Abstract
We present a case of neurotoxic effects in a pediatric patient after envenomation by a timber rattlesnake (Crotalus horridus) in the Appalachian upstate of South Carolina. Though some members of this species are capable of primarily neurotoxic envenomation, there is heterogeneity in venom composition, and neurotoxic timber rattlesnakes are not endemic to the Appalachian region. However, neurotoxic effects caused by C horridus species lacking typical neurotoxins have been suspected, though not previously confirmed in the medical literature. This case presents a patient who was envenomated by a genotypically confirmed non-neurotoxic C horridus but who nevertheless presented with symptoms consistent with primary neurotoxicity. Neurotoxic effects can be variable in their response to traditional antivenom, though this patient demonstrated rapid response to treatment, representing a novel case in the literature of neurotoxic effects from a snake lacking typical neurotoxins with documented improvement with traditional antivenom.
Introduction
The timber rattlesnake (Crotalus horridus) is a venomous species of pit viper (family Viperidae) geographically widespread throughout the eastern United States. Envenomation from these snakes can be severe, likely due in part to their size, which allows them to inject significant quantities of venom. 1 Venom from these rattlesnakes is often traditionally categorized as containing neurotoxins, known as type A venoms, or as lacking neurotoxins, known as type B venoms; the authors note, however, that this dichotomy is likely too restrictive to capture the extensive variation in venom composition and effects present in rattlesnakes.2–4 The known neurotoxin present in some C horridus is a potent heterodimeric phospholipase A2 (PLA2) also known as crotoxin, or termed canebrake toxin in the case of the timber rattlesnake. 5 This toxin inhibits acetylcholine release at the presynaptic neuromuscular junction, which can lead to effects including facial or extremity paresthesia, ptosis, lethargy, confusion, and difficulty speaking or swallowing. 6 Other clinical features that can be observed are myokymia or fasciculations of the muscles. This is hypothesized to be caused by the venom protein binding at calcium channels in peripheral nerves resulting in increased excitability. 7
Hemotoxic (type B) venoms often contain high levels of snake venom metalloproteinases (SVMPs), which are responsible for tissue damage. 8 Once injected, these enzymes cause local damage to the surrounding muscle and tissues producing erythema, edema, and in some cases muscle breakdown. Hypofibrinogenemia, prolonged prothrombin time, and thrombocytopenia are the laboratory hallmarks of this venom. 9 While most rattlesnake species possess hemotoxic venoms, neurotoxic venoms are recognized in at least 10 species.3,10 Several species (including C horridus) exhibit a polymorphism of venom type, meaning that different populations within this single species possess the neurotoxic canebrake toxin (type A venom), whereas other populations do not. In certain states, including Georgia, South Carolina, and others, both neurotoxic and non-neurotoxic venoms have been documented, though canebrake toxin–containing species in South Carolina are found only in coastal regions. 11 This polymorphism may make envenomation from C horridus challenging to recognize or treat and of clinical interest. Envenomation by individual snakes that possess neurotoxic PLA2-containing venom can produce significant neurotoxic effects as well as damage to the surrounding tissues. 12
Case Report
A 9-year-old female presented to an emergency department (ED) in the rural Appalachian region of South Carolina with a chief complaint of a snake bite. The family reported that she had been walking outside in sandals when she was bitten by a snake multiple times on the right ankle and foot. It was estimated that she arrived 45 min after the envenomation occurred. A family member apprehended and decapitated the snake and presented it to the ED for identification. The physician caring for the patient correctly identified the snake as a juvenile timber rattlesnake (Figures 1 and 2), and the carcass was subsequently donated to a regional academic organization for genotyping. On arrival, the patient was ill-appearing with nausea, vomiting, and shortness of breath. She was hypotensive with a blood pressure of 79/56 mm Hg and tachycardic with a heart rate of 175 beats/min, findings suggestive of acute shock. Multiple puncture wounds were noted on the dorsum of the right foot as well as the lateral aspect of the right ankle (Figure 3). There was no erythema or edema at the bite sites initially. At the time of presentation, the patient was found to have bilateral ptosis, facial diplegia, decreased pulmonary excursion, and diminished bilateral grip strength, and she complained of facial paresthesia. Her initial blood glucose level was noted at 103 mg·dL–1.

Head of the offending decapitated snake showing typical slit pupils, heat-sensing pits, and recurved fangs of pit vipers.

Portions of the snake body demonstrating typical C horridus patterning of chevron circumferential markings and a dorsal stripe.
The patient's foot at initial presentation demonstrating multiple dorsal wounds and lack of edema and erythema.
The patient was administered a 20 mL·kg–1 normal saline bolus for shock, 0.5 microgram·kg–1 fentanyl for pain control, and 4 mg ondansetron for control of nausea and vomiting just after arrival. The initial medical workup included a complete blood count (hemoglobin 16.2 g·dL–1, platelets 574,000 microliter–1), comprehensive metabolic panel (all values within normal limits), prothrombin time/international normalized ratio (PT/INR) (13.7 s/1.1 s), and a fibrinogen level (311 mg·dL–1), all drawn about 1 h after envenomation (Table 1). After receiving fentanyl, the patient developed mild sedation, bradypnea, and a right lateral rectus palsy. Naloxone was delivered to reverse the sedative effects, though her lateral rectus palsy remained. Similarly, the other neurologic findings persisted. During the time the patient was present in the ED, no swelling developed at the site of the bite. Despite the lack of local inflammatory signs, the combination of hypotension, tachycardia, and neurologic symptoms prompted the decision to initiate antivenom treatment. The patient was administered 6 vials of Crotalidae polyvalent immune fab (ovine) (FabAV, CroFab, BTG International Inc, Conshohocken, PA) at standard protocol rates over 60 min. All the neurologic symptoms resolved and the patient’s shock stabilized on completion of the FabAV infusion.
Timeline of laboratory values and fabav administration.
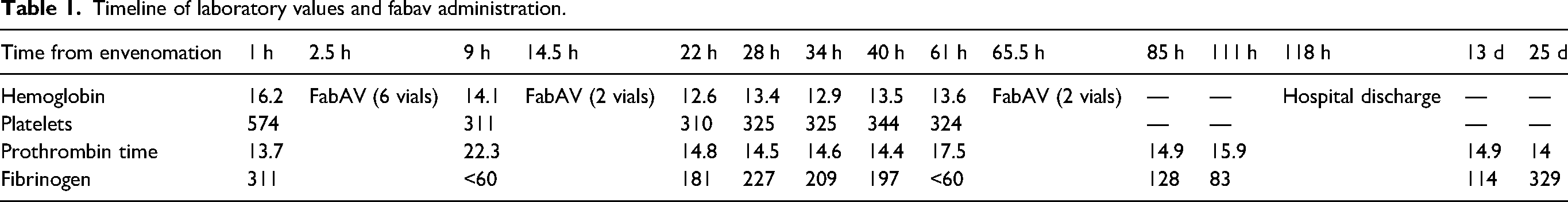
Following stabilization, the patient was transferred to a tertiary care center for admission to the pediatric intensive care unit. On the night of arrival, approximately 9 h following envenomation, repeat coagulation studies revealed development of coagulopathy with a fibrinogen level of <60 mg·dL–1 and a PT of 22.3 s. The patient's platelets were within normal limits at 311,000 microliter–1. A creatine kinase level was measured at 104 IU·L–1. With the development of coagulopathy, the patient was treated with 2 vials of FabAV. The coagulation studies stabilized, but 2 d later coagulopathy returned, and an additional 2 vials of FabAV were administered. Following this, the PT stabilized, though the fibrinogen level remained low (Table 1). The patient’s creatine kinase remained normal. The patient did well overall with continued clinical improvement and was ultimately discharged home on hospital day 5. She did not develop any additional coagulopathy or experience any neurologic symptoms after discharge, though her fibrinogen level remained below normal for 2 wk after envenomation.
Given the presentation of symptoms consistent with neurotoxic envenomation, genotyping was performed on the snake that was delivered to the ED. The snake was tested for the presence of both neurotoxic PLA2 subunits using polymerase chain reaction (PCR). 11 Following the methods described by Margres et al, 11 PCR revealed that the snake causing this patient's envenomation did not possess either subunit required for expression of the neurotoxic canebrake toxin.
Discussion
Because timber rattlesnakes are polymorphic for venom type, initially it was thought that the animal that envenomated this child possessed neurotoxic PLA2 capable of producing neurotoxic effects. However, in South Carolina, timber rattlesnakes containing canebrake toxin are not known to exist in the upstate region where this envenomation occurred. 11 Clinical suspicion at the time of the patient's arrival to the ED was that her symptoms represented a novel geographic presentation of a timber rattlesnake possessing canebrake toxin. However, the snake involved in this case was definitively shown not to possess this toxin. There are no other currently described venom components within timber rattlesnakes that have been proven to be neurotoxic in nature.
It is hypothesized that a myotoxin may have been responsible for the neurotoxic presentation witnessed in this patient. Juvenile snakes have been recognized as producing neurotoxic effects, and many timber rattlesnakes contain a large amount of myotoxin in their venom, which has been known to cause some neurotoxic effects in other Crotalus species.12–14 In addition, juvenile specimens of timber rattlesnakes tend to express different venom components than adults. 15 For example, adult timber rattlesnakes containing hemotoxic venom possess large amounts of SVMPs, whereas juvenile venoms are largely devoid of SVMPs. 16 The lack of clinically significant tissue damage initially observed in this patient was thought to be related to the offending snake's juvenile age and venom likely devoid of SVMPs. While fasciculations and myokymia are well-described neurologic complications in patients undergoing many crotalid species envenomation, including the timber rattlesnake, development of focal cranial nerve paralysis is rare, even among confirmed crotaline neurotoxic envenomation. When cranial nerve paralysis does occur, it is more specific for neurotoxic envenomation. 17
Several aspects of this patient's presentation make the case unique, irrespective of the presence of neurotoxin. Typical symptoms seen in neurotoxic envenomation are primarily paresthesia, headache, and myokymia. 18 Severe cases may present with total neuromuscular and diaphragm paralysis. Our patient presented with bilateral facial nerve involvement, which is rare. The only other documented case of this finding in C horridus envenomation occurred in a region known to harbor species containing canebrake toxin. 19 Facial diplegia has been documented in other species with primarily neurotoxic venom, however. 20 Interestingly, our case occurred in the Appalachian region of South Carolina, where C horridus populations have not shown canebrake toxin expression, 11 and previous clinical neurotoxic symptoms have not been recognized. Development of cranial nerve paralysis in timber rattlesnake envenomation is unexpected. Our case underscores this possible clinical neurologic presentation in C horridus despite the known lack of neurotoxic PLA2 within the offending venom. Additionally, this case represents the first incidence of FabAV resolving cranial nerve paralysis in C horridus envenomation. Reports of efficacy of FabAV in treating neurotoxicity are mixed, and when it is effective, it is usually limited to improvement of myokymia, fasciculations, or respiratory difficulty attributed to neurotoxic effect.21,22 In another C horridus neurotoxic envenomation described by Madey et al, 19 genotyping was not performed, but facial diplegia and ophthalmoplegia persisted, even after FabAV administration. Our patient's neurologic symptoms did not appear to be consistent with any sedative effects of treatment, and they all resolved very rapidly and simultaneously with the hemodynamic shock on completion of the FabAV infusion.
Finally, this patient did not demonstrate local cytotoxicity or hemotoxicity at the initial evaluation, which is atypical for envenomation in the region where the bite occurred. Delayed presentation of typical hemotoxic effects has been noted in other phenotypically neurotoxic presentations after crotalid envenomation, though in our patient's case this may be the result of relatively rapid initiation of antivenom. 23 It is unclear in snake species lacking neurotoxic PLA2 that still produce neurotoxic effects whether there is a delayed effect of the hemotoxic components of venom.
Conclusion
This case represents a rarely seen complication of timber rattlesnake envenomation. Although the snake in question ultimately tested negative for neurotoxic PLA2 by PCR and therefore definitively lacked the canebrake neurotoxin in its venom, this patient presented with prominent neurotoxic symptoms including bilateral ptosis, right lateral rectus palsy, bilateral facial weakness, facial paresthesia, decreased pulmonary excursion, and bilateral diminished grip strength. None of these findings were explained by other potential etiologies such as sedation, opioid effects, or rhabdomyolysis. This points toward the hypothesis that other venom components were creating the neurotoxic effects and represents the first case to show significant clinical support for this mechanism. This case also demonstrates the first documentation of cranial nerve paralysis reversal with FabAV in a timber rattlesnake envenomation. This patient's presentation and clinical course accentuate the importance that emergency and other acute care providers should be able to recognize atypical neurologic symptoms from rattlesnake envenomation, even in regions known not to have endemic activity of neurotoxic PLA2. This case also demonstrates the need to not delay treatment with FabAV in these patients.
This report was reviewed and approved by the Institutional Review Board of Prisma Health Upstate and was felt to have met all required ethical standards for a case report. Written parental consent for use of patient data and images was obtained.
Footnotes
Author Contribution(s)
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
