Abstract
Real-world utilization of 4-factor prothrombin complex concentrate (4F-PCC) and plasma for the management of oral anticoagulant (OAC)-associated bleeding in US trauma hospitals was described.
This is amulticenter, retrospective chart review evaluating the use of 4F-PCC and plasma in OAC reversal across medical specialties. Physicians completed a survey and extracted data from 3 to 5 patient charts. Variables of interest included medical specialty, urgency, and bleed type. Two hundred and thirty-five physicians completed the survey, and 861 patient charts were included in the study. 4F-PCC was commonly used in life-threatening or emergent indications, whereas plasma was used in emergent and urgent indications. Plasma was used mostly for patients on warfarin (53% vs 47% 4F-PCC) and 4F-PCC for those on apixaban (82% vs 18% plasma) and rivaroxaban (77% vs 23% plasma). This retrospective analysis showed that 4F-PCC was predominantly used for OAC reversal despite available specific reversal agents for some of the OAC. Although it is not recommended by any reversal guidelines, plasma is still used for OAC reversal. Plasma should be avoided in the management of OAC-associated bleeding.
Keywords
Introduction
Oral anticoagulants (OACs), including vitamin K antagonists (VKAs) and direct OACs (DOACs) such as activated factor IIa (FIIa) inhibitors and factor Xa (FXa) inhibitors, are standard preventive treatment for patients at elevated risk of arterial or venous thrombosis. 1 However, patients receiving OACs may require rapid anticoagulation reversal in cases of acute major bleeding or before urgent or emergent surgery.2–4 Management of bleeding in these patients depends heavily on the type and severity of bleeding, urgency, and anticoagulant medication.5–7 Several hemostatic and reversal agents are available, including plasma and prothrombin complex concentrates (PCCs). Plasma has historically been used, particularly for the reversal of VKAs, to restore vitamin K–dependent coagulation factors (VKDF) (II, VII, IX, and X). Plasma is slow at restoring VKDF and correcting the international normalized ratio (INR) in addition to several safety concerns regarding its use, including the risk of viral transmission, allergic reactions, transfusion-related acute lung injury (TRALI), and transmission-associated circulatory overload (TACO) due to the large volume necessary to raise factor levels.7–11 PCCs are purified concentrates of VKDF which can be administered in a smaller volume than plasma. 12 Clinical studies show PCC effectively restores VKDF, rapidly reverses VKA anticoagulation, and reduces subsequent risk of TRALI and TACO when compared to plasma.13–15
While the current standard of care for VKA reversal is 4-factor PCC (4F-PCC),7,16–19 specific reversal agents are recommended for the reversal of DOACs, including idarucizumab for FIIa inhibitors (dabigatran) and andexanet alfa for the FXa inhibitors apixaban and rivaroxaban.6,17,19–22 In emergency indications, the type of DOAC a patient has taken is not always known prior to administration of specific reversal agents.3,21 Off-label use of PCCs, as non-specific agents, is also recognized as a treatment option for the management of DOAC-associated bleeding, if specific reversal agents are not available.6,17,19–22 Plasma should have no role in DOAC reversal nor is recommended by international guidelines for the management of DOAC-associated bleeding, 21 including intracranial hemorrhage (ICH),23,24 gastrointestinal (GI) bleeding, 25 and trauma. 26 In this study, we aim to describe the real-world utilization data of 4F-PCC and plasma for the management of OAC-associated bleeding in US trauma hospitals based on specialty, type of bleed, and urgency level.
Materials and Methods
This study was a multicenter retrospective observational chart review which evaluated the use of 4F-PCC and plasma for the management of OAC-associated bleeding across medical specialties. Physicians from 11 different specialties were asked to complete a survey (Supplementary materials) and extract data from 3 to 5 patient charts. Participating physicians were selected based on meeting specific criteria such as a board-certified physician, a minimum number of 2 years in practice, majority of hospital working hours in direct patient care, trauma center affiliation, treatment of at least 3 patients who required OAC reversal with 4F-PCC or plasma in the past 6 months, and had no affiliation with pharmaceutical industries. Physicians received monetary compensation for their time. When completing the survey, physicians had the option to pick more than 1 option or most influencing reasons for their choices for some of the questions asked (see survey in Supplementary materials).
For the electronic data extraction, physicians were asked to randomly select 3 to 5 charts from a collection prioritized based upon the inclusion of adult patients (>18 years) from the last 6 to 12 months, who required OAC reversal with either life-threatening, emergent, or urgent indications and were treated with plasma or 4F-PCC. The variables of interest included medical specialty (ie, specialties most commonly using 4F-PCC or plasma), urgency (defined as life-threatening; emergent, where immediate attention was required but there was no threat to life; urgent, where quick, but no immediate action was required; or elective/non-urgent indication), and bleed type/patient condition (ie, most common type of bleed/patient condition treated with 4F-PCC or plasma).
Descriptive data were reported in frequency and percentages for categorical variables and mean, standard deviation (SD), median, and range for continuous variables.
Results
From April 2021 to July 2021, a total of 235 physicians extracted data from 861 patient charts and completed the survey. Physician characteristics are presented in Table 1.
Characteristics of Physicians Interviewed.
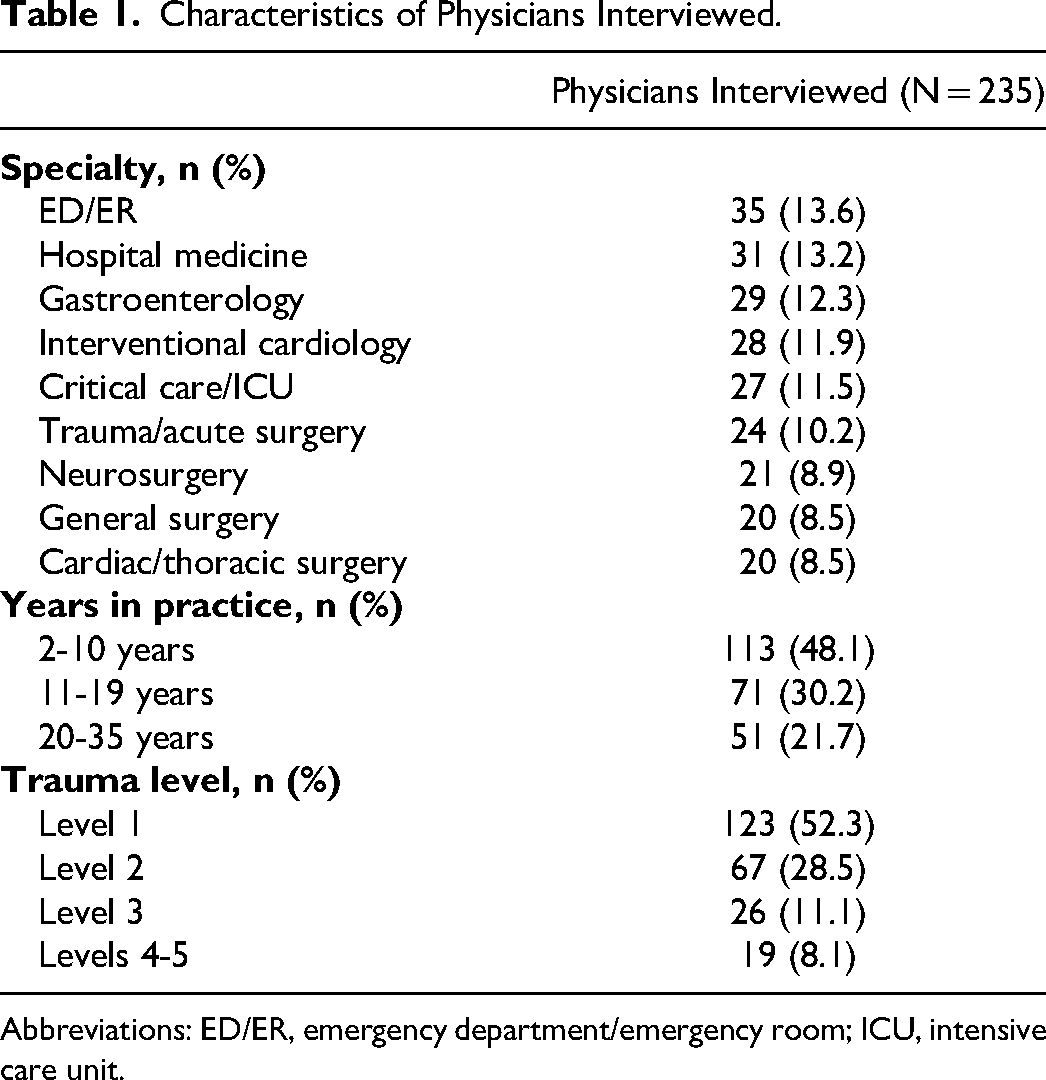
Abbreviations: ED/ER, emergency department/emergency room; ICU, intensive care unit.
Retrospective Patient Chart Review
A total of 861 patients who received 4F-PCC (n = 500) or plasma (n = 361) for major bleeding or need for urgent surgery were included in this study. Patient demographics and clinical setting characteristics are presented in Table 2.
Patient and Clinical Setting Characteristics.

aAge not available for 55 charts; gender not available for 18 charts; weight not available for 180 charts; INR data not available for 249 charts.
bIncluding warfarin (n = 413), apixaban (n = 153), rivaroxaban (n = 119), dabigatran (n = 44), none (n = 101), other (n = 7), and not available/not applicable (n = 24).
Abbreviations: 4F-PCC, 4-factor prothrombin complex concentrate; aPTT, activated partial thromboplastin time; ED/ER, emergency department/emergency room; ICH, intracranial hemorrhage; ICU, intensive care unit; INR, international normalized ratio; NA, not available/not applicable/not used; SD, standard deviation.
Characteristics of patients and clinical setting
The majority of patients who required bleeding management related to OAC use presented with ICH, GI-related bleeds, or blunt trauma or were in need of urgent surgery or invasive procedure (Table 2). Emergency department/emergency room (ED/ER), critical care/intensive care unit (ICU), and cardiac/thoracic surgery encountered more patients with a life-threatening indication. However, all medical specialties encountered all types of urgency levels when using 4F-PCC or plasma to treat OAC-associated bleeding (Figure 1). Nearly half the patients (n = 403/861, 46.8%) were not taking any other relevant medication. Other relevant medications patients were taking that had an influence on the OAC reversal agent selected included aspirin (n = 291/861, 33.8%), other anti-platelet (n = 89/861, 10.3%), vitamin K supplement (n = 52/861, 6.0%), and heparin (n = 42/861, 4.9%).

Urgency level in patients requiring treatment of oral anticoagulant (OAC)-associated bleeding across specialties.
Management of bleeding
A summary of urgency level, OAC medications, and patient conditions for 4F-PCC and plasma is outlined in Table 3. 4F-PCC was more commonly used when a patient's indication was life-threatening or emergent, whereas plasma was typically used in emergent or urgent indications (Table 3). Overall, 4F-PCC was mostly used for patients who were on warfarin, followed by apixaban and rivaroxaban, while plasma was primarily used for patients on warfarin (60.7%), although it was also used for those on DOACs (Table 3). A numerically higher proportion of patients received 4F-PCC compared to plasma across most specialties (Figure 2). When considering patient conditions, 4F-PCC was used more frequently for ICH and blunt trauma compared to plasma (Table 3). Additionally, there was similar usage of 4F-PCC and plasma for treating GI bleeds, but for specific types of GI bleeds, such as melena, plasma was used more often than 4F-PCC (Table 3).

Incidence of 4F-PCC and plasma use among specialties.
Incidence of Urgency Level, Oral Anticoagulant Medication, and Patient Condition by Treatment.

*Some patients may have received both 4F-PCC and plasma.
Abbreviations: 4F-PCC, 4-factor prothrombin complex concentrate; OAC, oral anticoagulant.
Surgery
Among patients requiring surgery (n = 524/861, 60.9%), 63.0% (n = 315) were administered 4F-PCC and 57.9% (n = 209) received plasma. Of note, some plasma utilized during surgery was for volume resuscitation, whereas 4F-PCC was used only for the reversal of anticoagulation. 4F-PCC was administered before the surgery nearly all the time (n = 287/315, 91.1%), and during surgery only 15.6% (n = 49/315) of the time and 6.3% (n = 20/315) after, while plasma was administered before 89.5% (n = 187/209), during 39.7% (n = 83/209), and after 9.6% (n = 20/209) of the time. 4F-PCC was the agent of choice when surgery was required within 1 h (n = 120/315, 38.1%) compared to plasma (n = 39/209, 18.7%). Both agents were similarly chosen when surgery was required within 2 to 3 h (4F-PCC: n = 103/315, 32.7% vs plasma: n = 73/209, 34.9%). Plasma was chosen more often than 4F-PCC when surgery was required within 4 to 6 h (plasma: n = 46/209, 22.0% vs 4F-PCC: 48/315, 15.2%).
Dosing and administration of treatment
The majority of time 4F-PCC was dosed by weight (n = 277/500, 55.4%) compared to fixed dosing (n = 143/500, 28.6%), or nominal dosing (n = 24/500, 4.8%), with an average dose by weight of 2432 IU, an average fixed dose of 2162 IU, and average nominal dose of 1572 IU. The remaining patients either had other dosing strategies (n = 4/500, 0.8%) or the dosing strategy was not available (n = 52/500, 10.4%). Looking across all dosing regimens, the average initial dose of 4F-PCC was 2186 IU. In life-threatening indications, the average dose, for all 4F-PCC use, appeared to be higher (2542 IU) than in emergent (1990 IU) or urgent (1661 IU) indications. Patients received a second 4F-PCC dose in 10.6% (n = 53/500) of the times. The average re-dose dose for these patients was 1328 IU (their average initial dose was 1653 IU). Re-dosing occurred most frequently in cardiac/thoracic surgery (n = 10/46, 21.7%) and least frequently in trauma/acute surgery (n = 1/55, 1.8%) compared to all other specialties (7.1%-13.2%). The 4F-PCC dosing strategy did not appear to differ substantially by urgency level, OAC medication, or patient condition (Supplementary Table 1). Similarly, the average number of plasma units administered to a patient was higher when the patient's urgency level was higher (life-threatening, 4.3 vs emergent, 3.9 vs urgent, 3.6 units). Overall, the average plasma units administered was 3.9.
When physicians were asked to indicate the primary reasons why a patient was treated with 4F-PCC or plasma, 4F-PCC was often used in life-threatening or emergent indications with the primary benefit cited as fast INR reduction, high efficacy, and fast administration, while the primary reason for using plasma was their experience or comfort with it (Supplementary Table 2).
Physician-Reported Information
A summary of physician-reported data can be found in Supplementary Table 3. In patients on an OAC, hemostatic/reversal agents are more likely to be used in life-threatening (74.9%) and emergent indications (77.0%) compared to urgent (59.1%) and elective/non-urgent (21.3%) indications. For all medical specialties, the majority of physicians (81.3%) reported that emergent indications typically needed to be addressed within 2 h (Supplementary Table 3). However, gastroenterologists provided more widespread answers with 51.7% (n = 15/29) reporting these indications needed to be addressed within 2 h, 24.1% (n = 7/29) within 3 to 4 h, and 24.1% (n = 7/29) within 4 h or more.
Details of the hemostatic/reversal agents physicians reported having used within the last 6 months for OAC reversal are outlined in Supplementary Table 4. In all medical specialties, the majority of physicians reported they independently decided which hemostatic/reversal agent was selected (n = 137/235, 58.2%), except for gastroenterology where 51.0% (n = 15/29) reported the decision was made in collaboration with other physicians. Hemodynamic instability was the most reported primary factor used to confirm the need for a hemostatic/reversal agent (79.6%) across all specialties, followed by lab results, INR levels, and risk of complications (Supplementary Table 3). Neurology and critical care/ICU also relied on neurological instability (n = 129/235, 54.9%), whereas gastroenterology relied also on patient co-morbidities (n = 115/235, 48.9%).
The most physician-reported influential factor when choosing a hemostatic/reversal agent was urgency (66.0%, see Supplementary Table 3). However, physicians indicated a preference toward using anticoagulation reversal agents they had the most experience with (68.9%). Physicians reported reasons for not using 4F-PCC were mostly due to cost (39.6%). Most physicians agreed that they preferred to use a specific reversal agent for DOACs (52.3%) (Supplementary Table 3). When physicians were asked how often 4F-PCC was utilized in conjunction with plasma, the majority of physicians stated that they sometimes utilized both products (40.9%), with 23.9% stating they used 4F-PCC in conjunction with plasma the majority of the time (Supplementary Table 3).
Discussion
In this retrospective study of 4F-PCC and plasma for OAC-associated bleeding, differences in the use of 4F-PCC or plasma were observed depending on medical specialty, level of urgency of treatment, patient condition, and current OAC medication. Urgency, type of bleeding, and OAC medication are the recommended primary considerations in managing bleeding and directing the use of hemostatic or reversal agents in bleeding patients taking OACs. 20 Local measures are recommended as sufficient for patients with non-urgent bleeding, and anticoagulation plans should be in place prior to elective or non-urgent surgery to avoid urgent reversals peri-operatively and risk of thrombosis extension or development.19,27 Results from the chart review in this study showed that plasma was most commonly used in emergent and urgent indications, whereas 4F-PCC was the preferred treatment option in life-threatening and emergent indications when rapid reversal was necessary, which reflects guideline recommendations. 6 4F-PCC can be stored at room temperature as a ready-to-reconstitute lyophilized powder that does not require ABO blood compatibility, in comparison to plasma; 6 therefore, it can be more readily available for use in life-threatening indications.
ICH, GI-related bleeding, and blunt trauma were among the top 6 conditions requiring OAC reversal in this study. Both ICH and GI-related bleeding are the most common major bleeding sites associated with OAC use.21,28 OAC-related GI bleeding management recommends a multidisciplinary approach.6,16,29 In this study, physicians from a gastroenterology specialty were the only group where the majority reported collaborative decision-making with other physicians for choosing hemostatic/reversal agents. Plasma was mostly used for GI bleeding in this study, despite 4F-PCC being the first-line treatment recommendation for GI bleeds 16 and allowing faster time to GI procedure, in comparison to plasma. 30 However, this may be due to time not being critical, as gastroenterology was among the 3 specialties to report the least incidence of life-threatening indications. 4F-PCC has been shown to be effective for treating VKA-associated ICH,13,31 as well as DOAC-associated ICH,32–35 and is recommended for the treatment of FXa inhibitor-associated ICH.17,36 Guidelines for urgent or emergent cardiac surgery recommend 4F-PCC, when a specific reversal agent is not available, 37 despite limited clinical data available. 38 Retrospective data have demonstrated no differences in key efficacy outcomes between the direct OAC reversal agent, andexanet alfa, and 4F-PCC.39,40 In addition, similar findings were observed in patients treated with PCCs, irrespective of which OAC therapy they were receiving.41,42 In this study, 4F-PCC was used most often within critical/intensive care, trauma/acute surgery, and neurosurgery specialties, aligning with its use in life-threatening indications and for the treatment of ICH and blunt trauma compared to plasma.
OAC medication was a major influential factor for physicians when choosing a hemostatic/reversal agent. As mentioned before, 4F-PCC is the indicated treatment option for VKA reversal; however, in our study, we observed that plasma was used more frequently in VKA-treated patients, most notably in urgent and emergent indications, as opposed to life-threatening indications. Notwithstanding that 4F-PCC has been found to be superior to plasma for both INR reduction and clinical hemostatic efficacy for urgent surgery, 13 and superior for INR reduction in major bleeding. 31 Preference toward utilizing plasma for VKA reversal was also found in a survey, in which 74% of respondents selected plasma for VKA reversal for emergency surgery or major bleeding. 4 Interestingly, physicians in this study reported using plasma mostly because of their experience and comfort with it, particularly for warfarin, while 4F-PCC was reportedly used mainly in life-threatening or emergent indications, and because of its fast action and high efficacy. These findings suggest there is a need for further education and understanding among physicians regarding the recommended reversal strategies for certain OAC agents.
Effective and prompt management of acute bleeding in patients on OAC therapy is vital. With the emergence of new anticoagulant medications, such as DOACs, it is imperative to share clinical experience on the management of DOAC-associated bleeding, particularly given the paucity of prospective and randomized studies in DOAC patients. Specific DOAC reversal agents are preferred but not yet broadly available, leading to 4F-PCC and even plasma use, though the latter should have no role in DOAC reversal, 21 but it may be used in bleeding settings for volume resuscitation or as part of massive transfusion protocols. Despite the fact plasma is neither recommended by DOAC reversal guidelines21,23–25,43 nor there is evidence of its effectiveness in DOAC reversal, plasma, as our study indicated, is still used in some cases for DOAC reversal. Since the concentration of coagulation factors in plasma is low, a large transfusion volume is required to increase coagulation factors; hence, plasma is not only unlikely to achieve the coagulation factor levels necessary for hemostasis, but it also comes with the risk of circulatory volume overload, which together with TRALI, is the leading cause of transfusion-related morbidity and mortality.9–11
Plasma was used around half of the time for VKA and FIIa inhibitor reversal and less than 25% of the time for FXa inhibitor reversal; 4F-PCC was used more than 75% of the time for FXa inhibitor reversal and around half of the time for warfarin and dabigatran reversal. The specific DOAC reversal agents idarucizumab and andexanet alfa target the FIIa and FXa inhibitors, respectively, which make them an effective treatment option when the DOAC is known.5,21 However, in life-threatening indications, testing for DOACs delays urgent treatment and puts the patient at risk. 5 4F-PCC provides the VKDF necessary to rapidly restore hemostatic balance in the event of major bleeding events without the need for DOAC testing.3,21 Despite being off-label, 4F-PCC has been shown to be a comparably cost-effective alternative agent for DOAC-associated bleeding in retrospective cohorts.39,40,44–46 There is no head-to-head prospective or randomized comparison data, though a trial assessing andexanet alfa versus standard of care is ongoing. 47
Besides the lack of evidence for a role in DOAC reversal, the limited historical role in VKA reversal, and the volume overload and acute lung injury issues,7–11 there are other reasons to avoid plasma in these settings. The coronavirus-19 outbreak in 2020 has had a significant impact on the amount of plasma available due to COVID-19 safety restrictions causing a reduction in donors, donor sites, and staffing at donor centers.48,49 Additionally, safety issues were raised for using blood products, as the risks of COVID-19 viral transmission, through allogeneic blood transfusions, were unclear. 49 Due to blood product shortages, and concern regarding viral transmission, patient blood management strategies outlined that other products, such as PCCs, should be utilized, if appropriate.48,49 4F-PCC is being used more readily, as not all specific reversal agents are widely available, 6 but clinical data evaluating its increased use over the pandemic are minimal to date. At the same time, physicians reported cost as one of the main reasons for not using 4F-PCC, and this was shown to be an influential factor in choosing plasma. However, utilizing 4F-PCC may circumvent the need for extra dosing, 50 and fixed dosing of 4F-PCC instead of weight-based dosing may be more cost-effective, while still as effective at controlling bleeding and restoring hemostasis across varied clinical settings for VKA reversal.34,51–54 Despite small cohort studies reporting hemostatic efficacy for low-dose 4F-PCC in DOAC-associated bleeding, 33 higher doses are still required within this setting. 21 Reducing blood product usage within clinical settings can alleviate the burden of supplying clinical demand that can make use of alternative, sometimes better recommended treatment options, as highlighted throughout the pandemic,48,49 in addition to donor exposure and associated risks of blood product transfusions for the individual patient.8,9 Often, patients may require OAC reversal and volume resuscitation simultaneously, and the latter should not preclude 4F-PCC use due to urgency or preference for plasma. PCCs can achieve hemostatic efficacy quicker than plasma while requiring fewer blood transfusion components needed for volume resuscitation, as shown in a recently published study comparing PCC and plasma in patients with post-cardiopulmonary bypass coagulopathy and bleeding. 38 Supplementation with fibrinogen in patients with trauma-induced coagulopathy has been shown to reduce the units of packed red blood cells, plasma, and platelets needed for transfusion, 55 emphasizing that blood product usage can be reduced in clinical settings, while maintaining or improving efficacious outcomes.
The main limitation of this study is its retrospective and observational nature. Other limitations include that only 2 hemostatic agents were studied, which precludes a more complete picture of the use of hemostatic/reversal agents in these institutions; however, both 4F-PCC and plasma were the most used agents, alongside vitamin K supplementation. Another limitation is that data from each chart was extracted only by 1 person and accuracy was not monitored. The method of chart selection was left to clinicians which introduced a degree of selection bias. Nevertheless, the multicenter nature of this study facilitated the participation and inclusion of a large number of physicians/patient charts, and thus, the large sample size may be able to overcome some of these limitations.
Conclusions
This retrospective chart review study has provided an in-depth evaluation of the real-world use of 4F-PCC and plasma for the treatment of OAC-associated bleeding and the clinical conditions in which they are administered. The results indicate that physician specialty has a significant impact on which hemostatic agents are used and that these decisions are governed mainly by the clinical presentation and urgency level, but that decision-making can also be affected by product availability and cost. The study showed that 4F-PCC was predominantly used for OAC reversal despite the available specific reversal agents for some of the OAC. Importantly, our study also showed that despite the fact plasma is neither recommended by OAC reversal guidelines nor there is evidence of its effectiveness in OAC reversal, plasma is still used for OAC reversal. However, plasma should be avoided in OAC reversal.
Supplemental Material
sj-docx-1-cat-10.1177_10760296231179682 - Supplemental material for Retrospective Analysis of the Real-World Utilization of 4-Factor Prothrombin Complex Concentrate and Plasma in Oral Anticoagulant-Associated Bleeding in US Hospitals
Supplemental material, sj-docx-1-cat-10.1177_10760296231179682 for Retrospective Analysis of the Real-World Utilization of 4-Factor Prothrombin Complex Concentrate and Plasma in Oral Anticoagulant-Associated Bleeding in US Hospitals by Majed A. Refaai, Paolo Bajcic, Robert McNeill, Christopher Hood and Truman J. Milling in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgements
Survey development, data collection, and analyses were performed by Elevate 42 (New York, US) and funded by CSL Behring. Medical writing support was provided by Meridian HealthComms Ltd (Plumley, UK) in accordance with Good Publication Practice guidelines and funded by CSL Behring. The authors authorized the submission of the manuscript by Meridian HealthComms and approved any statements or declarations.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MAR is in the advisory committee and is a consultant for Cerus, Stago diagnostic, CSL Behring, and Octapharma. TJM is a consultant for CSL Behring, Octapharma, Alexion-AstraZeneca, and Cellphire. PB, RM, and CH are employees of CSL Behring.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: survey development, data collection, and analyses were performed by Elevate 42 (New York, US) and funded by CSL Behring. Medical writing support was provided by Meridian HealthComms Ltd (Plumley, UK) in accordance with Good Publication Practice guidelines and funded by CSL Behring.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
