Abstract
Treatment of high altitude pulmonary edema (HAPE) can be challenging and is further complicated in the pediatric patient in the prehospital environment. The following case presents a decompensating pediatric patient with HAPE in the prehospital aeromedical environment. It illustrates the potential benefit of continuous positive airway pressure (CPAP) as a treatment modality in the treatment of HAPE.
Introduction
The patient was a 12-y-old female traveling with her parents from her home in Houston, Texas (approximately 80 ft/24 m) on a skiing vacation with her family. She had a history of a ventricular septal defect (VSD) repair at 18 mo of age with presurgical severe pulmonary hypertension (pHTN). After this procedure, the patient was documented as having no further pHTN and was cleared for all activity by her private physician. Her last echocardiogram was approximately 2 y prior to this incident and was reported to the sending clinic staff as “nonremarkable” by the patient's father. Previously, the patient had experienced altitude sickness requiring hospitalization but not intubation. Her father stated the patient's primary care physician had suggested acetazolamide for pretreatment; however, they did not obtain the prescription prior to this trip. Additionally, the patient was prescribed home supplemental oxygen to wear during periods of exertion and while sleeping. The family drove to the Rocky Mountains of northern New Mexico, where the patient spent one night at ∼6900 ft/2103 m and then continued to their destination at ∼8600 ft/2621 m. The patient spent one day skiing, at elevations ranging from 8750 ft/2667 m to 10 350 ft/3155 m and developed a dry cough overnight. The next morning, she developed worsening respiratory distress and was taken to a local clinic. The clinic advised that the patient's father had kept the patient on supplemental oxygen of an unknown amount throughout the entirety of the trip.
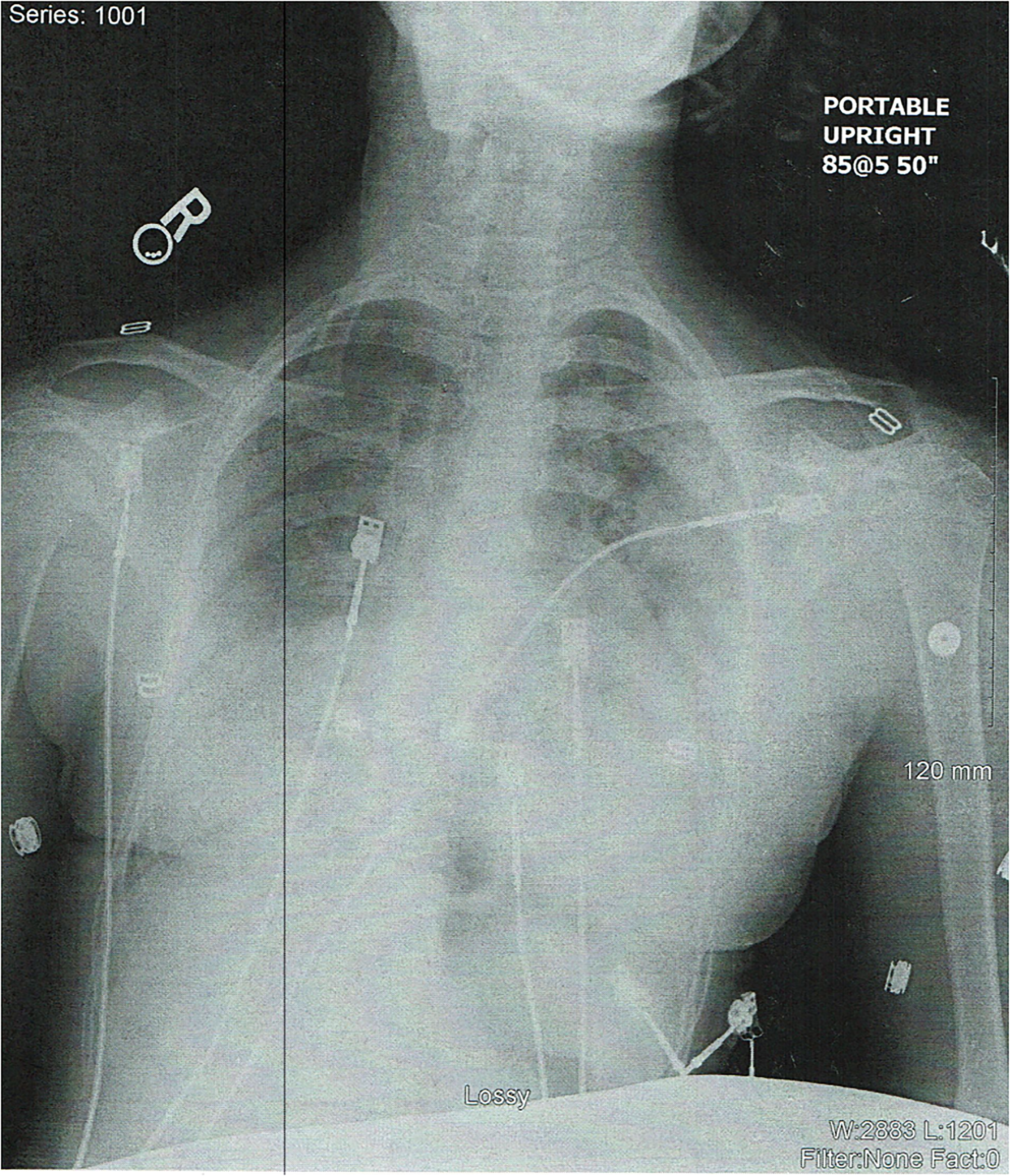
Receiving facility chest x-ray.
Case Report
Prehospital Course
An air medical crew was dispatched by helicopter for the 12-y-old female having difficulty breathing at a rural clinic in the southern Rocky Mountains of northern New Mexico. The patient presented to the clinic with peripheral cyanosis, an ECG showing sinus bradycardia at a rate of 36 beats per minute (BPM), a saturation of peripheral oxygen (SpO2) readings in the 40s, with tachypnea and altered mentation and a Glasgow Coma Scale (GCS) score of 12. The patient was placed on supplemental oxygen via nonrebreather mask at a rate of 15 liters per minute (lpm) approximately 1 hour before flight crew arrival, with little improvement of her symptoms. Her lung sounds were reported as “wet,” and a chest x-ray done at the clinic showed bilateral pulmonary edema, with no infiltrates. Despite the 15 lpm of oxygen being administered, the patient's SpO2 remained in the 40 s. Upon the arrival of the flight crew, it was determined that the patient might benefit from continuous positive airway pressure (CPAP). Initial settings were 100% fraction of inspired oxygen (FiO2) and positive end expiratory pressure (PEEP) of 10 centimeters of water (cmH2O) via a Carefusion LTV Series 1200 ventilator. Shortly thereafter, the patient experienced improving mentation and marked improvement in her oxygen saturation. Vital signs were blood pressure (BP): 114/77 millimeters of mercury (mm Hg); heart rate (HR): 154 BPM; respiratory rate (RR): 40; SpO2: 92% with no signs of cyanosis; and a temperature of 98.2 °F.
The patient remained tachypneic but was able to acknowledge subjectively feeling better with her GCS increasing to 14–15. The altitude of the landing zone was 8661 ft/2640 m. The nearest facility with a Pediatric Intensive Care Unit (PICU) and only hyperbaric chamber in the state was approximately 1 h and 20 min away. After initially going up to approximately 9000 ft/2743 m, the pilot was able to complete most of the flight over terrain at approximately 8000 ft/2438 m until closer to the destination where altitude was able to be maintained safely around 6000 ft/1829 m. Despite the improvement in the patient’s condition, the patient remained tachypneic on 100% FiO2. A good mask seal was maintained with minimal to no leak, and the patient tolerated the mask well.
Emergency Department and Hospital Course
The patient was transported to an emergency department (ED) in Albuquerque, New Mexico (5073 ft/1546 m) after a 1 h and 15 min transport. Her oxygen saturations had steadily increased during transport from 92 to 98% on CPAP. Upon arrival at the ED, the patient was alert and oriented with a GCS of 15. She continued to have a cough, shortness of breath, and tachypnea with no retractions. She remained tachycardic though her skin was pink, dry, and warm to the touch.
An attempt was made to remove the patient from CPAP by the ED staff and place the patient on a non-rebreather mask at 10 lpm, but her saturations dropped into the upper 80s. The patient was placed back on CPAP at a PEEP of 7 cmH2O. Labs and blood cultures were drawn, and ceftriaxone was administered. Chest X-Ray was repeated and initial labs revealed elevated B-type natriuretic peptide (BNP) and Troponin (Figure 1).
Pediatric cardiology was consulted, and an echocardiogram was ordered. The echocardiogram showed a pulmonary artery pressure (PAP) at ∼77 mm Hg with right ventricular thickening and dilation. The patient was admitted to the PICU, continuing with CPAP at 10 cmH2O and antibiotic therapy. The patient maintained adequate oxygenation on CPAP and did not require intubation or hyperbaric oxygen therapy. On day 2 of hospitalization, the patient was placed on a high-flow nasal cannula at 20 lpm. A repeat echocardiogram on Day 2 revealed a drop in the PAP to ∼60 mm Hg. The PAP continued to decrease to ∼40 mm Hg on Day 3. It was also noted that the BNP and Troponin levels dropped during this course. Oxygen weaning continued, at which time the patient was discharged home on Day 3 of hospitalization with oxygen at 1 lpm nasal cannula. The patient's primary discharge diagnosis was noted to be “High Altitude Pulmonary Edema.”
Discussion
HAPE is defined by noncardiogenic pulmonary edema. HAPE, like other high-altitude illnesses, is a result of the low-pressure (hypobaric) hypoxia encountered at high altitudes. Compounding the decreasing pressure of oxygen at altitude are factors such as acute hypoxic ventilatory response (AHVR), pulmonary hypertension (pHTN), patchy hypoxic pulmonary vasoconstriction (HPV), endothelial dysfunction, inflammation of the lung tissue, and capillary leakage.1,2 All of these factors play roles of varying degrees in HAPE cases.
Treatment of HAPE
High-flow oxygen is recommended as a first-line treatment for HAPE, when descent alone is unavailable or ineffective. Descent in altitude remains the primary goal of any treatment plan regarding HAPE. 3
Oxygen is the most common initial medical intervention for HAPE when descent is unavailable or unsuccessful. However, not all patients with HAPE are responsive to oxygen therapy alone. 4 Certain cases of HAPE require more than a simple increase in FiO2. CPAP therapy drives improvement in oxygen absorption by providing positive end expiratory pressure (PEEP) and establishing a better alveolar-capillary interface. CPAP recruits collapsed alveoli for functional use and more distally distributes inspired oxygen. For every 1 cmH2O increase in PEEP, the partial pressure of alveolar oxygen increases by approximately 1 percent. These all lend a benefit for CPAP use over just an adjustment in FiO2 alone. 5 Consequently, the use of PEEP in respiratory or ventilator cases has gained widespread usage, often delivered in stepwise increments alongside increasing percentages of FiO2. 6
The patient above improved dramatically on CPAP, in contrast to just the supplemental oxygen initially provided. Notably, she experienced a rapid desaturation and respiratory decline when she was removed from CPAP in favor of a simple oxygen mask at the receiving hospital. This reinforces the probability that PEEP can be essential to the recovery of these patients.
Interestingly, some medications have presented potential benefits in HAPE, including, but not limited to, nifedipine, tadalafil, and dexamethasone.3,7–11 Of note, none of the medications mentioned were available from either the clinic or the flight crew, and are not routinely caried by air medical crews in the region. Additionally, only dexamethasone and acetazolamide are currently recommended in pediatrics. 3
Inhaled nitric oxide (iNO) has also been used as a treatment for severe HAPE in previous studies. 12 iNO serves to promote vasodilation of the pulmonary vasculature, thereby reducing the pulmonary hypertension (pHTN) often seen in these cases.
All of the above treatments present legitimate options in a medically resource-rich environment. However, these patients are typically found in rural areas with limited medical resources.
Interestingly, the use of CPAP without supplemental oxygen has been shown to benefit certain HAPE patients. There have been several studies that have shown a clear improvement in symptoms of altitude illness, as well as oxygen saturation from the use of PEEP.13–15 Battery-operated CPAP machines are now available with a weight of as little as 2 lbs for some models—significantly less than a standard O2 tank.
Conclusion
Severe HAPE can be a life-threatening illness if not rapidly treated. CPAP shows potential promise in cases of HAPE where oxygen alone is insufficient to correct symptoms, particularly in the absence of medication therapy. Clinicians should carefully consider whether utilizing CPAP or other noninvasive positive airway pressure devices is appropriate when managing patients with refractory HAPE. In this case, prehospital providers were able to successfully utilize CPAP and improve a patient in extremis. Additionally, CPAP was continued when the patient was transferred to the ED and PICU, and intubation was avoided.
Footnotes
Author Contribution(s)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
