Abstract
Space exploration exposes astronauts to unique conditions such as microgravity and space radiation, potentially influencing gene expression and triggering carcinogenesis. Paradoxically, these extreme environments could uncover other pro-treatment aspects of cancer biology. Despite numerous reviews addressing these aspects in isolation, a comprehensive synthesis of the effects of space stressors on cancer development is incomplete. This scoping review aims to provide a holistic perspective on the influence of spaceflight and associated stressors—including gravitation (hyper- and microgravity), radiation, and vibration—on the potential for cancer development and altered cellular mechanisms. We adhered to the PRISMA-ScR checklist for our review. A multitiered search strategy was employed in English, starting with a preliminary keyword identification in Google Scholar and PubMed. Subsequently, the main search was conducted across five databases—Cochrane, Embase, PubMed, Scopus, and Web of Science—until 22 February 2024. All included studies were thoroughly assessed by two independent reviewers. Of the 15,553 identified articles, 158 were deemed eligible. The majority (92%) were experimental studies, predominantly cell-based (74.17%). Breast (15.19%), thyroid (13.92%), and hematopoietic (11.40%) cancers were the most frequently examined. Spaceflight stressors could affect different biological systems variably, with microgravity impacting spatial growth and metastasis, and cosmic radiation exerting both tumor-suppressive and mutagenic effects. Our findings highlight the need for large-scale, prolonged analog studies mimicking space conditions to enhance mission safety and shed light on the nuanced effects of space stressors on cancer. Additionally, further extensive studies need to be performed in the true weightlessness of spaceflight, both animal-based and on human tissue (cell cultures and potentially whole perfusion organ models), in addition to crew pre/intra/post-flight long-duration evaluations. Furthermore, this unique research avenue may reveal cancer cell sensitivities to these stressors, opening new pathways for innovative therapeutic strategies in cancer treatment.
Keywords
Introduction
Cancer is a leading cause of morbidity and mortality worldwide, resulting from various cell changes caused by endogenous and exogenous factors. 1 The extreme environment of space is a hostile setting that influences carcinogenesis.2,3 Evidence from aerospace medical research shows that, despite the natural adaptive capacity of the human body, the space environment exposes astronauts to multiple stressors challenging their health through various mechanisms.4–6 These include radiation, fluctuations in gravity force, and vibration.2,3
Evidence of space travel’s impact on cancer is inconclusive due to the variability of study methods and sample size. While some studies report no increased risk of cancer mortality among astronauts compared to controls, 7 others identified genetic mutations linked to cancer after space travel. 8 Recently, research revealed that spaceflight might inhibit tumor growth by upregulating certain tumor suppressor genes, highlighting the potential pro-treatment aspects of these extreme environments. 9 Additionally, parabolic flight studies have revealed that some cells and their gene expression are very sensitive to physical forces, including vibration, which results in a higher chance of carcinogenesis. 10
In addition, microgravity contributes to carcinogenesis by altering gene expression,2,11 cytoskeletal structure, signal transduction, and cell membrane permeability. 12 In contrast, some findings indicate that microgravity may improve cancer survival rates and aid in its regression. 5 Moreover, space radiation, especially ionizing types, can induce fibrosis through inflammatory responses 5 and DNA damage 2 while also negatively affecting the subsequent DNA repair, potentially resulting in carcinogenesis. 2
Furthermore, Cortés-Sánchez et al. reported that radiation and microgravity, when assessed in combination, can increase cancer risk. 2 Evidence also indicates that microgravity can modify cellular function, the cytoskeleton, and especially the morphology of cells. 13 According to a study conducted on human breast cancer cells, microgravity impacts adhesive molecules, the F-actin network, and the microtubule cytoskeleton involved in cell adhesion and migration. 14 Exposure of prostate cancer cells to microgravity induced by a random positioning machine (RPM) can result in the formation of two cellular populations: the adherent cells (ADs) that grow in a 2D monolayer and multicellular spheroids (MCSs) that detach from the cell culture flask and form 3D cellular structures. 15
Numerous studies investigated the independent effect of each space stressor on carcinogenesis; however, the literature lacks studies to assess the combined effect of various stressors, which is more representative of true spaceflight conditions experienced by crew members.
Conducting in vivo studies on humans or animals with active cancer is a less-discussed topic that can enrich our knowledge about cancer and long-duration spaceflight.2,11 In addition to the above-mentioned stressors, concerns exist regarding psychiatric and emotional stressors, cognitive impairment, the disruption of circadian rhythm, and the subsequent impact upon cortisol response and overall astronaut health. 16 On the other hand, the literature supports the potential role of prolonged psychological stress on cancer development and progression through various physiological and immune-mediated pathways. 17 Exploring the potential influence of psychological and mental stressors associated with space travel on cancer development remains an intriguing and important area of interest requiring further investigation.
We designed this study to systematically and critically review the available evidence regarding the effect of the space environment on cancer development. The results of this comprehensive study provide some degree of insight regarding space mission safety considerations and probable therapeutic or potential disease-mitigating effects in oncologic settings.
Methods
Review Question
We conducted a scoping review to investigate the current state of knowledge regarding the relationship between space missions and cancer. Our review question aimed to identify risk factors, signs and symptoms, pathophysiology, and gaps in knowledge related to this topic. We used the Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR). 18
Inclusion Criteria
To capture all studies, we included all types of original studies (cohort, RCT, case-control, case report, case series, and qualitative).
As we aimed to have a comprehensive view of this subject, all studies related to humans, animals, and cells, as well as those based on modeling and mathematics, were included.
Papers related to space and the space environment, with possible factors impacting the carcinogenesis process, were included to encompass all potential contributing factors.
Exclusion Criteria
Non-English-language papers
Studies not peer-reviewed
Unavailable full text
Reviews and editorials, viewpoints, guidelines, letters to the editor, and commentaries, as they were nonoriginal
Search Strategy
We performed a multistep search strategy. On May 10, 2022, a limited preliminary search was conducted by HA in multiple databases, including Google Scholar and PubMed, to identify the appropriate keywords. The following keywords for that preliminary query were used: (Microgravity OR Spaceflight OR Gravitation OR Weightlessness OR Atmosphere OR Cosmos OR “cosmic radiation”) AND (Cancer OR onco* OR tumor OR carcino* OR hyperplas* OR Neoplas* OR Malignan*). Also, to explore the impacts of elements such as acute or chronic stress, disruptions in circadian rhythm, and psychological challenges faced by astronauts during prolonged space missions, we conducted a preliminary search in PubMed using specific keywords (Supplementary Table 2).
On August 31, 2022, HA adopted search strategies to seek relevant articles in Cochrane Library, PubMed, Embase, Scopus, and the Web of Sciences. We applied available filters on each database to retrieve more relevant results (Supplementary Table 1). The results were then reviewed using Endnote 20. 19 Following the deletion of duplicates, two independent authors (MFK & NN) separately screened the titles and abstracts and removed irrelevant studies according to the inclusion and exclusion criteria (Figure 1). Discrepancies about the inclusion/exclusion of any paper were resolved through group discussion. On February 22, 2024, the same steps were implemented to update the systematic literature search.
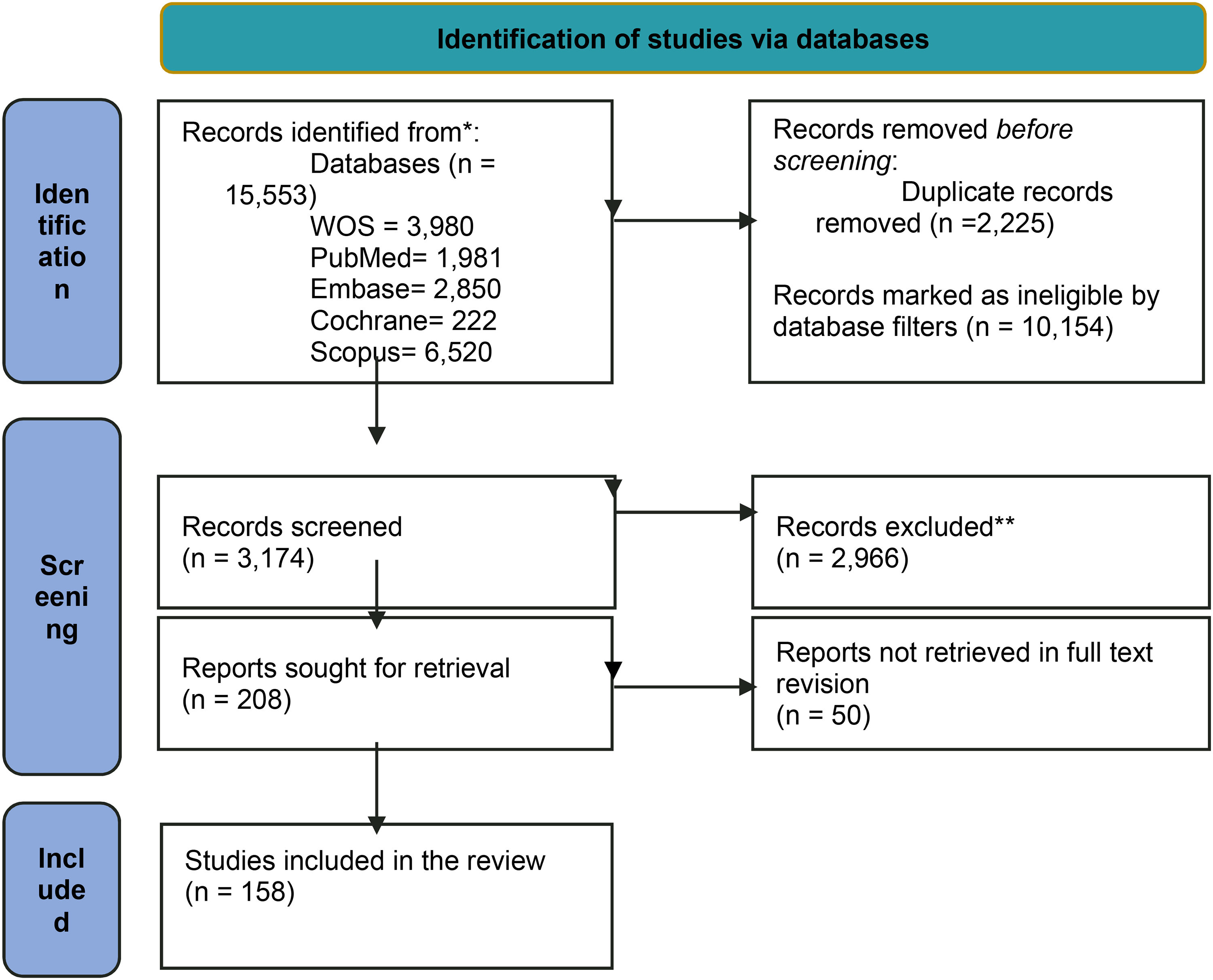
PRISMA 2020 Flow Diagram of the Present Study. 189
Data Management
Upon final article selection, the following elements were extracted from the included papers: primary author, publication date, methodology, study category (in silico, cellular, mathematical model, animal, and human), and type of cancer studied in the original investigations. The extraction form was designed in Excel, 20 and for each paper, two of the seven authors (HA, MFK, NN, MS, ZA, SAH, SMM) extracted the data independently. Discrepancies were resolved through group discussion.
Quality and Level of Evidence Assessment
Due to the methodological heterogeneity of the studies, an assessment tool introduced by Hawker et al. in 2002 was selected as the most suitable one to accommodate the majority of the studies. 21 This tool assesses articles by rating their titles, abstracts, introductions and aims, methods and data, sampling, data analysis, ethics and bias, results, transferability or generalizability, implications, and usefulness. Ratings ranged from Good (earning 4 points) to Very poor (earning 1 point). These ratings were collected to make up a total score ranging from 9 to 36, where 9 to 23 points were “low quality,” 24 to 29 points were “medium quality,” and 30 to 36 points were deemed “high quality.” 21 It is worth mentioning that the evaluation of quality and bias in mathematical modeling studies was not conducted due to their specific design.
Results
The search yielded 15,553 (13,837 and 1716 by the first and second searches, respectively) studies. Using database filters and deleting the duplicates, 3174 (2843 + 331) records were selected for title/abstract screening, resulting in the exclusion of 2974 (2660 + 306) records. Of the remaining 208 (183 + 25) records for full-text review, 158 (137 + 21) studies were ultimately included for data extraction (supplementary tables 3 and 4) based on the inclusion/exclusion criteria, as shown in the PRISMA diagram (Figure 1).
Our search on psychological stressors resulted in 29 studies retrieved from PubMed, but none of them were original studies specifically addressing the relevant topic.
Geographical Distribution of Studies
Studies were conducted in over 20 countries. Near 90% (139) of studies were performed in different individual countries, while 19 (12.02%) were carried out through international collaborations. The United States (n = 72), Germany (n = 33), China (n = 18), and Japan (n = 11) had the largest share of the included studies (Figure 2).
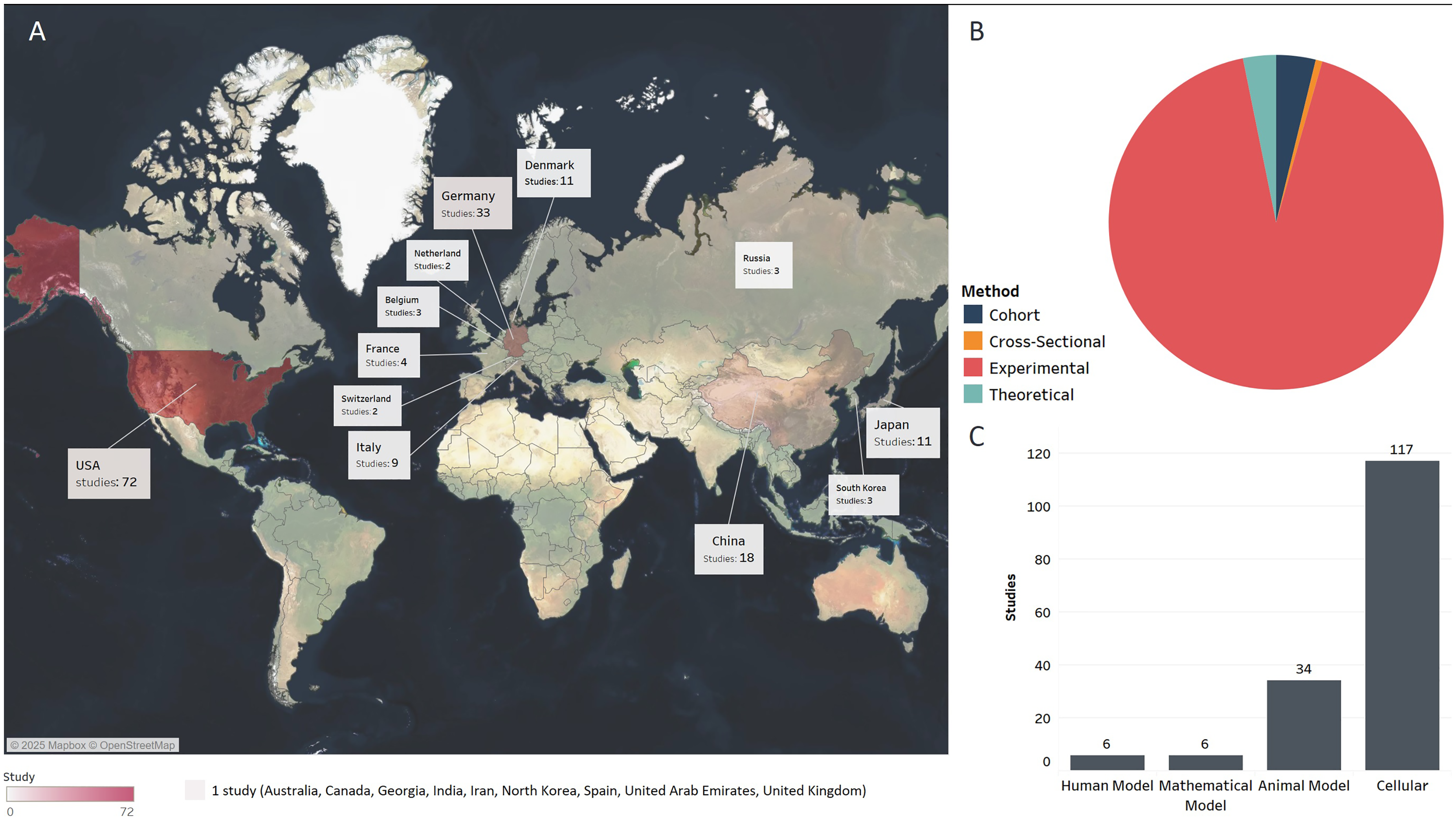
A. Geographical distribution of the studies. B. Distribution of publications among categories. C. Methods.
Methodologies and Categories of Studies
The studies varied in their methods, with a predominance of experimental studies (92%). Cohorts, cross-sectional, observational, and targeted effects modeling studies comprised 8% of the total share. Considering some studies used multiple methods, this included in vitro (n = 117), in vivo (n = 25), in silico (n = 6), and clinical (n = 6) (Figure 2).
Type of Cancer
Considering that some studies focus on multiple organs, the distribution of studies by cancer type is as follows, resulting in overlaps in the provided proportions. Breast (n = 24, 15.19%), thyroid (n = 22, 13.92%), hematopoietic (n = 18, 11.40%), and lung (n = 19, 12.02%) cancers were most frequently investigated. Urological (n = 13, 8.23%), male reproductive (n = 1, 0.63%), and female reproductive (n = 4, 2.53%) system; gastrointestinal (liver, gastric, and colorectal) (n = 14, 8.86%); neural (n = 9, 5.70%); skin (n = 4, 2.53%); and bone (n = 3, 1.90%) cancers were among the other cancers studied. Four animal studies investigated Harderian gland tumors. The other studies (n = 32, 20.25%) evaluated non-organ-specific aspects of carcinogenesis (Figure 3).
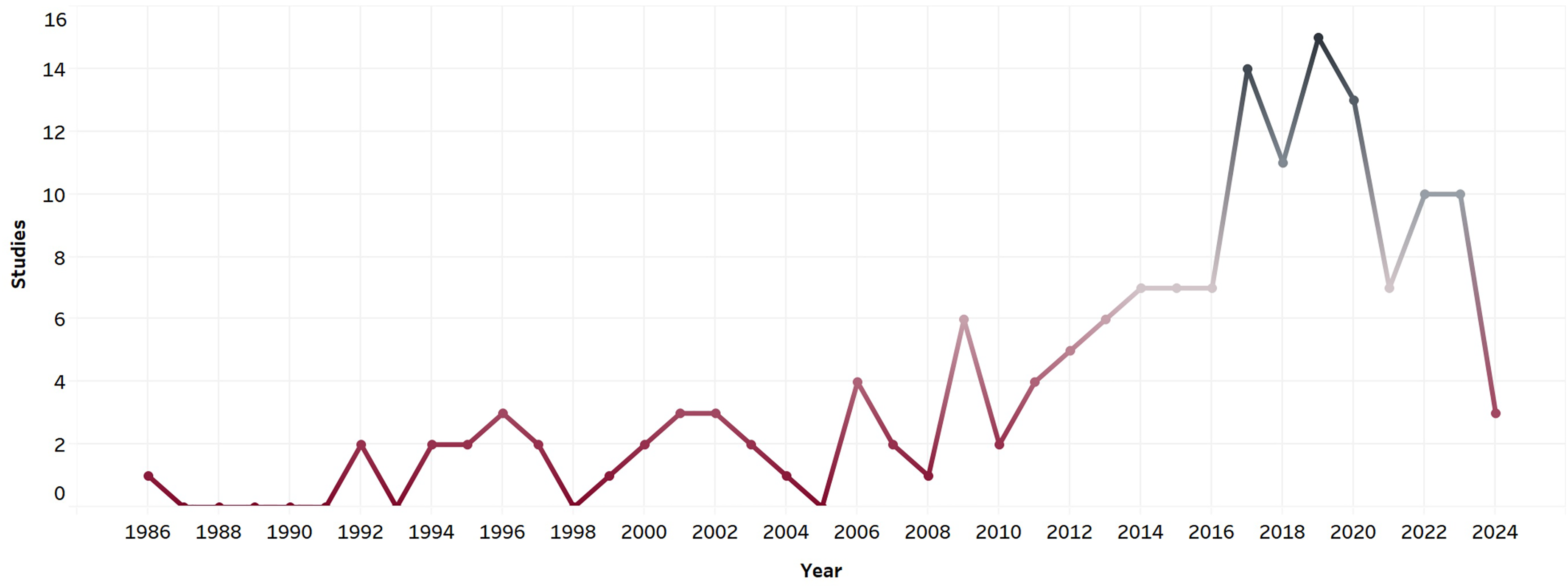
Time trend and studies on space oncology.
Time Trend of Studies
Overall, the number of studies on space oncology follows an increasing trend, with a higher slope after 2010. The number of included studies in 2020 has decreased, which can be due to the shift in medical research topics due to the COVID-19 pandemic or the fact that some papers may be in their final stages of publication or under review (Figure 4).
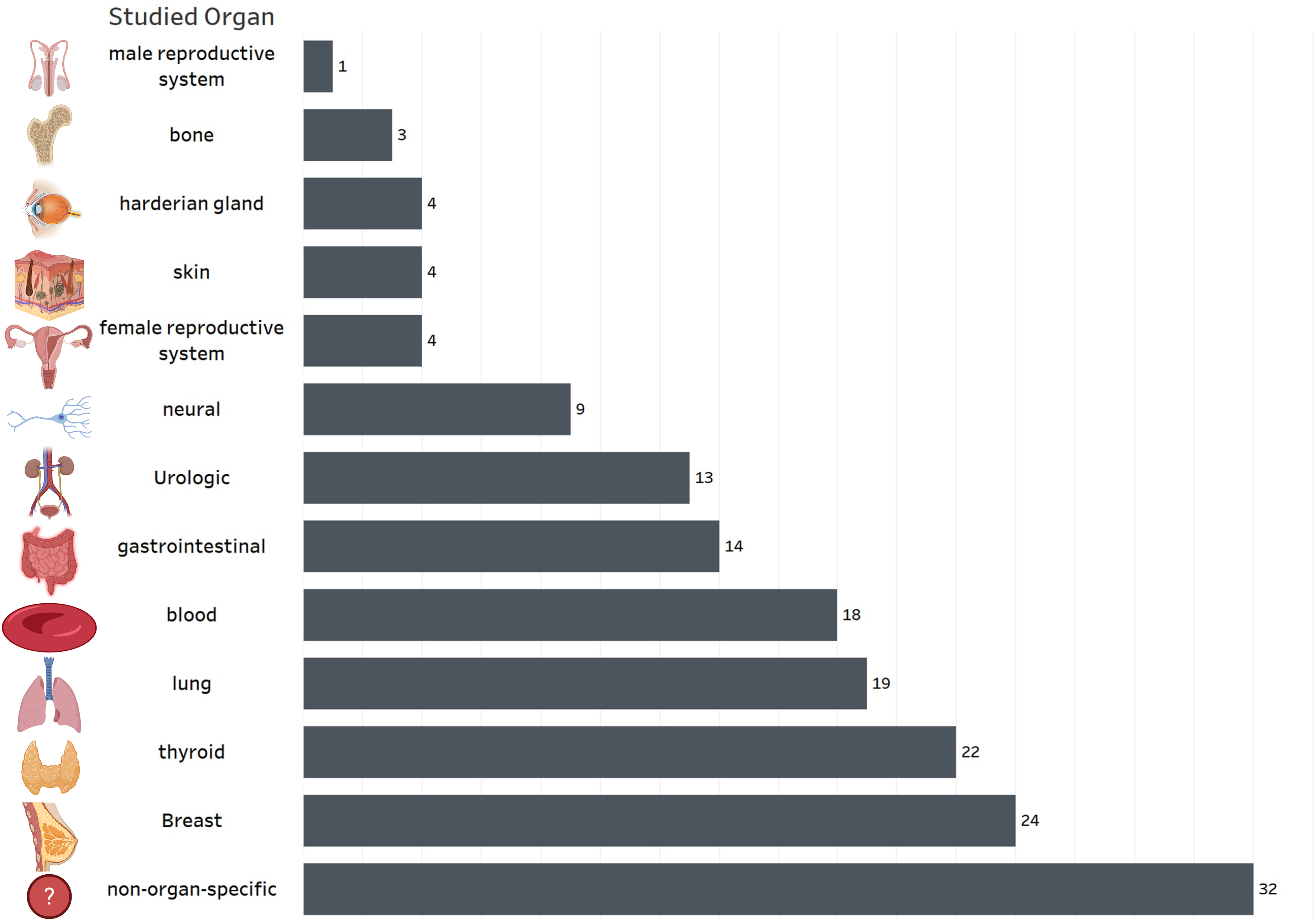
Distribution of publications based on studied organ.
Quality Assessment
Of the included studies, 92.71% (140 out of 151 non-mathematical studies) had high methodological quality, 7.28% (11 out of 151 non-mathematical studies) had medium quality, and zero had low quality. The ratings were calculated for each criterion: (i) analysis, ethics, and bias; (ii) introduction and aims, methods and data, sampling, transferability or generalizability, implications, and usefulness; and (iii) title, abstract, and results. Criterion (i) yielded a minimum rating of 1, (ii) a rating of 2, and (iii) a rating of 3 (supplementary files, Table 3).
The following paragraphs categorize the findings according to the cancer type and the stressors present in the space environment.
Microgravity & Cancer
Breast Cancer
Of 22 studies on breast cancer, 19 studies investigated the link between breast cancer and microgravity. Microgravity affected cancer cell lines on micro/macroscopic scales. 22 Microgravity could affect Michigan Cancer Foundation-7 (MCF-7) breast cancer cells by decreasing phosphotyrosine signaling, disrupting the perinuclear cytokeratin network and chromatin structure, prolonging mitosis, reducing proliferation through the cell-cycle blockage, and modifying microtubule organization.23,24 Using MCF-7 (less invasive) and MDA-MB-231 (highly invasive) cell lines, a study investigating prolonged simulated microgravity effects on breast cancer cells and the understanding of the MCS model in breast cancer patient contexts discovered a favorable connection between the actual metastatic microtumor environment and MCSs. This link involved factors like the cytoskeleton, extracellular matrix, and various other components, dependent on the type of breast cancer. 25 Keeping in mind the role of spatial cellular structure in cancer, simulated microgravity coincided with the downregulation of the genes involved in cellular shape, organization, and membrane interactions, resulting in multicellular spheroid formation. 26
Nassef et al.'s study on MDA-MB-231 cells, a triple-negative breast cancer (TNBC) cell line, revealed microgravity-downregulated ANXA2 and BAX genes while increasing pro-apoptosis factors, 27 raising questions about the role of gravity signaling pathways in spatial growth and metastasis.. 27 A recent study on MCF-7 cells (a type of breast cancer cells with estrogen receptor [ER] α and progesterone receptor [PR]) discovered notable changes in the F-actin and microtubule cytoskeleton and the role of focal adhesion molecules in detecting the effects of microgravity. The study also identified changes in gene expression and a decrease in E-cadherin levels, potentially leading to an elevated invasive capacity. 14 This supports a common pathway of gravity sensation. 14 Following exposure to simulated microgravity in an RPM, there were alterations observed in exosomal release and population in the breast cancer cell line MCF-7. 28 Furthermore, the MDA-MB-231 cell line of TNBC cells presented fewer malignant features at the cellular level by decreasing cell proliferation, migration, and extracellular vesicle (EV) secretion. 29 Another study found that Raf kinase inhibitory protein (RKIP), an anti-metastatic gene, was overexpressed 24 h after the exposure, while the expression level decreased back to the control level after 72 h of exposure, indicating a time-dependent effect of microgravity on the target gene expression. 30
Simulated microgravity (SMG) delayed key metabolic pathways, including glucose uptake, methionine uptake/incorporation, and thymidine incorporation, 31 with no impact on the cell cycle and apoptosis proteins in both normal vascular smooth muscle cells (VSMC) and neoplastic human breast cancer cells (MCF-7). 31 Microgravity may affect the expression of heat shock protein-60 (HSP-60), a stress protein regulating apoptosis in breast cancer.31,32 A study by Zheng et al. reported divergent outcomes of SMG on breast cancer cells. While ERα-positive MCF-7 cells exhibited increased mitochondrial activity when exposed to microgravity, the ERα-negative cell line MDA-MB-231 suffered oxidative damage under the same conditions. These findings suggest a potential protective role for ERα against oxidative stress induced by microgravity in the context of carcinogenesis. 33
Microgravity triggers irregular transformations in mammary epithelial cells, heightening the likelihood of cancer. 34 Random positioning machine (RPM)–induced SMG can result in the formation of two cellular populations: adherent (AD) and multicellular spheroids. 35 AD cells followed a suboptimal growth pattern leading to less viability compared to MCS cells. 22 SMG can cause the development of spheroid cells by affecting the proteins responsible for regulating E-cadherin and inducing modifications in gene expression and cytoskeletal structure, thereby altering cellular interactions.36–38 Microgravity led to decreased E-cadherin expression, increased cellular Src (c-Src), and higher levels of auto-degradation proteins in MCS. 36 A study evaluating the role of focal adhesions (FAs) and cytoskeleton in cell maintenance and movement in MCF-7 cells showed that modeled microgravity (MMG) induces cytoskeleton disorganization, indicated by atypical microfilament arrangement and microtubule disruption. FAs formed in MMG were less mature (reduced number and clustering) compared to the controls. Furthermore, the study revealed a decrease in integrins and FA kinase (FAK) activity under MMG. 39 Exposing MDA-MB-231 breast cancer cells to SMG resulted in two morphological phenotypes with considerable reversible adaptive alterations in proliferation, apoptosis, signaling pathways, and cytoskeletal reorganization. 40
Culturing MDA-MB-231 breast cancer cells in a rotary cell culture system bioreactor to simulate microgravity revealed disruptions in the cell cycle and ultrastructure. Cells exposed to SMG exhibited an increase in secondary lysosomes, higher apoptosis rates, and inhibited migration ability compared to normal gravity controls. Besides, the expression of BCL-2 and MMP9 was increased, while that of cyclin D3 diminished in the SMG group. 41
Thyroid Cancer
Of 21 studies related to thyroid cancer, 18 focused on the effect of microgravity on thyroid cancer. In one study, human follicular thyroid carcinoma cells showed decreased free T3 and T4 secretion and upregulated TSH receptors and extracellular proteins. 42 In addition, the level of apoptosis-associated proteins had increased, while the expression of B-cell lymphoma 2 (BCL-2) decreased. 42 These findings suggested that the decreased thyroid hormone levels in astronauts correlated with increased thyrocyte apoptosis under microgravity. 42 In another study, the synthesis of proteins responsible for cellular proliferation decreased, while NF-B p65 and apoptotic factors were elevated. 43 Four tumor markers of mouse thyroid showed decreased expression of MIB-1 and CK19 and overexpression of HBME-1 and Galectin-3. 44 Also, microgravity-induced membrane remodeling facilitated Galectin-3 diffusion into colloids. 44
A study investigating the impact of microgravity on differentiated follicular thyroid cancer cells (FTC-133 cell line) found that it regulates gene expression, indicating the cells’ ability to survive in space. Despite the gene analysis failure due to fixation issues, qPCR showed downregulation in most gene products, excluding VEGFA (vascular endothelial growth factor A).45,46 Additionally, the expression of cytoskeleton genes showed a nonsignificant decrease. 46 A study evaluated normal thyroid cells (HTU-5) and mitochondria-rich thyroid carcinoma (ONCO-DG1) cell lines under SMG. Apoptosis through mitochondrial redistribution and destruction occurs after multicellular tumor spheroids (MCTSs) formation. Extracellular matrix proteins were over-expressed, and both intrinsic and extrinsic apoptosis pathways were activated; 47 BCL-2 expression was also downregulated, with a significant increase in apoptosis in both normal and papillary cancer cell lines. 47 Genes responsible for diverse biological pathways exhibited different expression patterns. 48 The genes and proteins implicated in cancer proliferation and metastasis showed an anti-proliferative effect in the RPM and during spaceflight, contrasting the findings from parabolic flights. 48 In an experiment on FTC-133 human follicular thyroid cancer cells (TCC), the gene expression of proteins involved in cellular interactions (VCL, PXN, ITGB1, RELA, ERK1, and ERK2 mRNA) was downregulated in both adherent (AD) and MCS. 49 IK67 and SRC mRNA, both playing roles in cellular proliferation, were downregulated in MCS cells, while ICAM1, COL1A1, and IL6 mRNA (related to cellular adherence) were upregulated in AD cells. Protein secretion remained unchanged except for the upregulation of angiopoietin 2. TCCs predominantly showed antiproliferative response to microgravity, forming MCSs. ERK/RELA were introduced as the main regulatory pathways affected by microgravity. 49 Another study performed on FTC-133 cells evaluated alterations in exosomal miRNA composition under prolonged exposure to real microgravity. It showed more than a hundred differentially expressed miRNAs, predominantly involved in thyroid diseases. The study further highlights the ability of tumor cells to sense, adapt, and react to minimal environmental stressors, including gravity fluctuations. 50 A study by Ulbrich et al. evaluated the effects of short-term microgravity on ML-1 follicular thyroid cancer cells. It showed that microgravity affected the cytoskeleton by changing F-actin and cytokeratin cytoskeleton and upregulated ACTB (beta-actin protein-coding gene) and KRT80 (Keratin 80 coding gene) mRNAs. It downregulated COL4A5 (collagen type IV) mRNAs and conversely upregulated OPN (osteopontin) and FN (fibronectin) mRNAs.
Furthermore, microgravity downregulated MTSS1 (metastasis suppressor 1) and upregulated LIMA1 (a cytoskeletal-associated protein) as an adaptive response for cytoskeletal stabilization under microgravity conditions. 51 Pietsch et al. studied the effect of gravity on protein expression and interaction in normal (HTU-5) and cancerous thyroid cell lines (FTC-133 and CGTH W-1). They found most of these gravity-sensitive proteins involved in a network of interactions like carbohydrate and protein metabolic pathways, regulating cellular growth and taking part in cell membrane structure. 52
A study on metabolic enzymes of these thyroid cell lines found that removing gravity force affects gene expression, causes different modification patterns, and thereby alters cellular glycolytic enzyme content in both normal and malignant thyroid cell lines. 53 The endothelial growth factor (EGF) and connective tissue growth factor (CTGF) can reform the 3D structure of thyroid cancer cells under real microgravity. 54 In addition, caveolin-1 (CAV1) regulates the 3D growth of thyroid cancer cells under actual and simulated microgravity in another study. 55 MCS cells exhibited more significant changes in gene expression compared to the AD cells. 56
Genes responsible for 3D growth were upregulated, as were genes involved in regulating extracellular protein accumulation. 56 However, structural protein genes were downregulated. 56 A study of FTC-133 cells indicated that despite the phosphorylation of profilin-1, the heightened synthesis of extracellular matrix (ECM)-related proteins could hinder the formation of cell spheroids. 57 Notable temporal fluctuations in cytokine gene expression under microgravity indicate the potential significance of cytokines in initiating MCS formation via focal adhesion proteins. 58 Under SMG, CAV1 and CTGF were downregulated in multicellular tumor spheroids (MCTS) compared to the adherent cells, suggesting the association of these molecules with MCTS formation. 59
In the SNU-790 thyroid cell line, there was an increase in the expression of genes linked to histones, while genes related to microRNAs showed a decrease. Conversely, in SNU-80 cells exposed to simulated ground-based microgravity, genes associated with the cellular response to hypoxia were downregulated. 60
Lung Cancer
The effect of SMG on lung cancer varies based on the cell line studied. 61 The proliferation rate of adenocarcinoma (A549) cell lines was similar to controls, while this was lower in squamous cell carcinoma (H1703). 61 Meanwhile, microgravity exposure was associated with increased migratory ability in both cell lines. 61 Results of a study on non-small cell lung cancer (NSCLC) demonstrated various levels of promotion of gene expression and cell migration under microgravity. 62 The rate of cell proliferation was reported to be lower under microgravity than on Earth, possibly due to decreased initial cellular adhesion. 62 SMG decreases cellular proliferation, migration, invasion, and consequently, its metastatic ability in human lung adenocarcinoma. 63 Under microgravity, cancer stem cells (CSCs) undergo differentiation, resulting in the attenuation of their stem cell characteristics; moreover, microgravity exposure amplifies the apoptosis rate in these cells. 64 When mice with implanted human lung cancer were vaccinated with mesenchymal stem cells (MSC) under microgravity, the apoptosis was enhanced, along with the presence of proteins such as HSPs. 65 However, the levels of T-reg cells and cellular proliferation within the tumor tissue were diminished. 65
SMG could affect cell adhesion, increase apoptosis, and upregulate tumor suppressor gene (TSG) expression in human lung cancer cells. 66 Simulated microgravity was associated with changes in morphology; reversal of epithelial-to-mesenchymal transition (EMT); increased expression of epithelial E-cadherin; decreased expression of mesenchymal N-cadherin; and heightened levels of FCGBP, BPIFB, F5, CST1, and CFB in the progression of lung cancer using the human adenocarcinoma alveolar basal epithelial (A549) cell line. 67 The exposure of the A549 cell line (a model for lung adenocarcinoma) to microgravity results in the formation of poly-nucleated cells, indicating a free cell cycle and DNA replication. 68 It also results in growth inhibition and abnormalities in gross morphology. 68 Microgravity-induced damage to mitochondria may affect miRNA modulation and disrupt the cell cycle balance. 68
Neural Cancers
Redden and Doolin reported that the in vitro behavior of neuroblastoma varied under microgravity according to the cell line studied. 69 Neuroblastoma (SH-SY5Y) exposure to microgravity resulted in microfilament disruption and increased levels of reactive oxygen species (ROS), malondialdehyde, and free Ca2+ ions. 70 Conversely, adenosine triphosphate (ATP) levels diminished, with no significant change in cell apoptosis. 70
Microgravity may offer a therapeutic role in treating neural cancers like gliomas. SMG seems to decrease proliferation and induce apoptosis in malignant glioma (U251MG) cells. 71 A study on human glioblastoma U87 cells revealed that SMG decreased cell invasion and migration by downregulating store-operated calcium entry (SOCE), which regulates cell migration and calcium hemostat in non-excited cells. This suggests novel therapeutics for approaching glioblastoma. 72 After 24 h of SMG exposure, human glioblastoma cells (A-172) showed reduced activity of the yes-associated protein 1 (YAP-1) molecule, which plays a role in regulating cell migration and proliferation through adhesive mechanotransduction processes. 73 Furthermore, microgravity was associated with increased chemosensitivity to cisplatin and decreased malignant glioma proliferation, growth, and mitochondrial activity. 74 Results of another study supported the indispensable role of the FAK pathway in glioma SMG-induced cell suppression. 75 Under simulated zero gravity conditions, glioblastoma multiform (ANGM5) exhibited loss of cellular adhesion, a 32% reduction in apoptosis inhibition, and alterations in both morphology and behavior. 76
In another study, induced pluripotent human neural stem cells derived from skin fibroblasts, specifically the cell line “CS83iCTR-33n21,” exhibited abnormal cell division, including incomplete division, when exposed to microgravity in space. 77
Hemopoietic Cancer
SMG in murine lymphoma made cancer cells more susceptible to T-cell-mediated elimination by restoring the CD4+ T cell response and increasing CD8+ T cell–mediated cancer cell destruction. 78 SMG also hindered CD4 + activation by producing inhibitory factors against dendritic cells, reversing immune evasion. 78 The proliferation of human Hodgkin's lymphoma cancer cells (L-540 and HDLM-2) was significantly inhibited under time-averaged microgravity compared to controls. 79
A study on the effect of spaceflight on Epstein-Barr Virus (EBV) reactivation reported decreased DNA repair in human EBV-positive Burkitt's lymphoma cell line, suggesting that EBV-seropositive individuals may be at greater risk of accumulating DNA damage during space missions. 80 Interestingly, the anticancer effects of Daunorubicin on leukemic and erythroleukemic cancer cells were reversed under microgravity through enhanced cell migration before cell death. 81 ROS production was affected in a cell-type-specific manner. 81
Microgravity was also associated with the upregulation or downregulation of 16 p53-dependent genes. 82 TGF-β (transforming growth factor beta), TWEAKR (tumor necrosis factor-like weak inducer of apoptosis receptor), phosho-Pyk2 (proline-rich tyrosine kinase 2), and 14-3-3θ/τ were among p53-dependent downregulated proteins affected by microgravity in human cultured lymphoblastoid cell lines (TSCE5 and WTK1) during spaceflight. 83 Also, the expression of 14 lncRNAs and 17 mRNAs (differentially expressed genes, DEGs) was downregulated under SMG. 84
A recent study discovered that microgravity can modify the way K562 cancer cells react to drugs such as hydroxyurea and paclitaxel, particularly impacting the N/C ratio. Furthermore, untreated cells remained unaffected following exposure to microgravity, indicating potential mechanisms underlying drug response behaviors. 85
Bone Cancer
SMG suppressed EGF-induced c-fos gene expression in MC3T3-El pre-osteoblast cells. 86 Compared to the ground controls, mitogen-activated protein kinase (MAPK) phosphorylation was unaffected, suggesting that MAPK may be the signal transduction pathway's downstream microgravity action site. 86
In Ewing's sarcoma A673 cell line exposed to 1 day of SMG, plerixafor (AMD3100), a CXCR4 inhibitor, did not affect spheroid development. SMG altered cancer-related gene expression and promoted spheroidal adhesion. Despite overexpression of EWS/FLI1 (tumor-specific main translocation-protein), CXCR4, and CD44 (hyaluronic acid receptor), the associated protein levels were largely unaffected, and the amount of the CD44 protein decreased. Elevated EWS/FLI1 expression in adherent cells triggered protein synthesis. 87
In mouse MC3T3-E1 cells, SMG reduced focal adhesions, altered cytoskeleton structures, and inhibited key molecules involved in osteoblast differentiation. Additionally, it decreased alkaline phosphatase activity and matrix mineralization while also significantly reducing tibial trabecular bone loss. 76
Dermatologic Cancer
A study by Rijken et al. on human A431 epidermoid carcinoma cells under microgravity indicated that EGF-receptor clustering remained unaffected. Moreover, microgravity downregulated EGF-induced c-fos (proto-oncogene) and c-jun (proto-oncogene) gene expression. Although microgravity was associated with decreased expression of EGF- and TPA (12-O-tetradecanoylphorbol-13-acetate)-induced gene expression, forskolin- and A23187-induced c-fos expression was independent of gravity, suggesting that microgravity can influence some specific signaling pathways in this cell line and facilitate EGF-induced cell rounding as well. 88 Compared to the 1-G control experiment, simulated weightlessness was associated with changes in actin filament organization. 89 In the nonmetastatic melanoma cells, microgravity decreased cell motility and cancer-related gene expression. In particular, it downregulated the NOS-sGCMRP4/MRP5 pathway as well as iNOS and GC-A/GC-B. 90 Microgravity prevents the differentiation of stem/progenitor cells into dendritic cells, which are critical antigen-presenting cells. 91
Gastrointestinal Cancer
Microgravity could be a cancer-restraining factor by inhibiting cell proliferation and inducing apoptosis in gastric carcinoma cells. 92 Results of a study on HGC-27 (human gastric cancer) cell metabolism under SMG reported variable patterns of regulation in key metabolic pathways, mostly affecting glycerophospholipids and fatty acids. In fact, SMG was associated with upregulation of phosphatidyl ethanolamine, phosphatidyl choline, arachidonic acid, and sphinganine, while the following metabolites were downregulated: phingomyelin, phosphatidyl serine, phosphatidic acid, L-proline, creatine, pantothenic acid, oxidized glutathione, adenosine diphosphate, and adenosine triphosphate. 93
In HCT116 (human colorectal cancer) cells, SMG, as a stemness regulator, upregulated markers like CD133/CD44, YAP nuclear localization, and increased the number of polypoid giant cancer cells (PGCC). 94 SMG upregulated some TSGs in DLD1 (a colorectal adenocarcinoma cell line) cell lines but downregulated AKT (a driver for cell survival mechanisms) pathway and ultimately caused apoptosis in DLD1, HCT116, and SW620 colorectal cancer cells. 95 It also resulted in adaptive responses, with the PTEN/FOXO3/AKT pathway playing a pivotal role in cell function and viability under SMG. 95 In the DLD1 cell line, SMG altered cell morphology, decreased cell viability and colony-forming ability, and formed an aberrant cell cycle. 96 It also had an anti-proliferative role through the upregulation of microRNA-22 (tumor suppressor). 96
Turning to the liver, in the only study to investigate the HepG2 hepatoblastoma cell line, HepG2 mt cell (HepG2 cells transfected with a p53 mutant) proliferation was markedly reduced under SMG apart with or without CDDP (cis-diamminedichloroplatinum) therapy, suggesting the artificial control of gravity or gravity-influenced pathways as a potential anticancer treatment for those with a mutant p53. 97
In pancreatic cancer cells, SMG triggered a profound transformation in both behavior and metabolic activities, resulting in the development of a highly aggressive and metastatic phenotype reminiscent of stem cells. 98
Urologic Cancer
SMG alters the morphology and cytoskeleton of prostate adenocarcinoma-derived PC-3 cells as well as their ECM protein synthesis, focal adhesion complex, and growth. 15 It also upregulates PAM pathway genes, effectively leading to cellular-level alterations under microgravity. 15 Moreover, gene expression is affected, and thereby, cell adhesion molecules, E-cadherin, and CD44 are upregulated under microgravity. 99
A study used the high aspect rotating-wall vessel 100 to simulate microgravity and to evaluate its effect on DU-145 human prostate carcinoma cells. Based on the results and compared to the control cultivation methods, cell growth in the HARV was slower, with evident differences in the cell cycle and immunostaining. These findings suggest that HARV can produce less pliant, more differentiated cultures that are less aggressive in terms of proliferation. The HARV encouraged a less aggressive, slower-growing, and more differentiated cell type, resulting in a 3D growth model similar to natural tissue growth. The researchers noted several shifts in cellular signaling pathways during the HARV cultivation period, such as an active phase of ceramide, subsequent activation of PI3 K, and an increase in cAMP. These changes coincided with slower growth and 3D formation. The findings indicate that microgravity can influence cellular pathways related to apoptosis, survival, proliferation, and differentiation, providing a new perspective on prostate cancer cell growth.101,102
The three-dimensional proliferation of PC-3 prostate cancer cells under SMG resulted in varying levels of cytokines IL-1α, IL-1β, IL-6, and IL-8 expression, suggesting their involvement in the growth and progression of prostate cancer cells. 103 A parabolic flight investigation explored the gravitational response of prostate cancer cells, revealing distinct regulation of 5 cytokines, 1 zinc finger protein, 1 glycoprotein, and a microRNA. These findings hold promise for refining diagnostic methodologies in prostate cancer research. 104
Reproductive System Cancer
Cirelli et al. looked at the effect of SMG on aromatase gene expression in Sertoli cells. By upregulating aromatase gene expression, microgravity increased 17-estradiol release, which can negatively impact fertility and adversely affect testicular cancer. 105
Non-Organ-Specific Studies
In a study by Kumari et al., SMG on human lymphocytes affected DNA repair gene expression and, therefore, may result in DNA damage accumulation. Down-regulation of cell-cycle genes triggered a subsequent decrease in cell growth. Extended exposure to microgravity affected the apoptotic ability of the cells by downregulating the pro-apoptotic genes. 106
A study using planarians to assess the influence of gravity fluctuations on complex organisms indicates that alterations in gravity could affect gene expression and enhance cell proliferation. The affected genetic transcription predisposes molecules to abnormalities that can develop cancers. 107
Demonstrating the susceptibility of both cancerous and healthy cells, a 24-h exposure to SMG resulted in the remodeling of the VE-cadherin junctional protein in human umbilical vein endothelial cells (HUVECs). 73
Space Radiation & Cancer
Hematopoietic Cancer
Rodman et al. reported that sequential exposure to protons and iron ions (frequent in deep space) had a more damaging effect on the hematopoietic stem cells (HSCs) genome than either particle class alone. 108 However, a cohort study among children under 15 years of age found no clear evidence of an ecological connection between exposure to terrestrial gamma radiation and pediatric leukemia. 109 Space radiation can also affect p53-dependent gene expression by upregulating MECP2 (methyl-CpG binding protein 2) and Notch1 (neurogenic locus notch homolog protein 1), which are over-expressed in several human cancer types. Also, in their study, Takahashi et al. investigated the impact of space radiation and environment on p53 alterations in two human cultured lymphoblastoid cell lines. They found that pre-irradiated cells with wild-type p53 gene status showed maximum induced radio-resistance at 0.05 Gy, maximum depression of apoptosis at 0.02 Gy, and maximum depression of chromosomal aberrations at 0.02 Gy. Adaptive responses, including radio-resistance induction, depression of radiation-induced apoptosis, and chromosome aberration suppression, were observed in wild-type p53 cells exposed to space radiation. However, mutated p53 cells did not exhibit these adaptive responses. These findings demonstrate that space radiation induces p53-dependent adaptive responses within a specific low-dose range, even at very low doses present in space. 82 In another study, during the 133-day flight, the total equivalent space radiation dose was 71.2 mSv, and p53-dependent upregulated gene expression was observed for 111, 95, and 328 genes, and p53-dependent down-regulated gene expression was found for 177, 16, and 282 genes after exposure to space radiation, to microgravity, and to both, respectively. Data from this work revealed genes that might help advance basic space radiation biology, which is expected to help design physical protection from the harmful effects of space radiation during long-term stays in space. One additional study on p53-regulated proteins found that p53-dependent upregulated proteins in response to space radiation and environment were MeCP2 (methyl CpG binding protein 2), and Notch1 (Notch homolog 1), respectively. On the other hand, p53-dependent down-regulated proteins were DR4 (death receptor 4), PRMT1 (protein arginine methyltransferase 1), and ROCK-2 (Rho-associated, coiled-coil containing protein kinase 2) in response to space radiation. ROCK-2 was also suppressed in response to the space environment. The data provides the p53-dependent regulated proteins by exposure to space radiation during spaceflight. 83
EBV produces the ZEBRA (Z Epstein-Barr replication activator) protein during its active phase when the virus replicates. “EBV ZEBRA-infected cells” refers to cells infected with an active form of EBV, facilitated by the ZEBRA protein, leading to viral replication, potential host cell death, and viral spread to other cells. 110 EBV ZEBRA-infected cells do not undergo apoptosis as much as their counterparts. 111 Brinley et al. conducted a study on seropositive hematopoietic cells under radiation and demonstrated radiation as the main factor in upregulating EBV ZEBRA. Consequently, those cells have higher risks of DNA damage accumulation under space mission conditions. 80 Another study on hematopoietic stem cells revealed an increase in radiation-induced lymphomagenesis and microsatellite instability while also stunting mismatch repair. 112
High-energy galactic cosmic radiation (GCR) originates from outside the solar system and consists of alpha particles, protons, and high (H) atomic number (Z) and energy (E) particles (HZE). Gamma rays (a type of high-energy radiation) and those induced by HZE ions are two major radiation types to consider. 113
At the tumor histotype spectrum level, the spectrum of naturally occurring and gamma-ray-induced (3.0-Gy) cancers and their HZE (high-energy nuclei component) ion-induced (0.4 Gy) counterparts were not different, and each was maximally tumorigenic, suggesting the feasibility of gamma-ray exposure measurement as a mean to estimate cancer risk from galactic cosmic rays. 114
The results of a study concerning the mutagenic effects of particle radiation exposure on HAP1 (chronic myelogenous leukemia, blood cells) suggest that exposure to proton and alpha radiation does not markedly cause mutations in cultured human cells. 115
Lung Cancer
Compared to X-rays and low-LET γ-rays, HZE particles, regardless of dose and time, are more likely to result in lung and airways carcinogenesis. 116 Exposure to silicon predisposes individuals to lung cancers that are more aggressive among HZE particles, inducing mixed hypo- and hypermethylation at the genome level. Also, iron (Fe2+) ions cause genomic regions to become hyper-methylated, which helps distinguish between normal tissue and human adenocarcinoma and small cell carcinoma.117,118 In addition, exposure to a high-LET (linear energy transfer) dosage in a single or split dose increased tumor growth, cell proliferation, and upregulated DNA repair genes remained throughout time and might be heritable.117,119 While the type of cell that gives rise to K-Ras-driven tumors did not change. 120 Profiling analyses of bronchial epithelial cell exosomes also detected approximately 4-fold miR-1246, miR-1290, miR-23a, and miR-205 syntheses over HZE stimulation, which are non-small cell lung cancer–related microRNAs. 121 In an animal study, mice irradiated with 25 cGy and 100 cGy of GCRsim had shorter survival times compared to chronic exposure with acute exposure. The 100 cGy group showed double the adenocarcinoma formation compared to controls, while the 25 cGy group had a 21% increase. Acute exposure to 50 cGy reduced median survival compared to chronically irradiated mice. 122 Another study examined how low- and high-linear energy transfer (LET) radiation affects airway epithelial progenitor cells, which are important for maintaining the airways. Mice were exposed to XX-rays and Fe ions, and the researchers found that the ability of progenitor cells to form colonies decreased in a dose-dependent manner in lab conditions. Interestingly, after radiation exposure, the growth of progenitor cells in the mice increased, but there was no rise in epithelial cell growth, indicating that radiation may reduce the number of progenitor cells. These results provide important information about how ionizing radiation impacts the behavior of airway epithelial progenitor cells and their potential effects on long-term tissue maintenance. 123
Moding et al. explored the role of the p53 gene in lung cancer and lymphoma development in LA-1 KrasG12D mice by comparing normal mice to those with an extra copy of p53 (super-p53). Both groups were exposed to radiation, and their tumor growth was compared to mice that were not irradiated. The results showed that having an extra p53 copy reduced lung tumors and lymphoma in mice that were not exposed to radiation. However, after radiation, the extra p53 did not protect against lymphoma and even led to larger lung tumors in super p53 mice. These findings suggest that although p53 can help prevent some types of tumors, it does not protect against radiation-induced lymphoma and might worsen lung cancer associated with KrasG12D mutations. 124
The findings regarding the mutagenic impacts of particle radiation exposure on the human lung adenocarcinoma cell line A549 (CCL-185) indicate that proton and alpha radiation exposure does not significantly induce mutations in cultured human cells. 115
Urological Cancer
Low simulated space radiation (iron ions) doses, in comparison to similar doses of protons, were highly mutagenic and affected numerous chromosomes in mouse epithelial kidney cells, which survived for several months following irradiation. 125 Li et al. evaluated mutagenesis in a human bladder cancer cell line by HZE radiation and double-strand break induction (using an agent named I-SceI), which resulted in a 2-fold increase in translocations and a notable rise in deletions that increased the frequency of mutagenic repair. 126 I-SceI-stimulated translocations and deletions in human bladder cancer cells cultured with HZE-irradiated cells increased just like those cells that were directly exposed to HZE-irradiation. 127 On another note, in vivo and ex vivo ionizing radiation-induced foci were inversely linked with baseline DNA damage in lymphocytes from prostate cancer patients. Thereby, baseline DNA damage may predict radiation exposure risks. 128
Furthermore, 8.5 min of Pm-147 exposure (equivalent to 13.7 Gy of X-irradiation) reduced the relative growth of human embryonic kidney (HEK) cells to 37%. 129 This was also added to the fact that radiation doses are reportedly more accurate predictors than fluence for murine tumor prevalence. 130 Hellweg et al. used CAPE, capsaicin, and MG-132 to inhibit X-ray-induced NF-jB activation in HEK-PNF-jB-d2EGFP, which controls proliferation and suppresses apoptosis. 131
Reproductive System Cancer
Mishra et al. exposed 3-month-old female mice to 0 cGy (sham) or 50 cGy iron ions and aged them to 18 months, and as a result, a significant number of those irradiated with 50 cGy had unilateral and bilateral ovarian tumors. Their results brought some concerns about the rise of ovarian tumors as a late consequence of deep space travel in females. 132
Gastrointestinal Cancer
Roig et al. looked at two- and three-dimensional models for risk assessment of radiation-enhanced colorectal tumorigenesis. In colon biopsy samples, radiation-induced DNA damage was more prominent in the epithelium than in the stroma. Moreover, high-LET radiation damage was substantially more persistent than low-LET radiation damage. 133 However, Udho et al. showed that exposure to high-LET HZE radiation did not increase tumor aggressiveness compared to γ-rays or controls in murine models of hepatocellular carcinoma. No significant differences were observed in tumor grade, latency, metastasis, or biomarker expression. 134
Suman et al. investigated intestinal and colonic tumorigenesis in APC1638N/+ mice following high- and low-dose rate exposure to 56Fe and 28Si ions and found significantly increased tumor frequency regardless of dose rate, suggesting a dose rate–independent carcinogenic effect of heavy ions. 135 Altered expression profiles of various markers associated with intestinal stem cells (ISC), including ALDH1, BMI1, CD133, DCLK1, LGR5, and MSI1, as well as β-catenin, were noted in space radiation-induced premalignant intestinal tumors and nearby normal mucosa in the Apc1638N/+ mice model. 136 These findings indicate potential implications for assessing cancer risk following exposure to space radiation.
Breast Cancer
A study by Dicello et al. on adenocarcinomas and benign fibroadenomas breast cancer showed that the likelihood of mammary cancers forming in rats subjected to 1 GeV/nucleon 56 Fe ions and 250 MeV protons was less compared to additive radiation values. 137 Another study predicted that the maximum risk of breast cancer occurs at 220 keV/lm. 138
Research exploring the impact of galactic cosmic radiation (GCR) and γ-ray on the differentiation of mammary gland cells found notable increases in ductal growth and cell proliferation within the mammary tissues of GCR-irradiated mice. Moreover, elevated levels of Spp1 mRNA protein, known as a potential marker for preneoplastic changes in mammary cancer induced by GCR, were observed in the GCR-exposed group compared to the γ-ray-exposed group. 139 Similarly, Imaoka et al. demonstrated that carbon ion radiation induced mammary carcinomas in a strain- and dose-dependent manner in rats, with high relative biologic effectiveness (RBE), particularly at low doses. The induced tumors were predominantly ERα-positive and lacked H-ras and Tp53 mutations, with lung metastases observed only in the carbon ion–exposed group. 140 Meanwhile, Delhomme et al. reported that proton and alpha radiation exposure does not significantly mutate cultured human cells in the MCF7 (HTB-22) breast adenocarcinoma cell line, 115 similar to Udho et al.'s murine models of mammary tumors. 141
Neural Cancer
Chang et al. evaluated p53 deficiency and lacZ mutants in the brain of transgenic mice after radiation exposure, reporting that iron particles increase mutagenesis, especially point mutations and small deletions. These genome reorganizations were dependent on p53 alterations. 142
Thyroid Cancer
Stewart et al. studied thyroid epithelial cells subjected to a 20-cGy radiation dosage from iron ions with L-selenomethionine (SeM) supplementation. SeM has been recognized as an increasingly potent protective agent against the biological effects induced by radiation, and this study also revealed the effect of alteration of the transcriptional control of multiple genes involved in the cell cycle, proliferation, and death. SeM supplementation influenced the expression of 66% of upregulated and 82% of down-regulated genes at the 10-cGy radiation dose and 70% of upregulated and 93% of down-regulated genes at the 20-cGy radiation dose. 143
Harderian Gland Cancer (in Mice)
Edmondson et al. used a genetically diverse mouse population and reported that HZE ion and γ-ray exposures produced overlapping tumor spectra and shared genetic susceptibility loci for several tumor types. While certain tumors such as Harderian gland adenocarcinomas showed radiation-specific genetic drivers, overall metastatic potential and tumor latency were similar across radiation types, supporting the extrapolation of γ-ray data for modeling space radiation–induced cancer risk. 144 Alpen et al. investigated charged particle carcinogenesis in the Harderian gland of mice. They reported that the neoplastic effect sizes of all ions with linear energy transfer 145 greater than 100 keV/micron were identical, claiming that even a single hit could result in a neoplastic transformation. 119 In a similar study, Chang et al. found no changes in Harderian gland tumor frequency after exposure to fractionated low doses of titanium over 5 days with 24-h intervals between fractions. 146
Non-Organ-Specific Studies
Although a study on Escherichia coli by Harada et al. in 1997 reported that space and ground strains were identical in terms of mutagenesis and survivability, 147 recent studies like the one by Li et al. showed increased formation of the EML4-ALK, a lung cancer marker, resulting in fusion oncogene production. 148 It is observed that ionizing radiation has connections with different pathways, such as cellular signaling and the development of tumors. 149 Iron ions were also demonstrated to be mutagenic in situ and in vitro, while the cell toxicity from irradiation was also dose and time-dependent. 150
A study by Loria et al. investigated the effects of a single injection of androstenediol on mouse hematopoietic cell lines after a real-time galactic cosmic ray. Interestingly, the results showed reduced WBCs, lymphocytes, monocytes, granulocytes, RBCs, platelets, and hemoglobin following GCR and a significant increase in RBC, platelet, hemoglobin, and monocyte levels on 4, 7, and 14 days after radiation in the androstenediol group. 151
Also, we found 4 studies on mathematical models used to stratify space radiation risk of cancer by considering factors such as radiation quality factors (QFs), dose and dose-rate effectiveness, latency, and excess risk functions. These concluded that research on space radiation should focus on minimizing the uncertainties associated with high LET (linear energy transfer) risk prediction by QFs parameters, NTEs (nontargeted effects on risk estimates), increased tumor lethality, and decreased latency at high LET.152–155
Hyper-Gravity and Vibration
During spaceflight, notably in the departure and landing phases, the astronauts experience episodes of high force of gravity, which can disturb many physiological activities in their organs, even at the cellular level. 156 Hyper-gravity may alter structural organization by the heterogenous expression pattern of cytoskeletal proteins in thyroid cells. 157 Altered gravity could increase human A431 epidermoid carcinomatous cell adherence by EGF-induced c-fos expression upregulation. 88 In addition, elevated metastasis rates were also reported via upregulating the NOS-sGCMRP4/MRP5 pathway in human melanoma cells. 90 Moreover, vibration down-regulated and decreased cytoskeletal protein concentration. 157
Spaceflight (Microgravity + Galactic Radiation)
The effects of isolated stressors could be different from the cumulative effects of them. Yamanouchi et al. examined the occurrence of chromosome aberrations 158 in cultured human lymphoblastic TK6 cells. The cells were exposed to X-ray or carbon ion radiation while subjected to SMG (μG) conditions. It was observed that cells exposed to both radiation and μG simultaneously exhibited a higher frequency of both simple and complex types of CA compared to cells exposed solely to radiation. 159
In a separate study, Fu et al. found that under SMG, the expression of 14 long noncoding RNAs (lncRNAs) and 17 messenger RNAs (mRNAs) (referred to as differentially expressed genes, DEGs) was significantly downregulated. Conversely, irradiation resulted in the upregulation of 55 lncRNAs and 56 DEGs, with only one lncRNA showing downregulation and no DEGs affected. Moreover, in response to SMG after irradiation, two microRNAs, 70 lncRNAs, and 87 DEGs displayed significant alterations in expression. These changes were independently induced by both irradiation and SMG. 84 The profiles of lncRNAs and DEGs caused by SMG were unrelated to those induced by radiation, suggesting that the two variables may have additive and synergistic impacts on biological processes, with radiation making up most of the interaction.80,82,84,159
A study by Baba et al. on liver cells indicated that hepatocellular carcinoma biomarker (CYP4A1) and CIRP (cold-inducible RNA-binding protein) gene expression significantly increased during spaceflight; CIRP is a sepsis inflammatory mediator that blocks TNF release and induces macrophage translocation. However, there was a significant decline in p53 expression and heat shock protein-90 (HSP90) genes, stabilizing several proteins required for tumor growth. 160 However, there are several reports indicating a marked increase in p53 expression following space travel.161,162 MeCP2 (methyl CpG binding protein 2) and Notch1 (Notch homolog 1), over-expressed in several human cancer types, were identified as p53-dependent upregulated proteins in response to space radiation and the space environment. On the other hand, ROCK-2 (Rho-associated, coiled-coil-containing protein kinase 2) was downregulated in the space environment, 83 which has been proven to be related to neoplastic cell migration. 163
Spaceflight-induced changes in small nucleolar RNA (snoRNA) expression in peripheral blood suggest snoRNAs could be novel biomarkers for monitoring astronaut health, potentially relevant for various diseases, including cancer. 164 Mutations in 17 CH driver genes in astronaut blood samples taken after space travel were investigated, and 34 nonsynonymous mutations were identified. TP53 and DNMT3A mutations were the most prevalent, suggesting that space travel can lead to DNA methylation alterations and Li-Fraumeni syndrome. This rare genetic condition predisposes individuals to certain cancers. 8 Zhang et al. analyzed the rationale behind biological changes in cancer cells after spaceflight, revealing that neoplastic-related genes, including SUB1, SGEF, MALAT1, which are transcriptional coactivators, and cytoskeleton relative gene MYL6, as well as mitochondrial metabolism gene MTCO2, were upregulated by the space environment and for the first time, their findings demonstrate that spaceflight can inhibit the growth of tumor cells.. 9
CaSki cells are cervical cell lines infected with human papillomavirus type 16 (HPV-16). 165 Simulated microgravity and radiation exposure lead to various biological alterations in gene expression, tumorigenicity, cell proliferation, and cell morphology, altering CaSki cells’ tumorigenicity in vitro and in vivo. 166 After these cells were flown on the Shen Zhou IV space shuttle mission, they showed a change in morphology; they became rounder, smoother, and smaller with low adhesions and obscure and discontinuous band membranes. Also, they grew slowly compared to ground control, which suggested that the space environment decreased the malignant potential of CaSki cervical cancer cells. 167
In addition, QNS108 brain tumor cells from glioblastoma, subjected to suborbital rocket flight, showed increased tumor proliferation, migration, and stemness, as well as larger tumor-associated growth areas and lower survival rates. 100
Gridley et al. demonstrated that a 12-day shuttle mission decreased body mass and spleen size, elevated CD3/CD25 T cells, and NK1.1/CD2. No noticeable reduction in TNF secretion occurred. RBC and platelet levels were also higher, but MCV, MCH, MPV, and RDW levels were lower. 168 Bone marrow macrophages secreted more TNF alpha during Spaceflight. 169
Exacerbated by impaired NK-cell cytotoxicity and functional compromise, the astronaut cancer risk was found to be significant. 170 Furthermore, even though short-term flights reduced cell affinity, long-term ones reduced target-binding cells altogether.170,171
Bisserier et al. explored the impact of spaceflight on exosomal lncRNA levels in the peripheral blood plasma of three astronauts, which revealed alterations in the expression of 27 lncRNAs, with 15 showing increased expression and 12 showing decreased expression. 172
A research endeavor exploring mutations within peripheral blood mononuclear cells found 34 nonsynonymous mutations across 17 genes linked to clonal hematopoiesis. The TP53 and DNMT3A genes exhibited the most prevalent mutations. 8
Trinchant et al. analyzed clonal hematopoiesis before, during, and after human spaceflight and reported that it is increased during space travel, raising the risk of hematological cancers. 173 The dose-response data on space radiation collected under the 1G condition may have underestimated astronauts’ risk of health problems, including cancer. Microgravity and radiation together increased DNA damage and ROS.
During the Spacelab Life Sciences-2 (SLS-2) mission, a significant decrease in T lymphocyte activity was observed in rats compared to the controls, and cell-mediated immunity was suppressed. The fluctuations in immune parameters during the space mission suggest that these changes might reflect an immune system response to the spaceflight condition that potentially escalates over time. 174
Novoselova et al. studied the effects of a 30-day high-orbit space mission (BION-M1) on animals’ immunity. A reduction in splenic and thymic mass was observed, as well as lymphocyte counts. Such conditions increased p-53 levels, especially its activated phosphorylated form (ph-p53). The increased active/inactive p53 protein ratio (ph-p53/p53) after landing indicates apoptosis induction. The imbalanced immunity in mice returning from the BION-M1 space mission induced changes in signaling, apoptosis, and stress response that were irreversible even 7 days after landing. 175
Discussion
We provide a comprehensive overview of the scientific literature investigating the relationship between extreme space environments and oncology. We conducted an initial exploration using relevant keywords, informing the development of an exhaustive search strategy across multiple scientific databases.
Among the 151 studies reviewed, we found that about 90% were undertaken by a single country, with the remaining involving international collaborations. A similar percentage of studies employed experimental methodologies, and a quality assessment tool validated their high standards of research.
Several studies focused on specific cancers, including thyroid, breast, hematopoietic, and lung cancer (Figure 5). However, we identified a gap in the literature concerning studies that concurrently evaluate the impact of extreme space environments on multiple organs or cell lines. This underscores the need for increased international collaboration and the adoption of methodologies that yield higher levels of evidence to better understand the diverse effects of space conditions on various organs and cell lines.
Current research efforts primarily concentrate on individual aspects of space missions, typically through simulations. A notable deficiency lies in studies that investigate the interactions of multiple stressors through a blend of simulations and real-time space experiences. It is crucial to acknowledge that these factors could exhibit a synergistic effect on each other, necessitating meticulous attention. As an illustration, microgravity could augment radiation risks in astronauts by influencing multiple pathways. 91
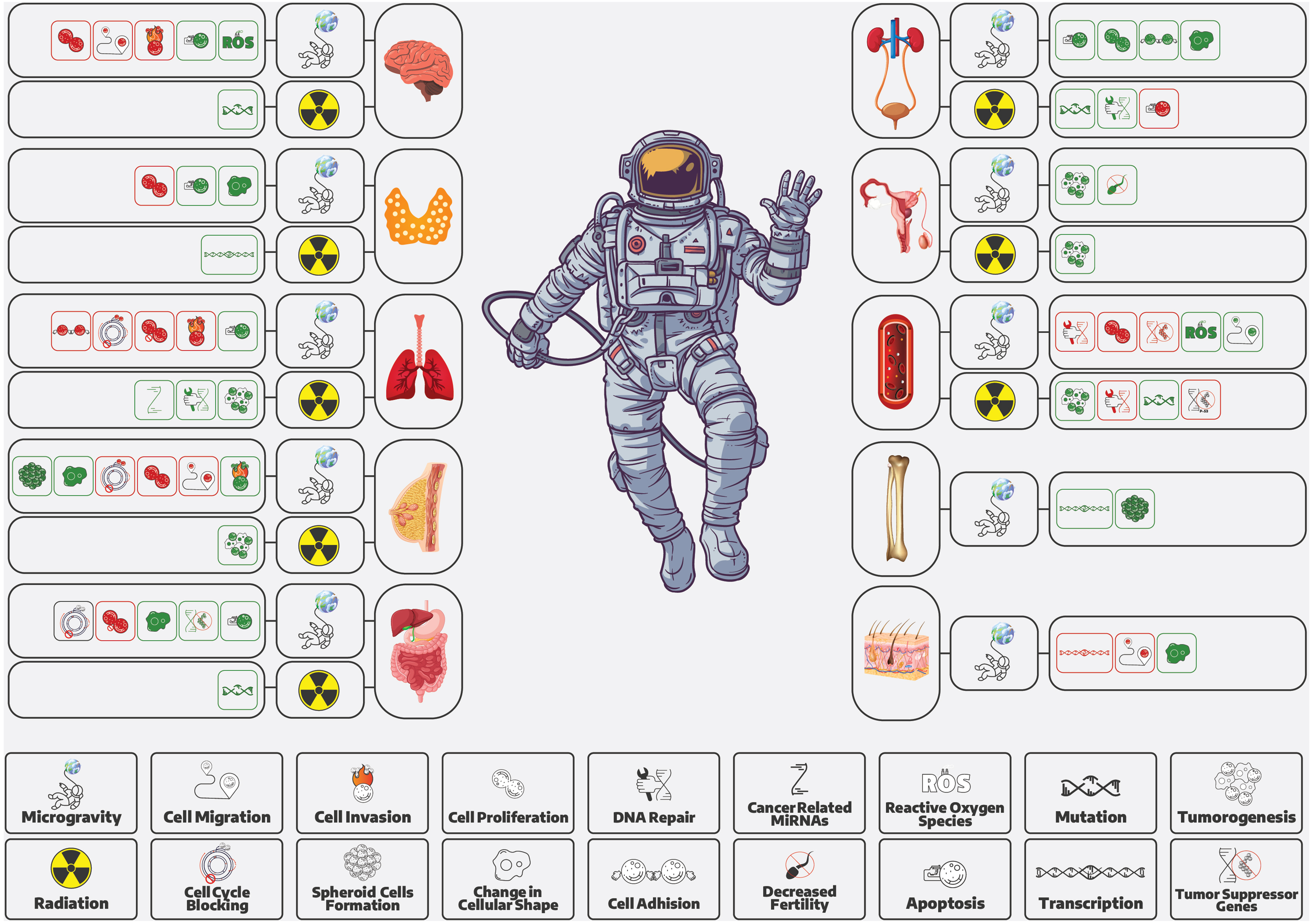
Organ-specific biological effects of spaceflight conditions contributing to cancer risk. This illustration summarizes how microgravity and space radiation may influence key oncogenic processes across multiple organ systems. Highlighted changes include alterations in cell proliferation, DNA damage and repair, oxidative stress (ROS), mutation accumulation, cell migration, apoptosis, cancer-related microRNAs, and tumor suppressor gene regulation. These effects point to a system-wide vulnerability to carcinogenesis under spaceflight conditions.
We discovered potential therapeutic strategies from studies on brain tumors and melanoma. For instance, microgravity was found to decrease invasion and migration ability in glioblastoma U87 cells 72 and to reduce cell mobility in nonmetastatic melanoma cells by downregulating the GC-A/GC-B pathway. 90 Similarly, microgravity was observed to regulate different metabolic pathways in HGC-27 gastric cancer cells, suggesting novel potential treatment targets. 93
Moreover, studies show that microgravity can affect cancer stem cells, potentially influencing cell viability, apoptosis, and the cell cycle. For instance, SMG increased cancer cell susceptibility to T-cell elimination and inhibited cancer cell proliferation in human Hodgkin's lymphoma, indicating potential new therapeutic strategies. 79
Microgravity influences signaling pathways related to spatial growth, cell proliferation, and apoptosis across nearly all organs.23,24 Investigations conducted on the breast, neurons, and skin reported a decrease in cell migration, invasion, and possible metastasis,14,27,29,30,72,90 while these parameters increased in hematopoietic cells. 81 Additionally, the expression of most cancer-related genes changed in bone and thyroid. Importantly, microgravity may affect the effectiveness of chemotherapy, with research showing increased sensitivity of glioma cells to cisplatin 74 and decreased anticancer effects of Daunorubicin on erythroleukemic cancer cells in microgravity. 81 These effects emphasize the need for ongoing research into the direct therapeutic potential of microgravity and its impact on the therapeutic window and effectiveness of chemotherapy agents.
The role of p53, a pivotal player in cancer biology, under space conditions warrants special consideration. Studies have shown downregulation of p53-dependent genes under microgravity or radiation exposure in hematopoietic cells,160,176–178 while other research found increased p53 expression in the gastrointestinal system under microgravity 95 and in hematopoietic cells exposed to radiation. 82 Evidence also suggests that gravity manipulation or targeting gravity-influenced pathways may serve as effective anticancer treatments, especially for those with mutant p53. 97
Radiation, in conjunction with microgravity, can impact tumor suppression through mechanisms like increased apoptosis, suppressed cell proliferation, and alterations in cell morphology and cytoskeleton. 179 However, it can also contribute to cancer development by inducing mutations, impairing mismatch repair, and accumulating DNA damage. 180 In contrast to the study by Evrard et al. reporting the absence of ecological association between terrestrial gamma radiation and leukemia in children, 109 a review by Little et al. supports the elevated risk of cancer and benign neoplasms especially CNS/brain tumors, thyroid cancer, and leukemia after exposure to moderate (0.1–1 Gy) and high (>1 Gy) acute doses of sparsely ionizing radiation. 181
The effects of hypergravity on cancer risk remain not fully understood, necessitating further research. Exposure to increased gravitational forces during space missions could disrupt cellular processes and elevate cancer risk.88,90 Even though the duration of hypergravity exposure during space missions is relatively short compared to radiation exposure, further studies are needed to better understand and mitigate potential risks.
Collateral findings of this study could extend beyond oncology; microgravity potentially may impact human fertility and other health conditions. For instance, SMG increased aromatase gene expression, leading to elevated 17-estradiol levels, potentially affecting fertility, testicular cancer, 105 and possibly several other androgen-estrogen-related conditions of human beings. The study on HTU-5 and ONCO-DG1 thyroid cells revealed that microgravity-induced apoptosis occurred by mitochondrial changes in distribution and structure, potentially leading to the development of hypothyroidism in space. Such impairments in thyroid function may provoke health-related challenges for those exposed to microgravity during space missions. 47
Moreover, the findings could have practical implications for astronaut selection and predeparture care. In one study, it was found that EBV-seropositive individuals face increased DNA damage risks during space travel. Adding EBV profiling to routine screenings for astronauts could be a new recommendation to address cancer-related challenges. 182
A study reports the isolation of four strains of Methylobacterium with unique coloration and differential genomic phylogeny of Methylobacteriacea family on the International Space Station.8,183 On the other hand, evidence supports the role of microbiome (bacteria, viruses, and/or fungi) in cancer development and immunotherapy. However, a limited number of microbes are recognized to directly cause cancer. 184 The literature lacks studies discussing the association between space environment-induced microbiome change and cancer. This could be a novel area of space oncology research in the future.
Most ground-based studies on dietary supplementation reveal little to no noteworthy impact on cancer incidence or mortality, occasionally showing adverse effects. 185 However, a study by Patel et al. considers inadequate food and nutrition as one of the stressors to human health during spaceflights. It also claims that many diseases of concern during space exploration, such as cancers, are nutritionally modifiable on Earth. 186 Therefore, providing the astronauts with an optimized nutritional supply could mitigate spaceflight-induced health challenges. 186 Flavonoids are a group of nutritional substances with anti-oxidative, anti-inflammatory, anti-mutagenic, and anti-carcinogenic effects by inducing apoptosis, modulating the autophagy network, and regulating NF-kB (nuclear factor kappa B subunit 1, an important signaling pathway in carcinogenesis).187,188 Considering past terrestrial experiments on dietary supplementation and the mentioned evidence from spaceflight, the authors propose that organizing such trials may not be a current priority but warrants investigation in the future.
Our literature search yielded no studies investigating the effect of psychological stressors on the development or progression of cancer during spaceflight or in simulated conditions. However, various factors during spaceflight may contribute to an extended range of psychological and psychiatric challenges. 16 Considering the suggested role of such stressors in the pathogenesis of cancer, 17 investigating the association of spaceflight-induced psychological stress and carcinogenesis is an area for future research.
Future research should consider various cell types, radiation doses, and types under different gravitational and radiational conditions. This could reduce uncertainty in high linear energy transfer 145 risk prediction and improve tumor lethality outcomes. Even with these suggestions for future research, it is important to note that progress in space oncology may be incremental and requires persistent, diligent investigation.
Despite the wealth of findings, our study had limitations. The small sample size affected statistical power, particularly in the three included animal studies. The conditions simulated in vivo studies that did not fully replicate the unique environments of space and human organisms. Also, the study durations may have been insufficient to observe the desired outcomes. Many studies were cross-sectional due to limited spaceflight access, which hampers the effective evaluation of cancer manifestation and progression under space conditions.
In conclusion, our investigation emphasizes the significance of sustained efforts in the field of space oncology. This field holds the dual potential of enhancing astronaut health safeguards and revealing novel cancer treatment strategies, as well as expanding our comprehension of cancer biology. The identified literature gaps and research requirements in this review are expected to guide forthcoming investigations, setting a trajectory for substantial progress in space oncology.
Supplemental Material
sj-docx-1-wem-10.1177_10806032251349442 - Supplemental material for Space Oncology: A Comprehensive Scoping Review
Supplemental material, sj-docx-1-wem-10.1177_10806032251349442 for Space Oncology: A Comprehensive Scoping Review by Nafise Niknam, Hossein Akbarialiabad, Mohsen Farjoud Kouhanjani, Lydia Johnson Kolaparambil Varghese, Amy Berrington de González, Matthew Mark Melin, Mohammad Shafie’ei, Seyedeh Maryam Mousavi, Seyed Ali Hosseini, Seyed Reza Taha, Thais Russomano, Dedee F Murrell, Ayman Grada, Sancy A. Leachman, Zahra Akbari, Armita Jokar Derisi, Marta Jurga, Ajay Kumar, Saswati Das, Jessica D’Urbano, Gabriella Hakim, Najmeh Sadeghian, Shahram Paydar, Michelle Tarbox, Cameron West, Seyyed Mojtaba Ghorashi, Seyed Hossein Hosseini, Timothy Squire, Christopher G. Bunick and Rowena Christiansen in Wilderness & Environmental Medicine
Supplemental Material
sj-docx-2-wem-10.1177_10806032251349442 - Supplemental material for Space Oncology: A Comprehensive Scoping Review
Supplemental material, sj-docx-2-wem-10.1177_10806032251349442 for Space Oncology: A Comprehensive Scoping Review by Nafise Niknam, Hossein Akbarialiabad, Mohsen Farjoud Kouhanjani, Lydia Johnson Kolaparambil Varghese, Amy Berrington de González, Matthew Mark Melin, Mohammad Shafie’ei, Seyedeh Maryam Mousavi, Seyed Ali Hosseini, Seyed Reza Taha, Thais Russomano, Dedee F Murrell, Ayman Grada, Sancy A. Leachman, Zahra Akbari, Armita Jokar Derisi, Marta Jurga, Ajay Kumar, Saswati Das, Jessica D’Urbano, Gabriella Hakim, Najmeh Sadeghian, Shahram Paydar, Michelle Tarbox, Cameron West, Seyyed Mojtaba Ghorashi, Seyed Hossein Hosseini, Timothy Squire, Christopher G. Bunick and Rowena Christiansen in Wilderness & Environmental Medicine
Footnotes
Acknowledgments
Some of our illustrations have been sourced via a Freepik licensed account and Data Wrapper account. We thank Razieh Kamali for designing the graphical abstract.
Author Contribution(s)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
