Abstract
Venous thromboembolism (VTE) is a life-threatening postoperative complication of malignant tumors. We identified risk factors for postoperative VTE in patients undergoing radical resection of colorectal cancer (CRC) and constructed and validated a clinical prediction model. Clinical data of 982 patients undergoing radical resection of CRC from September 1, 2020, to March 31, 2022, in Ningxia Medical University General Hospital were analyzed retrospectively. Patients were randomly divided into training (
Introduction
Cancer patients are at high risk of venous thromboembolism (VTE), including deep vein thrombosis (DVT) and pulmonary embolism (PE). 1 The incidence of VTE in cancer patients is 4–7 times that of non-cancer patients. 2 Geerts et al reported that the risk of DVT in patients with malignant tumors was at least twice than that of patients receiving surgery for similar benign diseases, and the risk of fatal PE was more than 3 times. 3 VTE is also an important cause of postoperative death in cancer patients. 4 VTE increases the risk of death in cancer patients 2- to 6-fold and is the leading cause of death within 30 days after surgery. 5
Colorectal cancer (CRC) is a commonly occurring malignant tumor in the pelvis and abdomen. In 2018, its morbidity and mortality ranked third and second among malignant tumors globally and fifth among malignant tumors in Taiwan, with an annually increasing incidence rate. 6 Surgery is the standard treatment for patients with CRC. Iversen et al reported that the prevalence of postoperative DVT was about 20% in patients undergoing radical resection of CRC. 7 At the same time, the incidence of PE after colorectal surgery was 4 times than that associated with other surgeries. 8
VTE can be effectively prevented by drug or non-drug methods.9,10 The guidelines recommend that physical processes and anticoagulant therapy should be used for hospitalized patients with a high risk of VTE to prevent its occurrence. However, bleeding complications associated with anticoagulant therapy are common in cancer patients, 11 and the potential harm associated with thromboprophylaxis may outweigh the benefits in patients at lower risk for VTE. 12 Therefore, antithrombotic therapy should be stratified according to risk in order to benefit more high-risk patients. Stratification of VTE risk establishing a predictive model in clinical practice may be 1 solution.
Although many VTE scoring tools are used clinically, including the Caprini score, 13 which is suitable for surgical patients; the Khorana score 14 and COMPASS-CAT, 15 which is ideal for outpatient cancer patients; the PROTECHT score, 16 which is suitable for chemotherapy patients; and the Padua score, 17 which is perfect for hospital inpatients. Still none of the available scoring methods are specific to cancer surgery patients. Currently, no simple, rapid, and effective VTE risk assessment tool is available for Chinese patients undergoing CRC surgery. Therefore, this study aimed to retrospectively analyze the potential risk factors for postoperative VTE in patients undergoing radical resection for CRC, evaluate the effects on morbidity and mortality, and construct and validate a predictive model for VTE risk.
Methods
Study Design
This retrospective, single-center, cross-sectional study collected the demographic and clinical data of patients who underwent radical resection of CRC in the General Hospital of Ningxia Medical University from September 2020 to March 2022. The follow-up time was 6 months after the operation. The inclusion criteria were as follows: (1) patients diagnosed with CRC in Ningxia Medical University General Hospital who underwent surgical treatment, (2) complete clinical data, and (3) VTE patients must have evidence of imaging diagnosis. The exclusion criteria were the following: (1) patients with severe liver and kidney diseases, (2) combined with other malignant tumors, (3) combined with hematologic diseases, and (4) patients who had been diagnosed with VTE on admission.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the Ethics Committee of Ningxia Medical University General Hospital (KYLL-2021-1046). Written informed consent was obtained from all patients.
Data Collection
Patients’potentially related factors of VTE were collected through the hospital electronic medical information system (HIS), including 6 main factors: (1) patient factors: age, sex, BMI, hypertension, diabetes, coronary artery disease, chronic obstructive pulmonary disease (COPD), cardiovascular and cerebrovascular accidents, atrial fibrillation, varicose veins of lower extremities, history of venous thrombosis, smoking history, thoracoabdominal pelvic effusion, history of blood transfusion in the past 3 months before surgery, and history of surgery in the past 3 months before CRC operation; (2) postoperative laboratory parameters: D-dimer, fibrinogen level, platelet count, hemoglobin, and white blood cell count; (3) tumor factors: tumor stage, distant metastasis, and lymph node metastasis; (4) surgical factors: ASA grade, operation time, intraoperative blood transfusion, intraoperative blood loss, surgical method, and postoperative admission to ICU; (5) postoperative complications: infection, hypoxemia, hypoalbuminemia, anastomotic leakage, intestinal obstruction, and heart failure; (6) treatment-related factors: preoperative neoadjuvant chemotherapy, postoperative chemotherapy, and postoperative radiotherapy; and (7) postoperative length of stay.
Primary Outcome Measures
The primary outcome measure was VTE diagnosed by imaging, including DVT and PE. All surgical patients underwent preoperative lower extremity venous color Doppler ultrasound examination to exclude preoperative VTE. Patients with lower extremity pain, swelling, and skin color changes underwent a Doppler ultrasound examination immediately to diagnose lighter extremity vein color. Postoperatively, occurrence of unexplained chest tightness, chest pain, hemoptysis, dyspnea, syncope, or decreased oxygen saturation was immediately examined by CT pulmonary angiography (CTPA) to confirm possible PE. When symptoms such as abdominal pain, abdominal distension, constipation, or vomiting occur postoperatively, an enhanced CT examination of the abdomen was performed immediately to determine whether or not intestinal obstruction was present. Hypoxemia was defined as partial arterial pressure of oxygen < 60 mmHg or oxygen saturation < 90%.
Statistical Analysis
All data analysis was performed using SPSS 26.0 software (IBM SPSS, Armonk, NY, USA) and R Studio 4.1.2 software. Among all included patients, 70% of samples were randomly selected for model establishment, and the remaining 30% were used for model validation. The best cut-off value was calculated for each continuous variable (corresponding to the maximum Youden index), and continuous variables were converted into categorical or rank variables, expressed in frequency and percentage using the chi-square test or non-parametric test between the 2 cohorts. All statistical tests were 2-sided, and
Univariate and multivariate logistic regression analysis were performed using SPSS 26.0 software to identify independent risk factors for VTE. Significant variables with
The accuracy of the risk prediction model was evaluated using multiple validation methods that incorporated data of the training set and validation dataset. The discrimination, calibration, and clinical applicability of the model were evaluated using the “pROC” package, “rms” package, and “rmda” package in R software. The receiver operating characteristic (ROC) curve was plotted, and the area under the curve (AUC) was calculated to evaluate the predictive ability of the model to distinguish true positives from false positives. Calibration diagrams were drawn, and Hosmer–Lemeshow (H-L) tests were performed for calibration. Finally, the clinical application value of the model was evaluated using the clinical decision curve (DCA).
Results
Sample Characteristics
This study collected the data of 982 patients who had undergone radical resection of CRC in Ningxia Medical University General Hospital from September 1, 2020, to March 31, 2022. Patients with severe liver and kidney diseases, malignant tumors, or hematologic diseases and patients who had been diagnosed with VTE on admission were excluded (

Flow diagram of study design. BMI, body mass index.
Baseline Patient Characteristic.

BMI, body mass index; COPD, chronic obstructive pulmonary disease; ASA, American Society of Anesthesiologists; VTE, venous thrombosis; DVT, deep venous thrombosis; PE, pulmonary embolism.
Establishment of VTE Prediction Model
The results of univariate and multivariate logistic regression analysis are shown in Table 2. Univariate analysis showed that age (
Results of Univariate and Multivariate Analyses in the Development Group.

BMI, body mass index; COPD, chronic obstructive pulmonary disease; ASA, American Society of Anesthesiologists.
Clinical Predictive Model Construction
Based on the results of multivariate analysis, the above 7 independent risk factors were selected to establish a clinical predictive model. We constructed a nomogram to predict the occurrence of VTE after radical resection of CRC. The nomogram was named CRSPOT nomogram representing “colorectal cancer surgery postoperative thromboembolism” (Figure 2a). The higher the total score for each patient, the greater the risk of postoperative VTE. As shown in Figure 2b, a patient with a BMI of 28.65 kg/m2 and no lower extremity varicose veins who underwent radical resection of CRC had a postoperative D-dimer level of 4.53 μg/mL and a postoperative hemoglobin level of 8.3 g/L. The time was 6 h, postoperative hypoxemia occurred, and postoperative intestinal obstruction did not occur. The probability of postoperative VTE in this patient was 90.4% (see Figure 2b), and the patient developed venous thrombosis on the second day after surgery.
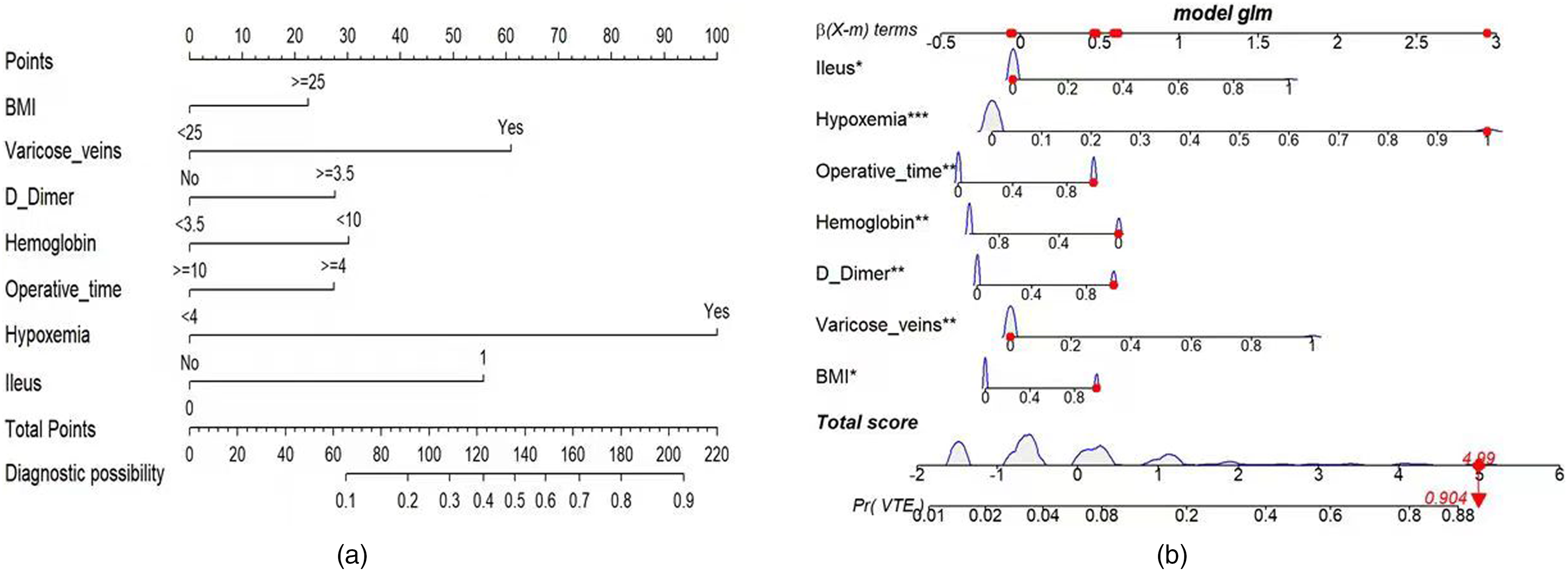
(a) Risk factors of BMI, varicose veins, D-dimer, hemoglobin, operative time, hypoxemia, and ileus for nomogram prediction model. (b) Dynamic nomogram used as an example. The significance of the asterisks beside each variable in part b represents importance of all the risk factors.
Prediction and Calibration Performance of the Nomogram Predictive Model
The ROC curve was used to judge the predictive ability and recognition performance of the nomogram model, as shown in Figure 3. The area under the ROC curve (AUC) is 0.826, indicating that the nomogram model has good recognition ability (the validation set AUC is 0.821). The calibration plot of the nomogram shows that the calibration curve is close to the ideal angle, indicating good agreement between prediction and observation, as shown in Figure 4. We obtained an excellent calibration map, and the Hosmer–Lemeshow test showed that the model fit well (

The receiver operating characteristic (ROC) curves for (a) development group and (b) validation group.
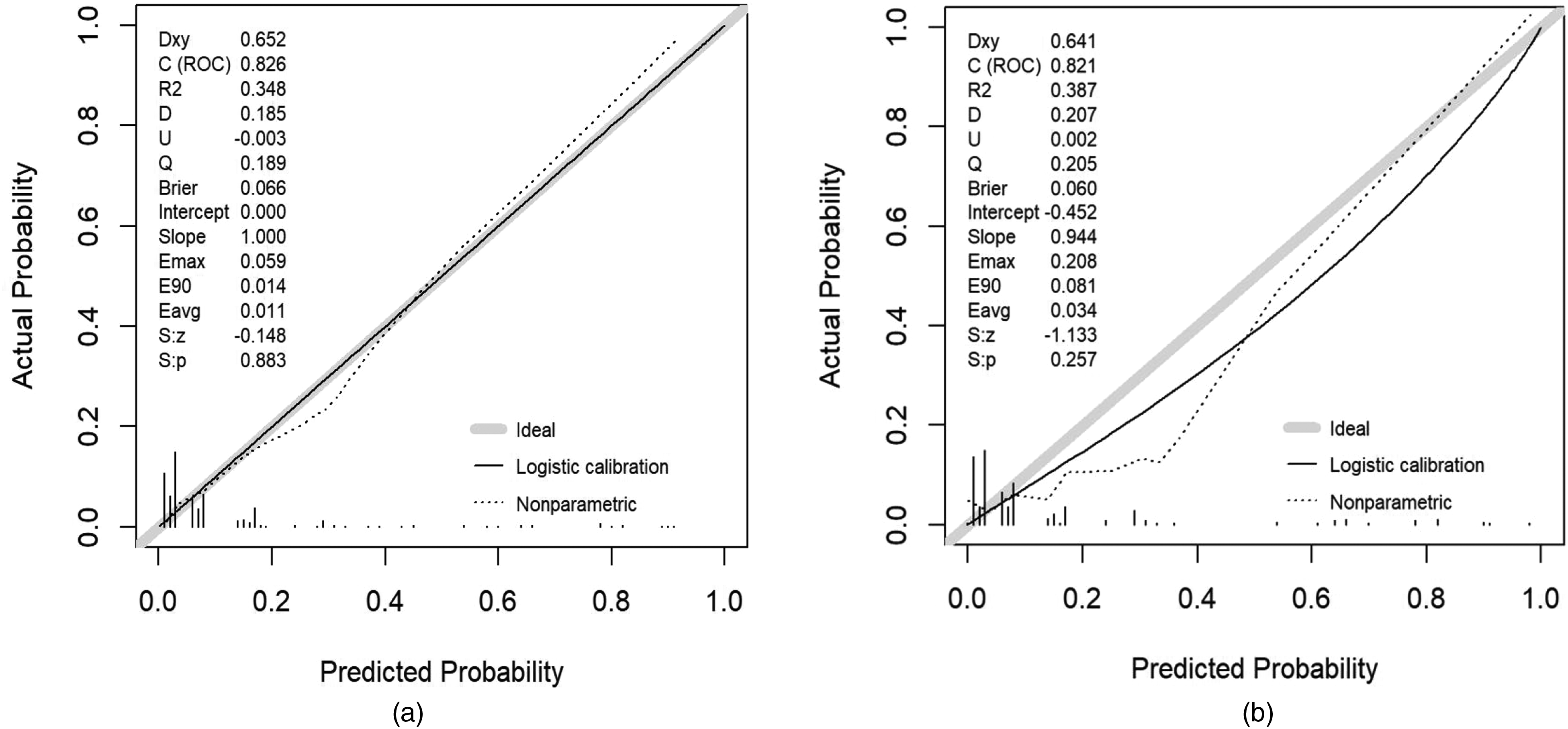
Calibration curve plot in each group. (a) The development group and (b) the validation group.
Clinical Application Value of the Nomogram Predictive Model
The decision analysis curve (DCA) of the nomogram model is shown in Figure 5. The abscissa of the DCA represents the threshold probability. For example, the likelihood of a patient diagnosis is recorded as Pi; when Pi reaches a certain threshold (Pt), it is defined as positive, and treatment measures are indicated. Currently, some patients are shown to benefit from the treatment (benefits), and some are shown to be harmed by the treatment (consequences). After subtracting the benefits from the risks, the vertical axis represents the patient's net benefit (net benefit, NB). The curved line in the figure represents the clinical predictive model. In addition, 2 extreme cases are shown. The horizontal line indicates that all samples are negative (Pi < Pt), no one was treated, and the net benefit is 0. A slash suggests that all models were positive, all received treatment, and the net benefit is a backslash with a negative slope. Setting the prediction probability threshold at 10.0% and 8.5% in the training set and validation set resulted in net benefits of 15.0% and 17.0%, respectively, which indicates the excellent potential of the nomogram model for clinical decision-making.

Decision curve analysis for the nomogram: (a) The development group and (b) the validation group.
Discussion
The present study reviewed relevant literature and recorded our clinical observations, including risk factors associated with VTE in patients undergoing radical resection of CRC, and established a postoperative VTE prediction model comprising 7 predictors of VTE. Increased BMI, lower extremity varicose veins, low postoperative hemoglobin and D-dimer level, as well as operative time over 4 h, postoperative hypoxemia, and posterior postoperative ileus are independent predictors of postoperative VTE in patients with CRC. These 7 factors were used to construct the risk prediction model for postoperative VTE in patients undergoing radical resection of CRC and constitute the CRSPOT nomogram.
VTE is a severe complication in patients with malignant tumors undergoing surgery, increasing the risk of postoperative death. 18 CRC is a common abdominal malignancy, and the prevalence of postoperative DVT is about 20% in patients undergoing radical resection of CRC. 7 Postoperative VTE in patients with CRC may be the result of abdominal trauma caused by surgery and the systemic hypercoagulable state caused by cancer itself. 19 The incidence of postoperative VTE in patients undergoing radical resection for CRC was 10.6% in the present study. However, the incidence of VTE after CRC surgery in South Korea and Hong Kong was only shown to be between 0.18% and 0.85%.20,21 The discrepancy in the incidence of VTE between the present study and the Korean/Hong Kong studies may be attributed in part to our inclusion of asymptomatic VTE patients in the present study and the improvement in precautions in the other studies. Varicose veins increase the risk of venous thrombosis due to venous blockage that causes blood vessels to bend and swell, obstructing blood return to the lower extremities. Yasui et al 22 studied the relationship between varicose veins and the risk of VTE in cancer patients but reported no conclusive findings. However, in the Vienna Cancer and Thrombosis Study (CATS), cancer patients with varicose veins had a 2-fold increased risk of VTE compared with patients without varicose veins. 23 In the present study, lower extremity varicose veins were an independent risk factor for postoperative VTE in patients with CRC and were included in the predictive model. Therefore, taking reasonable preventive measures to avoid VTE is essential for cancer patients with preoperative varicose veins.
D-dimer is a fibrin-related marker, and elevated levels often indicate systemic coagulation activation and fibrinolysis. Studies suggest that high concentrations of plasma fibrin-related markers reflect an increased risk of thrombosis. 24 The CATS study confirmed that D-dimer is an independent risk factor for VTE 25 and has a high predictive value. Cihan Ay et al added this parameter to the Khorana model to form the expanded “Vienna CATS Score” (D-dimer level was greater than 1.44 μg/mL). 26 However, the diagnostic threshold of D-dimer used to predict postoperative VTE in cancer patients is considered to be controversial. 27 Studies have shown that a postoperative D-dimer level of 1 μg/mL is an independent risk factor for VTE in urological tumors. 28 Intravenous ultrasonography in patients with D-dimer levels exceeding 3.3 μg/mL after brain tumor surgery may increase the detection rate of VTE. 29 However, the diagnostic threshold of D-dimer in patients with CRC has not yet been reported. In the present study, according to the ROC curve analysis, the optimal cut-off value of D-dimer after radical resection of CRC was 3.5 μg/ml (sensitivity 61.3%, specificity 76.5%) and was included in the final model. The cut-off value of D-dimer determined in the present study provides a reference for predicting postoperative VTE in patients undergoing radical resection of CRC to a certain extent.
Venous stasis is well recognized as a major factor in thrombosis. Prolonged operative time and general anesthesia induce vasodilation, which leads to stagnation of venous blood, thereby inducing DVT. 30 A large retrospective study showed that longer operative time was independently associated with an increased risk of postoperative VTE. For each additional hour of surgery, the risk of VTE increases by 15%. 31 Schlick et al 32 and Yamashita et al 33 showed in separate studies that operative times over 4 h and operative times over 5 h, respectively, were independent predictors of postoperative VTE. The present study also confirmed that an operative time exceeding 4 h is an independent risk factor for postoperative VTE in patients undergoing radical resection of CRC.
In patients who are overweight and obese, the risk of thrombosis may be tripled.34,35 The health status of obese patients is usually complicated by dyslipidemia, hyperlipidemia, and increased blood viscosity. At the same time, a higher BMI means that performing laparoscopic surgery becomes increasingly difficult, resulting in prolonged surgery and anesthesia time, and causing long-term muscle relaxation and local compression. The stasis of blood flow induced by surgery further increases the risk of VTE in the perioperative period. Studies have shown that BMI ≥ 25 kg/m2 is independently associated with a high risk of VTE,32,36 which was also confirmed in the present study.
To the best of our knowledge, previous studies have not reported postoperative hypoxemia as a predictor of PE. The clinical symptoms of patients with PE may manifest as dyspnea and hypoxemia in about 80%-90% of those affected, the severity of which is related to the size of the embolism. 37 The occurrence of hypoxemia in patients with PE is mainly associated with pulmonary vascular obstruction, and the decrease or loss of blood perfusion in the obstructed area leads to ventilation/blood loss balance and hypoxemia; in addition, pulmonary hypertension, decreased pulmonary blood flow, and decreased heart blood volume and blood pressure may further reduce blood volume in the pulmonary circulation and aggravate hypoxemia in patients with PE. In the present study, the incidence of hypoxemia in VTE patients after radical resection of CRC was about 45.2%. Cancer tissue is in a state of coagulation, inflammation, and hypoxia, prompting cancer proliferation genes to produce various substances to form a thrombus. Tissue factors (TF) produced by cancer cells are the starting point of the coagulation reaction that initiates cancer-associated thrombosis (CAT). 38 The expression of TF and its vesicle release are regulated by the tumor microenvironment (such as hypoxia), so the hypoxic environment caused by hypoxemia will accelerate the formation of CAT. 39 Therefore, the present study used postoperative hypoxemia as a screening index for postoperative VTE, confirming it to be a high-risk factor for postoperative VTE in patients undergoing radical resection of CRC. More extensive prospective controlled study is needed to verify the relationship between postoperative hypoxemia and postoperative VTE.
A previous study confirmed that postoperative ileus is a risk factor for postoperative VTE in patients undergoing orthopedic surgery; specifically, a 5.5-fold increased relative risk for DVT and a 19.5-fold higher risk for PE were found in patients with postoperative ileus. 40 That study also showed that pain, abdominal distension, increased abdominal pressure, and bed rest may induce postoperative VTE in patients with ileus. However, until the present study, no studies have confirmed the relationship between postoperative ileus and postoperative VTE in patients with malignant tumors. The present study showed that postoperative ileus is a prevalent postoperative complication in patients undergoing radical resection of CRC, a new concept for predicting postoperative VTE in this patient population.
Strengths and Limitations
In the present study, the clinical data of patients undergoing radical resection of CRC were screened to identify risk factors for postoperative VTE, supporting the establishment of the CRSPOT nomogram for postoperative VTE prediction. This visual, personalized model provides clinicians with an easy and intuitive practical predictive tool. The model is easy to use in the clinical environment and can effectively help clinicians identify high-risk patients with VTE after radical resection of colorectal cancer in the early stage and implement antithrombotic strategies early, which has a significant clinical value and social significance for saving medical resources.
Nevertheless, this study also has several limitations. First, the retrospective analysis used in this study cannot rule out the potential selection bias of retrospective research, and the cross-sectional design does not allow causal inferences. Therefore, the reliability and stability of the nomogram need to be continuously verified. Second, data reliability and availability are limited, resulting in some factors not being included, such as P-selectin, hyperlipidemia, Crohn's disease, etc. Third, the sample size of this study was small, and all patient data were retrieved from the same hospital, which may not allow results to be generalized to other populations. The universality of the model needs to be verified. Fourth, patients with no symptoms of VTE were not routinely screened after surgery, and the occurrence of VTE may be underestimated. Fifth, this study did not undergo external validation, and further prospective multicenter clinical studies are needed to demonstrate its clinical value.
Conclusion
Independent predictors of postoperative VTE in patients undergoing radical resection of CRC include elevated BMI, varicose veins of lower extremities, low postoperative hemoglobin, increased postoperative D-dimer, operative time greater than 4 h, postoperative low oxygen, and postoperative ileus. The CRSPOT nomogram was constructed using these 7 factors to predict postoperative VTE in patients undergoing radical resection of CRC. The model is verified to have good discrimination, calibration, and clinical applicability. The visualized and personalized predictive model provides clinicians with a simple, intuitive tool for early detection and identification of postoperative VTE high-risk groups. Applying the model may help clinicians take early and effective measures to prevent VTE and improve postoperative outcomes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
