6.8) then heated for 10 min in a water bath at 95 °C to complete denaturation, 10 µg of proteins were subjected to separation for 1.5 h at 130 V. After separation, the gels were fixed and then stained overnight with a 4.9 mM Coomassie Brilliant Blue G-250 staining solution in 50% (v/v) ethanol and 7.5% (v/v) acetic acid. Protein motifs were then visualized after decanting the gel until a clear background was obtained. The molecular weight of protein bands was estimated using reference proteins of known molecular weight (MW-GF-200, Sigma), using Un-Scan-It gel 6.5 analysis software (Silk Scientific, Orem, UT).
Statistical analysis
All data were subjected to Student's t-test to determine the effect of lairage time on animal physiological parameters and meat quality. Results were expressed as mean ± standard deviation. Pearson correlation analysis was performed to reveal relationships between the various parameters studied.
Principal component analysis (PCA) was applied to a matrix of 11 variables (COR, CPK, RT, HR, Glu, pH, H%, M.N, CL, WHC, MFI) using the XLSTAT, 2016 program to study the impact of lairage time after transport on animal physiological status and meat quality. The two most significant principal components were analyzed. p < 0.05 was considered statistically significant.
RESULTS AND DISCUSSION
Dromedary physiological parameters
The effect of lairage time after transport on the physiological parameters of Sahraoui dromedaries is shown in Table 1.
Physiological parameters measured (mean ± standard deviation (SD)) according to the dromedary lairage time before slaughter, between 1 and 8 h (G01, n = 6) and for 48 to 96 h (G02, n = 8).

Significance p < 0.05, assessed by Student's t-test.
Lairage time after transport had a highly significant effect on cortisol levels (p < 0.001). Indeed, dromedaries held in lairage from 1 to 8 h had high cortisol levels (874 ± 631 ng/mL) compared to dromedaries rested for 48 to 96 h (127.40 ± 93.9 ng/mL). These values were very high compared with the values reported for dromedary by Tabite et al. (2019), Lemrhamed et al. (2018a, 2018b), and El Khasmi et al. (2015). Several studies have shown that transporting animals to slaughter is a source of stress, assessed by the increase in cortisol levels after transport compared with before transport as observed in dromedary (El Khasmi et al. 2010), sheep (Parker et al. 2003), goat (Minka and Ayo 2010), and cattle (Gupta et al. 2007). This increase is due to the presence of animals in an unfamiliar environment and situation (Terlouw et al. 2007). Cortisol is a key hormone used to assess stress in animals. It is defined as an energy regulator, selecting the type and quantity of substrate (carbohydrate, lipid, or protein) required by the organism to meet its physiological demands (Burdick et al., 2011). Its secretion into the circulation induces catabolic activity in peripheral tissues (glycogenolysis, proteolysis, and lipolysis) and anabolic activity in the liver (gluconeogenesis and protein synthesis) to increase blood glucose concentration and provide the energy needed to manage with the stressor (Chulayo et al., 2016; Čobanović et al., 2020). In general, an increase in blood cortisol levels is recorded after 15 min of exposure to stress and subsequently returns to basal levels after around 3 h (Čobanović et al., 2020; Kumar et al., 2023). Stabling cattle in low-stress conditions can help them recover from the stress and strain of transport. Waiting for at least 24 to 48 h allows cattle to return to a normal behavioral, physiological, and metabolic state (Terlouw et al., 2007). Chulayo et al. (2016) also reported that increasing lairage time from 18 to 24 h led to a reduction in cortisol levels. For sheep, a lairage time of 12 h is recommended (Terlouw et al., 2015). In dromedaries, Lemrhamed et al. (2018b) reported that stress responses were significantly attenuated after 20 h of rest. Nevertheless, the effects of waiting time at the slaughterhouse also depend on conditions (new environment, mixing with unfamiliar animals, sounds, smells, food availability, etc.) which may instead generate long-term stress in the animals (Terlouw et al., 2007).
Serum CPK showed no significant difference between the two stall groups (G01: 192.40 ± 99.6 Ul/L and G02: 142.70 ± 51.1 Ul/L). These values are higher than those reported in dromedary by Aichouni et al. (2010), which ranged from 40 to 77.42 IU/L, and lower than those reported by Hamad et al. (2018), which were 386.46 ± 12.39 Ul/L. CPK is an enzyme of energy metabolism, and is released into the bloodstream in the event of muscle injury or intense physical exercise (Chai et al., 2010). Rabbits showed lower levels of CPK when they had short-duration stalling compared with long-duration stalling (2 vs. 8 h) (Trocino et al. 2018). Typically, after transport, animals need some time to adapt to the new situation and recover from the stress caused by loading and transport (Broom and Fraser, 2015).
Plasma glucose levels showed no significant difference between the two groups (G01: 1.34 ± 0.21 mmol/L and G02: 1.25 ± 0.33 mmol/L). Previous studies reported by El Khasmi et al. (2015) and Lemrhamed et al. (2018a, 2018b) have shown that road transport, storage density as well as a lairage period of less than 16 h at the slaughterhouse, cause high levels of stress in dromedaries that can be linked to elevated plasma glucose and cortisol levels. During lairage, the animals are supposed to recover the energy lost during transport. However, the rate at which energy is recovered depends on the level of transport stress and stabling conditions at the abattoir (Gregory, 2008; Grandin, 2010). A drop in plasma glucose levels has been observed in cattle transported for up to 31 h, even after 24 h of stalling (Knowles et al., 1999). In rabbits, on the other hand, plasma glucose levels did not return to baseline until 24 h after transport (Fazio et al., 2015).
Rectal temperature was unaffected by lairage time (G01: 38.32 ± 1.01 °C; G02: 38.22 ± 0.60 °C). However, the values recorded are included in the 37 °C to 42 °C range reported in dromedary by Souilem and Barhoumi (2009). According to Hamad et al. (2016), the rhythm of rectal temperature in dromedaries is controlled by a complex mechanism, strongly affected by the heatwave season, to minimize the heat gradient between the body and the environment. This rise in temperature can be induced to reduce heat stress in animals (El-Harairy et al. 2010).
HR showed no significant difference between the two groups (G01: 61 ± 16.3 beats/min; G02: 60 ± 7.53 beats/min). These values are higher than those reported by Souilem and Barhoumi (2009), which were 42.03 beats/min. This elevation from the norm can be justified by environmental changes (Hamad et al., 2016).
Meat quality parameters
The effect of lairage time on dromedary meat quality parameters is shown in Table 2.
Dromedary meat quality parameters (mean ± standard deviation (SD)) according to lairage time before slaughter, between 1 and 8 h (G 01, n = 6) and for 48 to 96 h (G 02, n = 8).
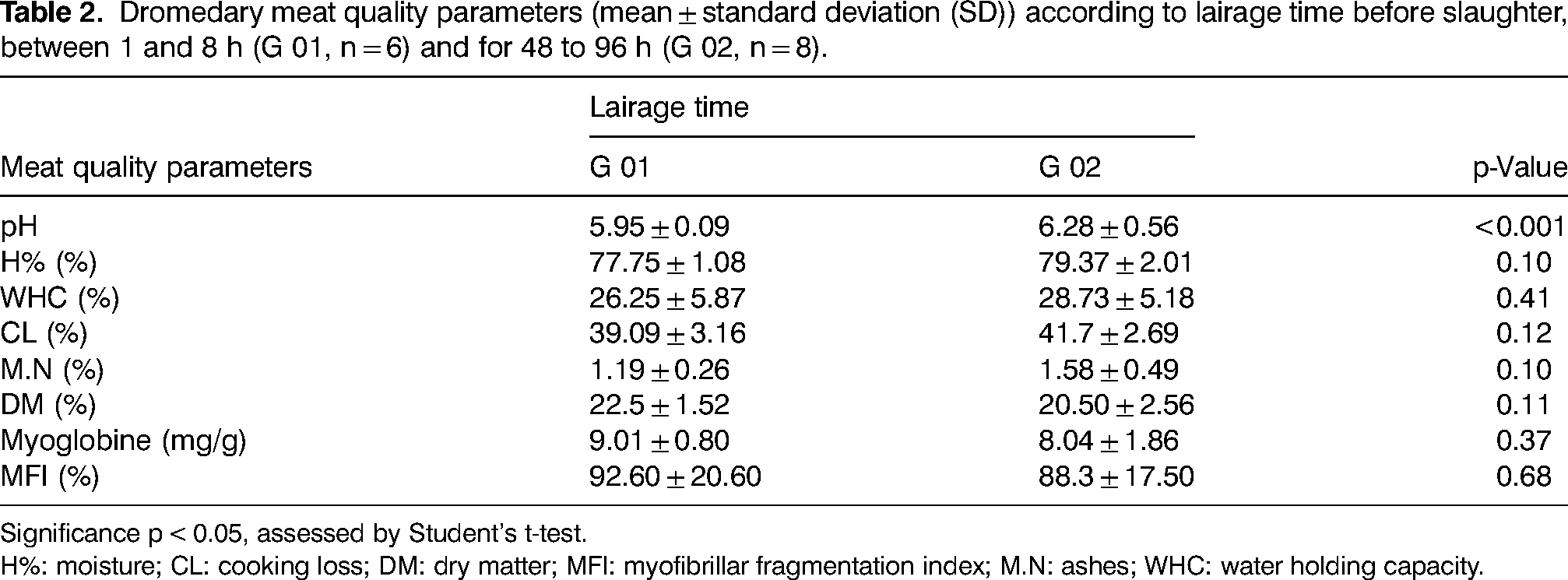
Significance p < 0.05, assessed by Student's t-test.
H%: moisture; CL: cooking loss; DM: dry matter; MFI: myofibrillar fragmentation index; M.N: ashes; WHC: water holding capacity.
Lairage time had a highly significant effect on the ultimate pH of the meat. Short-term lairage, from 1 to 8 h, produced meat with pHu = 5.95, significantly lower than long-term lairage, for 48 to 96 h, which produced meat with pHu = 6.28. pHu values similar to G01 have been previously reported for dromedaries in the same age category (Smili et al., 2022a, 2022b; Suliman et al., 2019; Kadim et al., 2006a). On the other hand, long-term lairage (G02) caused a higher pHu, characteristic of DFD (Dark, Firm, Dry) meats, probably due to depletion of glycogen reserves and animal tiredness. Indeed, it has been reported that the ultimate pH of muscle is largely determined by glycogen depletion and lactic acid accumulation in the pre- and postslaughter period, and is a major determinant of meat quality (Kadim et al., 2008; Febbraio et al., 2002; García-Torres et al., 2021). According to Chulayo et al. (2016), meats are classified into three categories according to their pHu: normal pHu (5.4–5.79), intermediate pHu (5.8–6.19) and high pHu (above 6.2). Variation in pHu is mainly associated with glycogen content and postmortem muscle metabolism, which are affected by many factors, including psychological stress, the animal's diet, transport and lairage time, affecting the meat's tenderness threshold (Bourguet et al., 2011; Teke et al., 2014). The ultimate pH of dromedary meat is between 5.5 and 6.6 affected mainly by age, with older dromedaries tending to produce lower pH values than younger ones, due to high glycogen levels (Kadim et al. 2006b). Stress also leads to muscle glycogen depletion and, consequently, increased pHu (Febbraio et al., 2002). Similar to our results, the study by Gallo et al. (2003), reported for cattle holded for 3, 6, 12, or 24 h after a 3 or 16 h journey, that the longer the duration of lairage, the higher the pHu of the meat. In addition, Tadich et al. (2005) found that long waiting periods at the slaughterhouse did not improve meat quality, since the waiting time depletes energy reserves and meat quality declines. Also in sheep, 48 h of lairage yielded meats with higher pHu compared to 24 h (Toohey and Hopkins, 2006). However, in cattle, Teke et al. (2014) reported that pHu after 24 h of lairage was higher compared to pHu after 72 h. One of the major defects of long-term stress is the generation of DFD meats, characterized by a high pHu (above 6.0) (Čobanović et al., 2020; Ferguson and Warner, 2008).
Meat moisture content and cooking loss showed no significant difference between the two groups (G01: 77.75%; G02: 79.37% and G01: 41.7%; G 02: 39.09%, respectively), with higher values than those reported by Moussahil et al. (2022), which were 76.03% and 30.23%, respectively. According to Barka et al.(2016), camel meat is rich in water compared to sheep and beef. This content depends on breed, sex, individual, age, health, diet, slaughter conditions and the amount of water consumed by the animal before slaughter.
Water holding capacity is an important meat quality characteristic because of its influence on meat yield and quality (Suliman et al., 2020). Water holding capacity showed no significant difference between the two stall groups (G01: 26.25%; G02: 28.73%). Cristofanelli et al. (2004) explain the high expression of juice in dromedary meat compared to other camelids such as llama and alpaca, due to their lower fat contents. Similar to our results, Teke et al. (2014) found no influence of lairage time (24, 48, and 72 h) on the water holding capacity and cooking loss of meat.
No significant differences were found for ashes and dry matter content between the two holded groups, respectively (G01: 1.19%; G02: 1.58% and G01: 22.5%; G02: 20.50%). The mineral content is similar to that reported by Al-Owaimer et al. (2014), which is 1.4%. In Camelus bactrianus, the latter ranges from 1.11% to 1.39% and is not influenced by age or muscle type (Si et al., 2022). Barka et al. (2016) reported that dry matter contents in dromedaries’ range between 25% and 29%, unaffected by transport distance and muscle type. Furthermore, Tabite et al. (2019) reported a significant decrease in glycogen content and an increase in pHu as transport distance increased, without significant variations in protein, ash, dry matter, and moisture.
Myoglobin content showed no significant difference between the two groups (G01: 9.01 ± 0.80 mg/g; G02: 8.04 ± 1.86 mg/g), with values higher than those reported by Gheisari (2011) and Maqsood et al. (2015) of 3.64 and 7 mg/g, respectively. These values are also higher than those reported for beef and sheep meat, which are around 5 mg/g (Manheem et al., 2023).
The tenderness threshold of meat during the tenderization process is highly affected by the activity of endogenous proteolytic enzymes, which degrade myofibrillar proteins, which is monitored by MFI measurement (Suliman et al., 2020). The myofibrillar fragmentation index showed no significant difference between the two lairage groups (G01: 92.60 ± 20.60%; G02: 88.3 ± 17.50%). These values are close to those reported by Kadim et al. (2006b) and Smili et al. (2022a) for the same age category and higher than those reported by Suliman et al. (2020).
The SDS PAGE profiles of myofibrillar and sarcoplasmic proteins are shown in Figure 1.
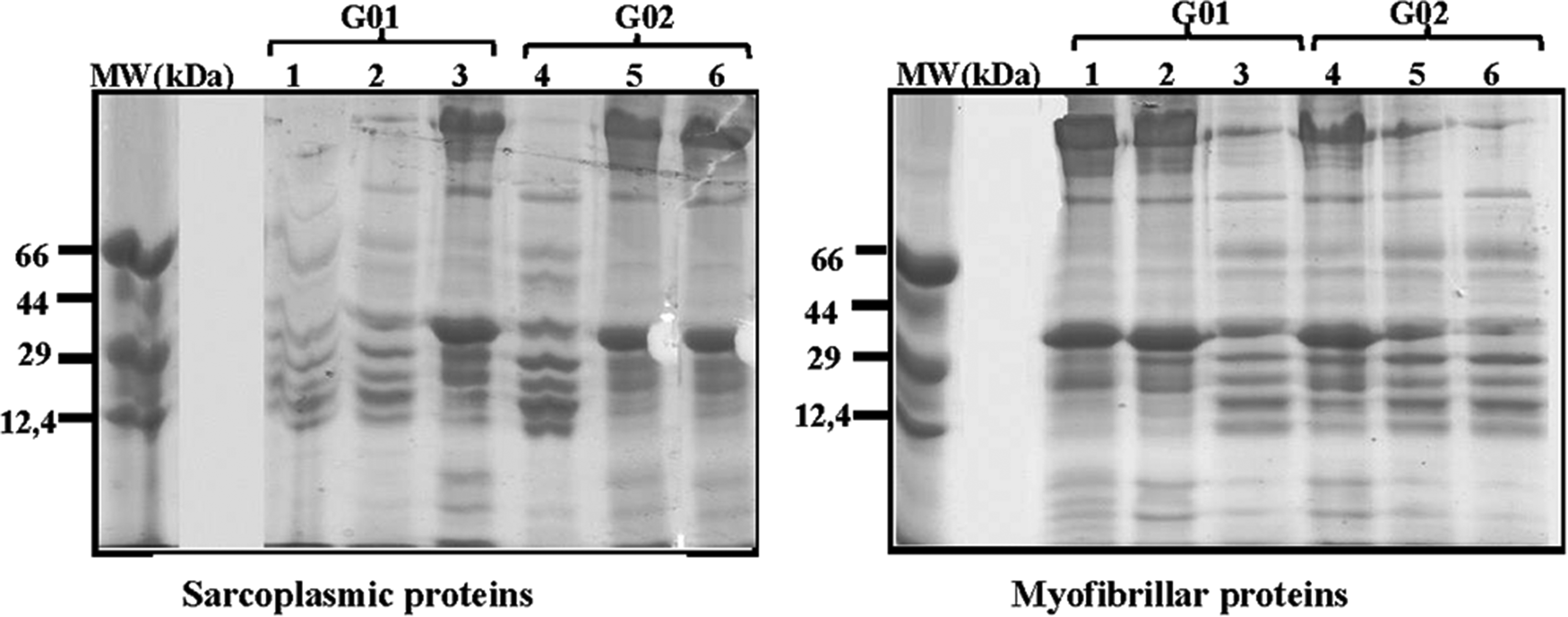
Electrophoretic profiles of sarcoplasmic and myofibrillar proteins in the Longissimus Lumborum muscle of dromedaries (from 1 to 6) stabled for 1 to 8 h (G01) and for 48 to 96 h (G02).
The electrophoretic profile of myofibrillar proteins revealed 15 protein bands from 2000 to 1.5 kDa. While the gel of sarcoplasmic proteins revealed the presence of 11 protein bands from 1000 to 3 kDa. The separated sarcoplasmic and myofibrillar proteins were subjected to densitometric analysis, density scores revealed no significant differences between protein bands, so variations in protein profiles are not significantly affected by the duration of lairage prior to slaughter.
Relationship between physiological parameters and meat quality
The correlations between physiological parameters and dromedary meat quality are shown in Table 3.
Pearson's correlation matrix for physiological parameters and meat quality.
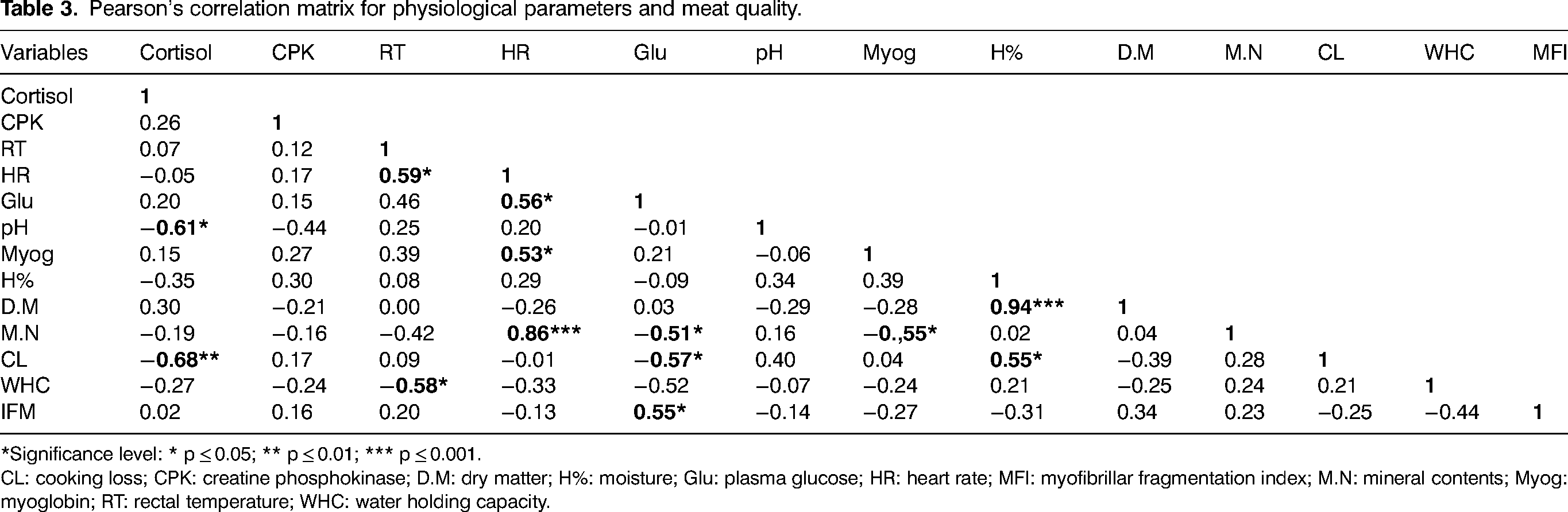
*Significance level: * p ≤ 0.05; ** p ≤ 0.01; *** p ≤ 0.001.
CL: cooking loss; CPK: creatine phosphokinase; D.M: dry matter; H%: moisture; Glu: plasma glucose; HR: heart rate; MFI: myofibrillar fragmentation index; M.N: mineral contents; Myog: myoglobin; RT: rectal temperature; WHC: water holding capacity.
Negative correlations were observed between cortisol and pH (r = −0.61, p ≤ 0.05) as well as cortisol and cooking loss (r = −0.68; p ≤ 0.01). Plasma glucose was negatively correlated with cooking loss (r = −0.57; p ≤ 0.05) and positively correlated with myofibrillar fragmentation index (r = 0.55; p ≤ 0.05). HR was positively correlated with plasma glucose (r = 056, p ≤ 0.05), myoglobin content (r = 0.53, p ≤ 0.05) and very strongly correlated with mineral content (r = 0.86 and p ≤ 0.001). Rectal temperature was positively correlated with HR (r = 0.59, p ≤ 0.05) and negatively correlated with water holding capacity (r = −0.58, p ≤ 0.05).
Several works have investigated the impact of preslaughter stress on animals, it causes an increase in cortisol, plasma lactate, muscle temperature, and pHu (Hambrecht et al., 2005; García-Torres et al., 2021), it influences meat quality by decreasing muscle glycolytic potential, water holding capacity and tenderness (Tabite et al., 2019). In contrast to our results, Maghfiroh et al. (2014) observed a positive correlation between cortisol and pH at 1 h but no correlation with pH at 24 h postmortem. And a positive correlation between cortisol and cooking loss. Cortisol injection in lambs resulted in a significant increase in glucose metabolism, temperature, pH, and water-holding capacity of fresh meat (Pighin et al., 2013). A negative correlation was observed in bulls between muscle pHu and their lairage times (1, 17, and 40 h) (Mounier et al., 2006). Water holding capacity, proteolysis, lipid oxidation, and meat color were largely influenced by the kinetics of postmortem pH and muscle temperature (Ferguson and Warner, 2008). Furthermore, it has been shown that an increase in lairage period increases the occurrence of dark cut meat (DFD) in cattle (Warner et al., 1998).
The principal component analysis (PCA) of physiological parameters: COR, CPK, RT, HR, and Glu, and of meat quality parameters: pH, H%, M.N, CL, WHC, and MFI, is shown in Figure 2, allowing visualization of the distribution of the variables studied and the animals of the two lairage groups.
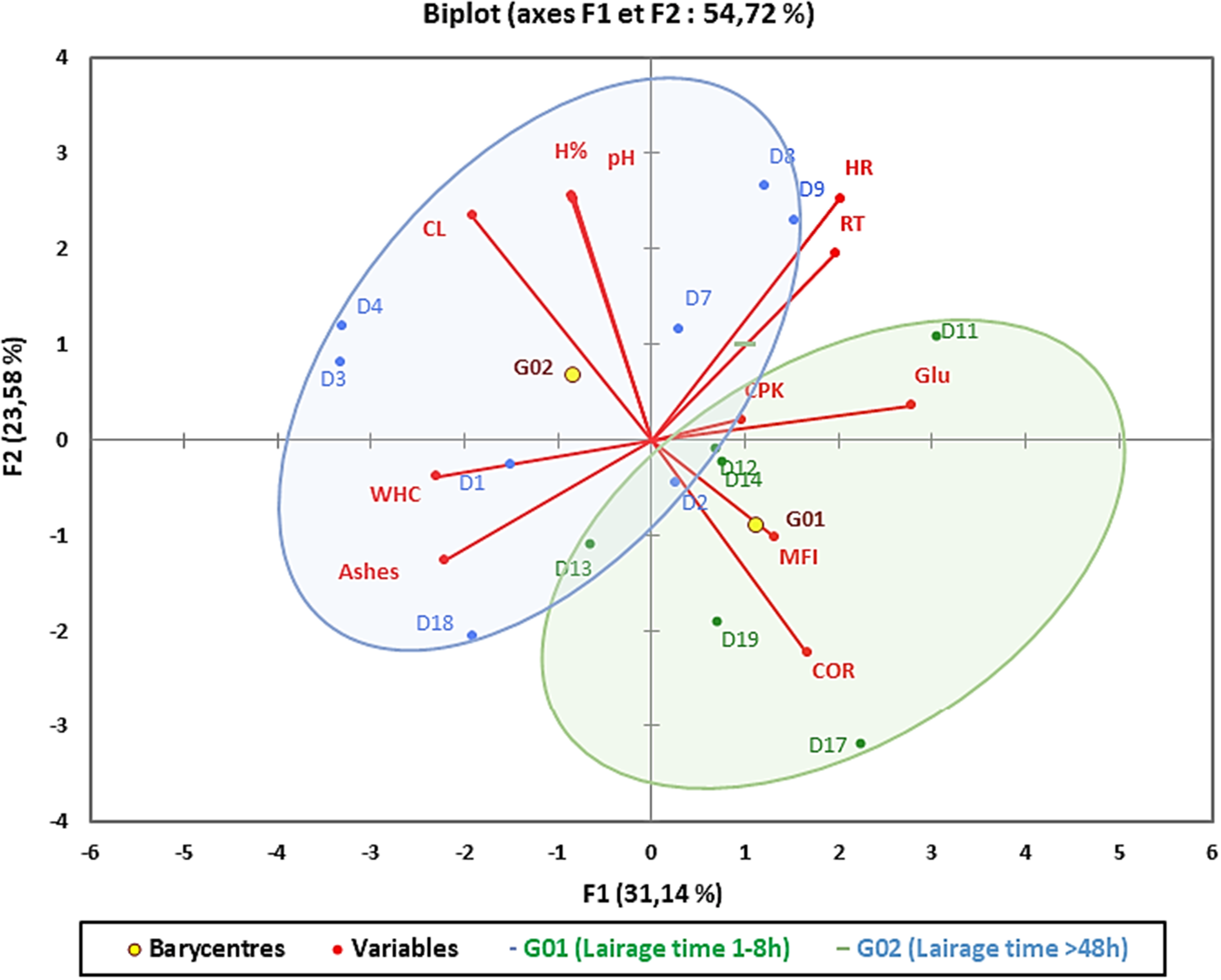
Principal component analysis of physiological parameters and meat quality parameters.
PCA applied to the variables mentioned accounts for 54.72% of the total variance, with 31.14% explained by F1 and 23.58% explained by F2. Plasma glucose, rectal temperature, ashes and WHC were the main variables explained by the F1 factor. The latter two were negatively correlated with this factor. Moisture, pH, cooking loss, cortisol, and HR were the dominant variables in F2, with cortisol negatively correlated with this factor.
Projection of the individuals on the factorial plane revealed a separation between the meat of dromedaries kept in lairage for less than 8 h and those kept for more than 48 h. Animals held in lairage for short period were characterized by high cortisol and plasma glucose levels and low meat pH, mineral content and moisture. These two physiological parameters were also negatively correlated with cooking loss and WHC, confirming an overall impact of lairage time on pH and meat water holding capacity. These results highlight the importance of controlling the duration and conditions of the lairage period in order to standardize dromedary meat quality.
CONCLUSION
Cortisol levels and pH of the Longissimus Lumborum muscle of Sahraoui dromedaries were significantly affected by the lairage time before slaughter. Increasing lairage time generated meat with high pHu, so this step may be a major source of stress for the animals. Further studies, in particular on optimizing lairage conditions and preslaughter treatment, are needed to improve dromedary welfare and meat quality.
Footnotes
ACKNOWLEDGEMENTS
The authors would like to thank the breeders and butchers of the Oued Souf City, especially Mr Ibrahim Touati, and the members of the Maquav/INATAA team who contributed to this work.
DECLARATION OF CONFLICTING INTERESTS
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
FUNDING
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Algerian Ministry of Higher Education and Scientific Research and Prima project CAMEL-SHIELD.