Abstract
Purpose:
This study aimed to elucidate the contribution of EP3 receptor agonism to the intraocular pressure (IOP)-lowering effects of sepetaprost, a novel dual agonist of FP and EP3 receptors, in monkeys.
Methods:
The agonistic activities of ONO-AG-367 (the active metabolite of sepetaprost) and latanoprost acid toward FP and EP3 receptors were assessed using a cell-based assay. The IOP-lowering effects of sepetaprost and latanoprost were compared in ocular normotensive monkeys. The involvement of EP3 receptor activation in sepetaprost-induced IOP reduction was evaluated by intracameral administration of an EP3 antagonist followed by topical sepetaprost. Aqueous humor dynamics (AHD) were assessed via fluorophotometry in ocular hypertensive monkeys. Outflow facility was measured by tonography in ocular normotensive monkeys. Additionally, the IOP-lowering efficacy of sepetaprost was examined in latanoprost low-responder monkeys.
Results:
ONO-AG-367 exhibited potent agonistic activity at both FP and EP3 receptors, whereas latanoprost acid selectively activated the FP receptor. Sepetaprost elicited greater and longer-lasting IOP reduction than latanoprost. Pretreatment with an EP3 antagonist significantly attenuated the IOP-lowering effect of sepetaprost at 26 h. In the AHD study, sepetaprost enhanced outflow facility and uveoscleral outflow. Tonographic assessment confirmed a significant increase in outflow facility following sepetaprost treatment. Sepetaprost also effectively reduced IOP in latanoprost low-responder monkeys.
Conclusions:
EP3 receptor agonism contributes to the longer-lasting IOP-lowering effect of sepetaprost in monkeys. These findings suggest that sepetaprost enhances uveoscleral outflow and improves facility in the trabecular outflow pathway, and may provide effective IOP lowering when there is a suboptimal response to latanoprost.
Introduction
Glaucoma is the leading cause of irreversible blindness worldwide, with the number of affected individuals projected to reach 111.8 million by 2040. 1 Reduction of intraocular pressure (IOP) remains the only evidence-based intervention for slowing glaucomatous visual field loss.2,3 Prostanoid FP receptor agonists, such as latanoprost, are widely employed as first-line therapy. 4 However, a subset of patients exhibits suboptimal responses to FP agonists, 5 and many require adjunctive therapy to achieve target IOP levels. 4 Consequently, there remains a critical need for alternative therapeutic options.
Preclinical studies in mice suggest that stimulation of prostanoid EP3 receptors, potentially via endogenous prostaglandin production, partially contributes to the IOP-lowering effects of FP receptor agonists.6,7 These findings imply that agents capable of activating both FP and EP3 receptors may provide superior IOP-lowering efficacy relative to selective FP receptor agonists. Sepetaprost (formerly ONO-9054 or DE-126), a novel dual agonist of FP and EP3 receptors, received manufacturing and marketing approval for glaucoma treatment in Japan on August 25, 2025.8–11 Sepetaprost is a prodrug that is hydrolyzed into the active metabolite ONO-AG-367 upon administration. Preclinical studies in monkeys have shown that sepetaprost produces greater and longer-lasting IOP reduction than FP receptor agonists such as latanoprost and travoprost. 8 Yamagishi-Kimura et al. demonstrated in mice that both FP and EP3 receptor activation contribute to the IOP-lowering effects of sepetaprost. 12 Their findings also suggested that EP3 receptor agonism may prolong the IOP-lowering action of FP receptor agonists. 12 Nevertheless, the role of EP3 receptor activation in IOP-lowering in nonhuman primates, whose ocular physiology and genetic background are more similar to those of humans, remains largely unexplored.
This study aimed to elucidate the contribution of EP3 receptor agonism to the IOP-lowering effects of sepetaprost in nonhuman primates. We first compared the receptor agonistic activities and IOP-lowering efficacies of sepetaprost and latanoprost. We then examined the impact of EP3 receptor antagonism on sepetaprost-induced IOP reduction and evaluated sepetaprost’s effects on aqueous humor dynamics (AHD). Finally, given emerging evidence that sepetaprost acts via mechanisms distinct from those of latanoprost, we investigated its efficacy in monkeys that exhibit poor responsiveness to latanoprost.
Methods
Materials
Sepetaprost, ONO-AG-367, and ONO-AE3-240 (an EP3 receptor antagonist) were synthesized by Ono Pharmaceutical Co., Ltd. (Osaka, Japan).
13
Sepetaprost ophthalmic solution and its vehicle were formulated by Santen Pharmaceutical Co., Ltd. The vehicle consisted of polysorbate 80,
Agonist activity at FP and EP3 receptors
Agonist activities of ONO-AG-367 and latanoprost acid toward FP and EP3 receptors were evaluated by Eurofins Cerep (Celle-Lévescault, France). Briefly, HEK-293 cells expressing human FP receptors and Chem-1 cells expressing human EP3 receptors were treated with the Fluo-4 Direct probe, followed by exposure to the test compounds. Intracellular Ca2+ signaling responses were measured via fluorometry. EC50 values were derived from concentration–response curves generated using mean replicate values and fitted with the Hill equation via nonlinear regression analysis (Hill software, Eurofins Cerep). Geometric mean EC50 values were calculated from three independent experiments performed in duplicate.
Animals
Male cynomolgus monkeys (Shin Nippon Biomedical Laboratories, Ltd., Tokyo, Japan; KEARI Co., Ltd., Osaka, Japan; Eve Bioscience Co., Ltd., Wakayama, Japan) were used for IOP and tonography studies. Female cynomolgus monkeys (Harlan Laboratories, Inc., Indianapolis, IN; Covance Inc., Princeton, NJ) were used for the AHD study. To induce unilateral sustained IOP elevation for the AHD study, the trabecular meshwork of the left eye was lasered. Details of the laser procedure are described elsewhere. 14 Lasering had been performed between 8 and 16 years prior to this study. All animal experiments adhered to the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research and were approved by the Animal Care and Use Committees of Santen Pharmaceutical Co., Ltd. or the University of Nebraska Medical Center.
IOP study
Monkeys were trained for conscious IOP measurement using a pneumatonometer (Model 30 Classical; Reichert, Inc., Depew, NY) following topical anesthesia with 0.4% oxybuprocaine hydrochloride (Benoxil Ophthalmic Solution 0.4%, Santen Pharmaceutical Co., Ltd.). IOP changes from baseline were calculated by subtracting baseline IOP from values obtained at each post-administration timepoint. The mean and standard error of the mean (SEM) were determined. All treatments followed a crossover design with a washout period of 5–14 days between administrations.
Each monkey received a 20 μL instillation of the test solution into one eye. IOP was measured prior to and at predefined timepoints after administration. In cases where two instillations were performed, the second was administered 5 min after the first.
In the EP3 antagonist study, 10 μL of 100 μM EP3 antagonist solution or its vehicle (saline containing 0.5% dimethyl sulfoxide) was intracamerally injected under general anesthesia. Test solutions were subsequently instilled, and IOP was measured at baseline and at designated timepoints. To assess treatment effects, the IOP change from baseline in the control group was subtracted from that in the test groups.
AHD study
The AHD study was conducted as previously described, with minor modifications.14,15 A masked, crossover design was employed with a minimum 1-week washout period between administrations. A 20 μL drop of test solution was instilled into hypertensive eyes once daily for 7 consecutive days. On day 7, the final dose was administered ∼4.5 h prior to AHD measurements. Thirty minutes after the last instillation, 10% fluorescein was applied to the cornea. Monkeys were sedated via intramuscular injection of ketamine HCl (10 mg/kg). Following topical anesthesia with 0.5% proparacaine HCl (Akorn Inc., Lake Forest, IL), IOP was measured using a pneumatonometer. Central corneal thickness and anterior chamber depth were determined via ultrasound pachymetry (Pac Scan; Sonomed Inc., Lake Success, NY), and corneal diameter was measured using calipers. Corneal and anterior chamber volumes were subsequently calculated. 16 Starting at ∼10:00 AM, fluorescein intensity in the cornea and anterior chamber was quantified using a scanning ocular fluorophotometer (Fluorotron Master; Ocumetrics Inc., Mountain View, CA). Measurements were repeated three times at 45-min intervals. Aqueous flow (Fa) was calculated from the decay curves of corneal and anterior chamber fluorescein concentrations and anterior chamber volume, as previously described. 17
Immediately after the fourth scan, IOP (denoted IOP1) was recorded, followed by systemic administration of acetazolamide (16 mg/kg) via intravenous or intramuscular injection to suppress aqueous humor production. Fluorescence intensity and IOP were reassessed at 45-, 90-, and 135-min post-administration. Fluorophotometric outflow facility (Cfl) was calculated at each timepoint as the ratio of the change in Fa to the change in IOP following acetazolamide administration. 18 If Fa or IOP decreased by <0.0001 μL/min or <0.0001 mmHg, respectively, Cfl was not calculated. The reported Cfl represents the average of two or three valid values; if only one or no valid Cfl value was available, it was excluded.
Uveoscleral outflow (Fus) was determined using a modified Goldmann equation: Fus = Fa − Cfl × (IOP1 − Pev), where Pev denotes episcleral venous pressure. Due to the inability to noninvasively measure Pev in monkeys with highly pigmented conjunctiva, its value was estimated. Based on previous literature, 19 Pev was varied between 14 and 18 mmHg, and the minimum value yielding positive Fus in all subjects was adopted, resulting in a final Pev estimate of 17 mmHg. Fus could not be calculated if any variable required for the equation was missing.
A total of 15 monkeys were included in the study; however, Cfl could be determined in only 8 animals. Thus, reported values represent the mean ± SEM of these 8 animals.
Tonography
Six hours after instillation of the test solution, monkeys were sedated with intramuscular injections of ketamine (9.2 mg/kg) and xylazine (0.3 mg/kg). Topical anesthesia was applied using 0.4% oxybuprocaine hydrochloride. IOP was initially measured in the seated position, followed by the supine position, using a pneumatonometer. The values were entered into the tonography program of the device. A 10-g-weighted probe was applied to the cornea, and IOP was recorded continuously for 2 min. Outflow facility (Cton) was calculated by the tonography software, and the results are presented as mean + SEM.
Statistical analysis
All statistical analyses were performed using EXSUS software versions 8.0.0, 10.0.3, or 10.0.7, or Microsoft Excel for Microsoft 365. For IOP and tonography data, variance equality was assessed using the F-test, followed by Student’s t-test or Aspin–Welch t-test as appropriate. The EP3 antagonist studies were analyzed using repeated measures or paired t-tests. Paired t-tests were used for the AHD data. For analyses in Supplementary Fig. S1, Bartlett’s test for homogeneity of variances was followed by Dunnett’s or Steel’s test. Statistical significance was defined as P < 0.05.
Results
Agonistic activity of sepetaprost and latanoprost at FP and EP3 receptors
ONO-AG-367 exhibited potent agonistic activity at both FP and EP3 receptors, with EC50 values of 1.6 and 38 nM, respectively (Table 1). In contrast, latanoprost acid showed strong agonistic activity exclusively at the FP receptor (EC50 = 1.2 nM), with negligible activity at the EP3 receptor.
Agonist Activities (EC50) of ONO-AG-367 and Latanoprost Acid to Human FP and EP3 Receptors

Data represent geometric mean of three independent measurements with duplicates.
IOP-lowering effect of sepetaprost and latanoprost in ocular normotensive monkeys
The IOP-lowering effects of sepetaprost and latanoprost were evaluated in ocular normotensive monkeys following single topical administration. IOP was measured over time, and changes from baseline were calculated (Fig. 1). Baseline IOP values (mean ± SEM) ranged from 19.0 ± 0.6 to 19.5 ± 0.7 mmHg, with no significant differences between vehicle and test solution groups. Sepetaprost significantly reduced IOP at 8, 10, 12, 26, and 28 h post-administration, while latanoprost induced significant reductions only at 8 and 12 h. The IOP-lowering effect of sepetaprost was consistently greater than that of latanoprost at all time points.
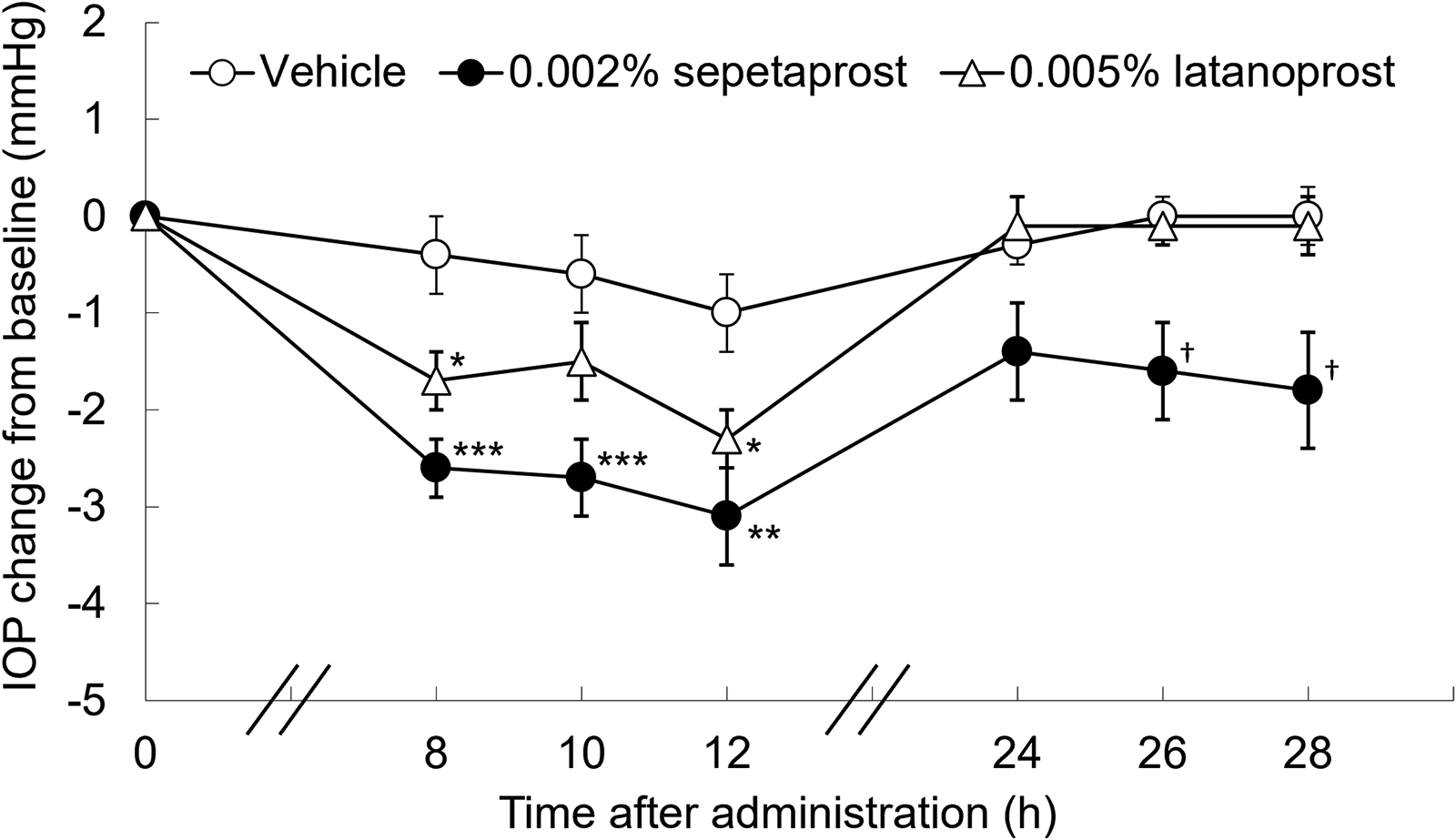
Data represent mean ± SEM of 10 eyes. *P < 0.05, **P < 0.01, ***P < 0.001, compared with vehicle-treated group (Student’s t-test). †P < 0.05, compared with vehicle-treated group (Aspin–Welch’s t-test). SEM, standard error of the mean.
Effects of EP3 antagonism on sepetaprost-induced IOP reduction in ocular normotensive monkeys
Despite similar FP receptor agonism between ONO-AG-367 and latanoprost acid (Table 1), sepetaprost demonstrated superior IOP-lowering (Fig. 1), suggesting a contribution of EP3 receptor activation. To evaluate this, monkeys received intracameral injection of the EP3 antagonist ONO-AE3-240 5 min prior to topical sepetaprost. IOP and IOP changes from baseline were recorded (Table 2). Baseline IOP values (mean ± SEM) ranged from 15.5 ± 0.7 to 15.9 ± 0.8 mmHg, with no significant differences among groups. The control group exhibited a significant IOP reduction at 24 h, likely due to the effects of intracameral injection or general anesthesia. To isolate treatment effects, IOP changes in sepetaprost- and sepetaprost plus EP3 antagonist-treated groups were normalized to the control (Fig. 2). There were no significant differences at 8 and 24 h; however, at 26 h, the IOP reduction was significantly smaller in the sepetaprost plus EP3 antagonist group compared with the sepetaprost-only group.
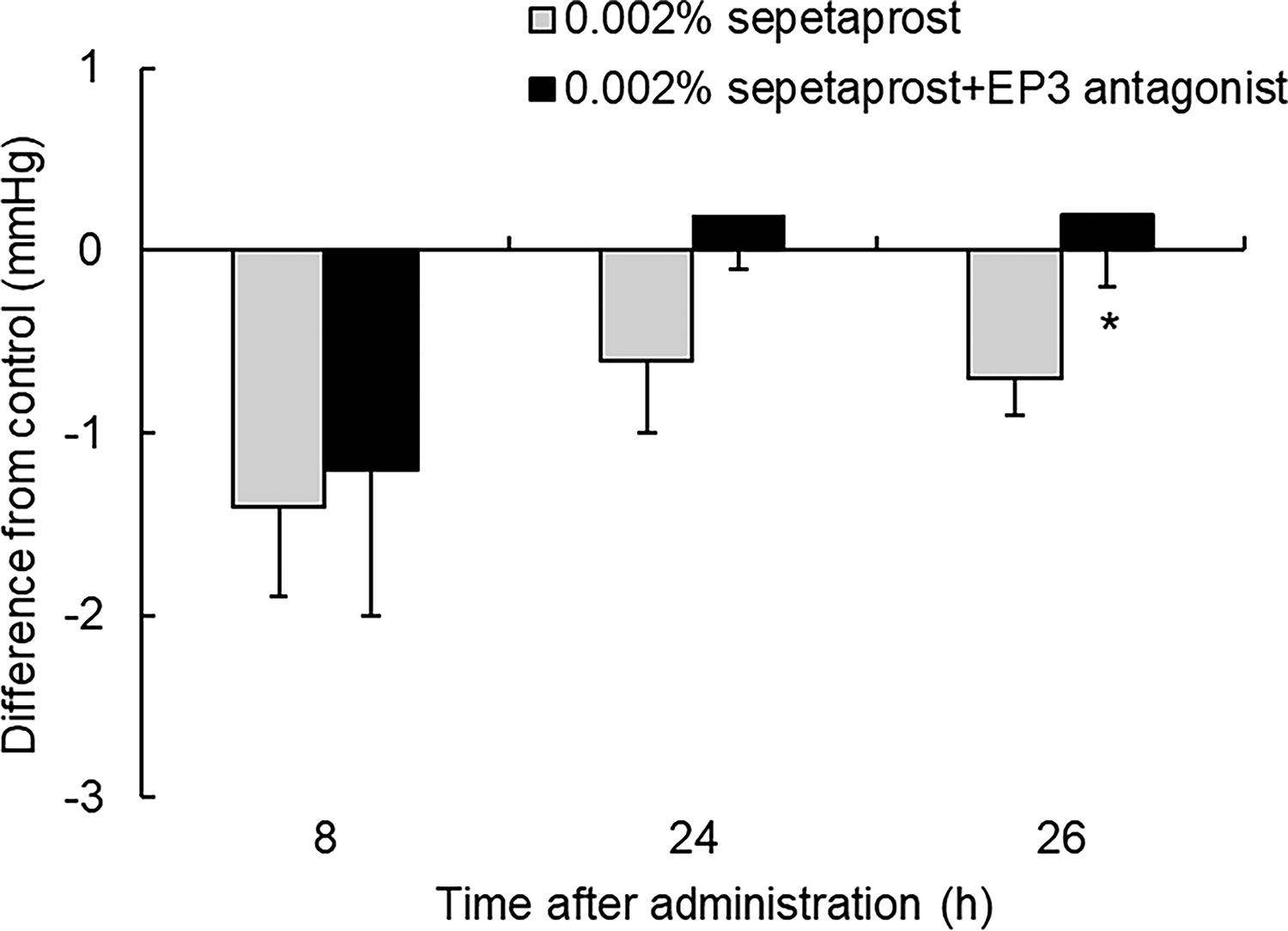
Difference from control was calculated by subtracting IOP changes from baseline of control group from that of 0.002% sepetaprost and 0.002% sepetaprost+EP3 antagonist groups. Data represent mean ± SEM of seven eyes only, for visual clarity. *P < 0.05, compared with 0.002% sepetaprost-treated group (paired t-test). IOP, intraocular pressure.
Effects of EP3 Antagonist on IOP-Lowering Effects of 0.002% Sepetaprost in Ocular Normotensive Monkeys

Vehicle for IC or EP3 antagonist (ONO-AE3-240) was intracamerally injected 5 min prior to vehicle or 0.002% sepetaprost ocular instillation.
IOP changes from baseline were calculated.
Saline containing 0.5% DMSO was used as vehicle for IC. Data represent mean ± SEM of seven eyes.
*P < 0.05, compared with baseline IOP (repeated measures t-test).
IC, Intracameral injection; IOP, intraocular pressure; SEM, standard error of the mean.
Effects of sepetaprost on AHD in monkeys
To investigate the mechanism underlying sepetaprost-induced IOP reduction, AHD were assessed in ocular hypertensive monkeys following once-daily sepetaprost instillation for 7 days (Table 3). Aqueous humor flow did not differ significantly between sepetaprost and vehicle groups. Although not statistically significant, sepetaprost numerically increased outflow facility from 0.25 ± 0.04 to 0.43 ± 0.13 μL/min/mmHg (68.4% increase; P = 0.199) and uveoscleral outflow from 2.15 ± 0.56 to 3.68 ± 1.24 μL/min (70.7% increase; P = 0.239). The absence of statistical significance may be attributed to the limited number of animals satisfying the strict criteria for calculating Cfl.
Effect of Once Daily 0.002% Sepetaprost Instillation for 7 Days on Aqueous Humor Dynamics in Ocular Hypertensive Monkeys

Aqueous humor parameters are expressed as the mean ± SEM of eight eyes.
P values were calculated by two-paired t-test between the vehicle and 0.002% sepetaprost-treated groups.
To further explore the increase in outflow facility, tonography was performed after a single administration of sepetaprost or latanoprost in ocular normotensive monkeys (Fig. 3). Sepetaprost significantly enhanced outflow facility, whereas latanoprost showed no significant change relative to vehicle, suggesting distinct IOP-lowering mechanisms.
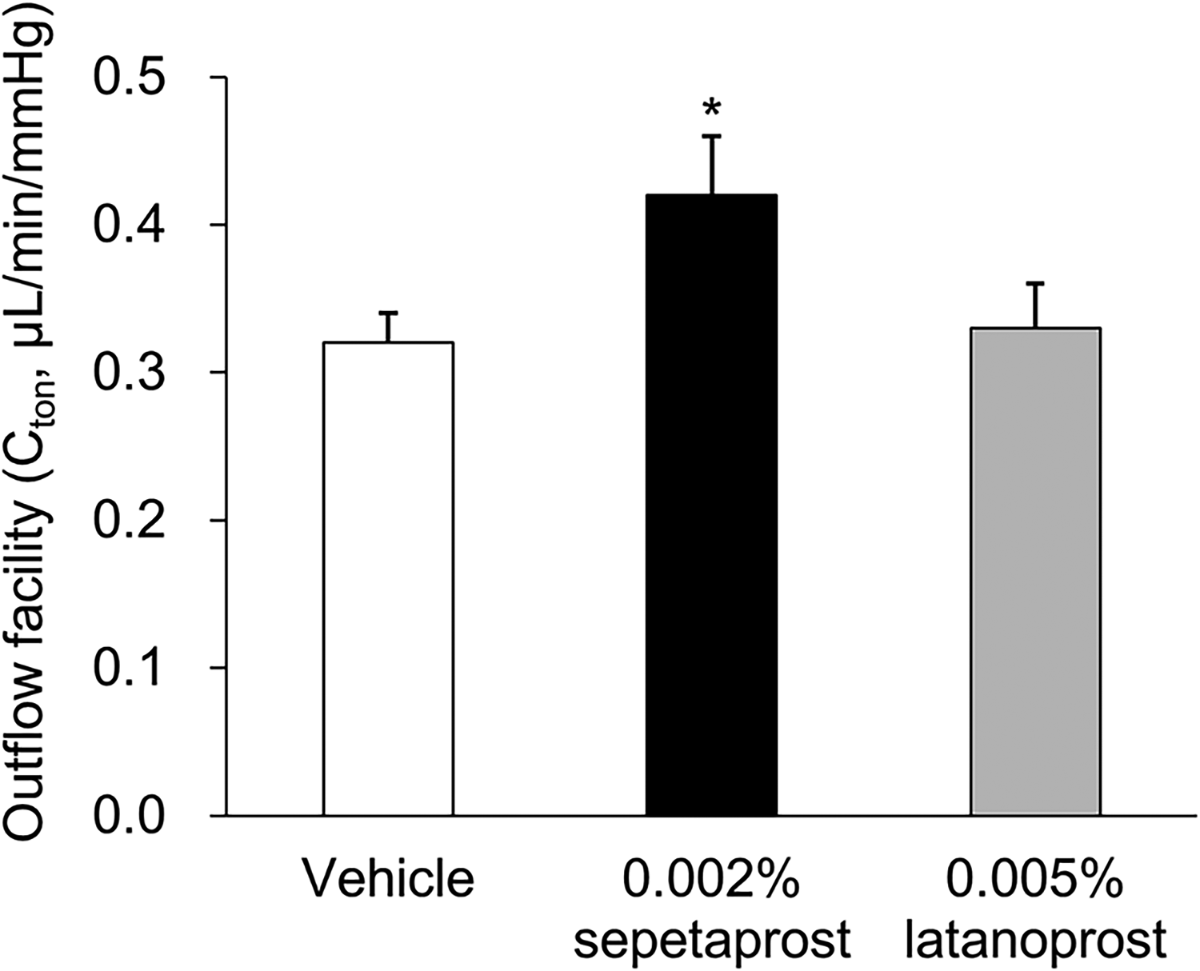
Outflow facility was evaluated by tonography at 6 h after administration. Data represent mean + SEM of 20 eyes only, for visual clarity. *P < 0.05, compared with vehicle-treated group (Aspin–Welch’s t-test).
IOP-lowering effect of sepetaprost in latanoprost low-responder monkeys
Given its distinct mechanism, we hypothesized that sepetaprost may be effective in latanoprost low-responder monkeys. Low responders were defined as those with an IOP reduction of <1 mmHg after latanoprost administration, based on a previous report. 20 Thirty-two monkeys were screened, and IOP responses to latanoprost were measured twice to ensure reproducibility (Fig. 4A). Seven animals that consistently exhibited an IOP reduction of <1 mmHg were classified as low responders (Fig. 4B). The IOP-lowering effect of sepetaprost was then evaluated in these animals, with measurements repeated for confirmation. Baseline IOP values (mean ± SEM) ranged from 16.7 ± 0.5 to 17.4 ± 0.6 mmHg, with no significant differences between vehicle and test groups. Sepetaprost significantly reduced IOP at 8, 10, 12, and 24 h post-administration in one or both tests (Fig. 5A–C), confirming efficacy in latanoprost low responders.
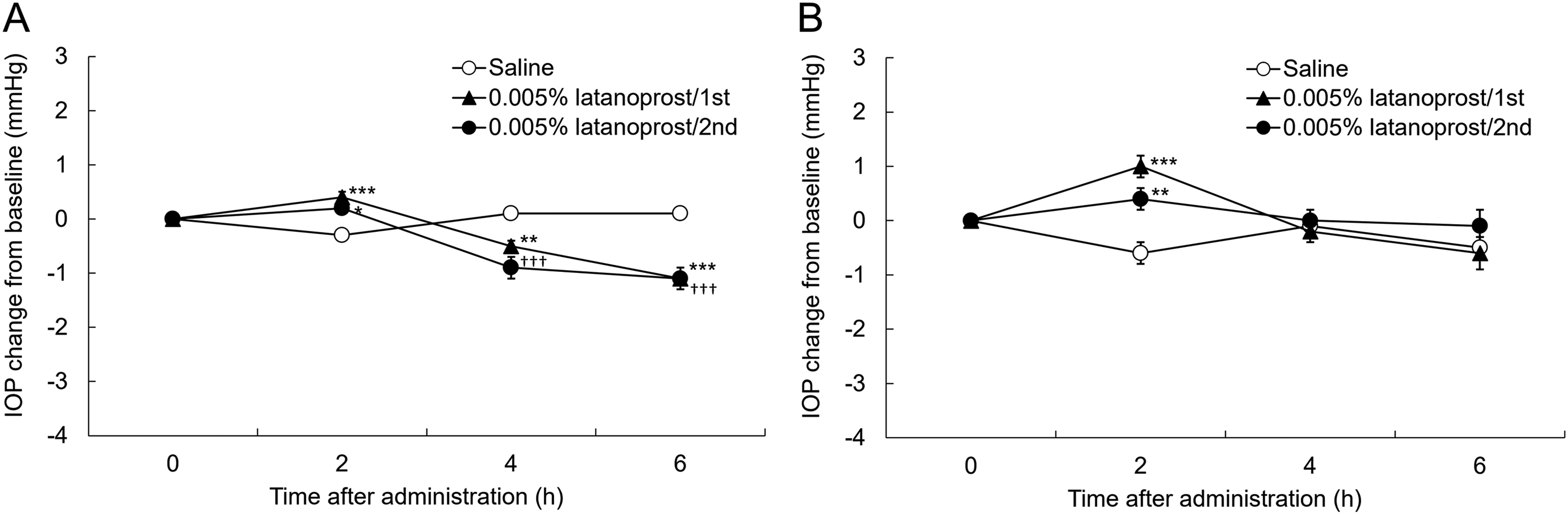
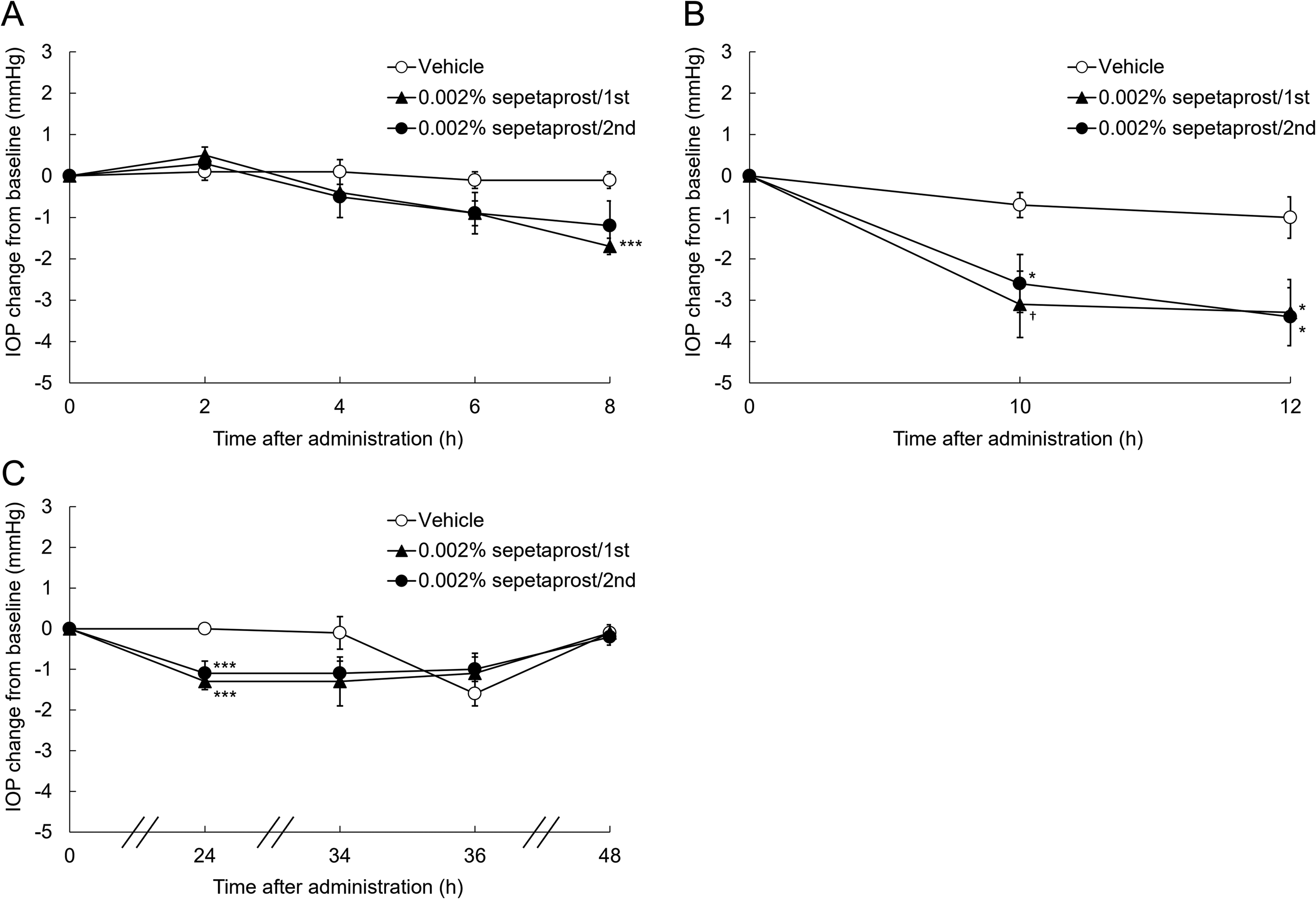
IOP changes from baseline after single administration of 0.002% sepetaprost in latanoprost low-responder monkeys at 2, 4, 6, and 8 h
Discussion
ONO-AG-367 and latanoprost acid exhibited comparable FP receptor agonist activity (Table 1); however, sepetaprost produced a more pronounced IOP-lowering effect than latanoprost (Fig. 1), implicating EP3 receptor activation in this enhanced efficacy. Administration of an EP3 antagonist attenuated the extended IOP-lowering effect of sepetaprost (Fig. 2), supporting the notion that EP3 agonism can prolong FP-mediated IOP reduction, as previously observed in rodents 12 and now confirmed in primates.
Sepetaprost did not alter aqueous humor production but increased both uveoscleral outflow and outflow facility in the AHD study (Table 3). FP receptor agonists primarily lower IOP by increasing uveoscleral outflow 21 ; thus, the observed increase in uveoscleral pathway is consistent with the pharmacologic profile of sepetaprost. The absence of additive IOP-lowering when sepetaprost was combined with latanoprost (Supplementary Fig. S1) further supports this mechanism. The measurement of outflow facility cannot distinguish trabecular from uveoscleral routes; rather, it represents the combined facility of both drainage pathways. However, in primates, the contribution of uveoscleral facility is small at normal to high pressures, so the outflow facility value primarily reflects that in the trabecular meshwork and downstream tissues. 21 Unlike latanoprost, sepetaprost significantly increased outflow facility in the tonography study (Fig. 3), suggesting it can reduce the resistance to drainage through both uveoscleral and trabecular outflow pathways.
Although topical EP3 receptor agonists alone are known to have minimal impact on IOP in rodents and monkeys,12,22,23 there are reports indicating that they can lower IOP in rabbits. 24 This suggests that species differences should be taken into account when understanding the role of prostanoid receptors. In monkeys, where EP3 activation alone does not directly contribute to IOP reduction, our findings imply that EP3 receptor activation may potentiate the pharmacological effects of FP receptor stimulation. Both FP and EP3 receptors are expressed in ocular tissues such as the ciliary muscle and trabecular meshwork cells. 25 FP agonists reduce IOP by promoting uveoscleral outflow through relaxation of the ciliary muscle and remodeling of the extracellular matrix (ECM) via induction of matrix metalloproteinase (MMP) expression.26–28 EP3 receptor activation has also been reported to enhance MMP expression, 29 suggesting a synergistic effect in ECM remodeling. Hence, EP3 agonism may amplify MMP-mediated ECM remodeling initiated by FP receptor activation, contributing to prolonged IOP reduction. Additionally, EP3 agonism may improve trabecular outflow facility by promoting ECM turnover in the trabecular meshwork. 30 Although latanoprost increases MMP expression in this tissue, 31 it may not induce sufficient remodeling to enhance trabecular facility. Sepetaprost, by engaging both FP and EP3 pathways, may achieve more robust MMP upregulation, thus reducing IOP more effectively and sustainably. Sepetaprost significantly lowered IOP in latanoprost low-responder monkeys (Figs. 4 and 5), suggesting potential utility in patients with suboptimal response to FP agonists. Since FP agonists act predominantly by enhancing uveoscleral outflow, 21 anatomical or molecular differences in this pathway may underlie nonresponse. One study showed that individuals with high baseline uveoscleral outflow exhibit minimal IOP reduction with latanoprost. 32 Alternatively, structural abnormalities or impaired remodeling capacity may limit latanoprost efficacy. Sepetaprost’s effect on trabecular outflow could compensate for such limitations. Another plausible explanation involves reduced FP receptor expression. Single-nucleotide polymorphisms in the FP receptor gene correlate with attenuated latanoprost responses, likely via decreased transcription. 33 Through EP3-mediated enhancement of FP signaling, sepetaprost may overcome low FP receptor expression and induce sufficient ECM remodeling to lower IOP.
Several limitations warrant consideration. First, while tonography indicated increased outflow facility with sepetaprost (Fig. 3), this method is influenced by confounding factors such as ocular rigidity and pseudofacility. 21 However, fluorophotometric measurements, unaffected by these confounders, also demonstrated numerical increases in outflow facility (Table 3), supporting the conclusion that sepetaprost can improve facility in the trabecular outflow pathway. In this study, strict criteria for Cfl calculation were applied to exclude values affected by animal conditions, limiting analyzable data. To improve data accuracy in future studies, stricter sample size planning, refinement of implementation criteria for potential re-measurements, and consideration of imputation for missing data may be effective. Second, studies were conducted using both male and female monkeys based on availability, precluding sex-balanced cohorts. We assumed no sex-related differences in IOP or drug response. Third, the mechanisms underlying latanoprost nonresponse in monkeys may not fully recapitulate those in humans; clinical trials are needed to confirm sepetaprost’s efficacy in this population. Finally, although our findings suggest EP3 agonism prolongs IOP-lowering, the precise molecular mechanisms remain undefined. Further investigation is needed to elucidate how EP3 agonism extends IOP-lowering at the molecular level, including the specific roles of EP3 receptor isoforms.
Conclusion
Sepetaprost provided greater IOP-lowering effects compared to latanoprost, with EP3 receptor agonism extending the effects beyond 24 h. Our AHD and tonography studies suggest that, unlike latanoprost, sepetaprost lowers IOP by increasing uveoscleral outflow and improving trabecular outflow facility in nonhuman primates. Sepetaprost also demonstrated efficacy in latanoprost low-responder monkeys. Collectively, these findings highlight the therapeutic potential of sepetaprost as a first-line therapy or as an alternative for patients inadequately controlled with FP agonists.
Authors’ Contributions
K.M., E.K., Y.Y., M.F., A.S., and M.K. contributed to the study design, data interpretation, and article preparation. C.B.T. and S.F. were responsible for designing, conducting, analyzing, and interpreting several of the experiments under a research agreement with Santen Pharmaceutical Co., Ltd., and also contributed to article review. All the authors reviewed and approved the final article.
Footnotes
Acknowledgment
The authors thank Lisa Reid, Toni Goeser, Masafumi Mieda, Naoko Yamashita, Isao Matsuoka, and Hitoshi Nakazawa for their excellent technical assistance in handling animals and collecting data.
Author Disclosure Statement
K.M., E.K., Y.Y., M.F., A.S., and M.K. are employees of Santen Pharmaceutical Co., Ltd., which funded the study. C.B.T. and S.F. conducted the aqueous humor dynamics study under a research agreement with Santen Pharmaceutical Co., Ltd. and received institutional research support. No personal financial compensation was received. The authors declare no other conflicts of interest.
Funding Information
The aqueous humor dynamics study was funded by Santen Pharmaceutical Co., Ltd. as part of its internal research and development activities. Experimental work was conducted under a research agreement with the University of Nebraska Medical Center.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
