Abstract
We extend Li et al.’s investigation of aqueous humor (AH) metabolomics in sequential cataract surgery by referencing our prior study on interocular symmetry/asymmetry in AH metabolomic profiles from simultaneous bilateral cataract surgery in emmetropic patients, which demonstrated similar AH compositions in fellow eyes. We also illustrate variability with 2 sequential-surgery cases and highlight the most and least variable metabolites across 6 biochemical classes. Taken together with Li et al., these observations support careful attribution of second-eye changes to surgery versus biology.
Purpose:
To extend the findings of Li et al. on AH metabolomics in sequential cataract surgery by incorporating reference data on interocular symmetry/asymmetry in AH metabolomic profiles and illustrating variability in sequential cases.
Methods:
We drew on our prior study of simultaneous bilateral cataract surgery in emmetropic patients, which demonstrated high interocular similarity, and examined AH metabolomic variability in 2 patients undergoing sequential cataract surgery.
Results:
Baseline interocular comparisons highlight metabolic symmetry in the AH among patients undergoing simultaneous cataract surgery. In 2 additional cases, we identified the most and least variable metabolites across 6 biochemical classes among patients undergoing sequential cataract surgery, complementing the observations of Li et al.
Conclusion:
Our reference data help contextualize Li et al.’s results. Although based on limited cases, our findings emphasize the need for caution when interpreting AH metabolomics in sequential surgery to distinguish true intra- and inter-individual biological variability from potential surgical effects on the second eye. Multimodal approaches integrating metabolomic and vascular metrics may improve biomarker selection and inform surgical timing.
Purpose
We congratulate Li et al. on their publication, “Changes in aqueous humor cytokines and metabolomics in contralateral eye after unilateral cataract surgery,” 1 published in BMC Ophthalmology. Their findings on altered cytokine and metabolite concentrations in the aqueous humor (AH) are pivotal in understanding the systemic and localized effects of surgical intervention on ocular health. These observations may influence risk assessment for complications, scheduling of second-eye surgeries, and postoperative healing monitoring.
The debate over simultaneous (same-day) versus sequential (separate-day) bilateral cataract surgery intensified during COVID-19 to reduce visits. Prior studies have addressed medical risks, patient comfort, and cost-effectiveness,2,3 while Li et al. provided metabolomic insights.
Building on Li et al., we previously reported in “Extent of interocular (a)symmetry based on the metabolomic profile of human aqueous humor” that simultaneous bilateral cataract surgery in emmetropic patients yielded highly similar AH composition in both eyes, with 67 metabolites precisely quantified. 4 These baseline symmetry data offer a vital reference for interpreting Li et al.’s results, particularly for metabolites with high interindividual (between patients) and intraindividual (right versus left eye) variability. Identifying such metabolites is crucial when proposing candidate biomarkers across diseases.
Aside from Li et al.’s study of 11 patients over a 2-week interval, no reports address AH metabolomics in sequential cataract surgery, likely due to the invasiveness of AH collection and logistical challenges. Here, we report on 2 patients who underwent sequential cataract surgery with 6- and 7-week intervals between surgeries.
Methods
Metabolite quantification was performed using liquid chromatography-tandem mass spectrometry (LC–MS/MS) (Agilent 6470) with the AbsoluteIDQ® p180 kit (Biocrates Life Sciences AG, Innsbruck, Austria). Sample preparation followed the manufacturer’s protocol with minor modifications, using 30 µL of AH instead of 10 µL recommended for plasma/serum. Data were processed using MetIDQ software (version Oxygen DB110-3005-290, Biocrates, Life Science AG, Innsbruck, Austria), with quality control-based normalization, exclusion of metabolites with a coefficient of variation >30% or those detected in <70% of samples, and imputation of values below the limit of detection (LOD) using extrapolated concentrations. Supplementary Data S1 describes the stepwise preprocessing, LOD filtering, and ranking procedures applied in the variability analysis. This methodology does not resolve the exact fatty acid composition, position of double bonds, or the presence of possible isobars/isomers. Consequently, the reported values should be interpreted as quantitative measures of summed species rather than fully structurally defined lipids.
Using our targeted AH metabolomics protocol, 5 we quantified 73 metabolites in Patient 1 and 81 in Patient 2 across 6 classes: acylcarnitines, amino acids, biogenic amines, glycerophospholipids, sphingolipids, and sugars. Patient characteristics and methods are summarized in Supplementary Table S1 in Supplementary Data S1; raw data and paired-sample % differences are provided in Supplementary Data S1 and S2. We defined the absolute relative % difference as |(second-eye concentration − first-eye concentration)/second-eye concentration| × 100%. The study was conducted in line with the Declaration of Helsinki and approved by the Local Bioethics Committee of the Medical University of Bialystok (decisions no. R-I-002/154/2014 and R-I-002/140/2018).
Results
Although 2 cases cannot yield definitive conclusions, they illustrate real-world intra-individual variability. Exploration of variability sources (comorbidities, medications) lies beyond our scope, but these findings, and those of Li et al., should be interpreted with caution for highly variable metabolites.
For each patient and each of the 6 classes, the median of all individual metabolites’ percent difference values within that class was calculated (Fig. 1). Class-stratified results for all metabolites are presented in Supplementary Figure S1 in Supplementary Data S1. Supplementary Table S2 in Supplementary Data S1 presents the top 10 and bottom 10 metabolites percentage difference for each patient. Glycerophospholipids and sphingolipids display the greatest variability, especially in Patient 1, whereas sugars vary least. Changes in acylcarnitines, biogenic amines, and amino acids are moderate. Overall, Patient 2’s lower average differences suggest greater metabolic stability. As detailed in Supplementary Table S2, glycerophospholipids dominated among the most variable metabolites, whereas amino acids and acylcarnitines were typically more stable. Supplementary Figure S1 further shows that the distribution of variability within classes was broader in Patient 1 than in Patient 2, which might result from their different clinical backgrounds. As shown in Supplementary Table S1, Patient 1 presented multiple systemic comorbidities, while Patient 2 was otherwise healthy. We presume that these clinical differences may partly explain the greater metabolic variability observed in Patient 1.
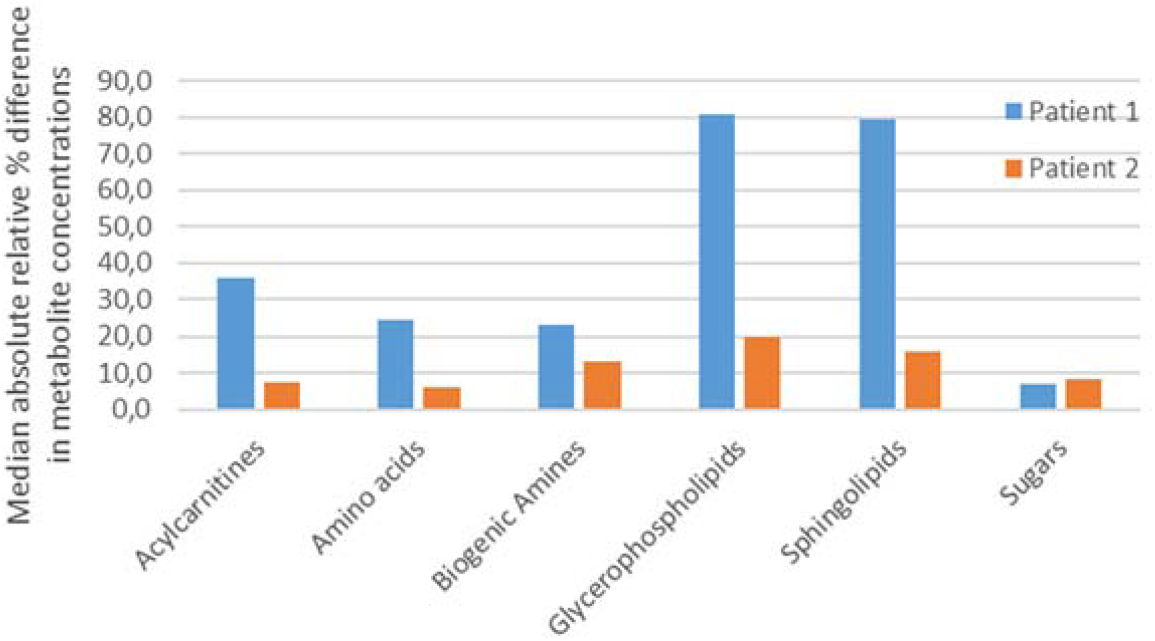
Median percent difference in metabolite concentrations by metabolite class. n = 67 metabolites common to Patients 1 and 2.
Li et al. propose delaying second-eye surgery by over 2 weeks to minimize the metabolomic impacts on clinical outcomes in the second eye; however, we suggest that prospective studies measuring macular thickness, choroidal metrics, and ocular blood flow before and after the second procedure are needed to determine the clinical translation of the metabolomic changes.
Conclusions
In summary, data from Li et al.’s study and our 2-case report provide unique insights into AH metabolomics in sequential cataract surgery. We advocate for cautious interpretation of AH metabolomics results to distinguish true intra- and inter-individual biological variability from surgical effects on the second eye. Further multimodal studies integrating metabolomic and vascular metrics are needed to optimize surgical timing and biomarker selection.
Authors’ Contributions
D.A.D.: Clinical oversight, patient recruitment, aqueous humor collection, conceptualization of the comment, design of the case reports, overall supervision of the project, formal data analysis and statistical interpretation, drafting of the article. J.G.: Formal data analysis and statistical interpretation, drafting of the article. P.M., K.P., and A.G.: Processing of aqueous humor samples, targeted metabolomics experiments, data curation. M.C.: Supervision of metabolomics methodology, critical revision of the article. J.K. and A.K.: Critical revision of the article. All authors read and approved the final article.
Footnotes
Acknowledgments
Portions of the text were edited with the assistance of an AI language model to improve clarity and flow; the authors accept full responsibility for the content.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This work was supported by the National Science Centre, Poland (grant ID 2021/05/X/NZ5/00723).
Ethics Approval and Consent to Participate
This study was approved by the Medical Ethics Committee of the Medical University of Bialystok, Poland (decisions nos. R-I-002/154/2014 and R-I-002/140/2018) and was conducted in accordance with the Declaration of Helsinki. All patients provided written informed consent to participate in the study.
Availability of Data and Materials
Data are provided within the article and Supplementary Data S1 and ![]() .
.
Supplemental Material
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
