Abstract
Objective
Catheter-related thrombosis (CRT) is a common complication in colorectal cancer patients undergoing chemotherapy, significantly impacting patient outcomes. However, effective predictive tools for identifying high-risk patients are currently lacking. This study aimed to develop and validate a predictive model, the TD score, to identify patients at high risk of CRT based on clinical parameters.
Methods
This single-institutional retrospective study included 730 colorectal cancer patients with intravenous catheters, divided into training (n = 624) and test sets (n = 106). The primary endpoint was CRT, diagnosed via imaging. Multivariable logistic regression analysis was used to identify independent predictors of CRT, and a predictive model (TD score) was developed based on T stage and duration of intravenous catheter use. The model's performance was evaluated using receiver operating characteristic (ROC) curve analysis.
Results
The TD score demonstrated good diagnostic performance, with areas under the ROC curve of 0.732 in the training set and 0.749 in the test set. Survival analysis revealed that patients with lower T stages had longer durations of non-CRT status. The study identified T stage and duration of intravenous catheter use as independent predictors of CRT.
Conclusions
The TD score is a promising tool for identifying high-risk patients for CRT. It may improve patient risk stratification and guide targeted prophylactic interventions. Future validation studies involving larger and more diverse patient cohorts are needed to confirm the clinical utility of the TD score and evaluate its performance across different TNM stages.
Article highlights
This retrospective study of 730 colorectal cancer patients developed a TD score using T stage and catheter duration, demonstrating good predictive performance (AUC 0.732-0.749). The TD score effectively stratifies catheter-related thrombosis risk, enabling targeted preventive strategies for high-risk individuals.
Introduction
Globally, colorectal cancer (CRC) ranks as the third most prevalent malignant tumor by incidence and the second leading cause of cancer-related mortality, posing a substantial threat to global public health. Systemic chemotherapy serves as a critical therapeutic modality in the management of colorectal cancer. At present, peripherally inserted central catheters (PICCs) and implanted ports are commonly used intravenous administration routes for patients undergoing malignant tumor chemotherapy. These devices offer several advantages, including simple operation, long indwelling time, safety, and efficiency. They can effectively avoid the pain caused by chemotherapy drug-induced vascular damage and repeated punctures. A small number of patients also utilize centrally inserted central catheters (CICCs). 1
However, despite the convenience of intravenous catheters, they are associated with complications such as infection and thrombosis.2,3 Cancer patients exhibit elevated thrombotic risk, which is further exacerbated by the presence of indwelling vascular access devices. Consequently, CRT represents a relatively common complication in this population. It is important to distinguish between asymptomatic CRT, often detected incidentally on routine imaging, and symptomatic CRT, which presents with clinical signs such as limb swelling, pain, or erythema. The reported incidence rates for CRT vary widely (5% to 28%) largely depending on the intensity of screening; asymptomatic cases are far more common than symptomatic ones.2,4 While asymptomatic CRT may often be managed conservatively, symptomatic CRT is a major cause of catheter removal of central venous access devices (CVADs). While catheter removal is sometimes necessary in CRT patients, this intervention disrupts ongoing therapies, prolongs treatment, and prolongs hospital stays, thereby worsening patients’ physical condition, psychological well-being, and financial burden. In critical cases, complications may escalate to fatal pulmonary embolism or life-threatening cardiac arrhythmias. In recent years, numerous studies on venous catheter-related thrombosis (CRT) have been conducted both domestically and internationally. However, the independent risk factors identified in these studies vary significantly.5,6 Additionally, the prophylactic use of anticoagulant therapy in patients with solid tumors undergoing chemotherapy remains controversial, particularly for gastrointestinal tumors.6–15 This is because malignant digestive tract tumors are prone to tumor-related bleeding, such as hematochezia.
Although clinical models for predicting thrombosis exist, such as the Wells score and modified Wells score, they are not suitable for evaluating venous access port-related thrombosis in cancer patients undergoing chemotherapy. In recent years, there have also been studies on CRT, but the applicable disease range is relatively broad.16–19 However, most CRT studies focusing on malignant tumors have investigated populations with lung and breast cancer, while colorectal cancer patients have been rarely studied specifically.20–25 This focus on CRC is warranted for several reasons. First, CRC patients have a distinct thrombotic risk profile and a high prevalence of underlying bleeding risks due to the nature of the primary disease (eg, hematochezia from the primary tumor or anastomotic sites).6–15 This creates a significant clinical dilemma regarding anticoagulant prophylaxis. Second, CRC is one of the most common malignancies globally, leading to a large population exposed to the risk of CRT. The lack of a validated, CRC-specific predictive model represents a critical gap in evidence, as risk factors identified in other cancer types may not be directly generalizable to this population.
Therefore, the aim of the current study was to develop and validate a predictive model specifically for identifying CRT risk in colorectal cancer patients.
Methods
Study Patients
The study was designed as a single-institutional retrospective analysis, incorporating both a training set and a test set. For the training set, 5-fold cross-validation was employed, where each fold involved using one of the five subsets as the validation set, while the remaining four subsets were utilized for training (Figure 1). The inclusion criteria for the entire study cohort were as follows: (a) Diagnosis of colorectal malignant tumor; (b) Presence of an intravenous catheter (including port, PICC, and CICC); (c) Availability of enhanced CT and/or color Doppler ultrasound for venous evaluation. The choice of imaging modality was based on clinical presentation and institutional protocols. Contrast-enhanced CT was typically performed as part of routine oncological staging or follow-up scans, which allowed for concurrent assessment of thrombus presence. Color Doppler ultrasound was the primary investigation for patients presenting with symptoms suggestive of CRT, such as limb swelling, pain, or erythema along the catheter path. The timing of imaging was either prompted by symptoms or performed at scheduled intervals during cancer follow-up. The exclusion criteria were: (a) Absence of the date of catheter implantation; (b) Poor imaging quality that rendered the images difficult to interpret; (c) Having received anticoagulant treatment. Specifically, the training set included consecutive colorectal malignant tumor patients with intravenous catheters from January 2022 to December 2022 at the study hospital. The test set comprised consecutive colorectal malignant tumor patients with intravenous catheters from January 2023 to February 2023 at the same institution.
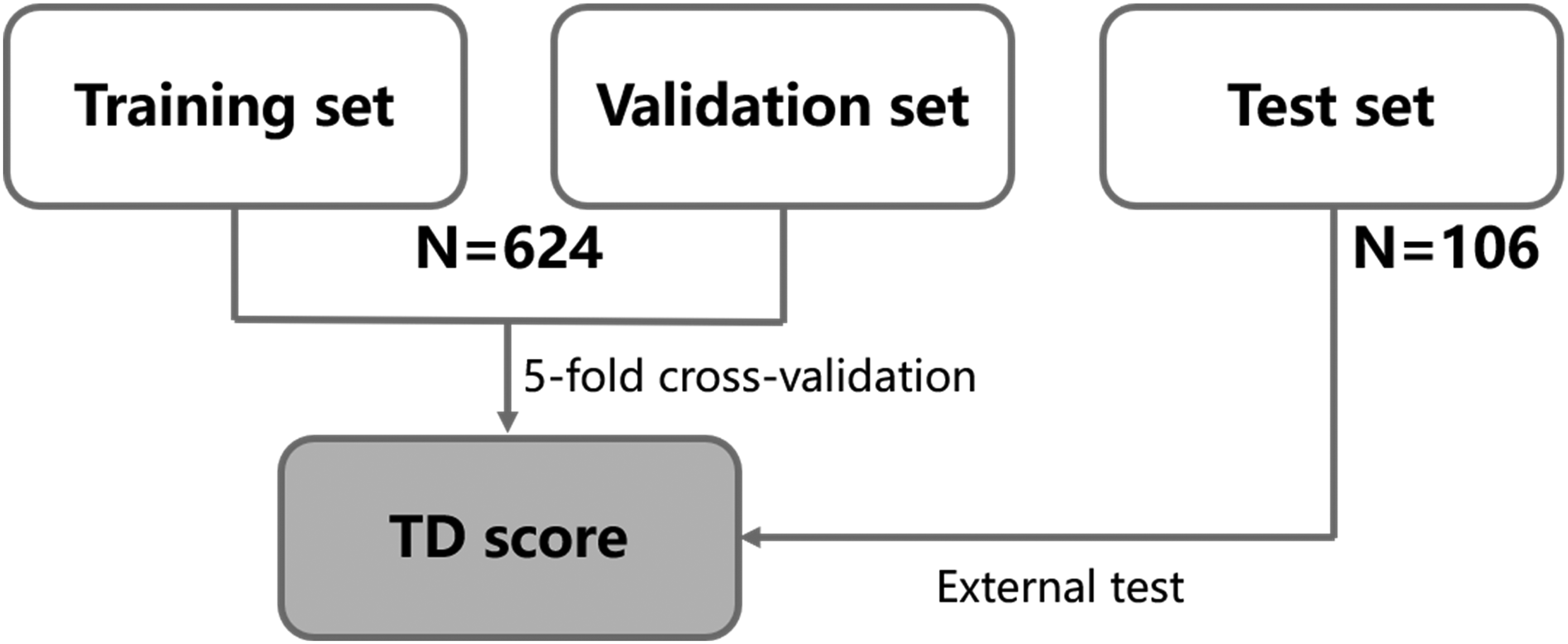
Study flowchart. Patients were divided into the training set (n = 624) and the test set (n = 106). The training set was divided into 5 samples. One part of the five samples was used as the validation set, and all the others were used as the training set. After five training sessions, five different models were obtained. The performance of five models was evaluated, and the best hyperparameter was selected. Using the optimal hyperparameters, all the five datasets were used as the training set to retrain the model, and the final required model was obtained. Finally, the model was tested on the test set. The Clinical Characteristics- and laboratory value-based predictive model, the TD score (based on the T stage, and days with intravenous catheter), was developed to identify catheter-related thrombosis (CRT) in the training set and externally validated in the test set.
Clinical data were meticulously collected, including age, sex, BMI, Basic Activities of Daily Living (BADL), TNM staging of colorectal cancer (according to the UICC/AJCC eighth edition, 2017), and history of diabetes, hypertension, hyperlipidemia, smoking, and chemotherapy. Additionally, laboratory data were gathered, encompassing leukocytes, platelets, hemoglobin, coagulation function, and D-dimer levels.
Variable Definitions
The duration of intravenous catheter use was defined as the time from catheter insertion to the occurrence of the primary endpoint (CRT diagnosis) or to the date of the last imaging examination for patients who did not develop CRT (censoring). In the predictive model, this variable was treated as a fixed, baseline characteristic (ie, the total observed duration for each patient) rather than as a time-dependent exposure. This approach was chosen for pragmatic reasons to facilitate risk stratification at the point of patient follow-up based on the elapsed catheter time.
Indications for Venous Assessment
The diagnosis of CRT was confirmed by imaging. The selection between contrast-enhanced CT venography and color Doppler ultrasound was guided by clinical considerations. Contrast-enhanced CT was generally preferred for asymptomatic patients undergoing routine cancer staging scans, as it allowed for a comprehensive evaluation of the central veins (eg, brachiocephalic vein, superior vena cava) and simultaneous assessment of thoracic anatomy and tumor status. Color Doppler ultrasound was the first-line investigation for symptomatic patients due to its accessibility, lack of radiation, and high sensitivity for detecting thrombi in the upper arm and jugular veins. It was particularly useful for evaluating the catheter insertion site and the adjacent peripheral veins.
Catheter Placement and Maintenance
All PICCs were inserted by an experienced (≥10 years) and qualified professional nurse under ultrasound guidance. The tip of the PICC catheter was located in the superior vena cava. All centrally inserted central catheters (CICCs) and implantable venous access port (PORT) patients underwent venipuncture using the modified Seldinger technique under ultrasound guidance or via blind insertion. The catheter tip position was confirmed by chest X-ray examination post-procedure.
The right subclavian vein was preferred, followed by the right internal jugular, and the left subclavian veins.
Predictive Model Derivation and Validation
Patient Allocation: The study cohort was split into derivation (training) and validation (test) sets using a temporal split. The derivation set comprised consecutive patients from January to December 2022 (n = 624), and the validation set comprised consecutive patients from January to February 2023 (n = 106). This temporal split was used to approximate external validation by testing the model on data from a later time period.
Model Development in the Derivation Set: Variable selection was performed exclusively within the derivation set. Univariable logistic regression was first used to assess the association between each clinical variable and CRT. Variables with a P value < .1 in the univariable analysis were included in the multivariable logistic regression model with forward stepwise selection. The final model retained only variables that were statistically significant (P < .05).
Model Specification and Validation: The regression coefficients (β) for the predictors in the final model were derived from the derivation set. These coefficients were then fixed and applied to the patients in the validation set to calculate each patient's TD score without any further re-estimation or adjustment. The model's performance was evaluated in both sets based on its discriminative ability, measured by the area under the receiver operating characteristic curve (AUC). Calibration, which assesses the agreement between predicted probabilities and observed outcomes, was evaluated using the Hosmer-Lemeshow goodness-of-fit test in the derivation set. A non-significant result (P > .05) would indicate good calibration.
Sample Size Calculation
Given that the incidence of CRT (catheter-related thrombosis) can be as high as 30%–50% among all intravenous catheter patients,4,26 the allowable error was set at 0.1 times the proportion of CRT cases. The width of the confidence interval was defined as twice the allowable error. Based on these criteria, the calculated sample size range was 402–928. Accordingly, we included a total of 624 patients in the training set, which satisfied the required sample size.
Follow-up
The primary endpoint of the study was the occurrence of CRT (catheter-related thrombosis). For patients who did not develop thrombosis, the endpoint was defined as the date of their last imaging examination (enhanced CT and/or color Doppler ultrasound) (not later than February 2024). The cutoff date for the training set was December 2022, while for the test set, it was limited to January–February 2023.
Statistical Analysis
Categorical variables were compared using the x2 test or Fisher's exact test, as appropriate. Continuous variables were compared using the Mann-Whitney U test or the t-test, depending on the data distribution. For the primary objective of developing a predictive model, we employed multivariable logistic regression. Although a time-to-event analysis (eg, Cox proportional hazards model) could be considered, logistic regression was selected for its direct interpretation of the predicted probability of CRT occurrence within the observable clinical timeframe. This approach aligns with our goal of creating a simple, static risk score based on baseline and readily available follow-up data (T stage and total catheter duration) to stratify risk at a given point in a patient's journey, rather than modeling the instantaneous hazard over time. All statistical analyses were performed using SPSS version 22.0 (IBM). A two-sided P value of less than 0.05 was considered statistically significant.
Ethics Statement
(i) The study protocol was reviewed and approved by the Institutional Review Board of the Sixth Affiliated Hospital of Sun Yat-sen University (Approval No. 2024ZSLYEC-208); and (ii) confirmed that all experiments were performed in accordance with relevant guidelines and regulations.
Given the retrospective nature of the study, the requirement for written informed consent was waived by the approval committee. This study was conducted in accordance with the principles of the TRIPOD (Transparent Reporting of a multivariable prediction model for Individual Prognosis or Diagnosis) statement to ensure comprehensive and transparent reporting of the development and validation of the predictive model.
Results
Baseline Patient Characteristics
A total of 730 patients (435 men and 295 women; mean age, 58 years ± 11 [SD]) were initially considered for inclusion. These patients were divided into a training set (n = 624) and a test set (n = 106) (Figure 1, Table 1). The diagnosis of thrombosis in all cohorts was based on imaging findings.
Baseline Clinical Characteristics of Patients in the Training set, and Test set.
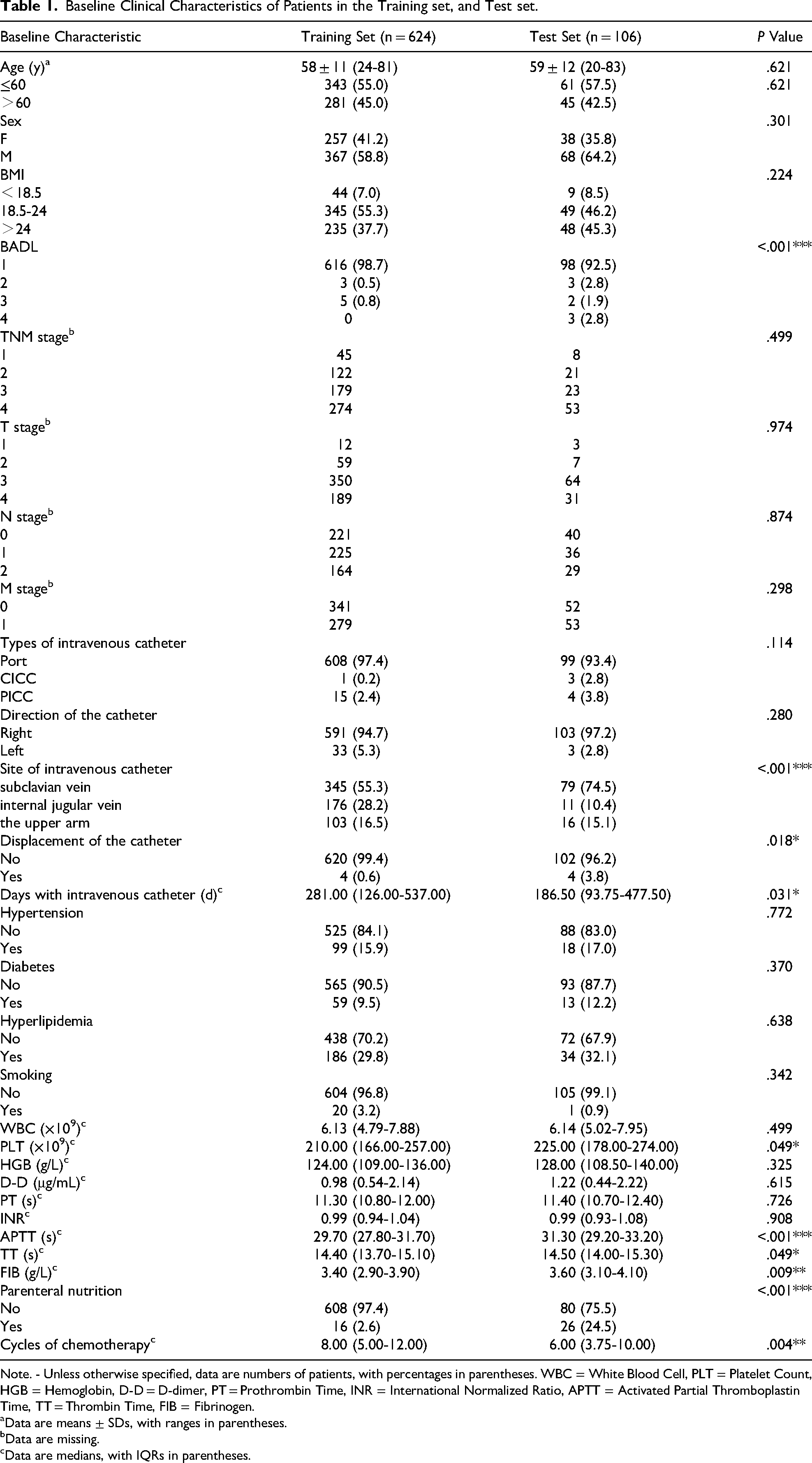
Note. - Unless otherwise specified, data are numbers of patients, with percentages in parentheses. WBC = White Blood Cell, PLT = Platelet Count, HGB = Hemoglobin, D-D = D-dimer, PT = Prothrombin Time, INR = International Normalized Ratio, APTT = Activated Partial Thromboplastin Time, TT = Thrombin Time, FIB = Fibrinogen.
Data are means ± SDs, with ranges in parentheses.
Data are missing.
Data are medians, with IQRs in parentheses.
To ensure that the “training” effect of the model could be reasonably generalized to the “testing” effect and ultimately applied to real-world scenarios, the data distribution of the training set, validation set, and test set was designed to be approximately similar (Table 1).
Development and Validation of a CRT Predictive Model
Based on all variables, the optimal regularization parameter (λ) in the regression model was determined through 5-fold cross-validation. This parameter was selected from all clinical features based on the best-performing λ value (Figure 2). When comparing the general clinical parameters between the positive and negative groups in the training set, significant differences were observed in age, TNM stage, T stage, M stage, duration of intravenous catheter use, white blood cell count (WBC), D-dimer levels, and cycles of chemotherapy (*P < .05) (Table 2).

Feature selection. (A) Vertical line is drawn when λ (optimal parameter value) = 0.020. (B) shows the relationship between each characteristic coefficient and log(λ) after adjusting λ.
Comparison of General Clinical Parameters Between Thrombosis Positive and Negative Patients in the Training set.
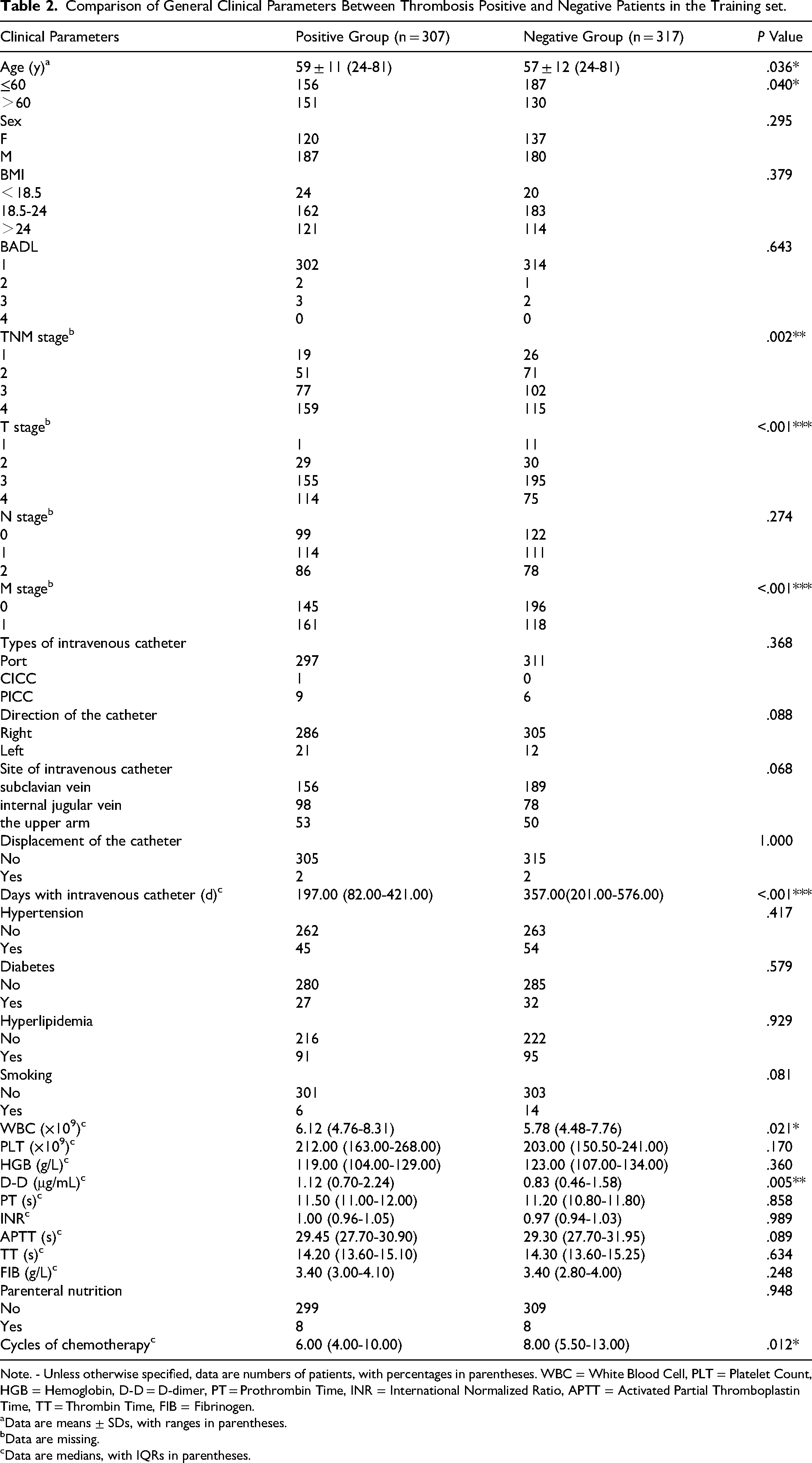
Note. - Unless otherwise specified, data are numbers of patients, with percentages in parentheses. WBC = White Blood Cell, PLT = Platelet Count, HGB = Hemoglobin, D-D = D-dimer, PT = Prothrombin Time, INR = International Normalized Ratio, APTT = Activated Partial Thromboplastin Time, TT = Thrombin Time, FIB = Fibrinogen.
Data are means ± SDs, with ranges in parentheses.
Data are missing.
Data are medians, with IQRs in parentheses.
In the multivariable logistic regression analysis, T stage (OR, 2.210 [95% CI: 1.257, 3.883]; **P = .006) and duration of intravenous catheter use (OR, 0.998 [95% CI: 0.997, 1.000]; **P = .028) were identified as independent predictors of CRT (Table 3). The higher the T stage, the greater the tumor burden. Previous studies had shown that tumor burden was positively correlated with the risk of thrombosis. That is, patients with large tumors, advanced (stage III/IV) or metastatic tumors had a higher risk of thrombosis. The mechanism was that as the tumor burden increased, it released more coagulation-promoting substances (such as tissue factor and inflammatory cytokines), which further activated the coagulation system. Therefore, this study showed that for every additional stage of T classification, the risk of thrombosis increased by a factor of 2.2. Furthermore, the result also indicated that for every additional day of catheter placement, the risk of thrombosis decreases by 0.2%. However, this effect was extremely weak compared to the T stage. Although data on catheter-related variables (eg, type [PICC, Port, CICC], insertion site, laterality) were collected, these factors were not retained in the final multivariable model. In the univariable analysis, these variables did not demonstrate a statistically significant association with CRT risk (P > .05) in our cohort. Consequently, they were not selected for inclusion in the stepwise regression model.
Predictors for Identifying CRT at Logistic Regression Analysis in the Training Cohort.

Note. - WBC = White Blood Cell, D-D = D-dimer, OR = odds ratio.
Based on the coefficients derived from the logistic regression analysis, a predictive model for CRT was developed, named the TD score. Here, “T” represents T stage, and “D” represents the duration of intravenous catheter use. The cutoff value for identifying thrombosis versus non-thrombosis was set at 0.605, with an area under the receiver operating characteristic (ROC) curve of 0.732 in the training set, indicating moderate discriminatory ability (Figure 3, Table 4).
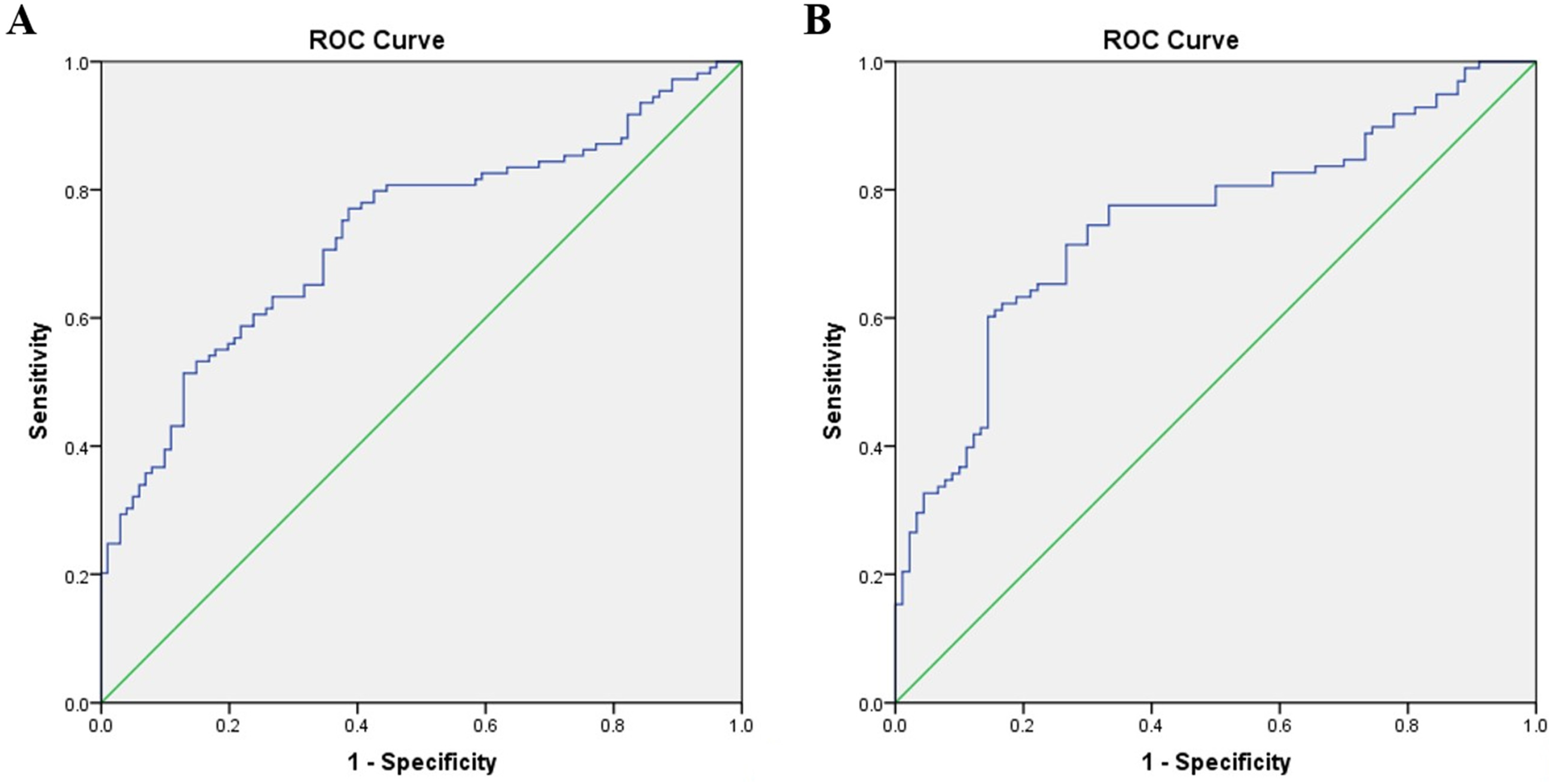
ROC curve validation plot. (A) The AUC of training set was 0.732, and the sensitivity and specificity were 51.4% and 87.1%, respectively. (B) The AUC of test set was 0.749, and the sensitivity and specificity were 60.2% and 85.6%, respectively.
Diagnostic Performance of the Predictive Model for Identifying CRT.

To externally validate the TD score model, the scoring wizard function in SPSS software was utilized for the test set. The results showed that the cutoff value for identifying thrombosis versus non-thrombosis remained 0.605, with an area under the ROC curve of 0.749 in the test set, which similarly represents a moderate level of discrimination (Figure 3, Table 4).
Kaplan-Meier Survival Analysis of CRT
Kaplan-Meier survival analysis of CRT-free survival was performed for the entire cohort. It is important to note that the CRT events included in this analysis comprised a mix of symptomatic and asymptomatic cases, as they were identified through clinically indicated imaging. Among the 730 patients in the training and test sets, the distribution of T stages was as follows: 15 patients in T1 stage, 66 in T2 stage, 414 in T3 stage, and 220 in T4 stage, with 15 patients having missing data. The overall median survival time was 568 days (95% CI: 461, 675). The mean survival times were 1344 days (95% CI: 1176, 1511) for T1, 738 days (95% CI: 584, 891) for T2, 905 days (95% CI: 747, 1063) for T3, and 684 days (95% CI: 554, 814) for T4 (Figure 4). A significant difference in survival was observed among the different T stages (*P < .05).

(A) median overall survival. (B) Kaplan–Meier curve demonstrating survival probability for the T stage.
Discussion
The GLOBOCAN 2020 database revealed over 1.9 million incident CRC (including anal carcinoma) diagnoses globally, with 935,000 associated fatalities during the same period. This dataset shows CRC ranks third by incidence but second by mortality. Regarding tumor treatment, most patients require venous catheters, resulting in a significant number of cancer patients at risk of CRT. Several CRT prediction models have been reported but did not give special attention to the CRC.16–19 For instance, a recent prospective machine learning study by Ma et al (2025) developed a nomogram for CRT risk in mixed-cancer patients with various cancers, identifying D-dimer and catheter indwelling time as key predictors. 18 The prominence of D-dimer, a global marker of coagulation activation and fibrinolysis, makes biological sense. Its absence in our final model may be specific to CRC. Colorectal tumors, especially with liver metastasis, can chronically elevate D-dimer, potentially reducing its discriminative power for a superimposed CRT event compared to other cancers. This highlights that a “one-size-fits-all” model may not be optimal. Similarly, Hu et al (2025) constructed a model for PICC-related thrombosis, highlighting platelet count and chemotherapy cycles as significant factors. 19 The role of chemotherapy cycles aligns with the known cumulative endothelial toxicity of certain agents. Our study, which included all catheter types and a CRC-specific population, found T stage to be a stronger driver than chemotherapy cycles. This suggests that in CRC, the intrinsic pro-thrombotic risk from the tumor itself (captured by T stage) may outweigh the incremental risk from cumulative chemotherapy, at least within the timeframe of this study. Our study contributes to this growing field by focusing specifically on colorectal cancer, a population with unique risks, such as a predisposition to bleeding, and by proposing a parsimonious model based on two readily available clinical variables. The comparable AUC of our simpler TD score (0.749) to these more complex models suggests that T stage and catheter duration are particularly potent predictors in the CRC population.
CRT pathogenesis aligns with Virchow's triad through three mechanisms: the placement of a venous catheter can cause local vessel damage; the presence of a venous catheter changes blood flow dynamics; and protein and blood cell adhesion on the catheter surface increases hypercoagulability.
Despite its prevalence, the management of CRT remains challenging due to the lack of effective predictive tools and the potential risks associated with prophylactic anticoagulation. Previous randomized controlled trials have evaluated the use of anticoagulation prophylaxis with warfarin and low molecular weight heparin, but the results showed no statistically significant difference in the incidence of CRT.27–30 Additionally, considering the relatively low incidence of symptomatic CRT, which is approximately 5%, 2 the American College of Chest Physicians Consensus Guidelines in 2008 recommended against the routine use of prophylactic anticoagulation in cancer patients.31,32 However, in clinical practice, when patients develop symptomatic CRT, anticoagulation alone often yields poor outcomes. Further treatments, such as thrombolysis and thrombectomy, are frequently required. These additional interventions not only increase the psychological and economic burden on patients but also heighten the risk of bleeding, especially for patients with malignant tumors of the digestive system. This study aimed to address this gap by developing a predictive model, the TD score, to identify patients at high risk of CRT based on readily available clinical parameters.
Therefore, the primary objective of this study was to identify risk factors for CRT (catheter-related thrombosis) in colorectal cancer patients undergoing chemotherapy and to establish an evaluation model using big data. By identifying patients at high risk of CRT through this model, we aimed to implement prophylactic anticoagulation to mitigate the incidence of thrombosis. The TD score was developed using a combination of T stage and duration of intravenous catheter use, both of which were identified as independent predictors of CRT through multivariable logistic regression analysis. The model demonstrated moderate diagnostic performance, with areas under the receiver operating characteristic (ROC) curve of 0.732 in the training set and 0.749 in the test set. These results suggest thatdiag TD score could potentially stratify patients based on their risk of developing CRT. However, given the retrospective design of this study, these findings are hypothesis-generating. Any consideration of targeted prophylactic interventions based on this score must await validation in prospective studies designed to evaluate its clinical impact and safety.
The results of this study showed that for every additional day of central venous catheterization, the OR value for thrombosis was 0.998 (95% CI: 0.997 - 1.000). This observation warrants careful interpretation and may be influenced by several factors. First, survivor bias and competing risks could play a significant role. This is a critical consideration in our retrospective, observational design. Patients who experience early complications like CRT, infection, or catheter malfunction have their devices removed, censoring them from the “long-duration” group. Therefore, the cohort with long-term catheters represents a selected population that has already passed an early, higher-risk period. The model does not adjust for these competing risks (eg, death, elective removal post-chemotherapy), which may truncate follow-up before a CRT event occurs. Second, the risk of CRT is likely not linear over time. The highest risk period is probably immediately post-insertion due to acute vessel trauma and the foreign body reaction. Patients who do not develop thrombosis in this initial phase may have better endothelial adaptation or less pronounced inflammatory responses to the catheter material, leading to a plateau or even a decrease in hazard over time. Our logistic regression model, which uses total duration as a static variable, may not capture this time-varying hazard, unlike a survival analysis model like Cox regression. Third, it is plausible that the highest risk for CRT occurs shortly after catheter insertion due to vessel trauma and initial endothelial irritation, with the risk potentially plateauing or decreasing thereafter in patients who adapt to the catheter. Finally, the statistical significance of this finding, driven by a large sample size, should be interpreted with caution given the extremely small effect size, which may not be clinically meaningful.
This highlights the dominance of T stage as the primary risk factor in our model. The strong association between a higher T stage and increased CRT risk likely stems from the complex interplay between tumor biology and systemic hypercoagulability. Advanced local tumor invasion (higher T stage) signifies a greater tumor burden. It is well-established that larger and more invasive tumors release higher quantities of pro-coagulant factors, such as tissue factor (TF), cancer procoagulant, and inflammatory cytokines (eg, IL-1β, TNF-α, VEGF). These substances directly activate the coagulation cascade and platelets, while also damaging the vascular endothelium, fulfilling Virchow's triad. In colorectal cancer specifically, the local inflammatory milieu and direct vascular involvement by the tumor may be particularly pronounced. This provides a plausible biological pathway explaining why T stage emerged as the dominant predictive factor in our model, overshadowing other variables like catheter type. Future research could validate this by correlating TD scores with circulating levels of these biomarkers.
Furthermore, our model did not incorporate specific catheter-related characteristics such as type or insertion site, which have been implicated in other studies.1,3 While biologically plausible, these variables were not statistically significant predictors in our cohort. This could be due to a lack of variation in practice at our center (eg, a strong preference for a specific insertion site or catheter type) or insufficient statistical power to detect such associations. Future studies with larger, more heterogeneous populations should re-evaluate the contribution of these factors to CRT risk in CRC patients.
The TD score may provide a valuable tool for clinicians to identify high-risk patients who could benefit from prophylactic anticoagulation. By focusing on patients with a higher predicted risk of CRT, this approach could minimize the unnecessary use of anticoagulants, thereby reducing the risk of bleeding complications. Additionally, the TD score could serve as a foundation for further research aimed at refining risk stratification and improving patient outcomes.
Survival analysis revealed that patients with lower T stages had longer durations of non-CRT status. This finding suggests that tumor stage may play a role in the pathogenesis of CRT, possibly through mechanisms related to tumor burden and associated hypercoagulability. Further investigation into the underlying biological factors contributing to CRT in different tumor stages could provide additional insights for developing targeted therapies.
Limitations
The primary limitation of this study was its retrospective design, which may be susceptible to uncontrolled biases. Additionally, this study was conducted at a single center, limiting the generalizability of the findings. The validation was performed using a temporal split from the same institution. Future validation studies involving larger and more diverse patient cohorts from multiple, independent institutions are imperative to confirm the clinical utility and transportability of the TD score across different healthcare settings and to evaluate its performance more robustly across different TNM stages. Furthermore, prospective studies could help elucidate the impact of the TD score-guided interventions on clinical outcomes, including CRT incidence and bleeding complications.
Conclusions
In conclusion, the TD score serves as a preliminary tool for identifying colorectal cancer patients at elevated risk for catheter-related thrombosis. Prospective validation is required. If validated, employing the TD score for risk stratification could support more informed treatment decisions and improve the clinical management of CRT in this patient population. Future work should focus on validating the TD score in broader populations and exploring its potential integration into clinical practice. The TD score should not yet be used to guide prophylactic anticoagulation decisions would further strengthen the clinical boundary of the findings.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Guangdong Provincial Clinical Research Center for Digestive Diseases, Guangzhou Municipal Science and Technology Program key projects, National Natural Science Foundation of China, (grant number 2020B1111170004, 202002030348, 81301978).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
