Abstract
Introduction
The purpose of this study was to assess and compare the effectiveness of seven different closed system transfer device (CSTD) product lines following the 2015 NIOSH Vapor Containment Performance Protocol, using a Gasmet™ DX5000 Terra multigas FTIR analyzer.
Methods
Seven different CSTD systems were assessed using a two-task evaluation on their capacity to contain the NIOSH-specified challenge agent, 70% isopropyl alcohol (IPA). Task 1 simulated reconstitution and compounding steps while Task 2 simulated compounding and administration steps. Vial adapters, syringe adapters, IV bag adapters, and y-site adapters from each CSTD product line were tested. All tasks were performed in a custom-built environmental test chamber as outlined in the 2015 NIOSH protocol, with isopropyl alcohol (IPA) release detected and measured by a Gasmet™ analyzer.
Results
The BD PhaSeal™ CSTD product line effectively contained IPA vapor during both tasks with a task performance metric of 0.00 ppm for each task. BD PhaSeal™ Optima, Equashield®, Yukon Medical Arisure®, ICU Medical ChemoLock™, BD Texium™, and ICU Medical ChemoClave™ non-vented CSTDs demonstrated detectable and quantifiable IPA leakage during each task. The leaks occurred at different steps within the protocol, and the amount of IPA vapor detected varied.
Conclusion
In this study, one CSTD product line (i.e., PhaSeal™) successfully contained IPA vapor below the Gasmet™ analyzer's limit of quantification (LOQ) for IPA of 0.04 ppm for both Tasks 1 and 2, demonstrating it to be a validated closed system per the study protocol. All other CSTD product lines demonstrated some level of IPA leakage with task performance metrics above the analyzer's LOQ. However, these results tell us little about how well CSTDs contain hazardous drugs (HDs), as a universal HD surrogate for effectively assessing the closedness of all types of CSTDs has yet to be identified.
Keywords
All CSTD product lines tested, except one, demonstrated quantifiable IPA leakage during testing. These results can inform CSTD manufacturers and end users of potential CSTD design issues, variations in manufacturing across different lots of the CSTDs, or compounding technique issues that need to be addressed. A universal HD surrogate and testing methodology are needed for assessing the effectiveness of barrier and air-cleaning types of CSTDs to contain hazardous drug vapors, aerosols, and liquids.
Introduction
Closed system transfer devices (CSTDs) have been on the market since 1998 to protect healthcare workers from exposure to hazardous drugs (HDs).1,2 The National Institute for Occupational Safety and Health (NIOSH) has defined a CSTD as a drug transfer device that “mechanically prohibits the transfer of environmental contaminants into the system and the escape of the hazardous drug or vapor concentrations outside the system”. 3 These devices protect users throughout the medication use process by establishing secure connections when syringes are linked to drug vials, intravenous (IV) bags, or IV administration sets. These devices, depending on design, minimize or eliminate the chance of HD vapors, aerosols, and liquids escaping, thus, minimizing or eliminating the exposure risk to healthcare workers.
CSTDs on the market use a variety of mechanisms to prevent exposure to HDs, with the two primary design types being physical barrier CSTDs (i.e., membrane-based) and air-cleaning CSTDs (i.e., filter-based).3–5 Within the two types of CSTDs, there are also different engineering designs and/or filter types employed by CSTD manufacturers, potentially affecting the overall performance of their products.2,5 Barrier type CSTDs on the market include Arisure® (Yukon Medical), ChemoClave™ Vial Spike with External Ballon (ICU Medical), ChemoLock™ Vial Spike with External Ballon (ICU Medical), Equashield®, Halo® (Fresenius Kabi), PhaSeal™ (BD), PhaSeal™ Optima (BD), and Texium™ & SmartSite™ VialShield (BD). 6 Air-cleaning type CSTDs on the market include ChemoClave™ Vented Vial Spike (ICU Medical), ChemoLock™ Vented Vial Spike (ICU Medical), OnGuard® Tevadaptor® (B. Braun), OnGuard® 2/Chemfort® ToxiGuard® (B. Braun/Simplivia), and Texium™ & SmartSite™ Vented Vial Access Device. 6
Since 2004, NIOSH has recommended that healthcare workers use CSTDs during the entire medication use process – from compounding to administration – alongside other engineering controls to enhance protection against HD exposures. 7 The United States Pharmacopeia (USP), through USP general chapter <800>, also recommends the use of CSTDs during compounding but mandates the use of CSTDs during administration. 8 Although not all CSTDs offer equal protection or completely prevent HD exposure, they do help to reduce HD contamination. 9 As the designs and available models of CSTDs evolved, NIOSH and industry partners recognized the need for an independent method to test CSTD performance. 3 This recognized need was further heightened by the absence of standards for assessing HD containment, despite existing standards focused on sterile compounding and patient protection. 10
To fill this gap, NIOSH introduced a draft Vapor Containment Performance Protocol in 2015, specifically designed to assess CSTDs’ effectiveness in protecting against occupational exposure. 3 This protocol aimed to provide “a methodology for evaluating challenge agent containment performance of CSTDs” while undergoing manipulations consistent with pharmaceutical compounding and administration. 3 For this protocol, NIOSH selected 70% isopropyl alcohol (IPA) as the challenge agent due to its favorable chemical and usability characteristics. 3 Specifically, IPA is liquid at room temperature, is relatively safe and commonly available, and has a high vapor pressure, thereby allowing for efficient vapor generation and detection via numerous analytical methods. 3 NIOSH also recommended use of a MIRAN® SapphIRe XL infrared analyzer to detect and measure the amount of IPA released during the testing manipulations due to its moderately low limit of detection and regular use in the industry.
The 2015 NIOSH Vapor Containment Performance Protocol was developed to evaluate the extent to which CSTDs contain vapors, aerosols, and liquids. However, due to the analytical methods used and the high vapor pressure of the testing agent (IPA), the protocol cannot distinguish between different types of leakage nor be used to effectively assess air-cleaning CSTDs. These limitations led industry experts, researchers, and manufacturers to advocate for an improved protocol with a different challenge agent that can be used to test all types of CSTDs and better reflect the properties of HDs.11,12
Based on this feedback, NIOSH generated a list of potential HD surrogates to test and considered making some key changes to their 2015 protocol 12 ; however, no official updates to the draft 2015 protocol have been published, and this protocol continues to be used by CSTD manufacturers in 510k premarket submissions to the U.S. Food and Drug Administration (FDA). 2
In addition to NIOSH's efforts in 2015, various groups have attempted to develop strategies for measuring CSTD “closedness”. These groups have explored different testing strategies, a variety of challenge agents such as titanium tetrachloride (TiCl4), fluorescein, and radioactive technetium (99mTc), and different HD surrogates such as 2-phenoxyethanol (2-POE), propylene glycol methyl ether (PGME), 1,1,3,3-tetraethylurea (TEU), and methyl tert-butyl ether (MTBE).13–19 However, none of the testing strategies or surrogates explored have adequately assessed HD containment performance across all CSTD types or successfully differentiated and quantified vapor, aerosol, and liquid leakage.
Findings from previous studies
Previous studies utilizing the 2015 NIOSH Vapor Containment Performance Protocol with a MIRAN® SapphIRe XL infrared analyzer have demonstrated varying effectiveness of CSTDs to adequately contain IPA vapor below the predefined testing criteria.19–21 Some product lines, such as PhaSeal™, Halo®, and Equashield® – all physical barrier type CSTDs – have consistently contained IPA vapor below the testing criteria, defined as a mean maximum background-adjusted, zero-corrected (BG-0max) vapor concentration measurement (in parts per million [ppm]) less than the limit of quantification (LOQ) for IPA on the MIRAN® analyzer (LOQ = 1.0 ppm).20–22 Other CSTD product types, however, have not been able to consistently contain IPA vapor below this threshold using the NIOSH testing protocol.20–22 The product lines that successfully contain IPA vapor throughout both Task 1 and Task 2 of the 2015 NIOSH protocol are considered to be validated closed systems per the protocol. However, it is important to note that the selection of the MIRAN® analyzer's LOQ of 1.0 ppm for IPA as the cutoff for determining how closed a CSTD is (or isn’t) has not been shown to correlate to a CSTD's ability to effectively contain HD vapors, aerosols, and liquids.
Purpose
The purpose of this study was to assess and compare the effectiveness of seven different CSTD product lines – Arisure®, ChemoClave™ Vial Spike with External Ballon, ChemoLock™ Vial Spike with External Ballon, Equashield®, PhaSeal™, PhaSeal™ Optima, and Texium™ & SmartSite™ VialShield – in containing IPA during pharmaceutical compounding and administration processes following the 2015 NIOSH protocol, but using an analyzer with a lower LOQ than was prescribed by the 2015 protocol.
Methods
Methodology
Seven different CSTD product lines, limited to the products that could be procured at the time of the study, were tested from June through December 2023 to determine their relative effectiveness in containing the challenge agent, 70% IPA, while following the 2015 NIOSH protocol. Task 1 of the protocol includes a series of vial to syringe and syringe to IV bag transfers and Task 2 includes a series of vial to syringe, syringe to IV administration line, and IV line to IV bag transfers.
IPA vapor released or generated during each task was detected and measured in real time by a Gasmet™ DX5000 Terra multigas FTIR analyzer (Gasmet Technologies; Oy, Finland). Although the 2015 NIOSH protocol calls for the use of a MIRAN® SapphIRe XL infrared analyzer (Thermo Scientific; Franklin, MA), the Gasmet™ analyzer was used in its place, since production of the MIRAN® analyzer has been discontinued and is of limited availability. Of note, the Gasmet™ analyzer, with an IPA limit of detection (LOD) of 0.012 ppm (LOQ = 0.04 ppm), has a lower LOQ than the MIRAN® analyzer (i.e., LOD = 0.3 ppm; LOQ = 1.0 ppm). High purity nitrogen gas was used for purging and zeroing the analyzer after each test per the analyzer's instructions for use (IFU).
The tasks were performed within a custom-built environmental test chamber, built per the specifications outlined in the 2015 NIOSH protocol. 3 A few modifications were made to the testing chamber to facilitate its use with the Gasmet™ analyzer. Namely, the particulate prefilter respirator cartridges as described in the NIOSH protocol were removed and the test chamber was modified to function with the Gasmet™ analyzer as a closed-loop system, given the analyzer specifications (Figure 1). The modified testing chamber and Gasmet™ analyzer were supplied by the study sponsor.

Modified NIOSH custom-built environmental test chamber connected to a gasmet™ DX5000 terra multigas FTIR analyzer.
Prior to testing, vials containing 50 mL 70% IPA were prepared for the completion of four tests per task per product line. Preparation was done in a clean fume hood at room temperature. Each 50 mL sample was placed in a 100 mL glass vial and sealed with a 20 mm septum cap and aluminum crimp seal with a crimping tool. The outside of each vial was wiped down with a nonalcoholic cleaner to ensure no residual IPA was on the outside of the vial before use. All other materials for the tasks (e.g., CSTDs, IV bags, IV lines, etc.) were kept at room temperature, in sealed packages, prior to the testing period. The packaged materials needed for each task were opened immediately before placing them in the testing chamber.
Test procedure
The testing was conducted in a laboratory setting at room temperature. The stepwise instructions provided by the 2015 NIOSH protocol for each task were followed throughout the testing process with CSTDs used in accordance with the manufacturers' IFU. The fundamental compounding and administration actions included in the 2015 NIOSH protocol – representing standard compounding or administration processes, such as reconstitution of a drug vial (e.g., Action Step #3, Task 1) or injection of a syringe dose into an IV bag (Action Step #5, Task 1) – are described in Table 1 and Figure 2. 3
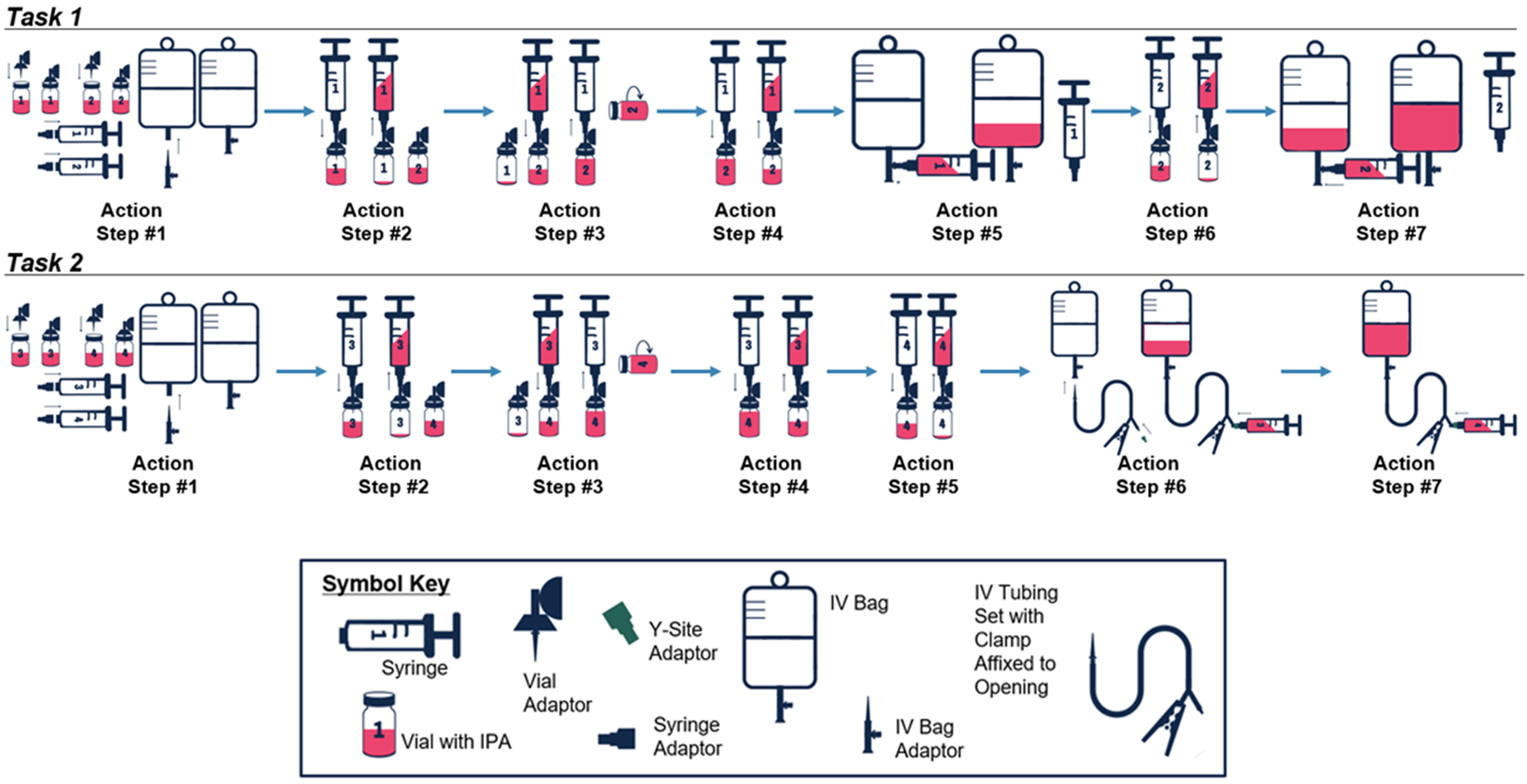
Graphical depiction of compounding and administration actions included in task 1 and task 2.
Compounding and administration actions included in task 1 and task 2 3 .
Task 1 simulated reconstitution and compounding steps that consisted of transferring 45 mL of IPA from one vial to another vial, followed by two transfers of 45 mL of IPA from the second vial to a 500 mL 0.9% sodium chloride IV bag using two syringes. Task 2 simulated compounding and administration steps that consisted of transferring 45 mL of IPA from one vial to another vial using a syringe, followed by two 45 mL transfers of IPA from a second vial to an IV administration line connected to a 500 mL 0.9% sodium chloride IV bag.
Both tasks were completed using 50 mL Luer-Lok™ syringes for all product lines except for Equashield® and ChemoLock™. These two product lines use system-specific 60 mL syringes, where the CSTD syringe adapter is directly bonded to the syringe.
Each task was run four times per CSTD product line, as prescribed by the NIOSH testing protocol. 3
Data collection and analysis
The amount of IPA vapor detected by the Gasmet™ analyzer was recorded in real-time, after each key step, following three minutes of continuous sampling. This methodology was a deviation from the 2015 NIOSH protocol which states to “pause for 30 seconds or until the IPA detector stabilizes” prior to recording IPA vapor concentration readings. 3 This protocol deviation was based on the differences in analyzers used in the 2015 NIOSH protocol and this study and on the analyzers’ IFU.
All vapor measurement data points collected were background (BG)-corrected prior to analysis. The maximum value observed for each task was averaged to give the mean maximum BG-0max concentration values (in ppm) for each CSTD product line. The 95% confidence interval (CI) was also calculated for each mean BG-0max, with the upper 95% CI of the mean BG-0max serving as the overall task performance metric. Calculations were performed using Microsoft® Excel® software (Microsoft 365 MSO Version 2401 Build 16.0.17231.20236).
Effective containment of IPA was defined as an average vapor concentration (i.e., an upper 95% CI value) less than the analyzer's IPA LOQ (0.04 ppm). Any IPA concentration measurements greater than the Gasmet™ analyzer's LOQ indicated ineffective containment of IPA by the CSTD. Tests were immediately suspended upon detection of vapor concentrations exceeding 5.0 ppm.
BD PhaSeal™, a CSTD product line currently FDA-cleared and sold in the United States, 2 was used as the study control. This product line has previously been shown to effectively prevent IPA vapor leakage using the defined task performance metric in the 2015 NIOSH protocol (i.e., an upper 95% CI value less than a MIRAN® analyzer's LOQ of 1.0 ppm).20–22
Results
Task 1
PhaSeal™ effectively contained IPA vapor during Task 1 with a task performance metric of 0.00 ppm. All other CSTD product lines demonstrated varying amounts of IPA leakage during Task 1 with task performance metrics above the analyzer's LOQ of 0.04 ppm.
The Texium™ and ChemoClave™ CSTDs demonstrated detectable and quantifiable IPA leakage during Task 1, with an average IPA vapor concentration greater than 5.0 ppm before the completion of Task 1, resulting in early termination of the task. All other CSTDs had task performance metrics between 0.17 and 3.49 ppm upon full completion of the task. Full results from Task 1 are provided in Table 2.
Summary of task 1 and task 2 results.
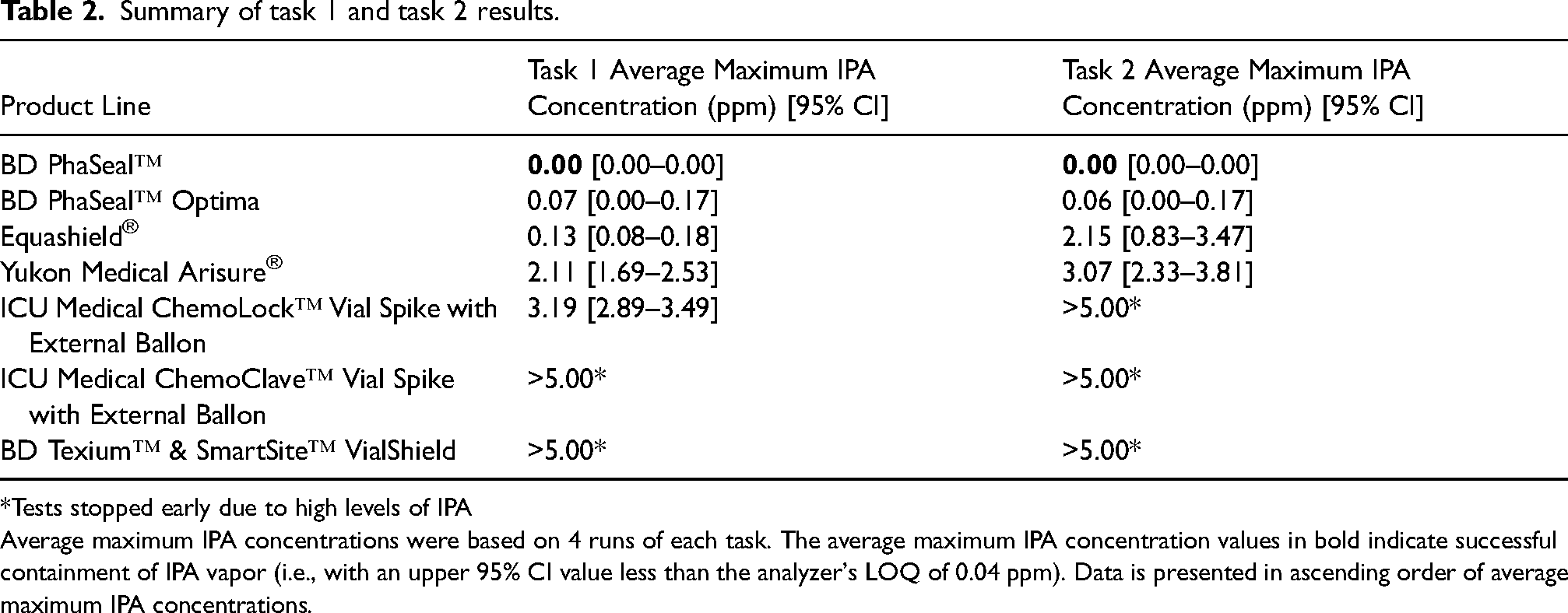
*Tests stopped early due to high levels of IPA
Average maximum IPA concentrations were based on 4 runs of each task. The average maximum IPA concentration values in bold indicate successful containment of IPA vapor (i.e., with an upper 95% CI value less than the analyzer's LOQ of 0.04 ppm). Data is presented in ascending order of average maximum IPA concentrations.
As illustrated in Figure 3, the ChemoClave™, Texium™, ChemoLock™, and Arisure® CSTDs initially exhibited IPA leakage in Task 1 during use of the vial and syringe CSTDs. The amount of IPA vapor detected increased progressively throughout the task. The PhaSeal™ Optima and Equashield® CSTDs did not show any IPA leakage during Task 1 until the IPA was transferred from a syringe to the IV bag.
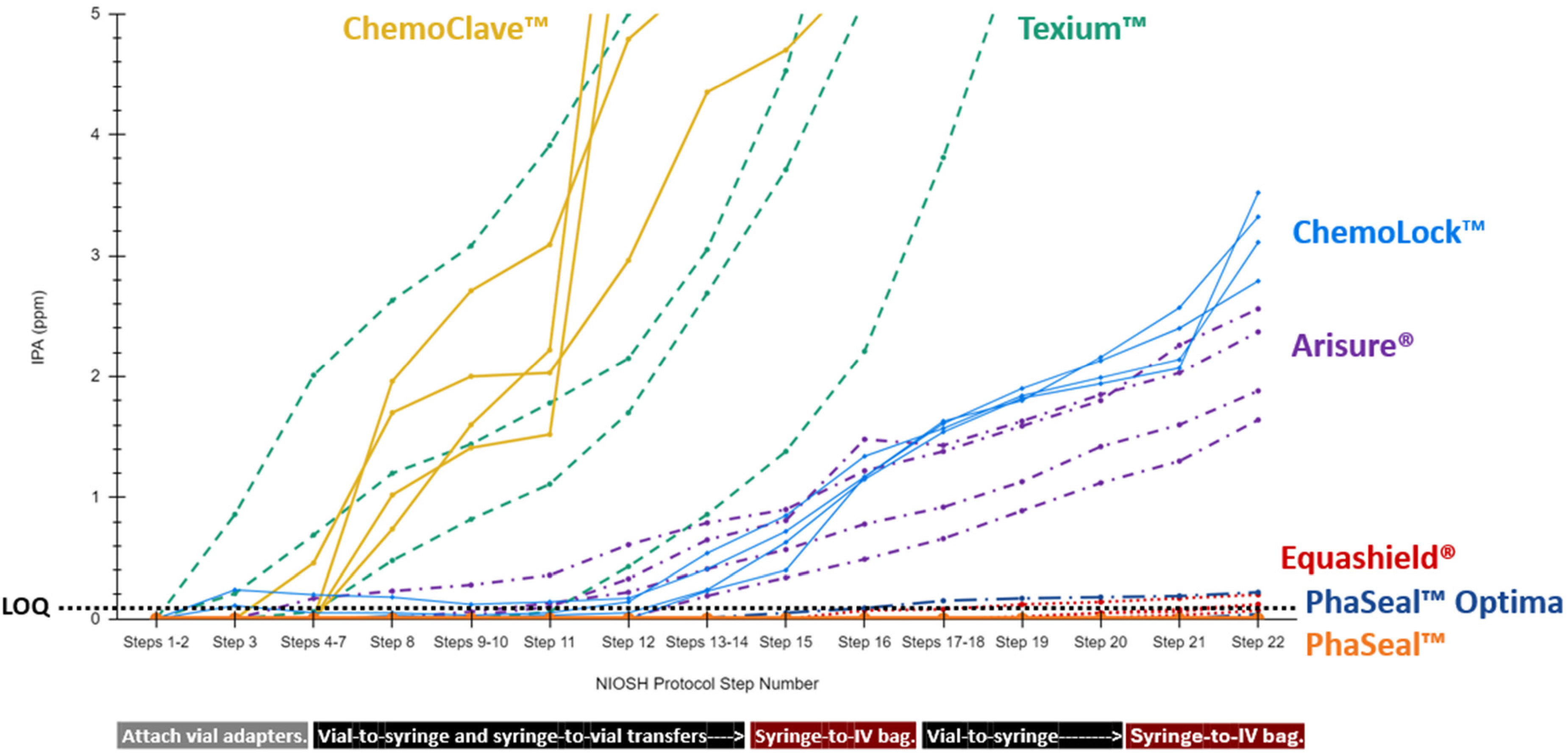
Comparison of task 1 results.
Task 2
During Task 2, PhaSeal™ also effectively contained IPA vapor with a task performance metric of 0.00 ppm. All other CSTD product lines demonstrated varying amounts of IPA leakage during Task 2 with task performance metrics above the analyzer's LOQ.
The ChemoLock™, Texium™, and ChemoClave™ CSTDs demonstrated detectable and quantifiable IPA leakage during Task 2, with an average IPA vapor concentration greater than 5.0 ppm before the completion of Task 2, resulting in early termination of the task. All other CSTDs had task performance metrics between 0.17 and 3.81 ppm upon full completion of the task. Full results from Task 2 are provided in Table 2.
As shown in Figure 4 and similarly to Task 1, the ChemoClave™, Texium™, ChemoLock™, and Arisure® CSTDs first displayed IPA leakage in Task 2 during manipulation of the vial and syringe CSTDs. The PhaSeal™ Optima CSTDs did not display IPA leakage until IPA was transferred from Vial 4 to a syringe a second time, and the Equashield® CSTDs did not display IPA leakage until IPA was transferred from the syringes to the IV administration line via the line's y-site.
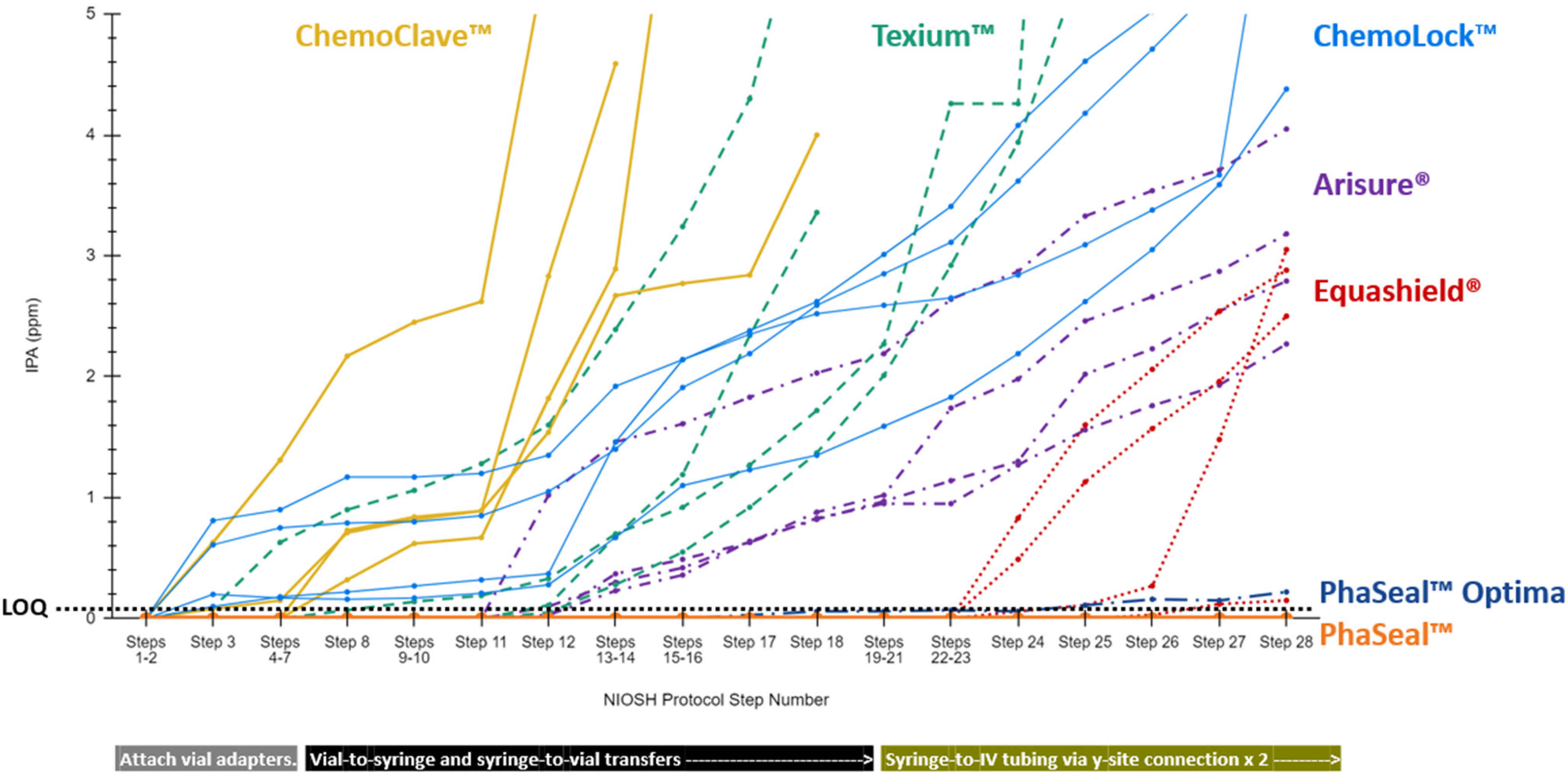
Comparison of task 2 results.
Discussion
In this study, the PhaSeal™ CSTD product line was able to effectively contain IPA vapor below the Gasmet™ DX5000 Terra multigas FTIR analyzer's LOQ of 0.04 ppm during both Tasks 1 and 2 of the 2015 NIOSH Vapor Containment Performance Protocol.
PhaSeal™ Optima demonstrated detectable and quantifiable IPA leakage during both Tasks 1 and 2, with task performance metrics above the analyzer's LOQ for IPA but not exceeding 0.17 ppm. Equashield® also showed detectable and quantifiable IPA leakage in Task 1, with a performance metric not exceeding 0.18 ppm. All other CSTDs evaluated showed detectable and quantifiable IPA leakage in both tasks but displayed differences when IPA vapor concentration increases were observed (i.e., at different steps of the compounding and administration processes tested) and the amount of IPA vapor detected.
In examining the differences between Task 1 and Task 2, all CSTD product lines tested in this study demonstrated relatively consistent results between the two tasks, except for Equashield®. The Equashield® CSTDs exhibited significantly more IPA leakage during Task 2 compared to Task 1. This discrepancy suggests a potential issue with the specific Equashield® y-site adapter tested or the connection of the Equashield® bag adapter to the IV administration line, both of which are involved when the excursion above the LOQ is observed (Figure 4). Further testing is required to determine whether the observed leakage was related to specific design features of the CSTDs, variations in manufacturing across different lots of the CSTDs, or the technique employed by the operators, especially given that leakage at either of these sites could result in occupational HD exposure. 7 Given the consistent performance of Equashield® CSTDs in containing IPA vapor below predetermined limits in prior studies,20–22 additional investigation is critical for fully understanding these observed results.
The four remaining CSTDs included in this study (Texium™, ChemoLock™, ChemoClave™, and Arisure®) displayed IPA leakage during both tasks. However, it is important to note that IPA vapor measurements exceeding this analyzer's LOQ do not necessarily indicate that a CSTD is unable to contain HD vapors in clinical practice. IPA, due to its higher volatility, smaller molecular size, and distinct chemical properties compared to HDs, is not an ideal surrogate for evaluating CSTD effectiveness. 5 Furthermore, no existing data correlates the extent of exceeding the Gasmet™ analyzer's IPA vapor LOQ with actual HD occupational exposure. Such excursions merely indicate that a given CSTD cannot be validated as completely closed under the protocol as used, since detectable and quantifiable amounts of IPA were observed during testing.
Previous studies using the 2015 NIOSH protocol with a MIRAN® SapphIRe XL infrared analyzer have consistently demonstrated that PhaSeal™ and Equashield® CSTDs successfully contain IPA vapor below the predefined testing criteria of IPA vapor concentrations less than the MIRAN®'s LOQ of 1.0 ppm.20–22 Similar to the findings of this study, the inability of some CSTDs to limit IPA vapor to levels below the LOQ of the MIRAN® analyzer during testing is not necessarily indicative of their effectiveness at containing HD vapors in clinical practice.
Additionally, it remains unclear how results using the Gasmet™ DX5000 Terra analyzer compare with those obtained with the MIRAN® SapphIRe XL in measuring IPA vapor concentrations. The impact of the closed-system configuration of the Gasmet™-chamber setup versus the open-system setup of the MIRAN®-chamber setup on the results is also not well understood. Without established performance standards for CSTDs under this study's testing conditions (i.e., measuring IPA vapor leakage with a Gasmet™ analyzer) or fully understanding the implications of the differences in sensitivity and specificity between the Gasmet™ and MIRAN® analyzers, these results, along with previous findings, do not support the establishment of a definitive “pass/fail” demarcation for CSTD vapor containment performance for HDs, especially considering the limitations of IPA as a surrogate for HDs.
While these results offer limited insights into the effectiveness of CSTDs to adequately protect users from HD exposure, CSTDs that demonstrate the least amount of IPA vapor leakage should be preferred to minimize the risk of HD exposure until a better CSTD testing protocol is developed. These results can also assist companies and end users in identifying and addressing potential vulnerabilities in the CSTD products they manufacture and/or use by enabling the association of specific CSTD product vulnerabilities with specific compounding or administration actions, as demonstrated in Figures 3 and 4.
The key limitation of this study was that testing was performed by two different individuals, which might allow for bias due to variations in technique. However, minor variations in technique between device users in the marketplace are expected, and since these devices are designed for such variations to be tolerated, this factor is not a concern to the investigators. As demonstrated in Figures 3 and 4, relatively consistent results were observed for each product line, regardless of user.
To better evaluate the effectiveness of CSTDs in containing HDs, additional testing is necessary using a surrogate that more closely represents HDs. It is also important to develop a testing methodology that can specifically differentiate between vapor, liquid, and aerosol leakage and can be used to effectively test physical barrier and air-cleaning types of CSTDs. While some progress has been made in identifying an effective universal surrogate for CSTD testing,4,13–22 an ideal surrogate (or combination of surrogates) has not yet been discovered. Further research is also needed to compare results obtained using the Gasmet™ and MIRAN® analyzers, to allow for comparison of results from studies using different analyzers, ensuring consistency and reliability across research findings.
Once an effective universal surrogate for testing CSTDs has been identified and a compatible testing methodology developed, the correlation between the amount of HD surrogate leakage and actual HD exposure needs to be delineated, as does the amount of HD exposure that would lead to deleterious effects in healthcare workers who use CSTDs in the compounding and administration of HDs.
Conclusion
In this study, one CSTD product line (i.e., BD PhaSeal™) successfully contained IPA vapor below the Gasmet™ analyzer's LOQ for IPA of 0.04 ppm for both Tasks 1 and 2, demonstrating it to be a validated closed system per the study protocol. All other CSTD product lines demonstrated some extent of IPA leakage with task performance metrics above the analyzer's LOQ.
While these results provide limited insight into the ability of CSTDs to adequately contain HDs during compounding and administration processes, those devices showing lower amounts of IPA vapor leakage should be preferred to minimize HD exposure, thereby enhancing the safety of patients and staff. These results also identify the specific steps or manipulations in compounding and administration processes where IPA vapor was detected, informing manufacturers and end users of potential CSTD design issues, variations in manufacturing processes across different lots of the CSTDs, or compounding technique issues that need to be addressed.
Despite the advances in CSTD development and testing, there is still a need to find a universal HD surrogate (or combination of surrogates) and testing methodology for assessing the effectiveness of physical barrier and air-cleaning types of CSTDs in containing HDs.
Footnotes
Author contributions
SFE, LTA, and MS: conceived the study and were involved in protocol development. LTA, MS, and MAE: collected the data; LTA, MS: researched literature, performed data analysis, and interpreted the results. LTA, MS: wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Acknowledgements
The authors would like to thank Lauren Hairston, PharmD Candidate at the UNC Eshelman School of Pharmacy for her assistance in conducting the experiments,
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
Declaration of conflicting interests
The authors declared following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This study was sponsored by Becton, Dickinson and Company (BD). The sponsor provided the testing chamber, Gasmet™ DX5000 Terra multigas FTIR analyzer, BD-specific CSTD products, and BD brand 50mL syringes used in this study. The Gasmet™ analyzer was calibrated by the study sponsor prior to the start of the study. The sponsor did not participate in the conduct or reporting of this study. Author S. F. E. entered into a consulting agreement with the study sponsor in January 2025 after the study was completed and the article had been accepted for publication. This conflict was disclosed by the author during the process of updating the end statements after publication. The authors have no other relevant conflicts of interest with respect to financial relationships in the research, authorship, and/or publication of this article to report.
Funding
This study was sponsored by BD (grant number CyberGrant #/Project: 76223579). The sponsor did not participate in the conduct or reporting of this study.
