Abstract
Background
Standard Modifiable Cardiovascular Risk Factors (SMurfs) influence Atrial fibrillation (AF). However, data on their cumulative prognostic effect in Asian non-valvular AF (NVAF) patients receiving oral anticoagulants (OACs) remain limited. This study investigated the association between the cumulative burden of SMurfs and the prognosis of these NVAF patients on oral anticoagulation (OAC) therapy.
Methods
This retrospective cohort study used Tianjin Health and Medical Data Platform (THMDP) data on 20 782 NVAF patients on OACs between 2015 and 2020. Patients were categorized into groups by the number of baseline SMurfs (0, 1-2, or 3-4: hypertension, diabetes, hyperlipidemia, smoking). Cox proportional hazards models assessed the association between SMurf burden group and the primary outcome (all-cause mortality) and secondary outcomes. Subgroup and sensitivity analyses were performed to evaluate the consistency and robustness of the findings.
Results
After adjusting for confounders, compared to the SMurf-less group, the hazard ratios (HRs) for all-cause mortality were 1.20 (95% confidence interval [CI]: 1.01-1.44) for the SMurf (1-2) group and 1.67 (95% CI: 1.33-2.08) for the SMurf (3-4) group. For ischemic stroke, the hazard ratio (HR) was 1.44 (95% CI: 1.03-1.99) for the SMurf (3-4) group, while the association was not significant for the SMurf (1-2) group. A more pronounced association between SMurfs and all-cause mortality was observed in patients with an estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73 m2.
Conclusion
In Asian NVAF patients receiving OACs, a higher SMurf burden is significantly associated with an increased risk of all-cause mortality and ischemic stroke.
What's New?
In a large-scale cohort (>20 000) of anticoagulated Asian patients with non-valvular atrial fibrillation (NVAF), this is the first study to quantify the cumulative, dose-response impact of standard modifiable cardiovascular risk factors (SMurfs) on prognosis. We demonstrate that a simple SMurf count is a powerful, independent predictor of all-cause mortality and ischemic stroke.
This prognostic impact is significantly amplified in patients with chronic kidney disease, identifying a critical subgroup for targeted intervention. These findings highlight the insufficiency of anticoagulation alone and provide robust, real-world evidence for implementing comprehensive risk management, reinforcing the “Comorbidity” pillar of the Atrial Fibrillation Better Care (ABC) pathway to address the patient's total residual risk.
Introduction
Atrial fibrillation (AF) is the most common type of cardiac arrhythmia globally, often linked to complications like stroke and systemic embolism.1,2 AF patients commonly have multiple comorbidities, leading to high disability and mortality rates, imposing substantial health and economic burdens.
The approach to AF management has evolved significantly towards a more holistic and integrated strategy. A pivotal development was the introduction of the “Atrial Fibrillation Better Care” (ABC) pathway in 2017, which established a simple yet comprehensive framework for AF management: “A” for anticoagulation/avoid stroke, “B” for better symptom management, and “C” for cardiovascular and comorbidity optimization. 3 The effectiveness of this integrated approach is now well-supported by a robust body of evidence. Its benefits have been validated in major randomized clinical trials, such as the mAFA-II and MIRACLE-AF trials in China,4,5 and have been consistently demonstrated in large-scale observational studies 6 and further substantiated by meta-analyses showing that structured risk factor management improves AF-related outcomes. 7 This integrated approach was formally incorporated into major international guidelines, including the 2020 European Society of Cardiology (ESC) guidelines. 8 More recent guidelines, such as the 2023 ACC/AHA guidelines (with its similar SOS pathway) and the 2024 ESC AF-CARE pathway, have further reinforced and built upon this established focus on integrated care, underscoring that comprehensive risk factor management is a central pillar of optimal AF care.1,2 Therefore, AF should not be viewed in isolation, a thorough evaluation and management of comorbidities and risk factors are essential for patients with AF.
While oral anticoagulation (OAC) is the cornerstone for preventing cardioembolic stroke in AF, a substantial residual risk for adverse outcomes persists, even in fully anticoagulated patients. This residual risk could be largely driven by the progression of underlying atherosclerotic disease, which is influenced by the burden of standard modifiable cardiovascular risk factors (SMurfs). These factors, comprising hypertension, diabetes mellitus (DM), hyperlipidemia, and smoking, are well-established drivers of atherosclerosis.9–11 Although the benefits of managing these risk factors individually in patients with AF are recognized, the cumulative prognostic impact of harboring multiple SMurfs on this residual risk is not well-defined.
Therefore, this study aims to investigate the association between the cumulative burden of SMurfs and adverse prognostic outcomes in a large, real-world cohort of non-valvular AF patients receiving oral anticoagulants. By quantifying the dose-response relationship between the number of SMurfs and the risk of adverse outcomes, we seek to underscore the importance of the baseline risk factor profile and provide a stronger evidence base for comprehensive risk management in this population.
Methods
Study Design and Population
The data for this study were primarily sourced from the Tianjin Health Medical Data Platform (THMDP), an electronic medical record-based healthcare data platform in Tianjin, a provincial-level municipality located in Northern China with a large, diverse urban population of 13.9 million. The THMDP includes electronic health information from 97.8% (261/267) of community healthcare centers and 78 public hospitals, including 100% (42/42) of tertiary general hospitals and 36 secondary hospitals. The system employs a unique personal identification mechanism that enables continuous monitoring and coverage of the entire healthcare pathway for patients in Tianjin, based on hospital visits and information from community healthcare centers. The initial cohort included 134 516 patients who were first received OACs between January 2015 and June 2020. The index date was defined as the date of the first OAC prescription. The final study population was identified from this cohort by applying a multi-step exclusion process based on International Classification of Diseases, 10th Revision (ICD-10) codes. To define the target population of NVAF, patients were first excluded if they had no prior hospital discharge diagnosis of atrial fibrillation (ICD-10: I48). Subsequently, to ensure a true NVAF cohort and remove other primary indications for OAC, patients were also excluded if they had a prior discharge diagnosis of significant valvular heart disease or mechanical valve implantation, or if their OAC prescription was documented solely for the prevention of venous thromboembolism (VTE) complications. Additional exclusion criteria included: a prior OAC prescription within 12 months before admission; initiation of OAC therapy more than 90 days after admission; a diagnosis of trauma; and missing low-density lipoprotein cholesterol (LDL-C) data at baseline. This 90-day interval from hospital admission to the initiation of OAC therapy was established to ensure that the baseline data recorded at admission accurately represented the patients’ clinical status at the time of treatment initiation, thereby minimizing potential misclassification bias. 12 The study was conducted in accordance with the ethical principles of the Declaration of Helsinki and received full approval from the Ethics Committee of Tianjin Medical University General Hospital (Approval No: IRB2022-YX-235-01). The committee waived the requirement for individual informed consent due to the retrospective nature of the research and the use of fully anonymized, de-identified data, which posed minimal risk to patient privacy. The study's flowchart outlines the detailed inclusion and exclusion criteria (Figure S1 in the Supplement).
Definition of Exposure and Outcomes
The exposure in this study was defined as the number of baseline SMurfs, which includes hypertension, hyperlipidemia, diabetes, and smoking. Hypertension and diabetes were identified based on ICD-10 codes. The smoking status of patients was obtained by searching the personal registration information at community health service centers. Hyperlipidemia was defined as LDL-C ≥ 3.37 mmol/L and/or total cholesterol (TC) ≥ 5.18 mmol/L. 9 Participants were grouped based on their baseline SMurf counts into three categories: SMurf-less, SMurf (1-2), and SMurf (3-4). This categorization, which groups patients into low, intermediate, and high-burden categories, is consistent with the methodology used in prior research on the cumulative burden of SMurfs. 9
The primary outcome of this study was all-cause mortality. Patient mortality data was primarily collected from the hospital electronic medical records system, in conjunction with the THMDP, and death registration records from community health care centers. The study also assessed secondary outcomes such as ischemic stroke (IS), intracranial hemorrhage (ICH), gastrointestinal bleeding (GIB), and acute myocardial infarction (AMI), as well as composite outcomes of ischemic events (IS and AMI) and bleeding events (ICH and GIB). Among them, ICH includes intracerebral hemorrhage, subarachnoid hemorrhage, epidural hemorrhage, or subdural hemorrhage. The identification of ICH, IS, and GIB was based on ICD-10 codes and clinically relevant examinations results such as computed tomography (CT), magnetic resonance imaging (MRI), endoscopy. The identification of AMI was based on ICD-10 codes. Follow-up began on the index date and continued until the first outcome occurred, the maximum follow-up period of 3 years was attained, until December 31, 2021, or discontinuation of OACs (no new prescription within 183 days after the last prescription date), whichever came first.
Study Variables
The study's baseline variables encompassed demographic factors, clinical parameters, scoring scales, medical history, and medication use. Demographic factors included age, sex, body mass index (BMI), smoking status, and alcohol consumption. The main laboratory test information included hemoglobin (Hb), TC, LDL-C, high-density lipoprotein cholesterol (HDL-C), triglyceride (TG), uric acid. Estimate Glomerular filtration rate (eGFR) was calculated using the equation provided by the Chronic Kidney Disease Epidemiology Collaboration. 13 The scoring scales included CHA2DS2-VASc score, HAS-BLED score, DOAC score. Most medical history of patients was determined by searching for diagnoses that appeared in the system before the inclusion date of the population, according to ICD-10 codes, including stroke, venous thromboembolism, ischemic heart disease, peripheral vascular disease, bleeding or coagulation disorders, hemiplegia, dementia, chronic pulmonary diseases, gastrointestinal diseases, liver diseases, anemia, cancer, heart failure, connective tissue diseases, psychiatric disorders, except for hyperuricemia (defined as a serum uric acid concentration ≥420 umol/L for both sexes) 14 and hyperlipidemia (as previously described). Baseline medication use was determined by having at least one relevant prescription record within 6 months prior to baseline exposure, including anti-platelet agents, oral anticoagulants (categorized by OAC type: non-vitamin K antagonist oral anticoagulants [NOACs] vs warfarin), non-steroidal anti-inflammatory drugs (NSAIDs), gastric mucosal protective agents, glucocorticoids, amiodarone, anti-thyroid agents, selective serotonin reuptake inhibitors, statins, anti-hypertensive agents, anti-diabetic agents. A detailed list of the specific drugs included in each of these classes is provided in Table S1 in the Supplement.
Statistical Analyses
The distribution of SMurfs and internal risk factor composition is visually represented using donut charts and bar graphs. Normally distributed continuous variables are expressed as mean ± standard deviation, while non-normally distributed ones are shown as median (25th-75th percentiles). Categorical variables were expressed as frequencies (percentages). Group differences are evaluated using analysis of variance (ANOVA) and Kruskal-Wallis test for normally and non-normally distributed continuous variables, and Chi-square tests for categorical variables. The extent of missing baseline data for study variables is detailed in Table S2 in the Supplement. MissForest was predominantly employed for imputing these missing values.
Kaplan-Meier curves were used to visualize survival for the primary outcome of all-cause mortality. For all non-fatal secondary outcomes, cumulative incidence function curves were generated to appropriately visualize event probabilities. The association between the SMurf burden group and primary and secondary outcomes is primarily evaluated using Cox proportional hazards models, utilizing SMurf-less as the reference. The proportional hazards assumption for all outcomes was verified using Schoenfeld residuals, with global tests indicating no substantial violation. Four models are developed in the study: Model 1: Unadjusted. Model 2: Adjusted for key demographic and clinical confounders, including age, sex, eGFR, alcohol consumption, and a comprehensive list of medical histories. Model 3: Further adjusted for most of baseline medication use, importantly including the specific type of OACs. Model 4: Additionally adjusted for anti-hypertensive agents, anti-diabetic agents, and statins. The results are presented as hazard ratio (HR) and 95% confidence intervals (CI).
This study categorized specific causes of death based on baseline SMurf distribution into circulatory system diseases, respiratory system diseases, cancer, and other specific causes. 15 Subgroup analyses were conducted to explore the association between the SMurf burden group and all-cause mortality across different population strata, including age, sex, eGFR, and various health conditions and medication use. A series of sensitivity analyses were performed to confirm the robustness of our findings. In particular, to rigorously control for baseline confounding and approximate causal inference, a propensity score-based inverse probability of treatment weighting (IPTW) analysis was conducted for the primary outcome. Simultaneously, to account for the high mortality rate as a competing event, Fine-Gray proportional sub-distribution hazard models were employed for all non-fatal secondary outcomes. Moreover, to clarify the causal framework regarding baseline medication use (anti-hypertensive agents, anti-diabetic agents, statins), a Directed Acyclic Graph (DAG) was constructed (Figure S2 in the Supplement). Within this framework, these specific medications were primarily conceptualized as markers of unmeasured disease severity (confounding by indication). Accordingly, this assumption was verified by repeating the primary analysis after systematically excluding these treatment variables. Beyond this, additional sensitivity analyses were performed to further assess the impact of controlling specific risk factors on outcomes within the SMurf framework, specifically examining the effects of anti-hypertensive agents in hypertensive patients, anti-diabetic agents in diabetic patients, and statins in hyperlipidemic patients. Finally, to further validate our results, we employed several other approaches, including: utilizing the Fine-Gray proportional sub-distribution hazards model to account for competing risks when assessing cause-specific mortality; excluding extreme populations (those with bleeding or coagulation disorders, hemiplegia, dementia, cancer, peripheral vascular disease, or Hb < 90 g/L) that could disproportionately influence overall mortality; and excluding populations with major nontraditional risk factors such as chronic pulmonary diseases, hyperuricemia, connective tissue diseases, and psychiatric disorders. All statistical analyses were primarily performed using STATA (version 16.0) and R (version 4.1.3). A two-tailed P-value ≤ .05 was considered statistically significant.
Results
Patient Characteristics
A total of 20 782 patients were finally included in this study. The proportions of the SMurf-less, SMurf (1-2), and SMurf (3-4) in the total population were 12.2%, 77.5%, and 10.4% respectively (Figure S3 in the Supplement). The median follow-up times for the three groups were 208, 225, and 225 days, respectively, and the prevalence of SMurf within the population as well as the SMurf (1-2) and SMurf (3-4) was ranked in the order of hypertension, DM, hyperlipidemia, and smoking (Figure S4 in the Supplement). The average age of the population was 68.4 years, with a majority of males (55.8%). Patients in higher SMurf burden groups showed elevated uric acid and lipid levels (TC, TG, LDL-C), along with higher CHA2DS2-VASc, HAS-BLED, and DOAC scores. Most patients (82.2%) used NOAC, predominantly rivaroxaban. As the number of SMurfs increased, there was a gradual rise in the proportion of patients with pre-existing comorbidities. Specifically, heart failure and hyperuricemia were more prevalent in the SMurf (1-2), accounting for 37.6% and 18.7%, respectively. There was a gradual rise in the number of patients using additional medications as the number of SMurfs increased, such as anti-platelet agents, NSAIDs, gastric mucosal protective agents, statins, anti-hypertensive agents, and anti-diabetic agents, among others. Baseline characteristics are presented in Table 1.
Patient Characteristics by SMurf Burden Groups at Baseline.

Abbreviations: BMI, body mass index; eGFR, estimated glomerular filtration rate; Hb, hemoglobin; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; TC, total cholesterol; TG, triglyceride; DOAC, direct-acting oral anticoagulants; OAC, oral anticoagulant; NOAC, non-vitamin K antagonist oral anticoagulants; NSAIDs, nonsteroidal anti-inflammatory drugs; PPI/H2RA, proton pump inhibitors/H2 receptor antagonists.
Associations Between SMurf and All-Cause Mortality and the Distribution of Death Reason
The study observed a total of 1831 cases of all-cause death over a maximum follow-up duration of 3 years, representing 8.8% of the study population. Kaplan-Meier curve for all-cause mortality is shown in Figure S5 in the Supplement. Table 2 displays the univariate and multivariate adjusted Cox models for all-cause mortality. Initially, without adjusting other factors, the HR for all-cause mortality were 1.42 (95% CI: 1.20-1.69, P < .001) for SMurf (1-2) and 1.89 (95% CI: 1.54-2.32, P < .001) for SMurf (3-4) compared to SMurf-less. Subsequent adjustments were made for age, sex, eGFR, alcohol consumption, and medical history. After these adjustments, the HR for all-cause mortality were 1.21 (95% CI: 1.01-1.44, P = .035) for SMurf (1-2) and 1.78 (95% CI: 1.44-2.20, P < .001) for SMurf (3-4). Additional adjustments were then performed for baseline medication use, anti-hypertensive agents, anti-diabetic agents, and statins. Following these adjustments, the HR for all-cause mortality were 1.20 (95% CI: 1.01-1.44, P = .041) for SMurf (1-2) and 1.67 (95% CI: 1.33-2.08, P < .001) for SMurf (3-4). The comprehensive results of the final multivariable model were presented in Table S3 in the Supplement, and the results of IPTW analysis also confirmed the robustness of these findings (Table S4 in the Supplement). To further explore this dose-response relationship, we also modeled the SMurf count as a continuous variable. In the fully adjusted model, each additional SMurf was associated with a 22% increase in the risk of all-cause mortality (HR per SMurf: 1.22, 95% CI: 1.14-1.31, P < .001). Furthermore, to provide a more granular view of the risk gradient, we also calculated the hazard ratios for each specific SMurf count (1, 2, 3, and 4) separately, with detailed results presented in Table S5 in the Supplement. This analysis clearly illustrated an incremental increase in risk with each additional SMurf. Additionally, an analysis deconstructing the SMurf burden into its individual components was performed, with the independent prognostic weights for each risk factor detailed in Table S6 in the Supplement.
Associations Between SMurf and All-Cause Mortality.

Results are presented as hazard ratios (HRs) with 95% confidence intervals (CIs).
Model 2 adjusted for age, sex, eGFR, drinking daily, stroke, venous thromboembolism, ischemic heart disease, peripheral vascular disease, bleeding or coagulation disease, hemiplegia, dementia, chronic pulmonary disease, gastrointestinal disease, liver disease, anemia, cancer, heart failure, hyperuricemia, connective tissue disease, psychiatric disorder.
Model 3 included all covariates in model 2 plus anti-platelet agents, OAC type, NSAIDs, PPI/H2RA, glucocorticoid, amiodarone, anti-thyroid agents, SSRIs.
Table S7 presents the distribution of specific cause of death, categorized as SMurf-less, SMurf (1-2), and SMurf (3-4). The number of deaths in each category were 140, 1435, and 256 respectively. Among all deaths, the highest proportion (68.7%) was attributed to circulatory system diseases, followed by cancer (14.7%), and the lowest proportion was due to respiratory system diseases (5.0%). The internal cause of death within each category is similar to the overall distribution of cause of death.
Associations Between SMurf and Secondary Outcomes
The study also examined other outcomes such as ICH, IS, GIB, AMI, composite ischemic outcomes, and composite hemorrhagic outcomes (Table 3). Cumulative incidence function curves for all secondary outcomes are shown in Figures S6-S11 in the Supplement. The numbers of these events were 196, 877, 388, 371,1212, and 568 cases respectively. For composite ischemic outcomes, in the unadjusted scenario, the HR for events in SMurf (1-2) and SMurf (3-4) were 1.51 (95% CI: 1.22-1.87, P < .001) and 1.82 (95% CI: 1.40-2.35, P < .001) respectively when using SMurf-less as reference. After full adjustment, the HR for SMurf (1-2) was not statistically significant, while for SMurf (3-4), the HR for composite ischemic events was 1.46 (95% CI: 1.11-1.94, P = .007). In terms of IS, the study found that without adjustment, the HR in SMurf (1-2) and SMurf (3-4) were 1.44 (95% CI: 1.13-1.85, P = .004) and 1.63 (95% CI: 1.21-2.21, P = .002) respectively. Upon further adjustment for demographic variables, clinical parameters, medical history, and medication use, the HR for IS in SMurf (1-2) was 1.22 (95% CI: 0.94-1.57, P = .128), while for SMurf (3-4) it was 1.44 (95% CI: 1.03-1.99, P = .031), showing statistical significance. After adjusting for medical history, the HRs for SMurf (1-2) and SMurf (3-4) in relation to AMI lost their statistical significance.
Associations Between SMurf and Secondary Outcomes.

Composite ischemic outcomes were defined as ischemic stroke and acute acute myocardial infarction.
Composite hemorrhagic outcomes were defined as Intracranial hemorrhage and Gastrointestinal bleeding.
Results are presented as hazard ratios (HRs) with 95% confidence intervals (CIs). Model 1 was unadjusted. Model 2 adjusted for age, sex, eGFR, drinking daily, stroke, venous thromboembolism, ischemic heart disease, peripheral vascular disease, bleeding or coagulation disease, hemiplegia, dementia, chronic pulmonary disease, gastrointestinal disease, liver disease, anemia, cancer, heart failure, hyperuricemia, connective tissue disease, psychiatric disorder. Model 3 included all covariates in model 2 plus anti-platelet agents, OAC type, NSAIDs, PPI/H2RA, glucocorticoid, amiodarone, anti-thyroid agents, SSRIs. Model 4 included all covariates in model 3 plus anti-hypertensive agents, anti-diabetic agents, and statins.
In terms of composite hemorrhagic outcomes, the HR for SMurf (1-2) and SMurf (3-4) did not show statistical significance compared to SMurf-less in both partially adjusted and fully adjusted scenarios. After adjusting for medical history, the HR for ICH in SMurf (1-2) and SMurf (3-4) lost statistical significance. However, the overall trend remained consistent with the primary outcome. In terms of GIB, the HR for SMurf (1-2) and SMurf (3-4) did not show statistical significance, regardless of adjustment levels. Moreover, after adjustments, the HR values changed direction but remained statistically non-significant. Interestingly, when further adjusting for anti-hypertensive agents, anti-diabetic agents, and statins in both ICH and IS (Model 4), there was a slight increase in the HR for these outcomes in SMurf (1-2) and SMurf (3-4) compared to Model 3. Consistent results were observed for all non-fatal outcomes when accounting for the competing risk of death using Fine-Gray models in Table S8 in the Supplement.
Subgroup Analyses and Sensitivity Analyses
The subgroup analyses indicated that the findings were generally consistent with the main effects across all stratified groups after full adjustment. Furthermore, these analyses revealed a more pronounced association between SMurf and all-cause mortality in patients with eGFR < 60 mL/min/1.73 m2 when compared to SMurf-less (Pint < .001), as presented in Table 4.
Subgroup Analysis of the Association Between SMurf and All-Cause Mortality.
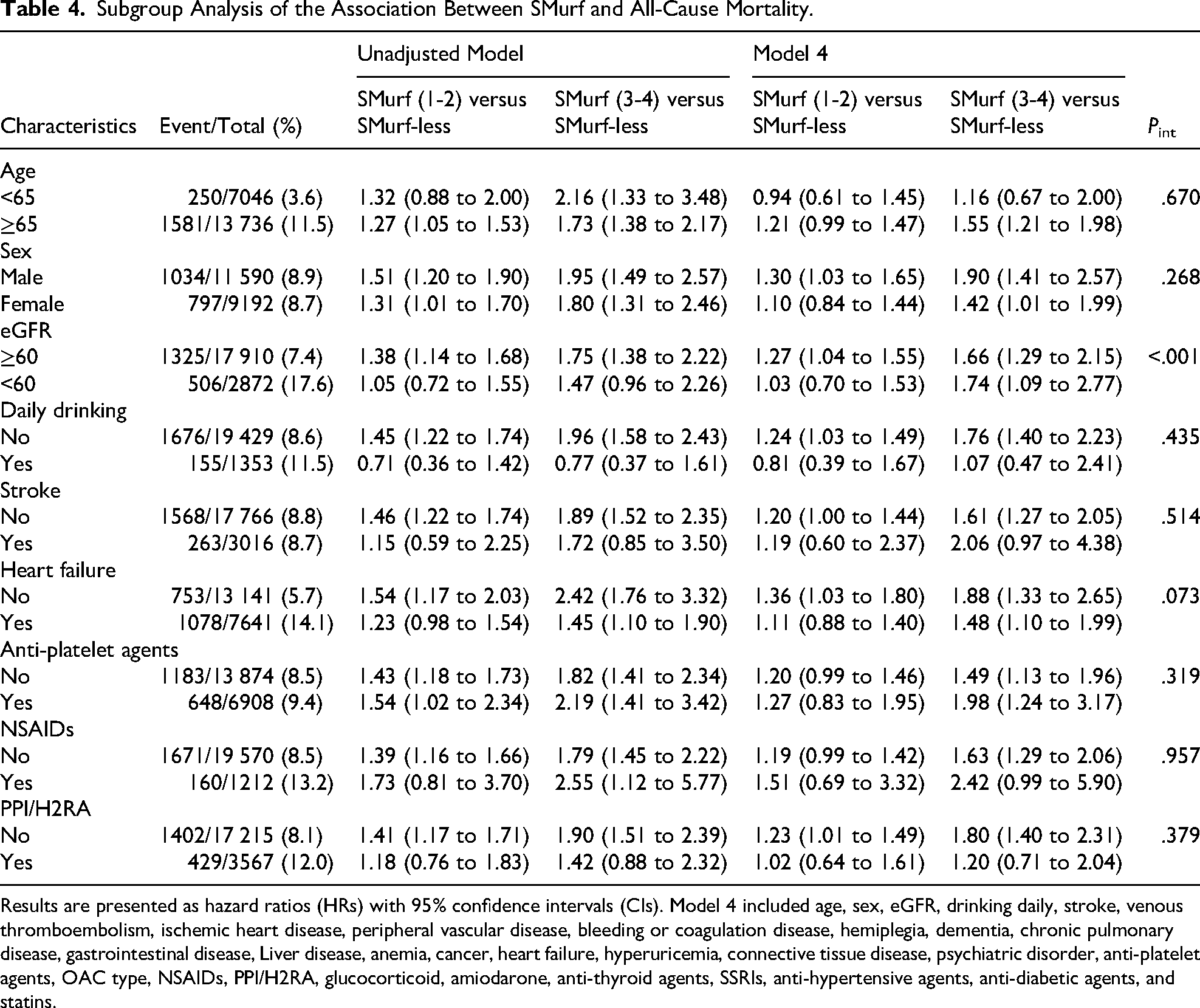
Results are presented as hazard ratios (HRs) with 95% confidence intervals (CIs). Model 4 included age, sex, eGFR, drinking daily, stroke, venous thromboembolism, ischemic heart disease, peripheral vascular disease, bleeding or coagulation disease, hemiplegia, dementia, chronic pulmonary disease, gastrointestinal disease, Liver disease, anemia, cancer, heart failure, hyperuricemia, connective tissue disease, psychiatric disorder, anti-platelet agents, OAC type, NSAIDs, PPI/H2RA, glucocorticoid, amiodarone, anti-thyroid agents, SSRIs, anti-hypertensive agents, anti-diabetic agents, and statins.
To test the robustness of our covariate adjustment strategy, we performed sensitivity analyses for the primary outcome (Table S9 in the Supplement). These analyses demonstrated that the association between a higher SMurf burden and all-cause mortality remained strong and statistically significant across all tested combinations of medication exclusion. Notably, the hazard ratios were modestly strengthened when these protective medication classes were excluded from adjustment, further supporting the robustness of our primary finding. Among hypertensive individuals, sensitivity analysis of anti-hypertensive medication use post full adjustment demonstrated a protective effect across most of outcomes compared to non-users besides ICH, albeit without statistical significance (Table S10 in the Supplement). In the diabetic population, sensitivity analysis of anti-diabetic medication use post full adjustment also showed a protective effect for most outcomes, including IS, AMI, composite ischemic outcomes, and ICH, but these effects were not statistically significant. However, there was a significant association between anti-diabetic medication use and higher all-cause mortality (HR: 1.45, 95% CI: 1.16-1.81). For composite hemorrhagic outcomes and GIB, there was a non-statistically significant increase in risk (Table S11 in the Supplement). For individuals with hyperlipidemia, sensitivity analysis revealed that statin use was associated with an increased risk of GIB (HR: 2.39, 95% CI: 1.22-4.67) (Table S12 in the Supplement). The results of competitive risk analysis indicate that SMurf primarily increases the risk of death related to circulatory system diseases (Table S13-S15 in the Supplement). After excluding for extreme populations that could potentially influence overall mortality (Table S16 in the Supplement), as well as baseline populations with nontraditional risk factors (Table S17 in the Supplement), the effect of SMurf on mortality remained consistent with the main findings.
Discussion
The study utilized data from THMDP for a retrospective observational study on non-valvular AF patients receiving oral anticoagulation therapy. Our findings suggest that in Asian populations with non-valvular AF, a higher burden of SMurfs is significantly associated with increased risks of all-cause mortality and ischemic events, particularly ischemic stroke. Subgroup analyses also revealed a more pronounced association between SMurf and all-cause mortality in patients with eGFR < 60 mL/min/1.73 m2 when compared to SMurf-less. Furthermore, competitive risk analysis indicated that SMurf primarily increases the risk of death related to circulatory system diseases. The sensitivity analysis for all-cause mortality further confirmed the robustness of the main results.
Patients with AF often present a complex clinical profile, characterized by multiple risk factors and comorbidities, advanced age, frailty 16 and inappropriate polypharmacy. This clinical complexity, also referred to as the clinical complexity of AF patients, 17 necessitates targeted treatment and management of relevant risk factors and comorbidities. Also, the burden of comorbidity and risk factors among patients with AF has risen in the last two decades, as highlighted by a cohort study from the UK. 18 In our study population, the baseline prevalence of hypertension, diabetes, hyperlipidemia, and smoking among AF patients was 76.5%, 27.3%, 26.2%, and 14.0%, respectively. Furthermore, a significant proportion of patients had a history of heart failure, peripheral vascular disease, liver disease, hyperuricemia, chronic pulmonary disease, and stroke. Analyses from the GLORIA-AF registry studies have revealed that AF patients with various patterns of comorbidities and more complex clinical phenotypes are associated with poor prognosis.19,20 Previous research has also confirmed that in populations with AF, the risk of all-cause death, cardiovascular death, major bleeding, and systemic embolism increases with worsened long-term health conditions, chronic diseases, and comorbidity burden.21,22 Recent ACC/AHA guidelines have highlighted the significance of risk factors and lifestyle choices in the secondary prevention of AF to avoid negative outcomes. They also stress the importance of multidisciplinary care for individuals with multiple chronic conditions. 2 Therefore, the management of pre-existing chronic conditions, risk factors, and comorbidities in AF is crucial. Our study uniquely incorporates SMurf into the secondary prevention strategies for AF populations. We investigate how SMurf, typically studied in coronary heart disease (CHD), impact the prognosis of NVAF patients on OAC therapy.
The importance of SMurf in managing CHD populations has been well established. Studies like the Framingham Heart Study have confirmed the impact of these risk factors on vascular health and the subsequent changes in prognosis post-intervention. 23 A recent study in China focusing on acute myocardial infarction further highlights the importance of managing these traditional risk factors in this population, 9 which emphasizes the critical role of SMurf in determining individual prognosis and stresses the need for timely and effective medical treatment. Furthermore, the “SMurf paradox” may be explained in this study by the possibility that individuals with fewer SMurfs may have worse outcomes due to a wider range of unfavorable and uncontrolled risk factors, as well as suboptimal evidence-based medical management. Previous research has shown limited evidence on the cumulative prognostic impact of SMurf on the prognosis of AF patients. A retrospective observational study from Australia suggested that a combination of traditional and nontraditional risk factors could impact the prognosis of AF patients, including all-cause death and major adverse cardiovascular events (MACE). However, the study did not find a statistically significant association between SMurf and AF prognosis in its primary outcomes. Additionally, the study was limited to a single tertiary center hospital with a small sample size. While nontraditional risk factors were categorized separately, the study only included a few factors (chronic kidney impairment, obstructive sleep apnea, obesity), potentially lacking reliability. 24 Prior research has also emphasized the impact of nontraditional risk factors in cardiovascular diseases. 25 Our study incorporates several nontraditional cardiovascular risk factors such as chronic pulmonary disease, hyperuricemia, connective tissue diseases, psychiatric disorders, etc, as covariates in the analysis. This is aimed at better controlling for the impact of these risk factors on the study population and elucidating the prognostic significance of SMurf.
In both ICH and IS outcomes, regardless of SMurf (1-2) or SMurf (3-4), the HR values in Model 4 showed a slight increase compared to Model 3. This increase may be attributed to the protective effects of the overall management practices such as BP reduction, glycemic control, and lipid lowering. Therefore, after adjusting for these factors, the HR exhibited a slight elevation. Subgroup analysis revealed a more evident association between SMurf and all-cause mortality in patients with eGFR < 60 mL/min/1.73 m2, but the effect of SMurf is diluted in the majority of other subgroups, thus further multidimensional assessments are needed in subsequent studies. As for the interaction between individuals with reduced kidney function and AF is currently intricate. Poor kidney function can lead to increased release of ACE, promoting atrial fibrosis and abnormal electrical conduction. 26 Uremic toxins in these patients may affect calcium ion levels in circulatory and myocardial cells, impacting cellular electrical activity. 26 Renal anemia and electrolyte imbalances from kidney dysfunction can worsen AF progression. 27 Conversely, AF can contribute to further kidney function decline through mechanisms like thrombus formation, microinfarctions, systemic inflammation, cardiac dysfunction, activation of the renin-angiotensin-aldosterone system, and nephrotoxic treatments, ultimately resulting in adverse outcomes. 28
Our analysis underscores the overarching importance of the cumulative SMurf burden, while also providing nuanced insights into the individual risk factors within our anticoagulated AF cohort. Hypertension, as the most prevalent SMurf, reaffirmed its critical role. Our sensitivity analysis, though not reaching statistical significance for all outcomes, suggested a beneficial impact of BP-lowering therapy on mortality and ischemic events, aligning with findings from large meta-analyses and the ARREST-AF study which demonstrate that aggressive risk factor management, including BP control, improves AF-related outcomes.29,30 The associations with diabetes and hyperlipidemia revealed greater complexity. In our analysis, the use of anti-diabetic agents was paradoxically associated with a higher risk of all-cause mortality, and statin use was associated with GIB. Crucially, as visualized in our DAG, these counterintuitive associations should not be interpreted causally, they represent classic confounding by indication. These findings likely reflect underlying clinical complexity in a real-world setting: anti-diabetic use serves as a marker for more advanced diabetes, or relates to treatment legacy effects and the specific risks associated with certain older drug classes, such as sulfonylureas.31–33 Similarly, the bleeding signal observed with statins was attenuated after excluding patients with pre-existing liver disease and gastrointestinal diseases (Table S18 in the Supplement), highlighting that such risks are likely driven by underlying comorbidities. Furthermore, pharmacological interactions may contribute. Given that the majority of our cohort received rivaroxaban, statins might competitively inhibit the CYP3A4 and P-glycoprotein pathways shared with rivaroxaban, potentially increasing anticoagulant plasma concentrations and contributing to the observed GIB risk. Smoking, another key modifiable factor, is a well-documented independent predictor of thromboembolic events and mortality in AF patients, and studies have shown significant benefits of smoking cessation.34–36 Our findings reinforce its importance as a critical component of the total risk burden. While these analyses of individual factors provide valuable clinical insights, they should not overshadow the central thesis of our study. The clear, dose-response relationship we observed between the total number of SMurfs and adverse outcomes demonstrates that the cumulative impact of these risk factors is more prognostically powerful than any single component. This finding reinforces the idea that a patient's long-term trajectory is shaped by their overall burden of atherosclerotic risk, shifting the focus from managing isolated conditions to assessing a patient's comprehensive risk profile.
Our findings should be contextualized by the known ethnic differences in AF. Asian patients often exhibit a higher intrinsic risk of ischemic stroke than Western counterparts with similar CHA2DS2-VASc scores 37 and may experience a greater prognostic impact from certain SMurfs like hypertension. 38 Concurrently, Asian populations have a demonstrably higher risk of intracranial hemorrhage with OACs. 39 This narrower therapeutic window between ischemia and bleeding underscores the heightened importance of managing the “residual atherosclerotic risk” in this population. Conceptually, while SMurfs are classically recognized as drivers of atherosclerotic disease, their prognostic impact in anticoagulated AF patients likely operates through dual pathways. First, a high cumulative SMurf burden accelerates coronary and vascular progression, which are major independent contributors to all-cause mortality in this cohort. Second, chronic metabolic stress from hypertension, diabetes, and smoking exacerbates systemic inflammation, endothelial dysfunction, and atrial structural remodeling. 40 These maladaptive changes promote a sustained pro-thrombotic milieu that persists despite oral anticoagulation, thereby driving the significant residual risk of ischemic stroke observed in our high-burden groups. By controlling the SMurf burden, clinicians can mitigate the underlying drive for atherothrombotic events, a strategy of particular importance for Asian patients. Our study provides crucial, large-scale data on the dose-response impact of SMurfs, offering a strong evidence base for prioritizing integrated care in this high-risk group.
A key methodological consideration was our decision to adjust for baseline SMurf-related medications. While our primary rationale was to control for confounding by treating medication use as an indicator of disease severity, these therapies can also act as mediators on the causal pathway. Our extensive sensitivity analyses provided clarity on this issue. The results showed that excluding these protective medications from the model strengthened the association between SMurf burden and mortality, as would be expected. This consistency across different adjustment strategies confirms that the association between a higher SMurf burden and worse outcomes is highly robust and is not dependent on the specific statistical model used.
The primary clinical implication of our study is the urgent need to move beyond an anticoagulation-only focus towards a holistic, integrated care model for AF patients. Our findings provide strong, real-world evidence from a large Asian cohort supporting the “C” (Cardiovascular and comorbidity optimization) pillar of the “Atrial Fibrillation Better Care” (ABC) pathway. By quantifying the substantial “residual risk” driven by the cumulative SMurf burden, our study complements the full spectrum of existing evidence from randomized trials like mAFA-II to observational studies and meta-analyses. While these studies have collectively shown what can be achieved with active management, our study shows what is at stake without it, reinforcing the importance of the ABC framework in routine practice. Streamlining this approach requires practical strategies. Key steps should include: (1) establishing multidisciplinary care teams involving cardiologists, primary care physicians, and other specialists; (2) implementing structured protocols for systematic risk factor assessment and management in AF clinics; and (3) empowering patients through education on lifestyle and treatment adherence. Leveraging digital health tools can further support these efforts. Adopting such an integrated model is crucial for addressing the patient's total cardiovascular risk and improving long-term outcomes.
This study has some limitations. Firstly, the research primarily utilized data from the THMDP, predominantly based on the Chinese population. The findings may not be generalizable to other population. Secondly, the medical history and medication data relied on ICD codes from electronic health records, which may have limited sensitivity and specificity, potentially introducing misclassification bias. Specifically, hyperlipidemia was defined primarily by baseline lipid levels to reflect the physiological status. However, this approach may underestimate the prevalence of hyperlipidemia among patients with well-controlled lipids on statins. Similarly, smoking status may be subject to under-reporting. Thirdly, the diagnosis of AF was also based on ICD codes without detailed classification of different types of AF. Fourthly, the study is retrospective and observational, therefore, causation cannot be determined. Furthermore, the observed hazard ratios represent a modest relative risk increase, suggesting that the findings should be interpreted as robust associations rather than definitive causality. Lastly, despite efforts to control for confounding factors, the study remains susceptible to unmeasured confounders. Furthermore, we were unable to assess patient adherence to OACs or account for dynamic treatment adjustments (eg, discontinuation or switching of therapies) during the follow-up period, which could influence outcomes. Our analysis also focused on the cumulative number of SMurfs, without investigating potential synergistic or interactive effects between specific combinations of these risk factors. Finally, our sensitivity analyses of concomitant medications were based on baseline use and could not capture subsequent changes in these treatments over time.
Conclusion
Our study demonstrates that in Asian patients with non-valvular AF on OAC therapy, a higher cumulative burden of SMurfs is associated with increased risks of all-cause mortality and ischemic stroke. These findings highlight the substantial residual risk conferred by baseline comorbidities and underscore the potential importance of implementing proactive, multifactorial risk management strategies. While our observational data cannot prove the effectiveness of such interventions, they provide a strong rationale for future trials and for prioritizing comprehensive risk factor control in clinical practice.
Supplemental Material
sj-docx-1-cat-10.1177_10760296261442055 - Supplemental material for Standard Modifiable Cardiovascular Risk Factors and Prognostic Outcomes in Non-Valvular Atrial Fibrillation Patients on Oral Anticoagulants
Supplemental material, sj-docx-1-cat-10.1177_10760296261442055 for Standard Modifiable Cardiovascular Risk Factors and Prognostic Outcomes in Non-Valvular Atrial Fibrillation Patients on Oral Anticoagulants by Linhao Liu, Hangkuan Liu, Pengfei Sun, Geru Aa, Yuanyuan Liu, Zhiqiang Zhang, Qing Yang and Xin Zhou in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Nonstandard Abbreviations and Acronyms
Acknowledgments
We sincerely appreciate all participants of this study for their invaluable contributions, which have been instrumental in advancing this research.
Author Contributions
XZ and QY contributed to the conception and design of the study. LL and HL contributed to manuscript drafting, data acquisition, analysis and interpretation of the data. PS, GA, YL, and ZZ contributed to the critical revision of the manuscript for important intellectual content. All gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Key Research and Development Program of China (No. 2022YFF0706000), National Natural Science Foundation of China (82320108001, 82270349, 82321001, 72274133 and 72304205) and Tianjin Key Medical Discipline (Specialty) Construction Project (Grant No. TJYXZDXK-069C).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data analyzed in this study were derived from the Tianjin Health Medical Data Platform (THMDP) and were rigorously de-identified prior to use by the research team to protect patient privacy. However, due to institutional policies of the THMDP, ethical approval stipulations, and overarching privacy safeguards designed to prevent any potential re-identification, the individual-level de-identified dataset underlying this article cannot be made publicly available.
Supplemental Material
Supplemental material for this article is available online.
