Abstract
Background
Cancer-associated atrial fibrillation (AF) is increasingly common, posing dual risks of thrombosis and bleeding. Long-term oral anticoagulation is often limited in these patients due to drug interactions, thrombocytopenia, and treatment-related bleeding. Left atrial appendage occlusion (LAAO) offers a non-pharmacological alternative for stroke prevention, yet its optimal use in cancer remains uncertain.
Methods
We conducted a narrative review of studies from 2009 to October 2025 across PubMed, Embase, and Google Scholar, including randomized trials, observational studies, registries, clinical guidelines, and expert statements addressing LAAO in AF patients, with emphasis on cancer and cardio-oncology populations. Case reports, non-English studies, abstracts without full text, and non-oncologic studies were excluded.
Results
Recent evidence suggests LAAO achieves high procedural success in selected cancer patients and reduces long-term bleeding compared with oral anticoagulants. Post-procedure management typically involves individualized anticoagulant or antiplatelet regimens adjusted for bleeding risk and hematologic status. However, short-term complications may be more frequent, likely reflecting cytopenias and patient frailty.
Conclusion
LAAO represents a promising stroke-prevention strategy for cancer-related AF when anticoagulation is unsafe or ineffective. Optimal outcomes require multidisciplinary evaluation, timing aligned with cancer status, and tailored post-implant antithrombotic therapy. Prospective studies are still needed to define patient selection, procedural timing, and post-procedural management in this high-risk population.
Keywords
Introduction
Mortality rate in cancer patients has been decreased in the past decades as a result of advances in screening, diagnosis, and therapy. 1 As the life expectancy of cancer patients and survivors increases, cardiovascular complications are becoming a major cause of morbidity and death in both the short-term and long-term. Atrial fibrillation (AF) is the most prevalent cardiac arrhythmia, especially in older adults, and it causes many adverse outcomes like stroke, heart failure, and hospitalization.2,3 Cancer patients have a high risk of developing AF compared with those without cancer, particularly in the first 3 months of cancer diagnosis.4,5 The risk of developing AF in cancer patients varies according to cancer type. As compared to people without cancer, patients with colorectal cancer have 54% increased risk of developing AF compared with individuals without cancer, and breast cancer patients have double the risk of AF development. 6
Many studies demonstrate there is a bidirectional relationship between cancer and AF as a result of shared common risk factors such as obesity, aging, smoking, and diabetes mellitus and the underlying pathophysiology of the two conditions.1,3 Treatment approaches for cancer, like chemotherapy, radiotherapy, and antineoplastic drugs, increase the risk of AF. 4 AF treatment in cancer patients is more challenging, particularly in management with anticoagulants. 1 Cancer patients have an increased risk of bleeding and thrombosis compared with the general population. 7 Anticoagulative drugs interact with cancer therapy and increase bleeding risk, enhanced thrombocytopenia, and coagulopathy.
Left atrial appendage (LAA) is a key site of thrombosis in non-valvular AF (NVAF). LAA is described by surgeons as “the most lethal human attachment” due to the ease of damaging during surgery. 8 Left atrial appendage occlusion (LAAO) is an alternative stroke prevention for patients who can’t tolerate anticoagulants. LAAO is indicated for patients with elevated stroke and contraindications of anticoagulants due to bleeding risk, and studies show LAAO outcomes in cancer patients are favorable. 9
We aim to provide a comprehensive overview of LAAO in cancer patients who developed AF as an alternative stroke prevention. The study focuses on patients who are candidates for LAAO, addresses the optimal time for the procedure to be done, reviews how antithrombotic therapy is managed before and after LAAO, synthesizes current evidence, highlights knowledge gaps, and proposes clinical and research pathways for improved integration of LAAO in cardio-oncology care.
Pathophysiology and Clinical Context
Cancer and AF share common risk factors, including aging, smoking, diabetes mellitus, and obesity. Recent studies demonstrate the association between the two conditions. AF incidence in cancer patients is 17.4 per 1000 person-years and 3.7 per 1000 person-years in patients without cancer. 7
Mechanisms Linking Cancer and Atrial Fibrillation
There are many mechanisms linking cancer and AF. The biology of these conditions, including inflammations, represents this association. Increased acute phase reactant proteins like c-reactive protein, high WBCs, and ceruloplasmin, and the production of chemokines and cytokines like interleukins 1 and 6. 4 Cancer treatment also increases the risk of AF development; the cytotoxicity of chemotherapy and radiotherapy and their potential to increase the risk of infection and sepsis will enhance AF.
Targeted therapy in cancer treatment, such as tyrosine kinase inhibitors, will promote AF due to inhibition of other tyrosine kinases in cardiac myocytes, and the use of immune checkpoint inhibitors increased the cardiovascular complications. 7 Some studies show ibrutinib will inhibit C-terminal Src kinase and lead to left atrial enlargement, fibrosis, and inflammation and, by production of ROS, lead to AF.10,11 (Figure 1) presents the pathophysiological link between cancer and AF.

Mechanisms linking cancer and atrial fibrillation (AF). Cancer-related inflammation amplifies thromboembolic and cardiovascular risk in cancer patients. Created in BioRender. Mahafdah, B. (2026) https://BioRender.com/9r5hj6f.
Thromboembolic and Bleeding Risk Profiles
Cancer affects hemostatic balance. Cancer increases the risk of the development of thrombosis as a result of over-expression of tissue factor driven by oncogenes, including KRAS, MET, or EGFR, and inhibitions of tumor suppressor genes such as TP53 and PTEN. Furthermore, venous stasis resulted from immobilization and compression by the tumor itself. 12 The risk of venous thromboembolism (VTE) is 12 times higher in cancer patients than in the general population. Additionally, chemotherapy and targeted therapy increase the risk of VTE 23 times. 13
Bleeding occurs in cancer patients as a result of multiple aspects, including long-term anticoagulative medications that are indicated for stroke prevention in AF and VTE and some dynamic factors such as thrombocytopenia and kidney function. 14
Assessment tools for stroke depend on the CHA2DS2-VASc score and HAS-BLED score, other important factors like LAA, left atrial size and function, AF itself (permanent/temporary), and the presence of other cardiac disease. 8 For balancing stroke prevention and bleeding risk, individualized assessment and management are important, and consider alternative treatment rather than anticoagulants.
A recent study showed the CHA-2DS2VASc score can predict ischemic stroke in patients with AF and certain types of cancer similarly to AF patients without cancer. Additionally, it found the HAS-BLED score in cancer patients is suboptimal because it may lead to over- or underestimating bleeding risk. 15 This finding supports another study that found the existing bleeding risk models cannot predict bleeding risk in patients with cancer, so there is no valid bleeding risk model for patients with cancer. 16 Another study demonstrated a strong linear relationship between CHA-2DS2VASc score and odds of stroke in AF patients with and without cancer. Notably, the odds of stroke were higher in patients with cancer than those without cancer. 17
Principles of Left Atrial Appendage Occlusion
AF increases the risk for non-valvular thrombosis and embolic stroke, which requires long-term oral anticoagulants (OACs), such as direct oral anticoagulants (DOACs) and vitamin K antagonists. In patients who are suffering from bleeding or are at high risk for bleeding, like cancer patients, these anticoagulants are used. So, an alternative interventional technique should be considered. 18
Device-Based Thromboembolic Prevention
Mechanical blockage of the (LAA) using a permanently implanted device may be an effective alternative for OACs. A number of percutaneous devices have been proposed to help in LAAO. There are many common devices used in LAAO, including Watchman FLX, Amplatzer Amulent, and LAmbre. A systematic review and meta-analysis show that the Watchman 2.5 and Amplatzer Amulet devices for LAAO in patients with NVAF do not significantly differ in safety or efficacy outcomes. 19
Procedural Overview
LAAO is carried out under general anesthesia and is guided by transesophageal echocardiogram (TEE). It is performed by femoral venous puncture under ultrasound guidance to minimize vascular complications, then transseptal puncture, is an important step to access the left atrium and implant the LAAO device. 18 Peri-procedural antibiotic prophylaxis to eliminate the potential source of sepsis and infective endocarditis. 20 TEE or CCTA should be done to assess for potential peri-device leaks. 21 However, in contemporary practice, many centers perform LAAO under monitored anesthesia care or nurse-led conscious sedation, frequently using intracardiac echocardiography (ICE) instead of TEE. These approaches reduce the need for general anesthesia and endotracheal intubation and may be particularly advantageous in frail patients or those with active malignancy who are at higher risk of anesthesia-related complications.18,22
Evidence in General AF Population
Randomized PROTECT-AF and PREVAIL clinical trials demonstrated LAAO provides stroke protection comparable to VKA. LAAO significantly reduced severe bleeding, hemorrhagic stroke, disabling/fatal stroke, cardiovascular (CV) death, and all-cause death, offering stroke protection.18,19
LAAO in Cancer-Associated Atrial Fibrillation: Clinical Evidence and Insights
LAAO represents a pivotal non-pharmacologic strategy for stroke prevention in AF patients who cannot tolerate long-term anticoagulation. The application of LAAO in the cardio-oncology population is critical but yet understudied. Patients who have cancer associated with AF are at high risk of both thromboembolism and bleeding due to malignancy itself and its treatments. Recent emerging data from observational studies and registries provide crucial insights in feasibility, safety and efficacy of LAAO in these populations, but the evidence base is still evolving.23–27 (Figure 2)

Left atrial appendage occlusion (LAAO) provides a non-pharmacologic stroke prevention option for atrial fibrillation patients, especially in the cardio-oncology population at high risk of bleeding or thromboembolism. Created in BioRender. Mahafdah, B. (2026) https://BioRender.com/6nh8iuv.
Epidemiology and Registry Data
Large U.S. database studies (NIS/NRD) report that approximately 2%–3% of contemporary LAAO hospitalizations involve patients with cancer.25,28 In a National Cardiovascular Data Registry (NCDR) analysis of over 130 000 LAAO procedures, approximate procedural success (∼94-98.5%) and in-hospital mortality below 1%.29,30 Recent observational studies and registry data have begun to fill this gap. NCDR LAAO and other Reports from large-volume centers and national registries suggested that successful device implantation procedures’ success rates are comparable to those in the non-cancer population, often exceeding 95%. 26 There is an increased need for prospective, multicenter, cancer-specific registries that would systematically collect data on cancer type, stage, ongoing therapies (chemotherapy, immunotherapy, radiation), hematologic parameters (platelet counts), and post-implant antithrombotic strategy and do stratified analyses to elucidate which subgroups of cancer patients derive the most benefit and which are at highest risk thus guiding more personalized clinical decision-making. 26 Several ongoing registries, such as the Amulet IDE Cancer Sub-analysis and the Cardio-Oncology LAAO Registry (NCT05764329) aim to prospectively evaluate outcomes in patients with active malignancy. These efforts aim and are expected to bridge current evidence gaps via systematically capturing cancer-specific variables (tumor type, treatment phase, and hematologic status) to guide more tailored procedural and post-procedural approaches. Despite the higher periprocedural risk, LAAO is still a promising strategy to decrease long-term complications in appropriately selected cancer patients. For procedure-related risk, active malignancy increases prothrombotic potential thus increases procedural risks with elevated risk of device-related thrombus (DRT) post-implantation.26,31 However, a higher incidence of short-term (in-hospital or 30-day) complications, slightly longer length of stay without increase in hospital mortality was highlighted in these studies.25,32,33 This higher risk is due to intrinsic vulnerability and high comorbid burden of cancer patients rather than technical failures. In terms of mortality and comorbidity, higher all-cause mortality is observed after LAAO in cancer cohorts which is attributed to the underlying malignancy rather than device failure or ischemic events. The more frequently reported complications were major bleeding (including pericardial effusion that needed intervention), vascular access site complications, and acute kidney injury.25,32,33 Factors like pre-existing anemia, thrombocytopenia, and cancer-related frailty amplify the periprocedural risk even if it was technically feasible. 11 Cancer patients are also at higher risk of impaired recovery, complications from anesthesia and the invasive nature of the procedure itself due to their cachexia and frailty. These factors must be weighed against the potential long-term benefits in decision making. Higgins et al, in their retrospective cohort study using real-world data from the NCDR, reported low long-term stroke rates but high competing mortality in an elderly, high-risk Medicare population undergoing LAAO. 34 In comparison, cancer patients undergoing LAAO tend to experience higher early mortality and similar or slightly elevated stroke rates, depending on cancer type, stage, and comorbidities.25,32,33 Thus, careful pre-procedure optimization (blood counts, renal status) and imaging-guided technique are essential.
Comparative Outcomes
Despite the higher periprocedural risk, LAAO is still a promising strategy to decrease long-term complications in appropriately selected cancer patients. Zhang et al, in their retrospective observational cohort study using NIS and NRD data (2016-2019) reported that In carefully selected patients with cancer, the rates of stroke or systemic embolism were similar to non-cancer patients over short to mid-term follow up. 25 This suggests that mechanically occluding the LAA provides effective long-term thromboembolic protection thus achieving the primary therapeutic goal even in hypercoagulable cancer patients. Anticoagulation and procedural outcomes are further complicated by the pro-thrombotic and pro-hemorrhagic milieu of malignancy which are driven by tumor-related cytokine release, endothelial activation, and chemotherapy-induced cytopenias. 26 LAAO in cancer patients can markedly lower the risk of major bleeding events making its utilization for this population strongly justified. 33 Gastrointestinal malignancy with history of intracranial hemorrhage or severe chemotherapy-induced thrombocytopenia patients have absolute or high relative contraindications for long-term oral anticoagulation. LAAO offers a strong alternative that circumvents the risk of hemorrhage of OACs in these patients. This enables cancer patients to discontinue OACs thus preventing life-threatening bleeds and facilitating uninterrupted cancer treatment. 26 For procedure-related risk, active malignancy increases prothrombotic potential thus increases procedural risks with elevated risk of DRT post-implantation.26,31 Also cancer-related coagulopathy and treatment-induced thrombocytopenia increase the risk of periprocedural bleeding. Cancer patients are also at higher risk of impaired recovery, complications from anesthesia and the invasive nature of the procedure itself due to their cachexia and frailty. These factors must be weighted against the potential long-term benefits in decision making. In terms of mortality and comorbidity, higher all-cause mortality is observed after LAAO in cancer cohorts which is attributed to the underlying malignancy rather than device failure or ischemic events. Higgins et al, in their retrospective cohort study using real-world data from NCDR, reported low long-term stroke rates but high competing mortality in an elderly, high-risk Medicare population undergoing LAAO. 35 In comparison, cancer patients undergoing LAAO tend to experience higher early mortality and similar or slightly elevated stroke rates, depending on cancer type, stage, and comorbidities.25,32,33 Thus, careful pre-procedure optimization (blood counts, renal status) and imaging-guided technique are essential.
Special Considerations in Active Cancer
The decision to perform LAAO in active cancer patients is highly individualized and complicated. The type, stage, and prognosis of the malignancy highly affect the decision. Patients with hematologic malignancies (eg, acute leukemia, multiple myeloma) have hazardous procedural risks of both oral anticoagulation and LAAO due to profound and prolonged periods of thrombocytopenia. While solid tumors patients (particularly adenocarcinomas of the pancreas or lung) are at very high risk of thromboembolism (Trousseau's syndrome). The potential benefit of stroke prevention must outweigh the immediate procedural risk. Patients with a life expectancy of less than one year are unlikely to gain significant advantage from LAAO.22,36 The coordination and collaboration between the cardiology/electrophysiology team and the oncology team is very important. Timing of the procedure must be planned strategically to align with the cancer treatment schedule. LAAO should be scheduled when chemotherapy is paused or just before starting a new cycle when platelets and neutrophils count recovered from their nadir minimizing bleeding and infection risk. Cardio-oncology recommendations provide some practical platelet thresholds for antiplatelet therapy (eg, dual antiplatelet therapy is generally acceptable at ≥30-50 × 109/L, while potent P2Y12 inhibitors should be avoided at lower counts) and are consistent with hematology guidelines for transfusion and supportive management of thrombocytopenia.37–40 To ensure a cohesive and safe management plan, the timing must account for other planned interventions like major surgery or radiation therapy.
Left atrial thrombus is one of the known complications of cardiac amyloidosis (CA). 41 Therefore, the risk of thromboembolic stroke in CA is challenging. A multicenter retrospective study has indicated that LAAO is effective and feasible in CA patients as an alternative to anticoagulants with the same success rate and complications as in non-CA patients. 42 Another study demonstrated there is no difference in bleeding/stroke-related readmissions between LAAO patients with CA and those without CA. 43
In summary, existing registry and observational evidence indicates stroke-prevention benefits in LAAO are comparable to those seen in non-cancer populations. However, higher short-term complication rates underscore the need for prospective, cancer-specific registries and individualized procedural planning.25–27,32,33
Definition of Endpoints and Imaging Follow-Up in Available Studies
In observational studies and registries assessing LAAO in cancer-associated AF, clinical objectives were delineated with significant diversity, leading to outcome heterogeneity. Stroke and systemic embolism were often characterized by established neurological and vascular criteria, including ischemic stroke validated clinically and/or radiographically, and non-central systemic embolism necessitating imaging or intervention.33,44 Most of the time, major bleeding was defined by the International Society on Thrombosis and Haemostasias (ISTH) or BARC criteria. This included fatal bleeding, symptomatic bleeding in important places like the gastrointestinal tract or central nervous system, or bleeding that needed a transfusion or hospitalization. However, some registries used administrative coding instead of clinical events that had been decided upon.25,33,37 DRT was consistently characterized as echogenic or hypoattenuating thrombus affixed to the device surface on subsequent imaging; however, the criteria for clinical significance and therapy varied across investigations.45,46
There were also significant differences in the follow-up imaging methods across groups, which made comparisons further harder. Most studies required TEE or CT around 45 days after implantation to check for DRT and peri-device leak (PDL). Some studies, on the other hand, did initial imaging at 60 to 90 days or only did imaging based on symptoms or bleeding risk.21,44,45 Peri-device leak was generally characterized by residual flow above 3–5 mm on color Doppler or CT; however, reporting criteria and clinical interpretations varied, with some studies deeming minor leaks clinically inconsequential while others included them as composite safety endpoints.21,46 Long-term imaging surveillance beyond the initial 3–6 months was applied inconsistently, especially in cancer cohorts with restricted life expectancy or competing mortality, highlighting the necessity for standardized endpoint definitions and imaging schedules to enhance interpretability across studies.25,33,46
Patient Selection: “who” Should Undergo LAAO
In the cancer population, rigorous assessment of thromboembolic risk, bleeding risk, life expectancy, and procedural feasibility is required to identify the ideal candidate for LAAO. The key consideration is identifying those whose expected long-term stroke prevention benefits clearly surpass the risks of the procedure and any limitations imposed by reduced life expectancy.22,36
Clinical Profiles Favoring LAAO
Patients who meet the following criteria are the strongest candidates for LAAO: Patients with high thromboembolic risk with oral anticoagulation contraindication (primary indication is no NVAF with high risk of thromboembolism [CHA₂DS₂-VASc score ≥ 2 for males, ≥ 3 for females] with absolute or prohibitive contraindication to long-term oral anticoagulation [previous life-threatening hemorrhage]), Patients with high risk of bleeding (HAS-BLED ≥3, previous significant bleeding, possible recurrent invasive procedures, cancer related bleeding diathesis like recurrent GI bleeding [primary GI tumor or radiation-induced enteritis], chemotherapy induced thrombocytopenia [platelet counts are chronically or recurrently below 50 000/μL], Intracranial metastasis or primary brain tumor, severe anemia that requires frequent transfusions which may be exacerbated by oral anticoagulation), patients with possible frequent interruption of anticoagulation (those who might undergo frequent surgeries [biopsies/lumbar puncture] and need repeated periods of subtherapeutic anticoagulation increasing the risk of stroke).22,36,47 (Table 1).
Proposed Clinical Profiles That Favor LAAO in Cancer Patients with AF.

Exclusion Criteria (or Major Relative Contraindications)
LAAO could be inappropriate or futile in some patients. Patients with limited life expectancy (<∼1 year of reasonable quality; several health-system frameworks use 2-3 years as practical thresholds) are often guided against LAAO.22,36 Patients with advanced metastatic disease, poor functional status (ECOG performance status 3-4) or patients who are transitioned to palliative care are also poor candidates for LAAO as the procedural risks are substantial and the potential to realize a long-term benefit from stroke prevention is minimal. Patients who undergo pre-procedural imaging (TEE or cardiac CT) and were found to have anatomical or technical contraindications (left atrial thrombus where device manipulation could precipitate a catastrophic embolic event, unsuitable LAA anatomy [excessively large or small ostium, complex multi-lobed structure, or very shallow depth]) are excluded from device implantation.47,48 Acute clinical instability (uncontrolled systemic infection or sepsis that could cause device endocarditis, uncontrolled active bleeding or severe coagulopathy/thrombocytopenia that would make procedure life threatening) defers LAAO as it is an elective procedure.
Patients who have indications for OACs like pulmonary embolism and deep vein thrombosis should not undergo LAAO. 49 LAAO addresses thromboembolic risk related to AF. Additionally, VTE is often linked to the venous access or postoperative immobility and DRT following LAAO making it not suitable for previous VTE. 50
In summary, the cornerstone in success is selecting candidates appropriately. Patients with high thromboembolic risk, contraindications to oral anticoagulation, and sufficient life expectancy to realize long-term benefits are expected to achieve the best outcomes. While those with advanced disease or prohibitive anatomy derive minimal advantage.22,36,47,48 (Figure 3) illustrates the candidate patients for LAAO in cancer-associated AF.

Candidate selection for left atrial appendage occlusion (LAAO) in cancer-associated atrial fibrillation. Created in BioRender. Mahafdah, B. (2026) https://BioRender.com/mba1btz.
Timing Considerations: “When” to Intervene
Deciding the optimal timing of LAAO in cancer patients is critical as this directly impacts both procedural safety and patient outcomes. This decision should be individualized according to the patient's specific course of cancer therapy. (Figure 4) presents the optimal timing for LAAO in cancer patients.
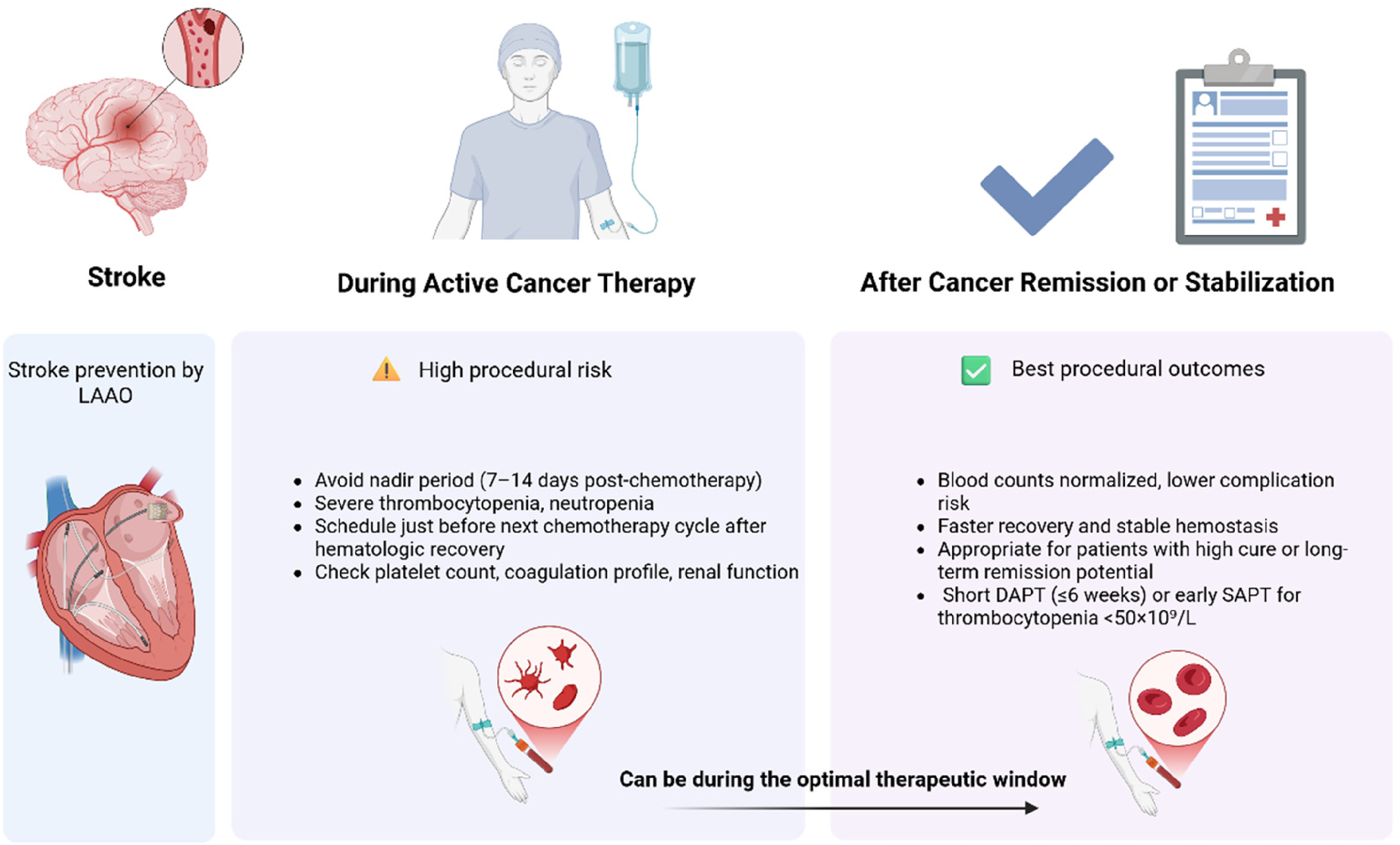
Timing of left atrial appendage occlusion (LAAO) in cancer-associated atrial fibrillation. The optimal window for LAAO is after hematologic recovery or during clinical remission. Created in BioRender. Mahafdah, B. (2025) https://BioRender.com/67q0qhc.
LAAO in patients with cancer-related AF by incorporating oncological status, bleeding tendencies, hematologic capacity, and overall prognosis.51,52 Patient selection should concentrate on individuals with NVAF and elevated thromboembolic risk who are ineligible for long-term oral anticoagulation due to cancer-related issues, including recurrent gastrointestinal bleeding, intracranial hemorrhage, central nervous system involvement, chemotherapy-induced thrombocytopenia, or the expected necessity for frequent interruptions of anticoagulation for invasive procedures.33,51,53 Patients exhibiting intact functional status and a life expectancy beyond one year are most likely to gain from LAAO, whereas those with advanced metastatic illness or low performance status see little benefit from an invasive stroke-prevention approach. These ideas align with current transcatheter LAAO consensus statements and cardio-oncology guidelines.51,52
The timing of intervention must be tailored to the specific cancer activity and treatment phase.26,52 LAAO is best done when the cancer is in remission or when the blood is recovering, because these are the times when the risk of bleeding and infection is lowest.26,53 For patients receiving active systemic therapy, LAAO may still be suitable when the bleeding risk associated with anticoagulation is excessive; however, implantation should be arranged to avoid chemotherapy nadir periods and coordinated with oncology teams and planned surgical or radiotherapeutic procedures.26,52 After the implant, the antithrombotic strategy must be based on the platelet count and take into account the bleeding sites and ongoing cancer treatment.53,54 In patients with moderate thrombocytopenia, shortened dual antiplatelet therapy followed by single antiplatelet therapy is appropriate. Conversely, in cases of severe thrombocytopenia or high-risk bleeding sites, such as the gastrointestinal tract or central nervous system,53,54 SAPT alone or a temporary interruption of antithrombotic therapy with close imaging surveillance may be required. Early and repeated imaging with TEE or cardiac CT remains essential to detect DRT and ensure procedural safety.51,54
During Active Cancer Therapy
Performing LAAO during active systemic cancer therapy is challenging but can be warranted in patients facing an imminent, high bleeding risk from oral anticoagulation. The key is to identify a period of relative clinical stability that serves as the optimal therapeutic window. The procedure should be timed to avoid the nadir period where bone marrow is maximally suppressed thus patients have severe thrombocytopenia, neutropenia leading to very high risk of periprocedural bleeding and infection (typically occurs 7-14 days after chemotherapy cycle). Timing LAAO just before starting the next treatment cycle where hematologic recovery has occurred is ideal. Also plan the procedure in coordination with any upcoming major surgeries to minimize the risk of interrupting antithrombotic therapy and to avoid competing bleeding/infection risks. Prior to the procedure, a thorough assessment of platelet count, coagulation profile, and renal function is required and the decision to proceed should take into account that the patient's physiological reserve is probably diminished.37,38,40
After Cancer Remission or Stabilization
The optimal strategy is deferring LAAO until the patient has completed their primary cancer treatment and is in remission or a stable phase whenever it is clinically feasible. Patients are no longer subject to the acute toxic effects of chemotherapy or radiation during remission. Their blood counts generally return to normal, nutritional status improves and overall frailty drops. The likelihood of periprocedural complications is profoundly dropped in this clinically stable period and recovery is faster and smoother. A patient's long-term prognosis determines whether to intervene after remission. Patients who have high probability of cure or long-term sustained remission from cancer (eg, early-stage breast or colon cancer) are evident to benefit from long-term stroke prevention. While patients who have malignancies with a high recurrence risk, the choice is more complex and nuanced. Doctors should carefully consider the patient's expected survival and the possibility of future cancer treatments to make sure the procedure truly supports their long-term health goals. Managing antithrombotic therapy after LAAO in cancer patients remains complex. Short-term DAPT followed by SAPT use is the standard approach. When thrombocytopenia or an elevated risk of mucosal bleeding is present, adjustments are often necessary. Recent expert recommendations support the usage of a shortened DAPT duration (≤6 weeks) or the early transition to SAPT in patients with platelet counts below 50 × 109/L.37,38,40 Collaboration between cardio-oncology and electrophysiology specialists within a multidisciplinary heart team is crucial to coordinate procedural timing, optimize antithrombotic management, and ensure alignment with the patient's overall cancer treatment plan.
In summary, optimal timing of LAAO depends on disease phase and hematologic stability. Whenever feasible, procedures should be scheduled during remission or recovery periods coupled with tailored antithrombotic regimens and multidisciplinary coordination.37,38,40
Antithrombotic Therapy After LAAO: “How” to Manage
Post implant Antithrombotic therapy, also known as post implant thrombo-protection, remains crucial for the prevention of DRT related to LAAO, especially within the first 45 to 90 days after the healing phase of the endothelial layer. 54 Current strategies focusing on the combination of both conventional antithrombotic therapies among non-cancer subjects have already shown efficacy; however, their administration among cancer patients entails additional concerns such as thrombocytopenia, mucosal injury, and chemotherapy.26,28
Standard Post-Implant Regimens
The initial goal of antithrombotic therapy in the setting of LAAO is preventing DRT during the course of endothelialization, which usually takes 45 to 90 day. 54 The existing approach for non-cancer subjects includes a brief course of OACs therapy with either warfarin or DOACs in the initial 45 days often in combination with low-dose aspirin, followed by DAPT for 1–3 months and finally SAPT as a long-term treatment. In those who develop intolerance to anticoagulants, DAPT alone for 1–3 months and subsequent SAPT therapy seem like a sound approach.54,55 The proposed treatment strategy aims to properly manage the dual threats of thrombo-blooming (progressive development or proliferation of DRT prior to endothelial tissue completely covering the device (endothelialization)) complications with an emphasis on comprehensive endothelialization of the device.
Current evidence also supports early de-escalation approaches like reduced DAPT or even SAPT alone for high-bleeding-risk patients. For instance, new randomized and meta-analysis evidence shows that reduced antithrombotic therapy may prove as effective as routine DRT prevention, as long as it is confirmed on strict imaging follow up by TEE or CT at 45 to 90 days. 56
Early randomized trials of LAAO such as PROTECT-AF and PREVAIL, mainly used warfarin immediately after implantation. however, most clinicians these days have shifted toward DOACs particularly apixaban and rivaroxaban. Observational studies and recent data show that DOACs match warfarin when it comes to rates of DRT and stroke, but they tend to cause less bleeding. Apixaban stands out for cancer patients; it carries a lower risk of GI bleeding and has fewer drug interactions than the alternatives. When oral anticoagulation is not feasible, the usual approach is dual antiplatelet therapy with aspirin and clopidogrel, then tapering to aspirin alone. Stronger P2Y12 inhibitors like ticagrelor or prasugrel are generally avoided in oncology patients because of excess bleeding risk.46,54
Tailored Strategies in Cancer Patients
Patients with AF pose a special problem for cancer patients due to the coexistence of hypercoagulability and high bleeding risk. 28 Issues of thrombocytopenia from chemotherapeutic drugs or from bone marrow suppression, mucosal lesions such as gastrointestinal or genitourinary lesions, and the usual fluctuations in coagulation factors due to treatment pose a challenge to the usual DAPT therapy. 57 In such circumstances, DAPT treatment may be reduced to 1 month along with SAPT or SAPT alone depending on the severity of the bleeding risk.
The coordination of multidisciplinary care is important cardiology, hematology, and oncology staff should work collaboratively to assess platelet levels and medication timelines for interactions with DOAC medications or antiplatelets. Imaging studies by TEE or CT should be carried out more often for thrombus formation and device leak.
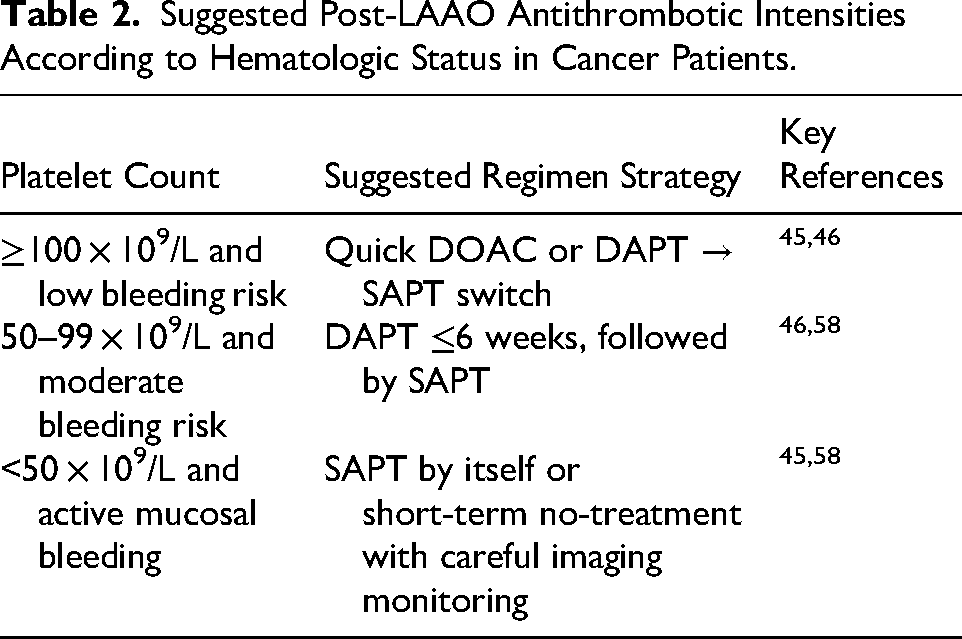
Increasing evidence indicates that LAAO can be performed safely and effectively in cancer patients with reduced risk of stroke without the long-term use of anticoagulation. Furthermore, it has been observed that cancer patients may remain on reduced-dose antithrombotic therapy without compromising immediate safety outcomes. However, the incidence of bleeding complications remains higher.58,59 (Table 2)
Suggested Post-LAAO Antithrombotic Intensities According to Hematologic Status in Cancer Patients.
Practical Guidance for High-Risk Clinical Scenarios
Several high-risk situations need an altered use of LAAO in cancer-related AF. Patients with severe thrombocytopenia (platelets <50 × 109/L) or hematologic malignancies are at a significantly elevated risk of bleeding when undergoing OACs and standard post-implant antithrombotic regimens. In such cases, LAAO may be selectively considered following hematologic recovery, with a preference for single antiplatelet therapy or a temporary interruption of antithrombotic treatment accompanied by close imaging surveillance.37,52 Likewise, in individuals with active gastrointestinal bleeding, LAAO offers a non-pharmacological stroke-prevention alternative after the bleeding is controlled, facilitating shortened antithrombotic treatment and diminishing the risk of recurrent hemorrhage in comparison to prolonged anticoagulation. 33
Patients with intracranial tumors or brain metastases necessitate heightened vigilance due to the dire ramifications of intracranial hemorrhage; should LAAO be undertaken following a multidisciplinary assessment, post-procedural antithrombotic intensity must be reduced, accompanied by prompt follow-up imaging to rule out device-associated thrombus.37,52 During times of peri-chemotherapy nadir, the risks of procedures and bleeding are most because of temporary cytopenias. Therefore, LAAO should be put off until the blood cells have recovered, and platelet counts and coagulation parameters should be checked again just before implantation. In all of these cases, early and organized follow-up using TEE or cardiac CT is necessary to weigh the danger of bleeding against the need for thromboembolic protection.37,46
Ongoing Trials and Evidence Gaps
Although promising data from observational cohorts exist, substantial evidence gaps persist in antithrombotic management in cancer-related AF. No randomized controlled trial has exclusively recruited cancer subjects or confirmed cancer-specific algorithms of antithrombotic treatment.60,61 Current recommendations primarily come from trials involving mixed populations of AF. 61
Biomarkers like D-dimer and platelet function/activation, as well as imaging factors like leak and peri-device flow, have been investigated as a potential component of personalized treatment duration and intensity of antithrombotic therapy. Various registries have been conducted to investigate the concept of abbreviated treatment regimens including monotherapy and reduced-dose DOACs. However, cancer-related validation has yet to be performed.
The next area of research should be prospective multi-center trials that assess not only thromboembolic and bleeding events but also outcomes such as quality of life, burden of treatment, and cost effectiveness in cancer patients. 62
Technical and Procedural Considerations
Technical aspects play an important role in the outcome of LAAO, particularly in cases of cancer patients who may have a complex anatomy and coagulation disorders. However, effective imaging and management are crucial for avoiding complications and providing a successful closure.60,63 In the context of oncology, the role played by radiation and thrombocytopenia increases importance.
Device Choice and Sizing in Altered Anatomy or Prior Radiation
Preprocedural imaging is a vital component of a successful and safe LAAO procedure. In particular, it is important in cancer patients who may have suffered from either radiation of the chest or chest surgery in the past. 63 Radiation of the chest may result in either fibrosis or calcification of the left atrium. Additionally, it may lead to anatomic changes within the left atrium. CT angiography and TEE offer the most precise determination of the appendage's anatomy. 64 In patients who have suffered from chest radiation and/or chest surgery, devices that possess high flexibility and conformability properties or newer occlude devices are considered ideal. These factors ensure better closure and prevent PDL.
Investigations have found that CT-perimeter-derived measurements of PAI size correlate better with successful implantation and lower leak rates than TEE alone. 64 The role of PAI implantation in cancer survivors may be especially important due to the potential for anatomic distortion.
Periprocedural Management of Coagulopathy and Platelet Dysfunction
Patients with cancer often present with hematologic abnormalities like thrombocytopenia, coagulation factor deficiency, or platelet functional defect due to chemotherapy. Careful assessment of platelet count, coagulation tests like PT and aPTT/INR, fibrinogen levels, as well as mucosal lesions that may pose a bleed risk should be done before undergoing LAAO. 58 The optimum timing of LAAO should be planned when platelet count remains relatively stable (>50 000/µL) and mucositis/ulcers have resolved. 60
In performing the procedure on the patient, while unfractionated heparin is the anticoagulant of choice, it may be necessary to adjust the dose in a patient with coagulopathy. When dealing with high-risk patients undergoing LAAO, transfusion support with platelets or fresh frozen plasma may be necessary in selected cases. For cancer patients undergoing the procedure postoperatively, monitoring for pericardial effusion, cardiac tamponade, and early bleeding complications have been found to be higher.28,60
Multidisciplinary Perioperative Planning (Oncology, Hematology, Anesthesia, Cardiology)
The ideal management of cancer patients undergoing surgery should incorporate the efforts of cardiologists, interventionalists, oncologists, hematologists, and anesthesiologists. 26 Through an integrated approach, it should be possible to determine the most appropriate timing of LAAO, preferably during the intracycle or stability phases. Pre-procedural conferences may help to prevent complications and address challenges relating to the use of either DOACs or antiplatelet medications.
Hematology data should always be included for the assessment of transfusion thresholds or thrombocytopenia management. 59 The oncologists’ input on pending therapy that could increase the risks of bleeding or coagulopathy should also be incorporated. The anesthesiologists may consider alternative forms of anesthesia such as ICE as opposed to TEE. 59
The coordination of various disciplines can also extend to the visits carried out post-procedure. For instance, cancer visits can be coordinated with cardiology imaging visits at 45- to 90 day post-implants. 60
Ethical, Prognostic, and Quality-of-Life Perspectives
Because there was no evidence of non-inferiority when compared to medical therapy over a median follow-up of roughly three years, the CLOSURE-AF trial raised questions about the long-term efficacy and safety of LAAO. 65 These results have significant ethical ramifications, especially for patients with short life expectancies. Patients with an expected survival of one to three years may not receive enough net benefit from the procedure because the clinical benefit of LAAO is expected to accrue over longer-term follow-up, while procedural and early post-procedural risks are immediate.65,66 Therefore, when considering LAAO, current expert consensus statements stress that life expectancy and overall prognosis should be crucial factors in patient selection and collaborative decision-making. 66
Decision-Making Should Incorporate Prognosis, Patient Goals, and Life Expectancy
Whether to undergo an LAAO procedure in a patient with active or existing malignancy should always be made on an individual basis. In those with existing malignancy who may gain a long-term survival advantage by preventing strokes via an LAAO procedure, such an approach may be indicated only if the underlying patient survival and quality of life justify the resource intensiveness of the procedure. In those who have limited survival expectation (< 12 months) or those who have rapidly progressive disease states, a more conservative approach may be more ethical.
In contrast, for those with controlled cancer disease or earlier disease states, LAAO can offer substantial thromboembolic risk reduction without the need for long-term anticoagulation. The important role of shared decision making among cardiology and oncology providers in coordinating treatment based on the patient's value and preferences should not be overlooked. 59
Ethical Considerations of Invasive Interventions in Advanced Malignancy
Invasive heart surgery in cancer patients who have an advanced stage of cancer raises several considerations. The challenges include the risks of such a surgery and the subsequent management of antithrombotic therapy. The principles of ethics applied here include beneficence. In some cancer patients who may be at an end-of-life stage, it may be better to adopt palliative or conservative options rather than proceeding with implantation.
Discussion of outcomes, risks, and quality of life considerations should be openly discussed before an intervention. The practitioner should ensure that consent for intervention is informed and that the patient recognizes that LAAO may not change survival but can decrease either stroke or bleeding events.26,67
LAAO May Reduce Hospitalization Burden and Improve Autonomy in Select Patients
Quality of life (QoL) is an important consideration in the management of AF related to cancer. Many cancer patients already suffer from substantial in-hospital time, drug toxicity, and psychological distress. In addition, LAAO can prevent recurrent admissions for bleeding and stroke and obviate the need for repeated measurements of INR in selected candidate patients undergoing chemotherapeutic treatments.
Observational data indicate that LAAO patients have fewer admissions for bleeding than patients on long term anticoagulation therapy. Simplified forms of antithrombotic therapy post-implantation (eg SAPT only) may add additional improvements in activity and independence. For highly motivated and stable disease patients with adequate functional capacity remaining, the balance of effectiveness, safety, and independent function made possible by LAAO may prove very beneficial to these cancer patients.28,67,68
Future Directions and Research Gaps
Randomized Trials in Cancer-Associated AF Populations
Randomized controlled trials (RCTs) assessing LAAO in patients with cancer-associated AF are scarce, despite mounting observational evidence. The majority of the data available today come from small registries or subgroup analyses that either restrict follow-up to brief periods or exclude patients with active cancer.28,33,69 To thoroughly assess the procedural safety, long-term stroke prevention effectiveness, and comparative clinical outcomes of LAAO in comparison to DOACs and conservative management techniques, RCTs are needed. It is crucial that future studies use inclusive and stratified designs that take into account the diversity of cancer patients, including solid and hematologic cancers, different disease stages, and dynamic treatment phases like immunotherapy, chemotherapy, and remission. The clinical applicability would be further improved by including cancer-specific endpoints, such as bleeding events during myelosuppression or DRT under concurrent anticancer therapy. adequate enrollment, longitudinal follow-up, and subgroup analyses across cancer phenotypes may be made easier by cooperative multicenter, registry-linked randomized frameworks.
Risk Prediction Models Using Cancer Biomarkers and Computational Modeling in Procedural Planning
Due to tumor-related hypercoagulability and systemic inflammation, patients with cancer-associated AF are at increased risk for thrombosis and bleeding, which reduces the usefulness of standard scores like the CHA₂DS₂ VASc. These findings were validated by the Cardio CHUVI-AF (Retrospective Observational Registry of Patients with Atrial Fibrillation From Vigo's Health Area) registry. 16 056 AF patients (1137 AF and cancer patients) were included in this retrospective observational Spanish cohort; the median follow-up was 4.9 years. The authors concluded that the CHA2DS2-VASc score had a limited discriminative capacity and underestimated the S/SE risk in patients with AF and cancer who were not receiving anticoagulation treatment. The S/SE risk of cancer patients with a CHA2DS2-VASc score = 1 was similar to patients with a CHA2DS2-VASc score ≥2, and only patients with a CHA2DS2-VASc score = 0 presented with a very low risk of embolic events. In addition, a HASBLED score >3 was not associated with a higher bleeding risk in patients with cancer compared with a noncancer population. 70 The performance of traditional models can be affected by a number of mechanisms that cancer itself introduces that increase the risk of stroke and bleeding, including endothelial dysfunction, platelet activation, chemotherapy-induced cytopenias, and tumor-related cytokine release. Next-generation risk prediction models should include cancer-specific biomarkers that represent tumor biology, coagulation activation, and systemic inflammation in order to overcome these constraints. Clinical factors like cancer type, stage, treatment method, and hematologic parameters could be combined with biomarkers like D-dimer, high-sensitivity troponin, C-reactive protein (CRP), vascular endothelial growth factor (VEGF), and circulating tumor DNA (ctDNA).71–75 These kinds of multidimensional models would allow for dynamic risk stratification, which means that they would change based on how the disease or treatment changes over time. New computational and machine learning-based methods present a chance to combine these disparate data and produce personalized estimates of bleeding and thromboembolic risk. 76 These systems could improve long-term anticoagulation strategies, choose the best patients for LAAO, and schedule procedures in relation to immunotherapy or chemotherapy cycles by continuously learning from changing datasets. Therefore, incorporating biomarker-driven analytics into standard cardio-oncology procedures is a critical step in preventing cardioembolic stroke in patients with AF linked to cancer. Computational fluid-dynamic (CFD) simulations provide patient-specific hemodynamic indices (mean residence time, LAA-appearance-complexity index) that capture stasis beyond morphological classification. Machine learning frameworks combine clinical and biomarker data with these CFD metrics, exposing nonlinear interactions that improve the prediction of adverse events following LAAO. 77 Ngufor et al (2024) 78 applied a causal machine learning framework to optimize patient selection for LAAO compared with DOACs among more than 740 000 patients with AF using the OptumLabs Data Warehouse. The causal forest model calculated individualized treatment effects for a composite endpoint of stroke, embolism, major bleeding, and mortality following extensive propensity matching. By two years, LAAO was linked to a significantly lower composite risk, with about 30% of patients identified as likely to derive net benefit, even though outcomes were comparable at one year. This study shows how artificial intelligence-powered causal inference models can be used to select LAAO candidates precisely. A precision medicine workflow that customizes LAAO procedural planning to the distinct risk profile of cancer-associated AF patients is made possible by the combination of biomarker-incorporated risk prediction, CFD-derived stasis indices, and causal machine learning algorithms.
Development of Individualized Post-LAAO Antithrombotic Algorithms
In oncology patients, both thrombotic and hemorrhagic risks are amplified, and the optimal intensity and duration of antithrombotic therapy after LAAO remain undefined. Incorporating biomarker-guided approaches (eg, D-dimer, fibrinogen, and platelet function assays) and dynamic clinical scoring systems could refine real-time adjustment of antithrombotic intensity to maintain stroke protection while minimizing bleeding risk. The primary objective of post-LAAO therapy is to prevent DRT and systemic embolism while ensuring adequate device healing (endothelialization). The complexity in oncology patients necessitates a focus on aggressively managing DRT risk. A critical component of the individualized algorithm is an accelerated imaging surveillance schedule (eg, routine TEE or cardiac CT) to detect DRT early. When DRT is found, the algorithm dictates an immediate intensification of antithrombotic therapy alongside a parallel assessment of cancer activity and concurrent oncologic treatment, acknowledging that the thrombotic event may be a signal of underlying disease progression.45,46,79,80
The core of the individualized algorithm lies in leveraging biomarkers to guide therapeutic decisions and duration after the procedure. Platelet function assays are essential for measuring the efficacy of antiplatelet agents (like aspirin and P2Y12 inhibitors). For instance, if a patient exhibits residual high platelet reactivity, the algorithm may call for an extension of dual antiplatelet therapy or a switch to a more potent antiplatelet agent to promote proper device sealing. Conversely, the detection of low platelet reactivity—especially when coupled with a high clinical bleeding risk or chemotherapy-induced low platelet counts provides objective justification for safely de-escalating the antiplatelet regimen.81–83 Furthermore, monitoring D-dimer and fibrinogen levels offers a dynamic indicator of persistent hypercoagulability. A sustained or rising D-dimer post-LAAO acts as a therapeutic trigger, suggesting a need to extend or adjust the intensity of the DOAC regimen, independent of the standard protocol duration, to maintain stroke protection.84–86 Ultimately, a truly effective algorithm requires close integration with the patient's cancer care, establishing a unified cardio-oncology treatment calendar. This coordination allows for the management of the antithrombotic regimen around peak bleeding or thrombotic risk periods associated with specific chemotherapy cycles. The algorithm defines protocols for planned, temporary interruptions or dose reductions of antithrombotic agents during high-risk oncologic treatment phases and guides the transition between antiplatelet and anticoagulant strategies based on the current phase of cancer treatment.82,87–89 This system transitions post-LAAO care from a fixed, standardized protocol to a precision medicine workflow that continuously adapts the antithrombotic regimen based on the dynamic interplay between the LAAO device, cancer biology, and systemic drug effects.
Available evidence for LAAO in cancer-associated AF is derived mainly from retrospective and registry-based studies and is therefore subject to selection bias, residual confounding, and competing non-cardiovascular mortality related to cancer progression.25,33,90 Future prospective studies should incorporate cancer-specific stratification, including cancer type and stage, treatment phase, and dynamic hematologic parameters, while adopting standardized antithrombotic regimens and uniform follow-up imaging schedules with TEE or cardiac CT to improve comparability and clinical interpretability across studies.33,46
Conclusion
Cancer-associated AF represents a unique clinical intersection, because patients are prone to both clotting and bleeding. For many people with cancer, especially those with GI tumors, low platelets, brain involvement, or a history of bleeding, long-term oral anticoagulation just is not safe or reliable. In this case, LAAO provides a critical non-pharmacologic alternative, giving ongoing protection against stroke, even during chemotherapy or remission, without the constant bleeding risk.
According to recent evidence, LAAO works well, provides good safety outcomes, and actually decreases the bleeding risk among cancer patients who are chosen carefully. Despite that, the cardio-oncology population still considered as underrepresented in major trials. Additionally, specific cancer-targeted strategies, including shortened dual antiplatelet regimens, increased imaging frequency, or therapy modification based on biomarkers, have not yet been approved in randomized studies.
Footnotes
Abbreviations
Acknowledgments
None.
Ethical Considerations
Ethical approval is not required for this review article.
Informed Consent Statement
Not applicable.
Authors Contributions
A.A. and A.E. conceived the study and contributed equally as co-first authors. M.A., K.I., and S.A. participated in data collection and literature review. M.N.S. B.A., H.A.A., and N.A. contributed to data interpretation and manuscript drafting. B.M. contributed to data interpretation and creation of figures. M.S.E., A.S., and M.B provided critical revisions for important intellectual content and supervised the project. All authors reviewed, edited, and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
This study did not involve the creation or analysis of any new data. Therefore, data sharing is not applicable.
Provenance and Peer Review
Not commissioned, externally peer-reviewed.
