Abstract
Commercially available insulin pumps for treatment of diabetes mellitus are currently not qualified to operate in the space environment. This work rigorously tested the fluid delivery performance of a Tandem t:slim X2 insulin pump in both micro- and hypergravity during a parabolic microgravity research flight. The parabolic research flight environment serves as an analogue to the types of transient gravitational loadings experienced during human-led missions, which provides a foundation to expand testing to suborbital and orbital flights in addition to other extreme environmental tests for wilderness dependency. The results of the flight data showed no significant difference between fluid delivery performance at 0, 1, and 2g acceleration regimes, nor at the transitions between gravity environments. Recommendations are made for further experimentation and qualification tests before use in future spaceflight missions.
Introduction
With >150 million cases globally, diabetes mellitus (DM) is both the most prevalent endocrine disorder worldwide 1 and a major barrier to access for human spaceflight. 2 DM is a disorder that primarily affects a patient's blood glucose levels due to a functional deficiency of insulin within the body. 1 In most cases, the deficiency of insulin stems from either a lack of insulin production at the pancreas (type 1 diabetes) or an insufficiency in insulin metabolization and use (type 2 diabetes). 1 Long-term physiologic complications of DM can include peripheral neuropathy, retinopathy, cardiovascular disease, hypertension, and nephropathy. 1 These complications have elevated DM as a recognized disability in countries such as the United States and have erected barriers to access for individuals living with the condition. 3 As access to space continues to increase, there is apparent value in creating safe access to spaceflight opportunities both for the millions of individuals living with DM and for companies looking to expand their market.
Many questions about the physiologic impacts of the space environment as it pertains to DM have been raised, and recent work has addressed this as a critical research area for spaceflight accessibility4,5 and space medicine. 6 What remains unanswered in the current literature is how the technology individuals with DM rely on in their daily lives will fare in space and other extreme environments.
There are two major axes of diabetes management technology: glucose measurement and insulin delivery. State-of-the-art insulin delivery technology is mainly comprised of insulin delivery pumps, with >350,000 individuals in the United States using the technology as of 2019. 7 Insulin pumps feature a reservoir of insulin that is incrementally dosed by preset controls and commands issued by the user, often separated by a length of flexible tubing to the subcutaneous infusion site where the medication is administered. Insulin pump designs and actuation methods vary based on the manufacturer, but all mechanisms must operate functionally to deliver precise amounts of medication on the order of microliters, thus motivating a concern for the performance of these devices in extreme environments. Some insulin pump manufacturers explicitly state that the system is susceptible to large changes in pressure and acceleration loadings and advise that the pump be disconnected for activities such as amusement park rollercoasters and skydiving. 8 As such, the environmental conditions of spaceflight exist outside the approved use cases for the device.
This report assesses the functionality and fluid delivery performance of a state-of-the-art commercial insulin pump in the micro- and hypergravity environments of a parabolic research flight. Parabolic research flights execute a series of high angle dives and ascents to create a microgravity state in the cabin of the plane for up to 25 s. 9 Each parabola first induces a hypergravity state in the aircraft of ∼1.8g for 40 s followed by 20 s of microgravity. This parabolic maneuver is then repeated up to 5 times before returning to a short duration of 1g flight. This set of 5 parabolas is repeated 4 times for the research portion of the parabolic flight. The acceleration loading gathered from the research flight is presented in Figure 1.
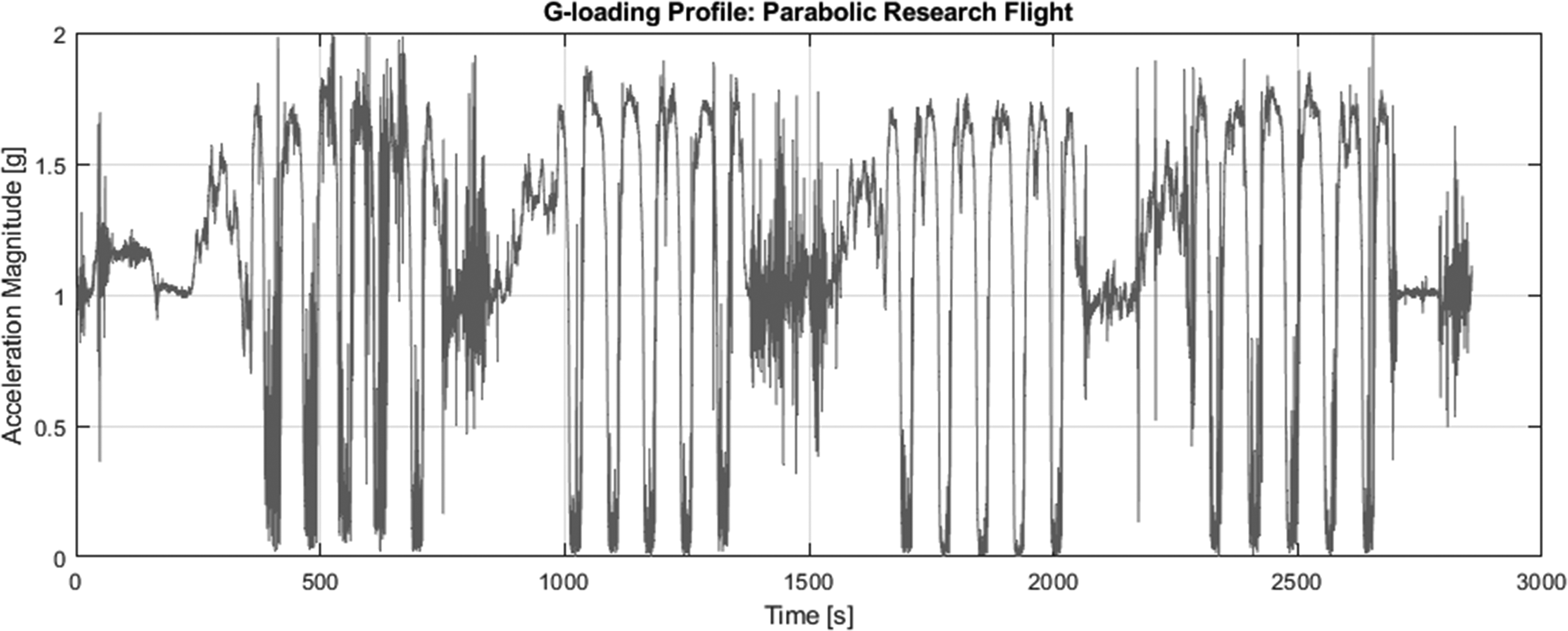
Acceleration loading during parabolic research flight, measured in g's (9.80665 m·s2). Note the 4 sets of 5 parabolic maneuvers accompanied by brief 1g interludes. Time is measured in seconds from the start of the flight.
Through the rigorous testing of a commercial insulin pump in an environment akin to the loadings of spaceflight, significant insight is garnered about performance characteristics. It is important to note that the parabolic flight environment is limited in its ability to adequately reproduce spaceflight conditions. Notably, hypergravity loadings experienced during launch and reentry exceed that of the 1.8g seen during a parabolic flight. Because pumps are a critical life support device for individuals living with DM, these data provide a foundation to conduct further qualification tests in environments more analogous to the loadings of spaceflight and to eventually use insulin pumps in space medicine, creating an accessible path to space for the DM community.
Experimental Setup and Methods
A Tandem t:slim X2 insulin pump (Tandem Diabetes Care, San Diego, CA) was tested in a flying laboratory setup onboard a Boeing 727 as part of a parabolic research flight operated by Zero-G. This specific pump was selected because it has been shown to be globally accurate in both basal and bolus deliveries, 10 and it was one of limited options available during the flight hardware procurement phase of this study. The experimental apparatus consisted of an aluminum support structure that mounted a GoPro HERO11 Black camera (GoPro Inc, San Mateo, CA) and calibrated camera board (seen in Figure 2). Below the calibrated board was the insulin pump, accelerometer, barometer, and lights. The tubing from the insulin pump was attached to the calibrated camera board and given multiple passes across the board in the pattern shown in Figure 2. The insulin tubing terminates with a free end under the calibration board attached to an absorbent fluid sink. The free end of tubing creates a source of error between the intrinsic flow resistance of the test apparatus and that of an insulin pump being used by a patient. Although this discrepancy is captured by baseline experimentation, it is an obvious limitation of the experiment.

Experimental setup and camera calibration board with tubing attached. Red-dyed saline solution fills approximately half the tubing line on the calibration board. Insulin pump, accelerometer, and lights are shown below the calibration board, and the aluminum/acrylic containment box sits behind the experimental apparatus.
The entire apparatus was enclosed in a 16-in cube-shaped aluminum/acrylic housing and mounted to the aircraft hull via an aluminum baseplate. The t:slim pump was filled with a dyed saline solution (consisting of 95% normal saline and 5% red food dye by volume) in lieu of insulin. The discrepancy in viscosity between the two fluids is controlled for through a baseline experiment (detailed below). The error emerging from viscosity differences is further discussed in the Results section of this report, and future experimentation should make use of insulin to eliminate this discrepancy.
The technique used to measure the fluid-delivery performance of the insulin pump was meniscus progression through a fine capillary. The flow rates for insulin-delivery pumps are on the order of microliters per minute—a range undetectable by most mass flowmeters. Thus, for this experiment, a camera was placed overhead and observed the progression of the dyed solution through the insulin tubing against a calibrated board for later image processing. Knowing the inner diameter of the tubing and the size of the camera calibration pattern, the flow can be calculated using simple geometry.
The concept of operations for the flight experiment examines 2 major questions: 1) will the insulin pump inadvertently deliver insulin when not told to do so, and 2) will the insulin pump deliver the proper amount of insulin when commanded in the adverse conditions. To address these core questions, the proposed flight delivery profile was developed, as shown in Figure 3.
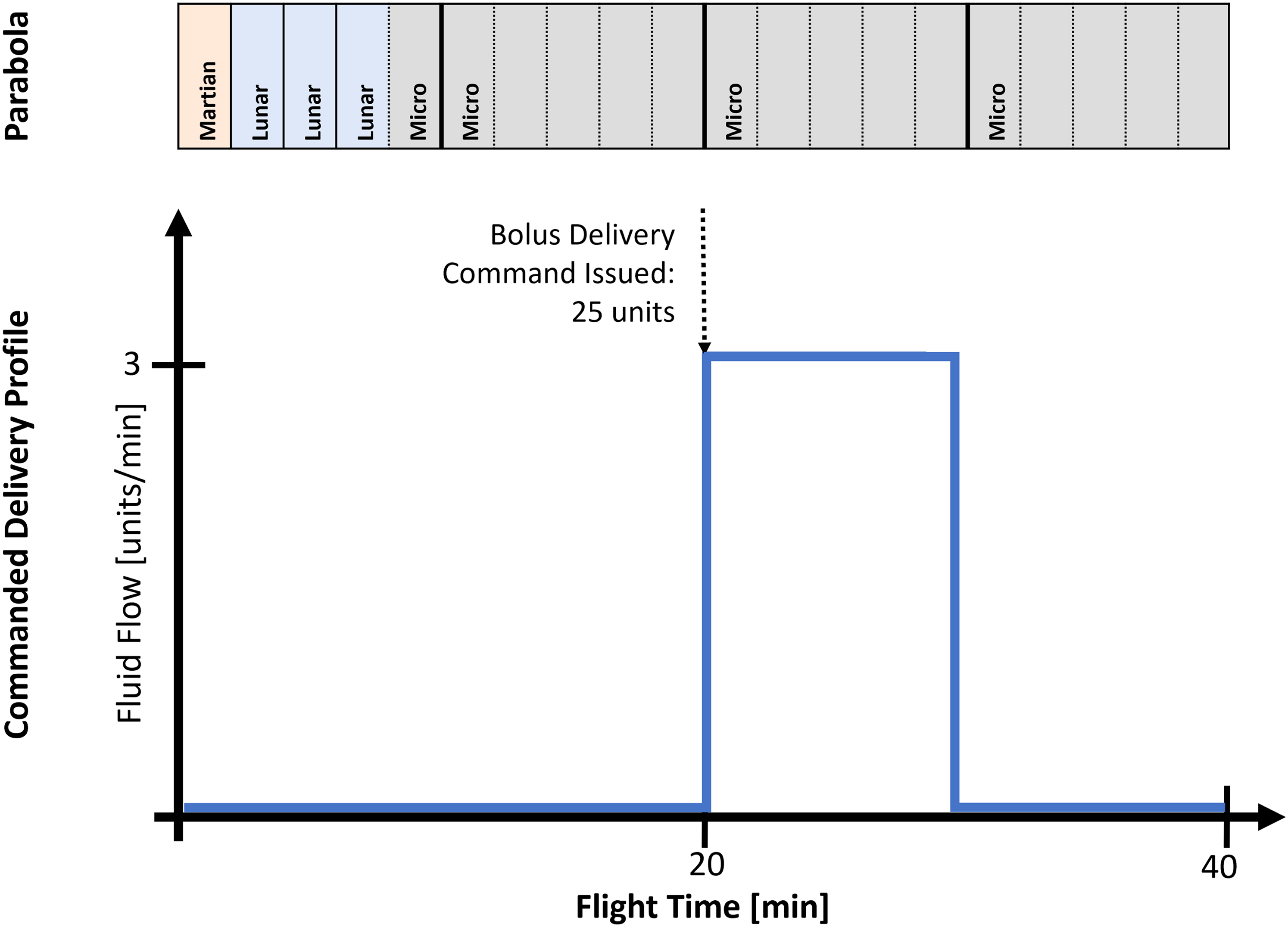
Concept of operations for microgravity pump performance test. The parabolic profiles are given in text, denoting Martin (0.38g), Lunar (0.16g), and microgravity (0g) trajectories and the corresponding delivery rates commanded to the insulin pump. The parabolic flight profile shows that 5 microgravity parabolas, each lasting ∼25 s, occur during the 10-min delivery window in the commanded delivery profile. Note that the unit of fluid volume for insulin pumps is the U-100 unit, equal to 1/100th of a milliliter.
The first 2 sets of 5 parabolic maneuvers investigated how the pump responds to the hypergravity and microgravity transitions. Note that the first set of maneuvers includes 1 Martin (0.38g), 3 Lunar (0.16g), and 1 microgravity (0g) profile. The second set consists entirely of microgravity parabolas. No delivery was commanded, and the pump was observed for inadvertent fluid delivery. For the third set of parabolas, the pump was instructed to deliver a single dose of 25 units of saline. Given the characteristics of the pump delivery system, a single bolus of this size takes ∼10 min to complete. The final set of parabolas again looked for safety concerns with inadvertent dosing. The structure of this experiment is inherently limited in its ability to adequately capture true spaceflight conditions due to the parabolic flight environment. These limitations are acknowledged with the design of the experiment and expanded on in the Discussion of this report. Note that the unit of measure for U-100 insulin is the unit and can be easily converted to milliliters in a 1-mL:100-unit conversion factor.
The delivery metrics of the specific t:slim X2 insulin pump used in the apparatus was first characterized in a 1g baseline test. The baseline test featured the flight-ready experimental setup operating in a controlled laboratory environment. A full operational cycle was performed to yield a complete set of baseline data for the system. Figure 4 shows the fluid-delivery baseline characteristics.
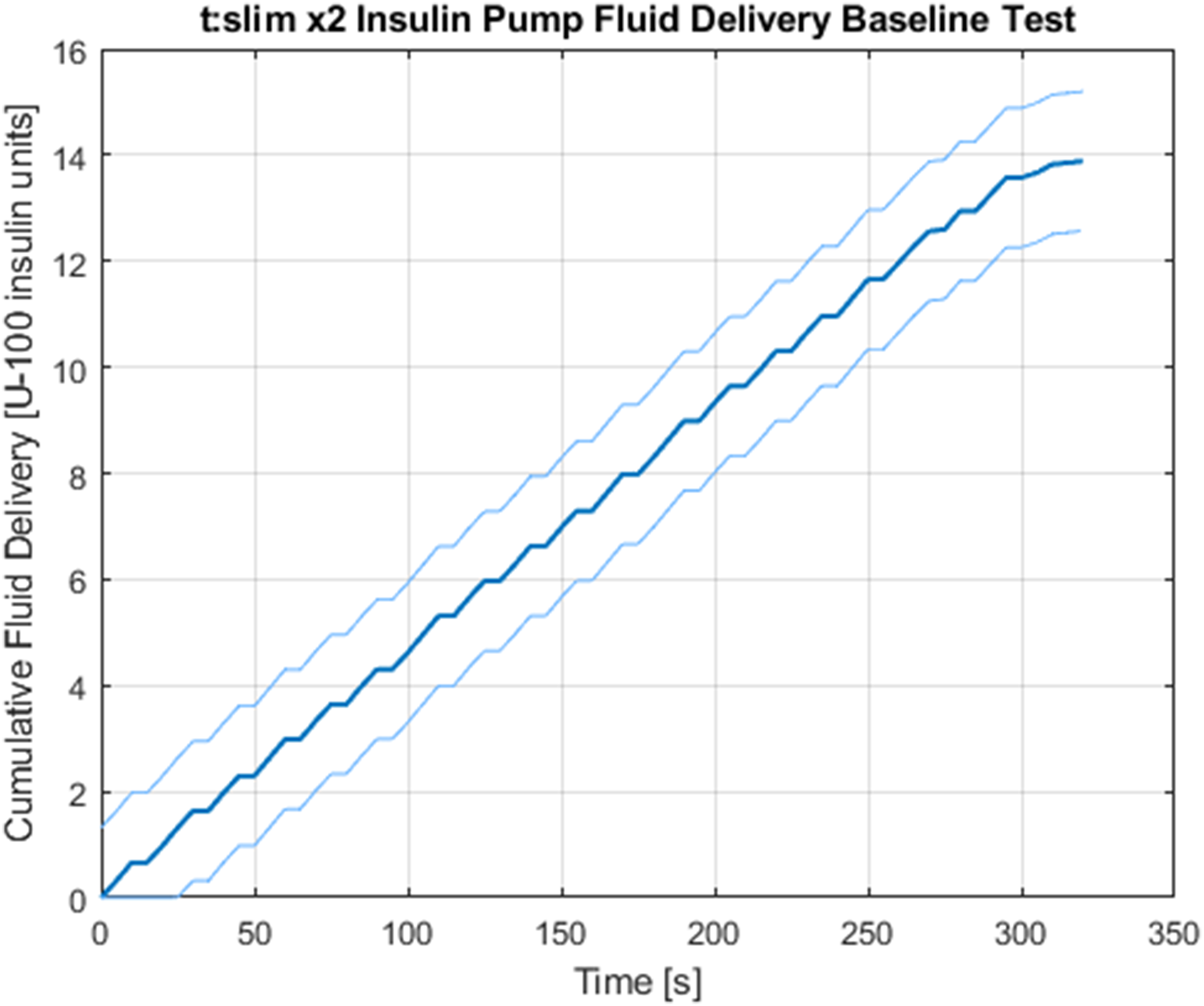
Tandem t:slim X2 insulin pump baseline characteristics delivering saline in a controlled 1g environment. The linear behavior of the delivery system shows regular dosing intervals and short breaks to avoid fatigue at a subcutaneous infusion site. The end of the dosing profile tapers off to deliver a decreasing rate of saline, and a discrepancy emerges with the commanded bolus of 15 units and the observed total delivery. Light bounding lines signify regions of uncertainty due to measurement error.
A bolus of 15 units was commanded and measured using the camera capture technique described earlier. Data from the camera capture were processed in 5-s intervals. That is, from the continuous video feed, 1 frame was extracted every 5 s, and the progression of the dyed fluid was measured against the calibrated board. This granularity provides a sufficient resolution to assess the delivery characteristics of the pump.
From these data, it is apparent that slight discrepancies emerge in the issued delivery command versus the observed delivery amount. An error of 7.43±0.21% is seen at the conclusion of the bolus. This error has 2 probable sources. The first is due to manufacturing tolerances of the internal diameter of the inner tubing that is used as a capillary to calculate volumetric flow. Small deviations from the specified diameter, on the order of micrometers, can disturb the cumulative bolus delivery by up to 1 unit. The second most probable source of discrepancy is from the difference in viscosity between insulin and the dyed saline solution. The viscosity of insulin varies by manufacturer, and the exact values are not widely accessible; however, characterization efforts of analogue insulins show that the dynamic viscosity ranges between 1.064 and 1.146 mPa·s–1, 11 which contains the viscosity of normal saline at 1.07 mPa·s–1. 12 Thus, the dyed saline solution is substituted for insulin in the fluid circuit, and the baseline test is used to characterize any residual discrepancy it causes. With these sources of error acknowledged, the flight data can be interpreted with more accurate insight.
Results and Discussion
To address the first major research question, the performance of the insulin pump was assessed during the first 2 sets of parabolic maneuvers while no bolus commands were issued. Inadvertent insulin delivery is a major safety concern, and thus, the pump was first tested to ensure that no fluid delivery occurred unless explicitly commanded by a user input. The pump performance over the entire flight duration is shown in Figure 5 alongside the acceleration profile and internal aircraft cabin pressure that correspond with the research period.
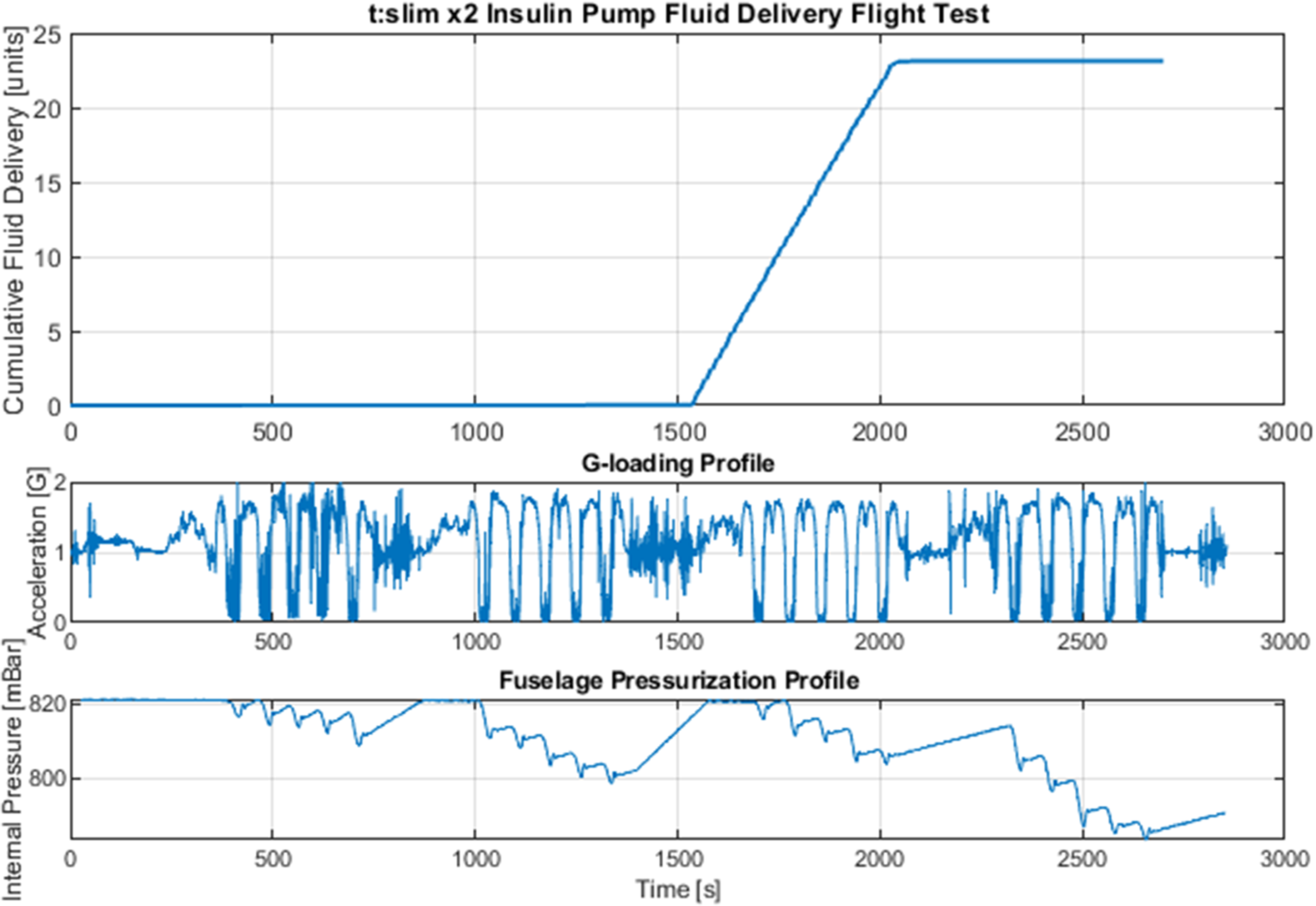
From top to bottom: Insulin pump performance across entire flight duration. acceleration loading inside aircraft, and measured ambient pressure of the aircraft cabin. The first, second, and fourth sets of parabolic maneuvers show the ability of the device to prevent inadvertent insulin delivery. The third set details the insulin-delivery performance during microgravity and hypergravity.
As shown, the pump maintained the displacement of the fluid in the insulin tubing with precision during the parabolic maneuvers. Total insulin increment during the elapsed time of the first 10 parabolas was 0.03±0.03 units. These results suggest that the pump is reasonably adept at preventing accidental dosage in the 0- to 1.8-g range of acceleration loadings. Another consideration for the pump delivery characteristics unique to the parabolic flight environment is the rapid cyclic change of ambient pressure. Due to the large aircraft altitude changes during the flight profile, internal cabin pressures also fluctuate. These fluctuations, shown in Figure 5, yielded no major points of concern in the performance of the insulin pump.
To address the second major research question, the pump performance was assessed during the third set of parabolic maneuvers with a commanded 25-unit bolus delivery. The delivery metrics from this bolus can be seen alongside the corresponding acceleration loading in Figure 6.
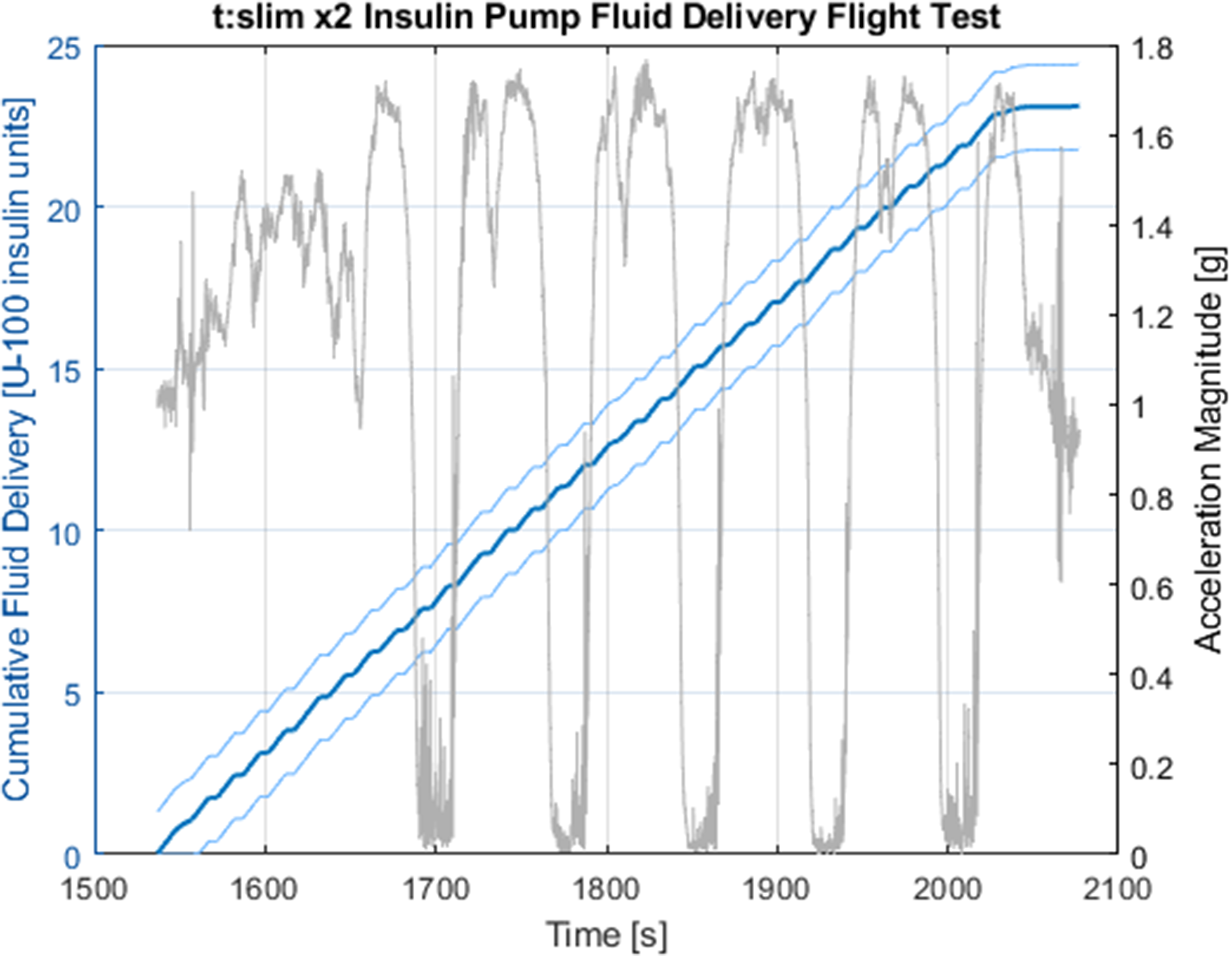
Pump performance shows accurate delivery of insulin in microgravity and hypergravity. The acceleration profile is shown over the cumulative fluid delivery. The linear nature of the delivery is consistent with baseline testing, and a similar discrepancy to the baseline is apparent from the 25-unit commanded delivery.
From Figure 6, it is clear that the discrepancy observed in the baseline testing exists in the flight data as well. The error in commanded versus measured fluid delivery is 7.64±0.13%, similar to the baseline test. Because of this, the underlying error source is likely common between the 2 tests, although further investigation is required for certainty.
The relationship of fluid delivery over time is seen to be linear, both in the baseline and in the flight data. From this the average rate of flow can be found through simple linear regression. Table 1 details the baseline average flow rate and flight test average flow rate.
Average volumetric flow rates.

The flow rates between the baseline and flight tests show a discrepancy of 1.85%. The performance qualitatively appears similar as well, with incremental dosing observed between the 2 tests. Due to the low sample size of these flow rates, conclusions about performance must be approached with caution, and further testing is required to make informed decisions on volumetric flow rates for the pump in the given environmental conditions.
The results demonstrate that the insulin pump selected for testing can both resist adverse dosing and administer the correct dosage of fluid during times of sustained hypergravity, sustained microgravity, and the transitions between micro- and hypergravity. Further testing is required to validate these results, and such testing should incorporate larger sample sizes and varying designs of insulin pumps.
The parabolic flight environment is limited as an analogue to genuine launch and reentry loadings. Peak reentry loading depends on the vehicle used, but in the case of the SLS Orion capsule, loadings peak at ∼4g and are sustained over 2g for >200 s. 13 This limitation identifies the need for further investigation of sustained hypergravity, such as with centrifugation studies, prior to use in the space environment.
Conclusions and Future Work
The ability for a consumer insulin pump to perform adequately in adverse environmental conditions was demonstrated. The fluid-delivery performance of the t:slim X2 insulin pump in both micro- and hypergravity conditions maintained consistency with the baseline test in a static 1g laboratory environment. Coupling between ambient pressure changes and gravitational loading on delivery metrics was not observed, suggesting robustness of the pump design. The tests completed on the parabolic research flight are promising steps toward flight qualification of this critical life support technology for individuals with DM.
Further testing in suborbital and orbital flight environments is needed to better understand the technology's limitations in sustained spaceflight conditions and genuine launch loadings. In addition to studies that investigate insulin pump performance under altered gravitational loadings, the technology also must be tested in other adverse conditions of the space environment, such as changes in ambient radiation, gas concentrations, and total pressure. The applicability of this work further extends to use of these devices in extreme environments on Earth as well. Such applications could include primary use for wilderness medicine and medicine in austere communities. Further testing is needed to fully assess the applicability of these devices in these situations. For the application of human spaceflight, insulin pump technology is a promising solution to enable safe management of DM in the space environment and overcome a previously insurmountable barrier to access to space for millions of people worldwide.
Footnotes
Acknowledgments
The authors thank all the funding organizations. This work would not have been possible without the support, guidance, and investment made by each of you.
Author Contribution(s)
Data Availability
The data associated with this research is available on request.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was sponsored by the Massachusetts Institute of Technology Space Exploration Initiative and the Department of Aeronautics and Astronautics. Further flight and mission support was provided by Mission: AstroAccess, and flight test materials were donated by Tekni-Plex Healthcare. This work also was supported by the National Science Foundation Graduate Research Fellowship Program (Grant No. 2141064).
