Abstract
An exposure assessment model for industrial use has been developed by using kinetic data from inactivation and growth of
INTRODUCTION
Rice is undeniably one of the most consumed foods globally, serving as a staple in the diet of many countries, particularly in Asia, with Bangladesh standing out as the world's largest producer and consumer of rice (USDA, 2024). In the European Union (EU), rice production in 2022 reached 1.1 million tons, with Italy accounting for 50% and Spain for 30% of the production area dedicated to rice cultivation (European Comission, 2023). It is an important source of energy in human nutrition due to its high carbohydrate content (Juliano, 1993). However, its composition, particularly the fact that almost 90% of its dry matter is starch, makes it an ideal substrate for the growth of certain pathogenic microorganisms such as
Vegetative cells of
Chitosan has developed as a natural antimicrobial with various potential applications in the agri-food industry. Chitosan has been obtained from the exoskeletons and shells of crustaceans and has demonstrated excellent antimicrobial and antioxidant properties (Goy et al., 2009). Based on those previous studies with crustacean chitosan, Valdez, Garcia, et al., (2022) and Valdez, Úbeda-Manzanaro, et al., (2022) carried out studies to assess whether chitosan obtained from insects exhibits the same antimicrobial properties as chitosan derived from crustaceans. The results suggested that insect-derived chitosan could be an interesting alternative as a natural antimicrobial, while also offering opportunities for the valorisation of insect-based industry by-products. These findings are significant as they imply that chitosan derived from insects might be equally effective in inhibiting the growth of pathogenic microorganisms, thereby expanding the sources of this important antimicrobial substance. Furthermore, the repurposing of industry by-products, such as insect exoskeletons, for chitosan production, can promote sustainability and the circular economy within the food chain and other industries (Valdez, Garcia, et al., 2022).
When dealing with a new product and a novel control measure, it is crucial to conduct an exposure assessment at an industrial level to complement measures such as HACCP. This assessment would enable the implementation of safety management actions for final products before they are released to the market. Additionally, it is essential to evaluate the impact that the refrigerated storage process may have on the final microbial load of the product before consumption. By conducting an exposure assessment, potential risks associated with the new product and control measures can be identified and addressed proactively. This helps ensure that the final products meet safety standards and are safe for consumption. Moreover, assessing the impact of refrigerated storage on microbial loads can provide valuable insights into the effectiveness of this storage method in controlling microbial growth and ensuring product safety throughout the supply chain. Overall, a comprehensive evaluation of exposure and risk management measures is essential for safeguarding the safety of new products and maintaining consumer confidence in the food supply.
Microbiological risk assessment (MRA) is indeed a systematic approach used to evaluate the potential risks associated with biological hazards in food. While its primary focus is on food safety, it is important to recognize that in an industrial level, the concept of “risk” can extend beyond just safety concerns to include aspects related to food quality as well. Microbial spoilage, for instance, can compromise the quality of food products, and depending on the microbial ecology promote the growth of pathogenic microorganisms. Therefore, when conducting MRA in an industrial context, it is essential to consider both safety and quality aspects to ensure the overall integrity of the food supply chain.
When transferring the risk assessment concept to the industry, exposure assessment becomes the main component. Exposure assessment involves understanding and quantifying the likelihood of consumers being exposed to microbiological hazards present in food. This step is essential because it provides crucial information about how and to what extent consumers may be exposed to microbiological risks. It considers changes from raw materials, processes or the use of new preservation methods through to consumption. This process involves evaluating various factors such as food processing methods, storage conditions, handling practices, and consumption patterns to estimate the level of microbial contamination that consumers are likely to encounter during the entire food chain (Membré and Boué, 2018).
In this context, the main objective of this study is to perform an exposure assessment by developing a modular model for assessing exposure to
MATERIAL AND METHODS
Building the model: structure, equations, inputs, outputs
The model structure plays a critical role in understanding how the inputs of the model influence the response of the model's output variables. Figure 1, shows a scheme that illustrates the relationships between the various components of the exposure assessment model.
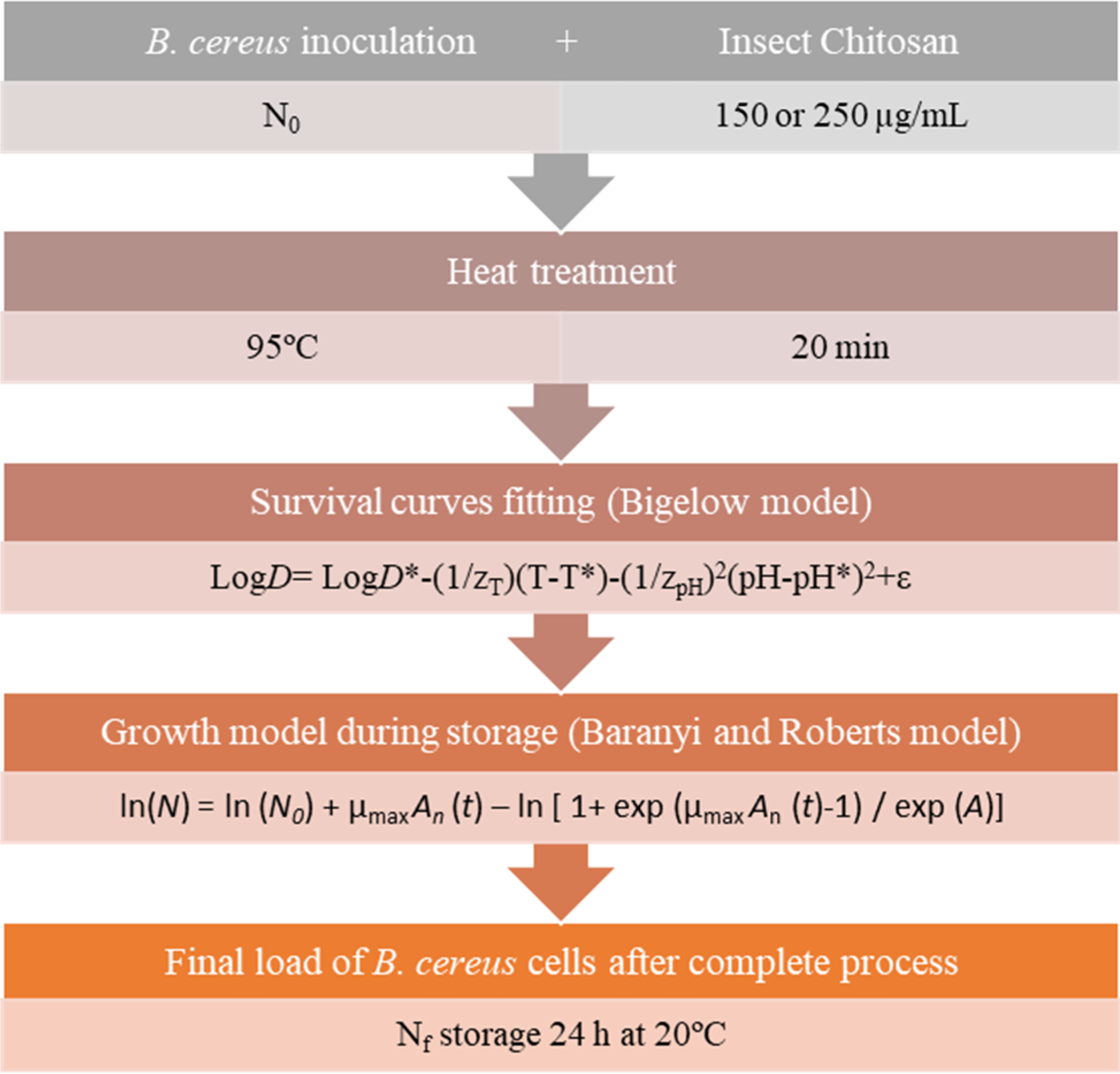
Flowchart for risk assessment model developed based on the final load of
A modular exposure assessment model was constructed, considering the kinetic parameters obtained from mathematical models of inactivation and growth. This modular approach integrates the results of microbial inactivation and growth models into a coherent and understandable exposure assessment framework. By combining these models into a modular approach, the exposure to pathogenic or spoilage microorganisms in foods can be comprehensively assessed throughout the entire process, from production to consumption. This provides a powerful tool for food safety professionals and regulators to understand and manage microbiological risks in the food chain.
The selection of equations depends on the specific aspects of the system being modelled. For example, in a model assessing microbial growth, equations describing microbial growth kinetics such as the (Baranyi and Roberts, 1994) model and modified Gompertz equation (Bhaduri et al., 1991) may be used to predict population dynamics over time. Similarly, equations describing microbial inactivation kinetics such as Weibull (Weibull, 1951) or first-order Bigelow models (Bigelow, 1921) may be employed to predict the reduction of microbial populations under different processing conditions.
Model equations
Inactivation model
In general, when a heat treatment step is involved in microbial inactivation, the resulting reduction in microbial populations is often described using a first-order kinetic model. In this case, the Bigelow model was used (equation 1).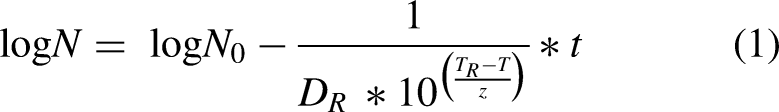
Growth model
A growth model was developed to estimate the final number of microorganisms after the storage. The Baranyi and Roberts, (1994) growth model (Equation 2) was employed for this purpose. This model used experimental data obtained under the same substrate conditions as those used in the heat resistance studies, allowing for the determination of the specific growth rate and the lag phase of 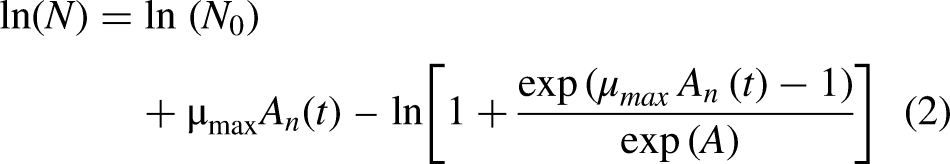
Industrial exposure assessment model input
The Industrial exposure assessment model developed has two types of inputs: variables and settings.
Table 1 shows the model inputs considered in the industrial exposure assessment model.
Model inputs are considered in the industrial exposure assessment model.

Industrial exposure assessment model output
The output obtained from an Industrial exposure assessment model, typically provides a quantity or concentration of microorganisms in a food product and/or in a consumer's portion. This output essentially represents an exposure assessment, as it quantifies the level of microbial contamination that consumers may be exposed to throughout the food chain. Table 2 shows the outputs considered in the model.
Model outputs are considered in the industrial exposure assessment model.

Simulation
Monte Carlo simulation was carried out by using the
Interpretation of industrial exposure assessment model output, what-if scenarios
An industrial quantitative microbial risk assessment (IQMRA) model aims to assess the risk associated with the transmission of microorganisms from their source (such as raw material for industry) to the final consumption (fork). The output of such a model typically includes the estimation of the quantity or concentration of microorganisms present in the food product or the consumer's portion. By quantifying this microbial load, it helps in understanding the potential health risks associated with consuming the food product. Therefore, in essence, a processing-to-fork model is an exposure assessment model, as it evaluates the potential exposure of consumers to microbial hazards throughout until consumption.
What-if scenarios are indeed powerful tools in risk assessment, offering insights into how different variables or parameters can affect outcomes. They allow analysts to explore a range of possibilities and make informed decisions. In the context of food safety, these scenarios can take various formats, such as:
RESULTS
Industrial exposure assessment model
Table 3 shows the output data from the exposure assessment model after the complete process. The final microbial load (
Summary of the input and output parameter values of the exposure assessment model for a cooking process of 95 °C for 20 min and storage temperature of 20 °C for 24 h.

However, by adding insect chitosan at different concentrations, this risk significantly decreases. For instance, at an insect chitosan concentration of 150 µg/ml, the final
Similarly, in Table 3, it can be seen that if the insect chitosan concentration increases to 250 µg/ml, the
Sensitivity analysis
Additionally, a sensitivity analysis was conducted, revealing that the variable that most affect the final number of microorganisms in the event of a cold chain breach (
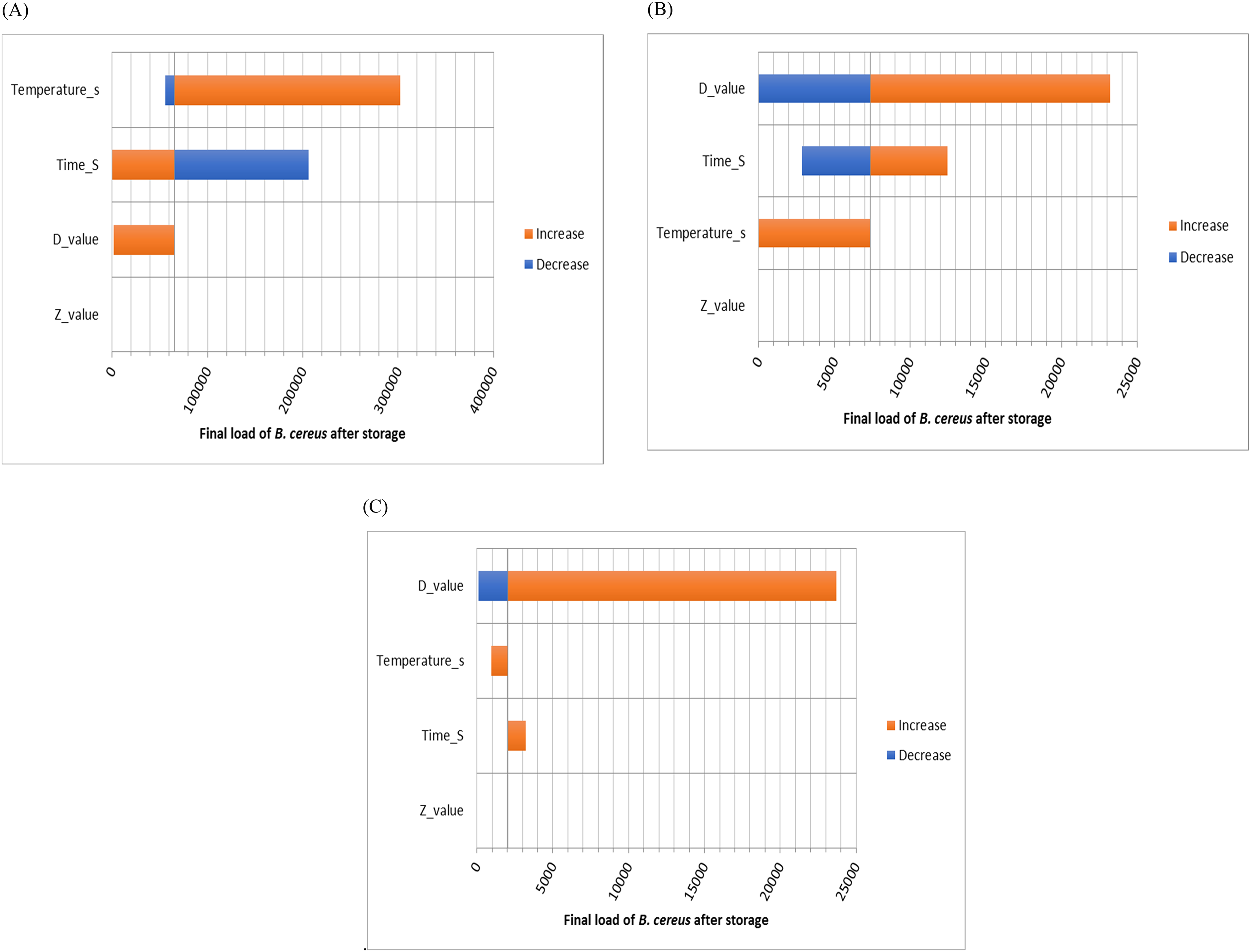
Tornado plot of the sensitivity analysis is performed when control samples and chitosan samples are stored at 20 °C for 24 h. A (Control), B (insect chitosan concentration 150 µg/ml), C (insect chitosan concentration 250 µg/ml).
Figure 3 shows the effect of a 2% deviation in cooking temperature on the residual number of microorganisms after the process, when the rice matrix contains 250 µg/ml of insect chitosan as a natural antimicrobial. As can be observed in the figure, a 2% decrease in temperature (93 °C) impacts on the number of residual microorganisms increase them on more than 4 log, unless it does not reach the infective dose of 105 CFU/g (EFSA Panel on Biological Hazards (BIOHAZ), 2016).
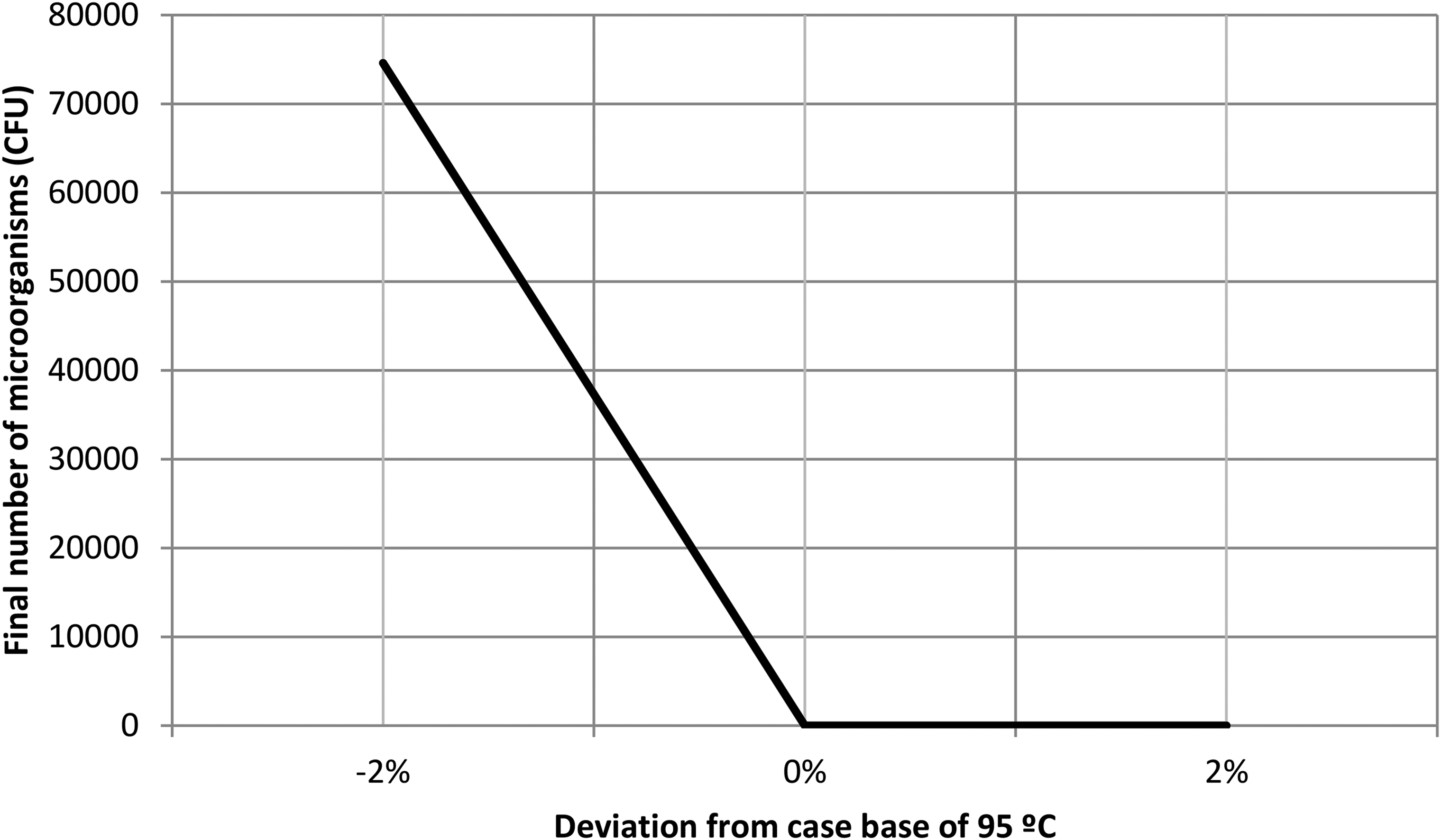
Effect of a deviation of the 2% on temperature in relation to the case base of 95 °C.
While, Figure 4 illustrates the effect of a 10% deviation in cooking time on the residual number of microorganisms after the process when the rice matrix contains 250 µg/ml of insect chitosan as a natural antimicrobial. As depicted in the figure, a 10% decrease in time (18 min) barely affects the number of residual microorganisms, failing to reach the infective dose of 105 CFU/g (EFSA Panel on Biological Hazards (BIOHAZ), 2016).
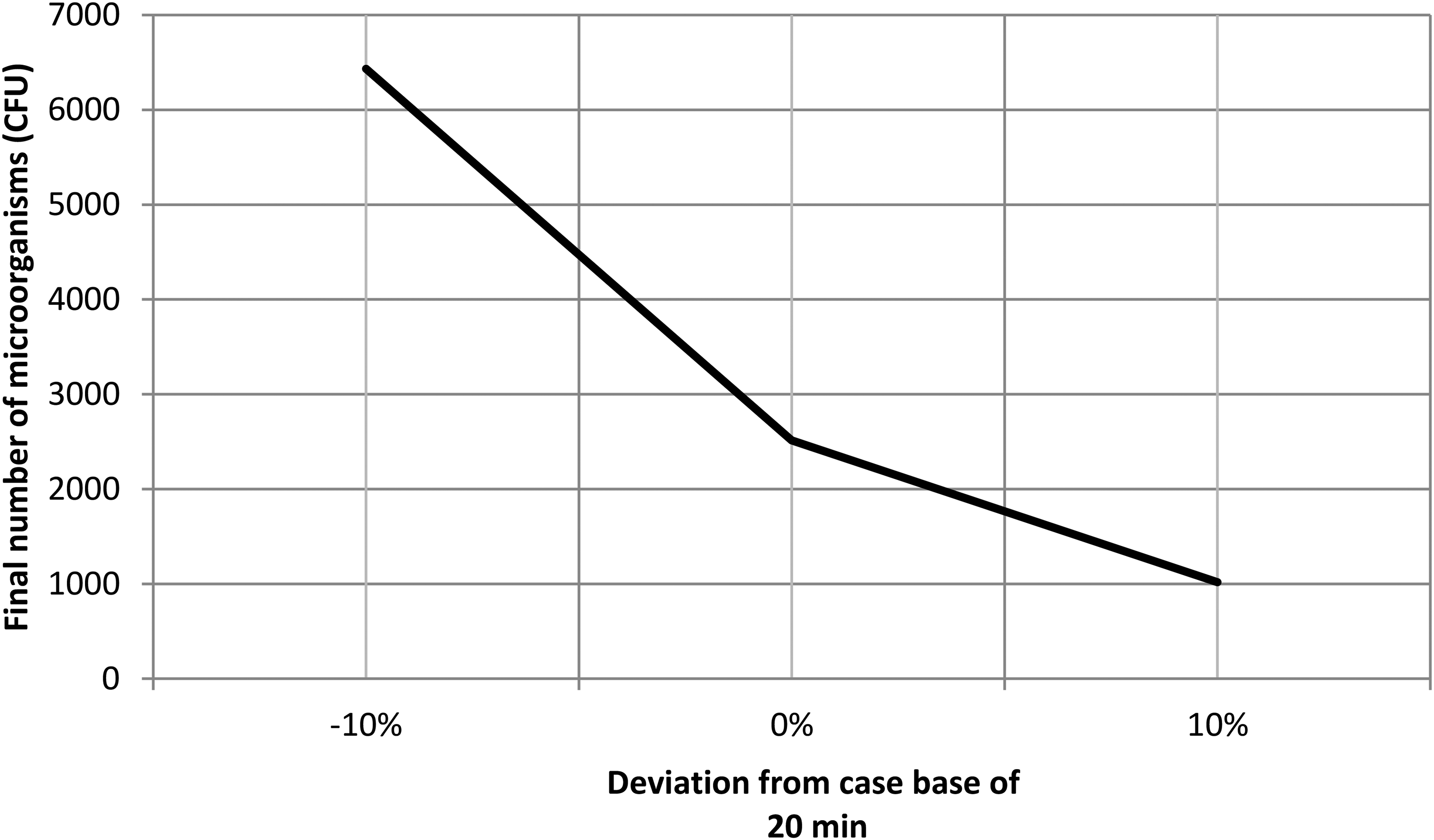
Effect of a deviation of the 10% on the cooking time in relation to the case base of 20 min.
Prediction of the number of contaminated units
The exposure assessment model has an additional module that allows for determining the percentage of units contaminated considering a performance criterion (PC) defined by the ratio of treatment time to the
First scenery: initial contamination of 104 cells/portion
The first scenery was considered the control samples. The simulation results can be seen in Table 4 and Figure 5. If the initial contamination is
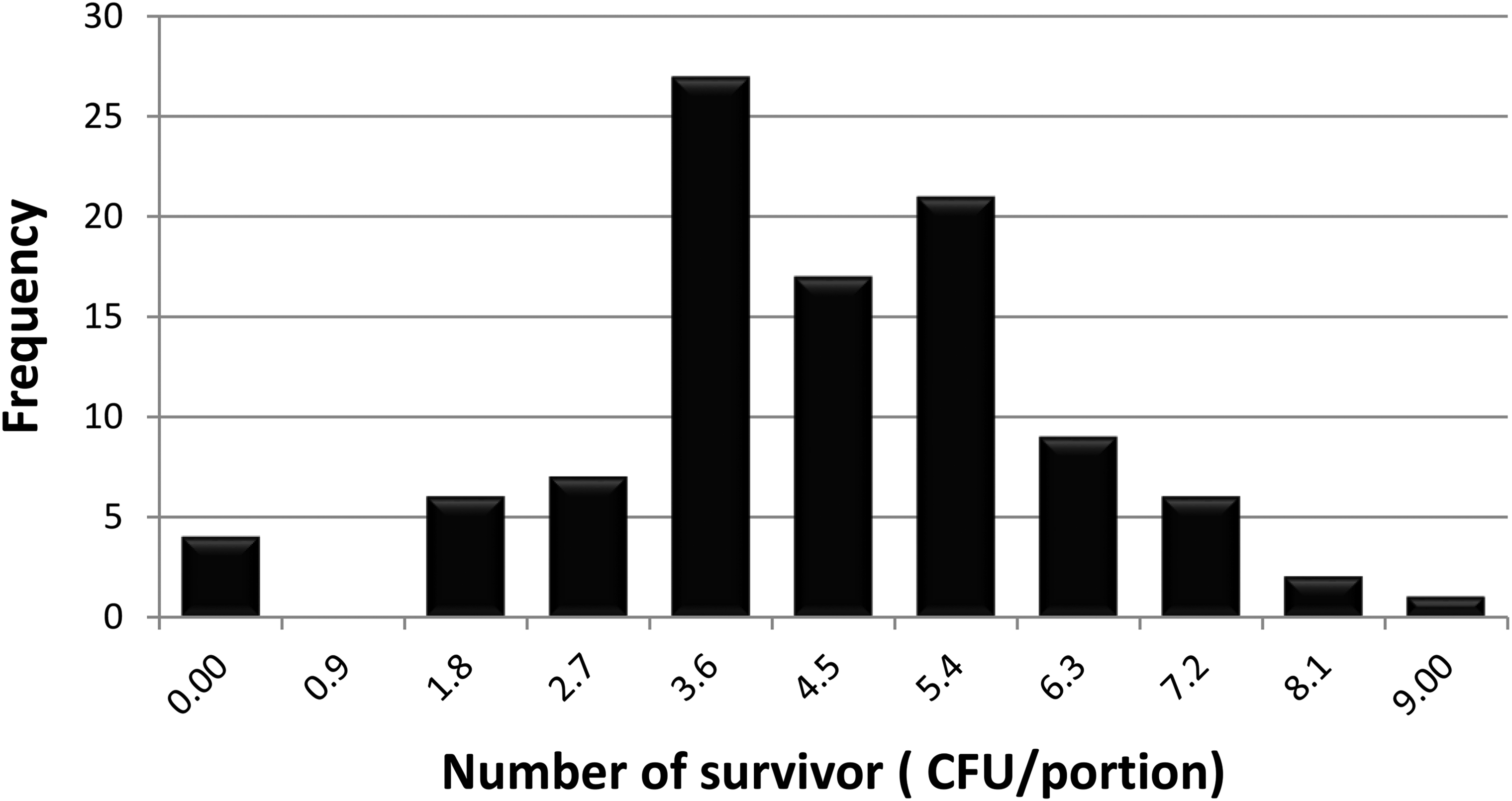
Frequency chart for an initial contamination of 104 cells/portion
Results of simulation scenery 1.

Second scenery: initial contamination of 104 cells/portion
However, in the second scenery if the initial contamination is
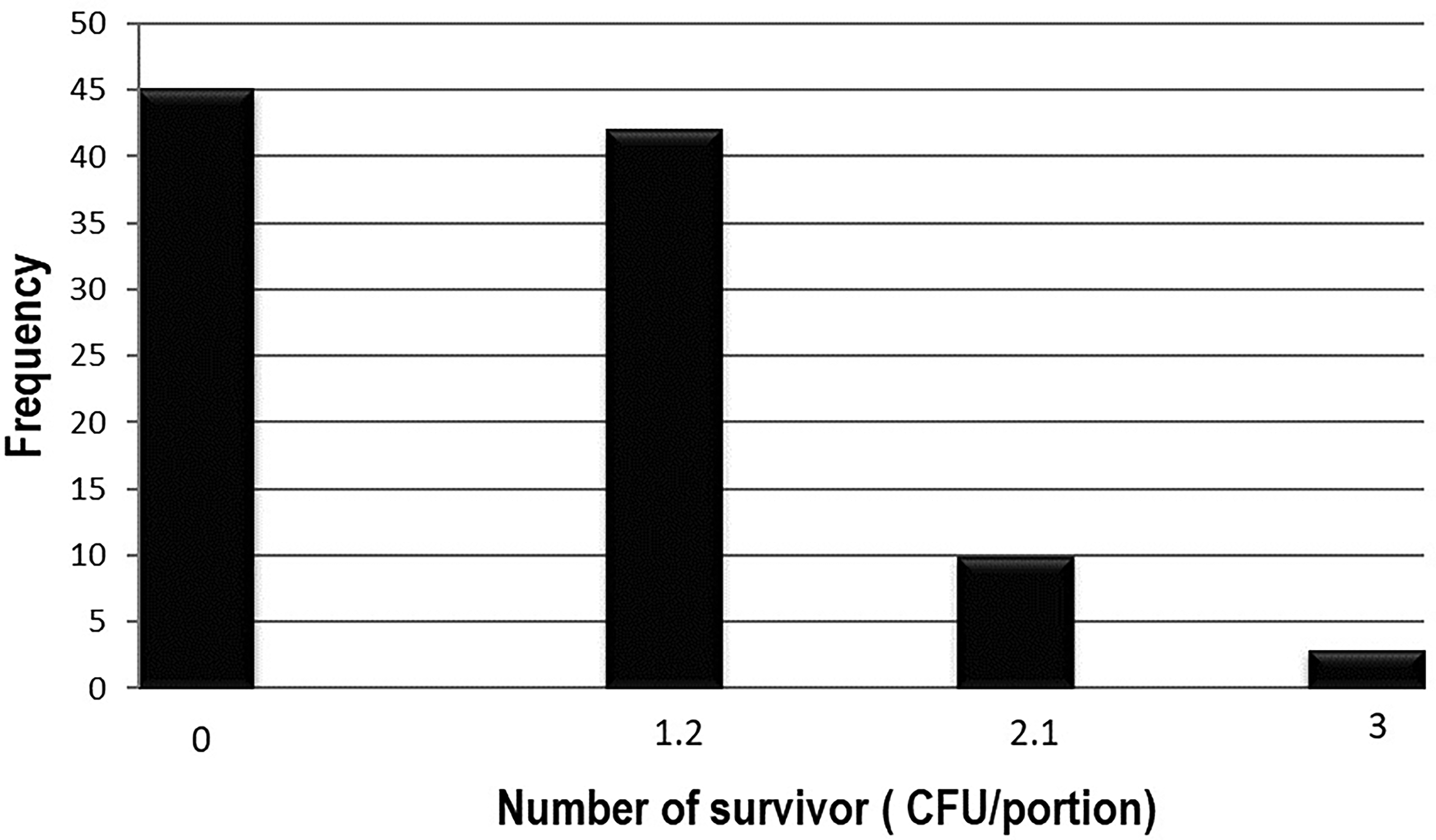
Frequency chart for an initial contamination of 104 cells/portion
Results of simulation scenery 1.

Third scenery: initial contamination of 107 cell/portion
Results of simulation for third scenery can be seen in Table 6 and Figure 7. In the case of an initial contamination of 107 microorganisms, the simulation indicates that 100% of the units would be contaminated after treatment with a mean of 729.48 microorganisms per container, even if the rice matrix contains 250 µg/ml of insect chitosan. This could be due to bacteria's resistance to thermal treatment and the survival probability established by the performance criterion, hence the importance of reducing the higher initial microbial load.
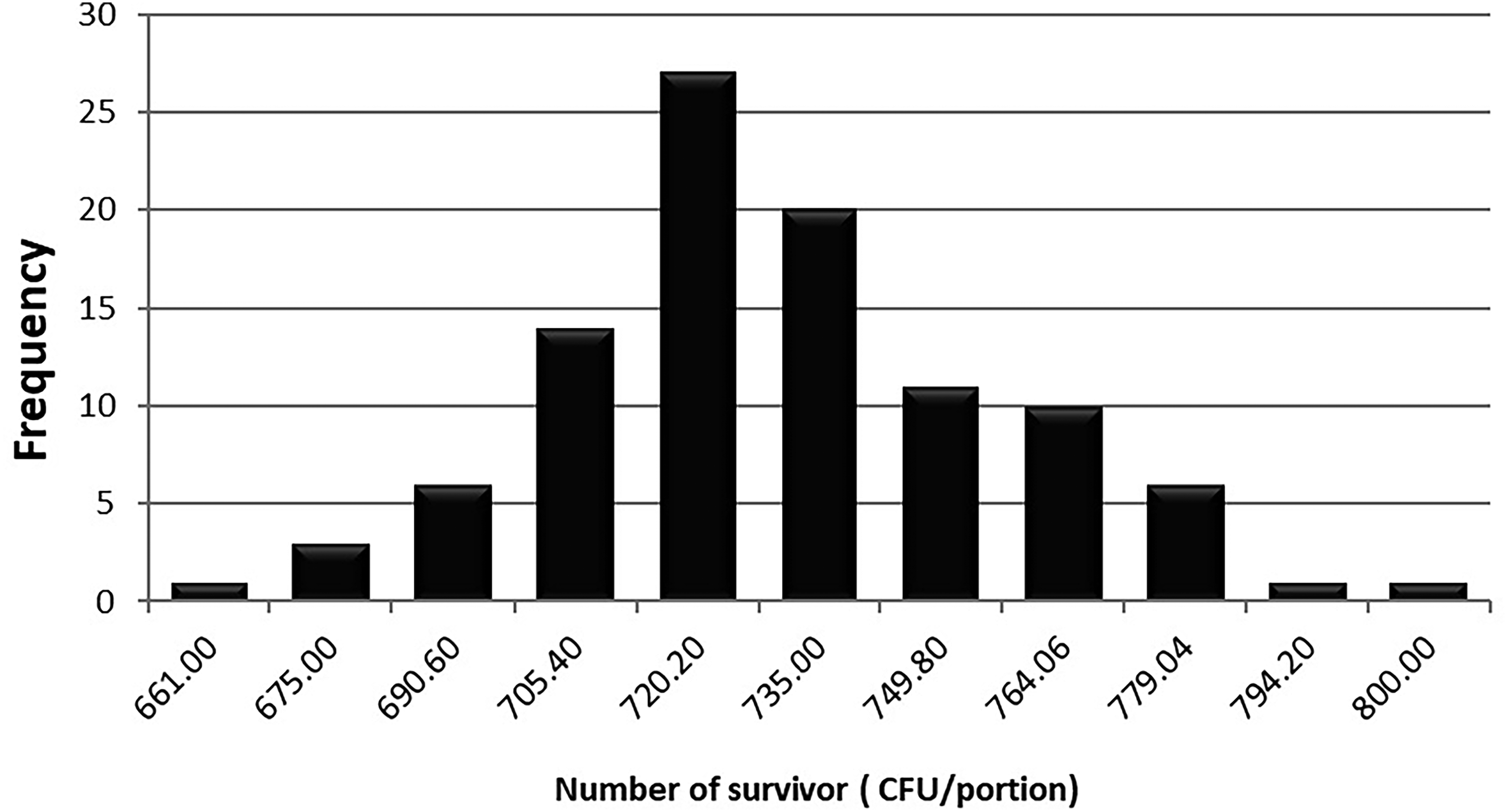
Frequency chart for an initial contamination of 107 cells/portion.
Results of simulation scenery 3.

CONCLUSIONS
The exposure assessment model built in this work allows obtaining an estimate of the final load levels of
Using a minimum concentration of insect chitosan (150 µg/ml) that allows controlling the final load of
It is essential to consider that food safety is a paramount concern in the food industry, and any measure that reduces microbial burden and minimizes the risk of food poisoning is beneficial. The utilization of insect chitosan as an antimicrobial agent can be an effective and natural strategy to enhance food safety, provided it is applied appropriately and complies with food safety regulations and standards.
Footnotes
Acknowledgements
This work was supported by MCIN/AEI/10.13039/501100011033, “ERDF A way of making Europe” and by the “European Union’’ [grants PID-2020-11631RB-C31 and PRE-2021-098627]. M. Inés Valdez-Narváez also acknowledges her scholarship from the Conselleria d’Innovació, Universitats, Ciència y Societat Digital ACIF/2021/424 of Generalitat Valenciana. The Instituto de Agroquímica y Tecnología de Alimentos-CSIC is an accredited Center of Excellence Severo Ochoa [CEX2021-001189-S], funded by MCIU/AEI / 10.13039/501100011033.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MCIN/AEI/10.13039/501100011033, “ERDF A way of making Europe” and by the “European Union’’ [grants PID-2020-11631RB-C31 and PRE-2021-098627] and by the Ministerio de Ciencia e Innovación, Conselleria de Innovación, Universidades, Ciencia y Sociedad Digital, Generalitat Valenciana [grant ACIF/2021/424].
