Abstract
Introduction
Buthus species, including B paris, are classified as one of the most dangerous scorpion genera in Morocco, implicated in several cases of human death. Our objective is to characterize, for the first time, the toxicity and histopathologic and biochemical impacts of B paris venom.
Methods
We investigated the experimental pathophysiology of B paris venom by examining histologic changes in vital organs (heart, kidneys, liver, and lungs) and assessing biochemical enzymatic markers (alanine aminotransferase, aspartate aminotransferase, lactate dehydrogenase, creatine phosphokinase, urea, and creatinine) in mice injected subcutaneously with 2 doses of 400 and 450 mg·kg–1.
Results
Our results showed that the subcutaneous median lethal dose of B paris venom was around 0.52 mg·kg–1. Histologic findings revealed significant tissue damage in the previously mentioned vital organs, confirmed through biochemical analysis indicating impaired heart and liver functions. Additionally, an increase in urea, creatinine, and glucose levels occurred following B paris venom injection.
Conclusion
Our findings show that B paris venom exhibits a high level of experimental toxicity. These results highlight the potentially lethal nature of this venom and emphasize the potential medical importance of this species.
Introduction
The scorpion fauna of Morocco is known for its richness, diversity, and significant endemism.1,2 Buthus spp are widely distributed in Morocco and account for 29.5% of the country's scorpion fauna.2,3 Some Buthus venoms are rich in potent, medically relevant toxins, and several species within the genus have caused fatal human envenoming.4–6
Several experimental studies have investigated the pathophysiologic effects of venoms from some Moroccan scorpion species fauna. Bouimeja et al7,8 reported that venoms from Buthus occitanus and Buthus atlantis induce severe alterations in the tissues of vital organs in mice, including the heart, liver, lungs, and kidneys. Ait Laaradia et al 9 indicated that venom from Buthus lienhardi causes extensive tissue damage in organs such as the lungs, heart, kidneys, and liver along with hematologic impairments manifested by a decrease in red and white blood cell counts.
Other studies conducted on the venoms of Hottentotta gentili, Androctonus liouvillei, and Androctonus mauritanicus have demonstrated highly toxic effects, including physiologic, histologic, biochemical, and behavioral disturbances.10–12
Following Vachon's famous monograph in 1952 13 on the systematics and distribution of North African scorpions, Lourenço (2003) 14 revised the North African scorpions of the genus Buthus. Consequently, the subspecies Buthus occitanus ssp malhommei and Buthus occitanus malhommei and Buthus occitanus paris were reassigned as B occitanus (Amoreux, 1789), B malhommei (Vachon, 1949), and B paris (C. L. Koch, 1839).
This study aims to characterize for the first time the toxicity and histopathologic and biochemical impacts of B paris venom in the vital organs of mice, and the findings from this study will enhance our comprehension of B paris venom toxicity in the mouse model. This knowledge should be useful in the development of targeted treatment approaches, refinement of antivenom therapies, and optimization of intervention strategies aimed at mitigating the deleterious effects of scorpion envenomation.
Methods
Statement of Ethics Approval
Throughout this study, we rigorously adhered to the ethical guidelines outlined by the World Health Organization for the ethical treatment and use of laboratory animals. The procedure was carried out in accordance with the institutional protocol following the 1986 Animal Welfare Act of the European Council Directive: 86/609 EEC. This protocol is approved by the local ethics committee of the Research Laboratories of the Faculty of the Polydisciplinary Faculty at the University of Beni Mellal to minimize animal suffering and ensure that the number of animals used does not exceed the statistical requirements of the experiment.
Study Area
Scorpions were studied in the mountainous regions of Zaouiat Ahansal (31° 49′ 53.1′′ O 6° 2′ 26.999′′) in the Azilal Province of central Morocco. They were collected from different microhabitats within the study area, under stones, rocks, and tree bark and even in litter.
Scorpion and Venom Extraction
In this study we are concerned with the scorpion species B paris (Figure 1). Morphologic determination of the species was carried out by Professor Oulaid Touloun (Sultan Moulay Sliman University) following the identification key described by Lourenço. 14 At the laboratory, specimens were kept alive in individual containers pending venom extraction, and venom was collected by electrical stimulation of the telson, as described by Ozkan et al. 15 The collected venom was diluted with sterile distilled water and centrifuged for 15 min at 15,000g. The protein content of the supernatant was determined using the method of Bradford. 16 Diluted venom was stored at −20 °C until further use.

Male B paris from Zaouiat Ahansal in Azilal Province.
Animals
Male Swiss mice (24–28 g) were used for determining the median lethal dose (LD50) by subcutaneous injection and determination of histopathologic and biochemical changes after venom injection. The animals were kept at a constant room temperature (25°C) with a 12-h dark-light cycle and free access to food. All procedures were conducted in accordance with approved institutional protocols and animal care and use provisions prescribed in the Scientific Procedures in Live Animals Act 1986 (European Council Directive: 86/609 EEC).
Determination of LD50
Five groups of 6 male Swiss mice were subcutaneously injected with increasing doses of venom (450, 500, 550, and 600 micrograms·kg–1) diluted in 200 mL physiologic saline solution. An equivalent volume of saline solution was injected into 6 mice as a control group. After injection, the animals were monitored for 24 h, and the number of deaths was recorded as a function of the dose. The dose required to kill 50% of the animals (LD50) was calculated using probit analysis.17,18
Histologic Study
For histopathologic study, 2 groups of 6 mice were subcutaneously injected with 400 and 450 micrograms·kg–1 of venom, respectively, while a control group of 6 mice received a 0.9% NaCl solution. At 3 h postinjection, the animals were sacrificed, and their vital organs such as heart, liver, kidneys, and lungs were dissected and fixed overnight in 10% formalin solution. After 24 h of fixation in formalin, the collected organs were cut along a transverse axis to obtain fragments with a thickness of 4 mm. The tissues were subsequently dehydrated in a series of increasing alcohol concentrations and then impregnated with paraffin. The sections were stained with hematoxylin-eosin for pathologic studies as described by Kiernan. 19
Biochemical Analysis
Biochemical analysis was conducted on the sera of mice that were subcutaneously injected with 400 and 450 micrograms·kg–1 of venom or a 0.9% NaCl solution. A blood volume of 0.7 to 1.0 mL was obtained from the jugular vein of each mouse and centrifuged at 3000 rpm for 10 min. Aspartate aminotransferase (ASAT), alanine transaminase (ALAT), lactate dehydrogenase (LDH), creatine phosphokinase (CPK), urea, and creatinine levels were measured in sera of both the control and test groups. The analyses were determined using standard methods with a biochemical analyzer (cobras c311, Roche Diagnostics, Indianapolis, IN).
Statistical Analysis
Data are reported as mean ± SEM and were subjected to a one-way analysis of variance. Post hoc differences between group means were tested with the Tukey test. Values of P <0.05 were considered significant. Statistical analyses were performed using IBM SPSS version 21 (IBM Corp, Armonk, NY).
Results
LD50 Determination
The LD50 was obtained when mice were inoculated by 0.52 mg·kg–– of B paris venom subcutaneously (Table 1).
Comparison of B paris venom experimental toxicity with venoms from some toxic scorpion species found in Morocco.

In Vivo Effects of Envenomation
After being injected, mice showed various signs of intoxication following all injections (ranging from 0.45 to 0.6 mg·kg–1 of venom), particularly with doses of 0.55 and 0.6 mg·kg–1. The observed signs included the following: grooming, chewing, weakness, mouth rubbing, hypersalivation, rotating behavior, tremors, and convulsions leading to death.
Histopathologic Analysis
Histopathologic analysis of some organs from the mice after subcutaneous injection of 400 and 450 micrograms·kg–1 of B paris venom for 3 h showed noticeable pathologic changes in the liver, lungs, kidneys, and heart tissues (Figure 2 and Table 2).
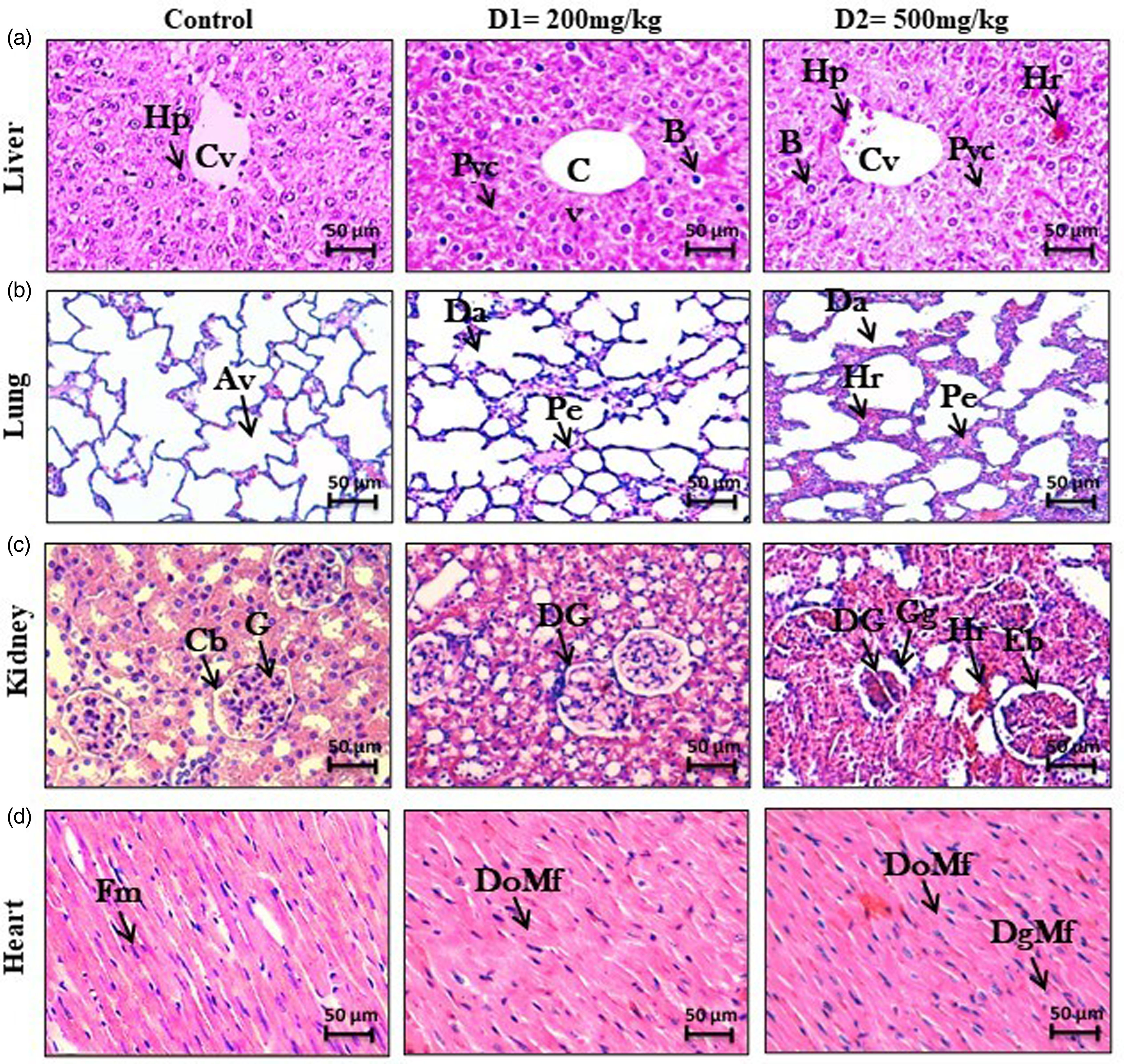
Histopathologic alterations of samples from mice inoculated with 400 or 450 µmicrograms·kg–1 of B paris venom. Light microscopic analysis of liver (a), lungs (b), kidneys (c), and heart (d) tissue specimens in healthy mouse as control. (a1, a2) Liver showed noticeable pathologic changes consisting of balloonization of hepatocytes (
Histopathologic changes in selected organs of adult male mice injected with different B paris venom doses (D1=400 micrograms·kg–1 and D2=450 micrograms·kg–1).
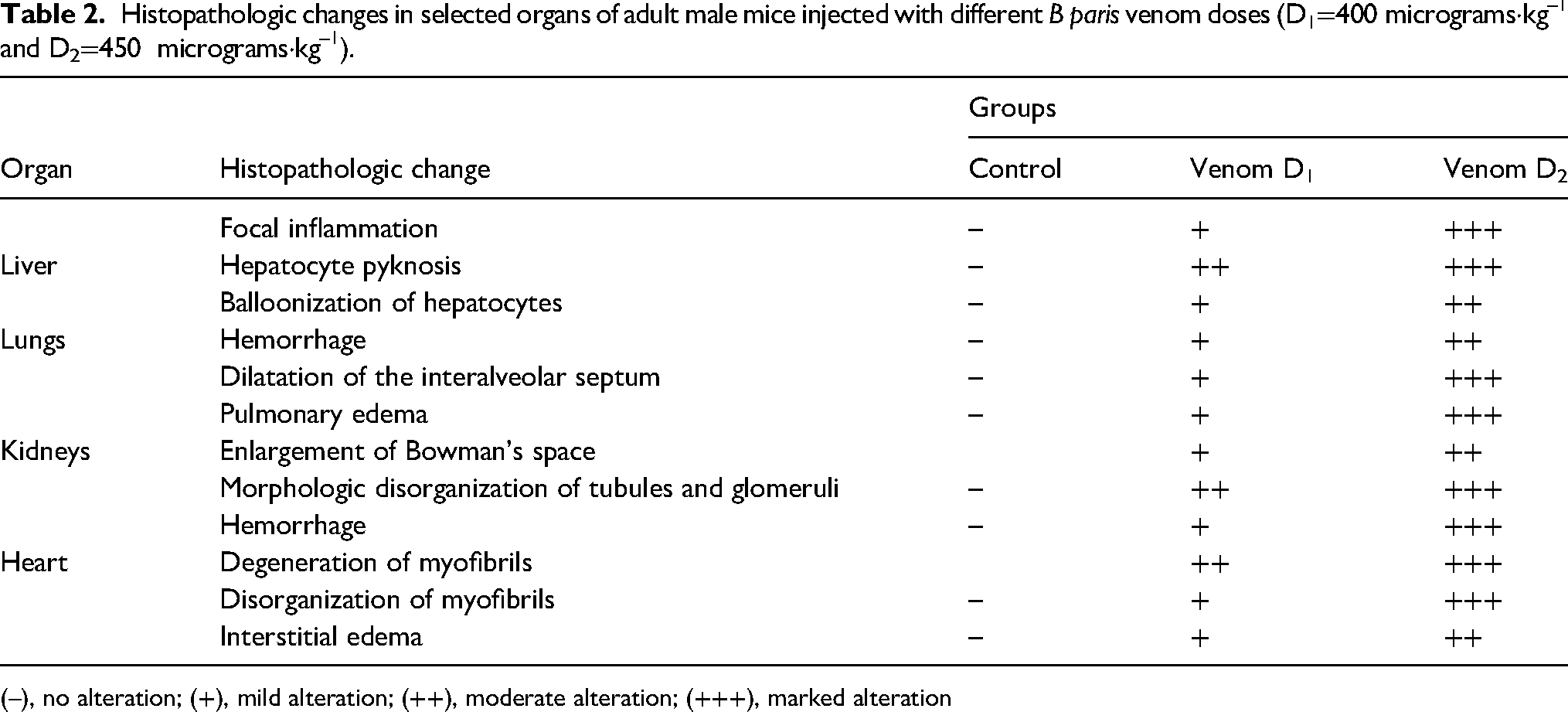
(–), no alteration; (+), mild alteration; (++), moderate alteration; (+++), marked alteration
In the liver, various lesions were observed, characterized by a balloonization of hepatocytes, hemorrhage, and hepatocyte pyknosis (Figure 2, a1, a2). Examination of the pulmonary tissue revealed an enlargement of the interalveolar septum and pulmonary edema. Additionally, hemorrhage also was detected following the injection of a dose of 450 micrograms·kg–1 (Figure 2, b1, b2). The renal examination revealed an enlargement of Bowman's space accompanied by abnormalities in the glomeruli. When animals were injected with a dose of 450 micrograms·kg–1 of B paris venom, glomerular swelling and signs of hemorrhage were observed (Figure 2. c1, c2). The pathologic effect of B paris venom on heart showed disorganization and degeneration of myofibrils (Figure 2, d1, d2).
Biochemical Analysis
Biochemical analysis of envenomed mice sera has shown a significant increment in the level of ALAT (P<0.05), ASAT (P<0,05), and LDH (P<.05). We observed an elevation in CPK levels in both groups of venom-injected mice, but this increase was not statistically significant (P=0.065). Regarding the metabolites analyzed, urea, creatinine, and glucose showed significant alterations (P<0.05) in the sera of envenomed mice.
Discussion
The genus Buthus is represented in Morocco by 18 species, 2 including B paris, which has a wide distribution in the country. 20 It is suspected of being the cause of several human deaths in the country.21,22
To date, few studies have been published on the toxicologic effects of B paris venom. However, some studies on the venom of other Buthus spp may provide insights into the pathophysiologic, histologic, and biochemical characterization of B paris venom. The study of the toxicity of B paris venom from the Azilal Region of Morocco fits into this context.
Previous investigation of Moroccan scorpion venom toxicities including that of B paris 21 have reported the subcutaneous LD50 of B paris scorpion venom as 0.45 mg·kg–1. This differs slightly from that found in our study (0.52 mg·kg–1) but is similar to the LD50 of other Buthus species, such as B occitanus. 7 This difference could lend some support for the idea that venom toxicity varies according to species size, age, nutrition, climatic, and geographic conditions.21,23 The LD50 value (0.52 mg·kg–1) suggests that B paris venom is highly toxic and supports the probable medical significance of the species and the importance of further research to understand and address the potential medical consequences associated with its venom.
Histopathologic analysis of the vital organs (heart, kidneys, liver, and lungs) of mice injected with 400 or 450 micrograms·kg–1 of B paris venom showed severe damage to all vital organs. In the renal tissue we noted enlargement of Bowman's space, glomerular disorganization, hemorrhage, and inflammation. Indeed, previous studies have shown the same alterations in the kidney. 24 Therefore, these results are concordant with those reported in other studies (eg, the study by Mirakabadi et al 25 ). Thus, histologic observations of the kidneys showed a correlation with the results of biochemical analysis of creatinine, whose increase signifies renal damage. In a similar study, venom from the Brazilian scorpion Tityus serrulatus caused glomerular degeneration, reduced blood flow to the kidneys, and reduced filtration levels. 26 In another study, during experimental scorpion envenomation of rabbits injected with Androctonus mauretanicus venom, elevations were seen in urea and creatinine, both known to be good biochemical markers for renal dysfunction, thus implying kidney impairment. 27
B paris venom also induces histologic alterations in the lungs, reflected in thickened alveolar septa and hemorrhages. These results are in line with those described by Khemili et al 28 and Emamet and Al-Otaibi, 29 who showed that the lungs exhibit alveolar wall thickening and polynuclear infiltration following a DL50 injection of Androctonus amoreuxi venom. We also observed histologic lesions in the heart following injection of B paris venom into mice. These pathologic changes included degeneration and disorganization of myofibrils and inflammatory infiltrates. These results confirm those of Adaika et al. 30 This lung damage is confirmed by elevations in biochemical markers (LDH and CPK) implicated in lung and heart dysfunction. Our results are in line with those of de Oliveira Yamashita et al 31 and Bakir et al. 32
Microscopic observation of the liver revealed numerous alterations, including hemorrhage and edema. Similar results were reported by Zangiabadi et al 33 and Darkaoui et al, 10 who confirmed the presence of several lesions in the liver such as hepatic congestion with hemolysis after envenomation by the scorpions Tityus discrepans and Androctonus mauretanicus. Elevated serum transaminases (ASAT and ALAT) correlated with the histopathologic findings; similar results have been reported in pathophysiologic studies of venoms from the medically important scorpion Leiurus quinquestriatus and the viperid snake Echis pyrimidium, respectively.34,35
Previous studies have found similar pathophysiologic effects induced by venom from the most medically important Moroccan scorpions, including A mauretanicus and B occitanus, such as irregular breathing, hypersalivation, grunting, jumping, tremors, hyperactivity, weakness, and coma.7,9 Our research in the Azilal Region of Morocco on the toxicity of B paris venom sheds light on the connection between histopathologic changes, observed symptoms, and clinical outcomes. With B paris being one of the 18 scorpion species in Morocco and suspected of causing human fatalities, our study highlights the need for further investigation in regions where these scorpions are prevalent.
Conclusion
This study aimed to investigate the experimental toxicity and pathophysiology of B paris venom following subcutaneous injection of mice at 2 dose levels. The subcutaneous LD50 of B paris venom was 0.52 mg·kg–1, indicating its high potency. Histologic examinations revealed significant impacts on various vital organs in envenomed mice following scorpion venom injection. These findings were further supported by biochemical analysis, which demonstrated an elevation in specific enzymes (ALAT, ASAT, and CPK) as well as urea and creatinine levels. Notably, B paris venom induced extensive tissue damage in the lungs, heart, kidneys, and liver. Overall, the results of this study highlight the highly toxic nature of B paris venom and its detrimental effects on vital organs in the experimental mouse model. The histologic and biochemical analyses provide valuable insights into the specific organs affected and the biochemical changes induced by the venom. These findings contribute to a better understanding of the pathophysiologic mechanisms underlying the toxicity of B paris venom and provide a basis for further research and the development of appropriate treatment strategies.
Footnotes
Author Contribution(s)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
