Abstract
Background
The clinical relevance of the early coagulation disorder (ECD) score, derived from platelet count, international normalized ratio (INR), and activated partial thromboplastin time (APTT), in patients with stroke remains unclear. This study aimed to evaluate the association between ECD score at intensive care unit (ICU) admission and all-cause mortality (ACM) in patients with stroke.
Methods
This retrospective cohort study utilized data from the MIMIC-IV database. The primary outcome was 30-day all-cause mortality; secondary outcomes included 90-day and 1-year all-cause mortality. Kaplan–Meier curves and multivariable Cox models were used to assess associations between ECD scores and outcomes. Subgroup analyses and interaction tests were conducted to verify robustness.
Results
Among 5753 participants (median age 70.97; 52.03% male), the 30-day mortality rate was 17.17%. Kaplan–Meier analysis showed significantly higher mortality in patients with ECD (log-rank P < 0.01). Multivariable analysis indicated increased risks of mortality within 30 days in patients with ECD (HR = 1.374, 95% CI: 1.174-1.608) and those with ECD scores of 5–6 (HR = 1.374, 95% CI: 1.011-1.868) compared to those without ECD or with a score of 0, respectively. Subgroup analyses supported these findings.
Conclusion
Elevated ECD scores are associated with increased mortality in critically ill patients with stroke. Comprehensive coagulation assessment may improve clinical management and risk assessment.
Introduction
Stroke is an acute neurological disorder caused by occlusion of cerebral blood vessels (ischemic stroke, IS) or rupture of cerebral vessels (hemorrhagic stroke). It ranks among the predominant causes of global mortality and disability. 1 In high-income countries, IS accounts for approximately 74.9% of strokes, compared to 63.4% in low- and middle-income countries. 2 In 2021, approximately 3.6 million deaths worldwide were attributed to IS. 3 Though the incidence and mortality of age-standardized stroke have declined since 1990, the absolute number of cases and deaths has continued to increase owing to population aging. 4 Given its profound public health burden, identifying and managing clinically relevant indicators is critical for reducing stroke-related disability and mortality.
In the acute early stage of stroke (typically 24-72 h post-onset), coagulation disorder rapidly occurs. These dysfunctional changes reflect disease progression and might influence clinical outcomes. Upon vascular injury, platelets tether to the vessel wall via the glycoprotein (GP) Ib-IX-V receptor complex, bind to von Willebrand factor, and then firmly adhere through glycoprotein VI (GPVI), the principal platelet collagen receptor. 5 This increases the difficulty of thrombolytic therapy and vascular recanalization. Moreover, a higher incidence of hemorrhagic transformation (HT) is observed after recanalization therapy. HT is more likely to occur after recanalization therapies and is associated with poorer stroke outcomes. 6 Coagulation disorders can render HT more difficult to control, thereby adversely affecting the clinical outcomes in patients with stroke.
Recent evidence has shown that IS is commonly associated with coagulation system activation and a hypercoagulable state. 7 Barakzie et al reported that various coagulation factors influence the risk, severity, and prognosis of IS. 8 Consequently, anticoagulant and thrombolytic therapies targeting hypercoagulability are standard strategies for IS. However, these treatments and individual patient variations might disrupt coagulation balance, inducing a hypocoagulable state and considerably elevating the risk of HT.9,10 This evidence underscores that dynamic coagulation changes after stroke (from initial hypercoagulability to treatment-induced hypocoagulation) represent critical determinants of patient outcomes.
Laboratory indicators like PLT count, international normalized ratio (INR), and activated partial thromboplastin time (APTT) reflect coagulation status and are commonly used to assess the severity of sepsis-induced coagulopathy (SIC). 11 Studies have shown that INR (≥1.7) and prolonged APTT (≥51.45 s) are considerably linked to the risk of mortality within 60 days among patients with trauma. 12 Nevertheless, the association between early coagulation disorder (ECD) and poor clinical outcomes in critically ill patients with stroke remains underexplored. Based on the Medical Information Mart for Intensive Care IV (MIMIC-IV) database, this research sought to ascertain the association between ECD scores and all-cause mortality (ACM) among individuals with stroke, providing guidance for optimizing the health management of these populations.
Method
Data Source
This retrospective study leveraged data from the Medical Information Mart for Intensive Care IV (MIMIC-IV, version 3.1) database, which was released on October 11, 2024. MIMIC-IV, version 3.1 contained de-identified electronic health records of patients admitted to the Beth Israel Deaconess Medical Center (BIDMC) from 2008 to 2022. As a contemporary critical care dataset, MIMIC-IV complemented the growing body of publicly accessible resources for critical care research. To access the data, one of the authors (Q.Y.Z) underwent formal training (record ID: 65255490). This study received ethical exemptions from the Massachusetts Institute of Technology and BIDMC, and no additional ethical approval was required.
Study Design and Population
This research included 10314 patients with stroke (≥18 years) who were admitted to the ICU for the first time. This study identified stroke cases based on International Classification of Diseases (ICD) codes from the MIMIC-IV database, covering both ICD-9 (codes 430, 431, 432, 433, 434) and ICD-10 (codes I60, I61, I62, I63) coding systems. These codes corresponded to the following subtypes of stroke: subarachnoid hemorrhage, intracerebral hemorrhage, other intracranial hemorrhage, and cerebral infarction. The exclusion criteria were outlined below: (i) Patients discharged or deceased within 24 h of ICU admission (n = 1574); (ii) Patients without essential data on PLT count, APTT, or INR within 24 h of ICU admission (n = 1295); (iii) Patients with comorbidities including malignant tumors, liver cirrhosis, or end-stage renal disease (n = 1675); (iv) Outliers in the covariates were excluded (n = 17). Finally, 5753 eligible patients were included (Figure 1).
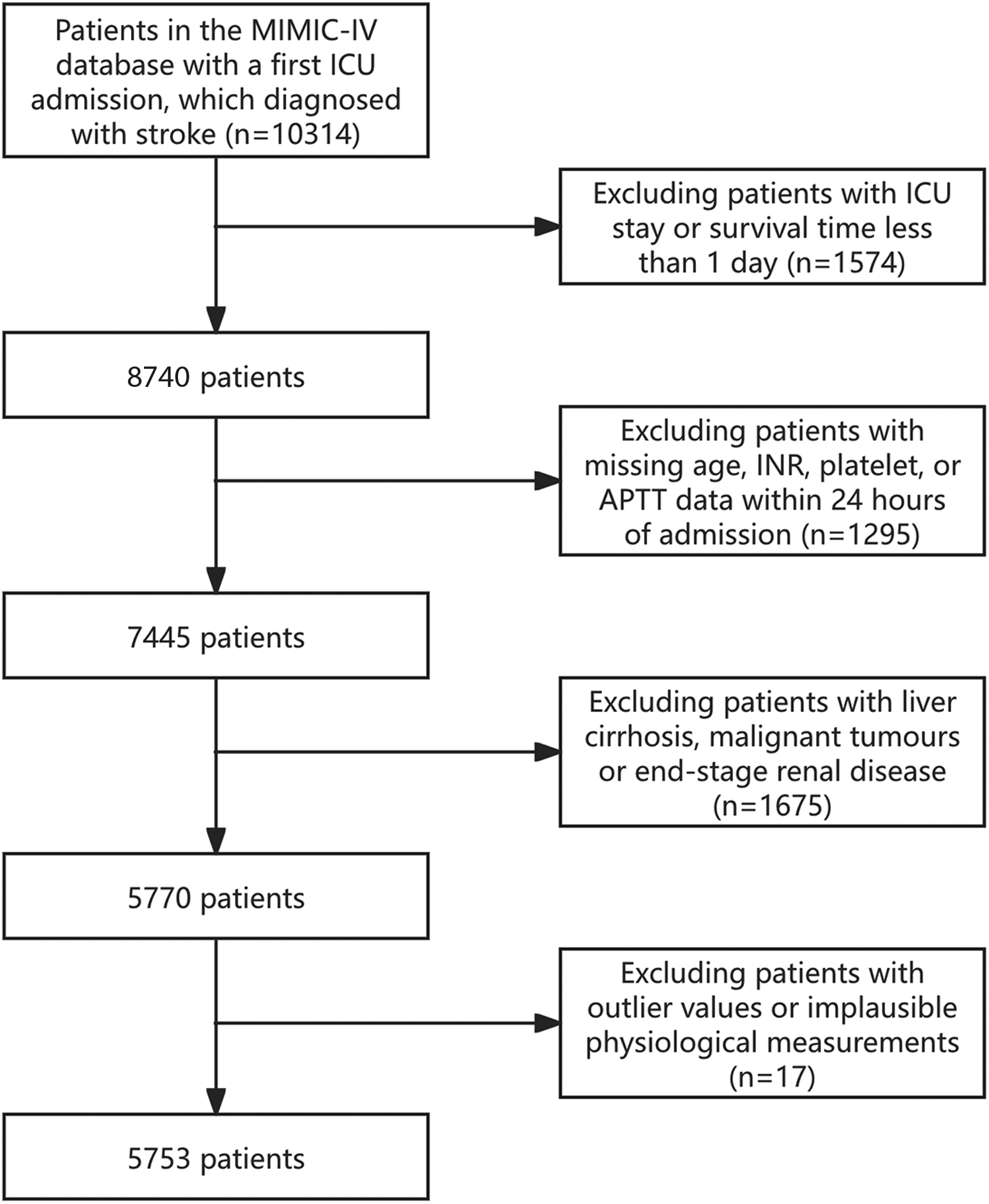
Flow of included patients through the trial.
Data Extraction
Data extraction was implemented utilizing Navicat Premium (Version 17.3.1) with Structured Query Language (SQL). This research ascertained variables categorized as follows: (i) Demographics included age, sex, and race; (ii) Past medical history included heart failure, atrial fibrillation, diabetes, hypertension, and sepsis; (iii) Vital signs included heart rate (HR), respiratory rate (RR), systolic blood pressure (SBP), diastolic blood pressure (DBP), and mean blood pressure (MBP); (iv) Laboratory indicators included glucose, red blood cell (RBC), hemoglobin, sodium, white blood cell (WBC), creatinine, PLT, APTT, and INR; (v) Disease severity score included Acute Physiology Score III (APSIII), Glasgow Coma Scale (GCS), Oxford Acute Severity of Illness Score (OASIS), Simplified Acute Physiology Score (SAPSII), and Sequential Organ Failure Assessment (SOFA); (vi) Therapeutic measures included alteplase, aspirin, clopidogrel, continuous renal replacement therapy (CRRT), invasive mechanical ventilation (IMV), non-invasive mechanical ventilation (NIV); (vii) Outcomes encompassed length of ICU stay (LOS), and mortality within 30 days, 90 days, and 1 year. Baseline blood indicators were measured within the first 24 h of ICU admission and before the initiation of any therapeutic or interventional procedures. Variables with > 20% missing values were excluded. The specific missing information and proportion of covariates are detailed in Supplementary Table S1. For continuous variables with < 20% missing values, missing data were imputed utilizing a random forest–based multiple imputation method.
Outcomes
The primary outcome was ACM within 30 days. The secondary outcomes included ACM within 90 days and 1 year.
Definition of ECDs
Additive PLT, INR, and APTT scores were utilized to define ECD with reference to SIC and coagulopathy.13,14 The most extreme values for PLT, INR, and APTT were collected within the first 24 h after patients’ ICU admission. PLT scores >150 × 109/L, 100–150 × 109/L, and < 100 × 109/L were assigned 0, 1, and 2, respectively. INR scores <1.2, 1.2–1.4, and > 1.4 were assigned 0, 1, and 2, respectively. APTT scores <37 s, 37–39 s, and > 39 s were assigned 0, 1, and 2, respectively. An aggregated score > 0 signified an ECD. 15
Statistical Analysis
First, normality tests were implemented for continuous variables. The results showed that all continuous variables were non-normally distributed. Non-normally distributed continuous variables were presented as median and interquartile range (IQR). Group comparisons were implemented utilizing the Wilcoxon rank-sum test. Categorical variables were presented as absolute numbers with percentages. Group comparisons were implemented utilizing Pearson's Chi-squared test.
Kaplan-Meier (K-M) curves were utilized to ascertain the incidence of primary and secondary outcomes. Patients were stratified based on ECD scores. Prior to inclusion in the multivariable Cox proportional hazards model, all covariates were assessed for multicollinearity. Variance inflation factors (VIFs) were calculated from the model design matrix, with VIF > 5 indicating significant multicollinearity. When multicollinearity was detected, variables were retained only if they showed lower correlations, smaller missing rates, or stronger clinical relevance.
Subsequently, univariable and multivariable Cox proportional hazards models were leveraged to estimate the association between ECD and adverse outcomes among individuals with stroke. Covariates were adjusted stepwise, and hazard ratios (HRs) with 95% confidence intervals (95% CIs) were reported. Variables included in the multivariate Cox model were selected based on clinical relevance, established literature support, and their potential role as confounders in the association between ECD and outcomes. Age, sex, and race were incorporated as basic demographic variables. Vital signs (HR, RR, SBP, DBP), comorbidities (atrial fibrillation, diabetes, heart failure, hypertension, sepsis), medications (alteplase, aspirin, clopidogrel), organ support therapies (CRRT, NIV), and laboratory parameters (creatinine, glucose, RBC, sodium, WBC) were included because they represented the severity of illness and had been shown to influence the risk of mortality. GCS and LOS were included as indicators of neurological and overall clinical status.
Model 1 was unadjusted. Model 2 was adjusted for age, sex, and race. Model 3 was further adjusted for vital signs, comorbidities, medications, organ support therapies, laboratory variables, neurological status, and ICU LOS, as listed above. In all three models, the group without ECD (ECD score = 0 and without ECD) served as the reference group. Moreover, subgroup analyses were implemented by sex (male, female), age (<70 years or ≥70 years), and the presence of comorbidities including hypertension, diabetes, heart failure, atrial fibrillation, or sepsis, to validate the reliability of the results.
All analyses were conducted using R (version 4.4.2, R Foundation for Statistical Computing, Austria). P < 0.05 signified statistical significance.
Results
Baseline Characteristics of Study Individuals
This research included 5753 eligible individuals with stroke from the MIMIC-IV database, with 2993 males (52.03%). The median age was 70.97 years (IQR: 59.05 to 81.04). Among these, the ACM within 30 days was 988 cases (17.17%) in the overall cohort and 749 cases (19.09%) in patients with ECD (P < 0.001). The ACM within 90 days reached 1230 cases (21.38%) overall relative to 938 cases (23.91%) in the ECD group (P < 0.001). The ACM within one year was 1530 cases (26.59%) overall and 1167 cases (29.75%) among those with ECD (P < 0.001). In total, 3923 patients (68.19%) had ECD. The baseline characteristics for each group are demonstrated in Table 1. Relative to the group without ECD, the group with ECD had a significantly higher proportion of males and older age, higher prevalence of past medical history or comorbidities, more frequent use of medications or interventions, elevated disease severity scores, longer LOS, elevated HR, RR, APTT, INR, creatinine, blood glucose, and WBC counts, and lower SBP, DBP, MBP, PLT count, hemoglobin, and RBC counts. At all measured time points, mortality was considerably higher in the ECD group. Intergroup comparisons showed no statistically significant differences in RR (P = 0.279), hypertension (P = 0.478), sodium (P = 0.835), GCS (P = 0.386), or LOS (P = 0.387) in the baseline table. Statistically significant differences were observed in all other variables during intergroup comparisons (P < 0.05).
Baseline Characteristics of Patients.
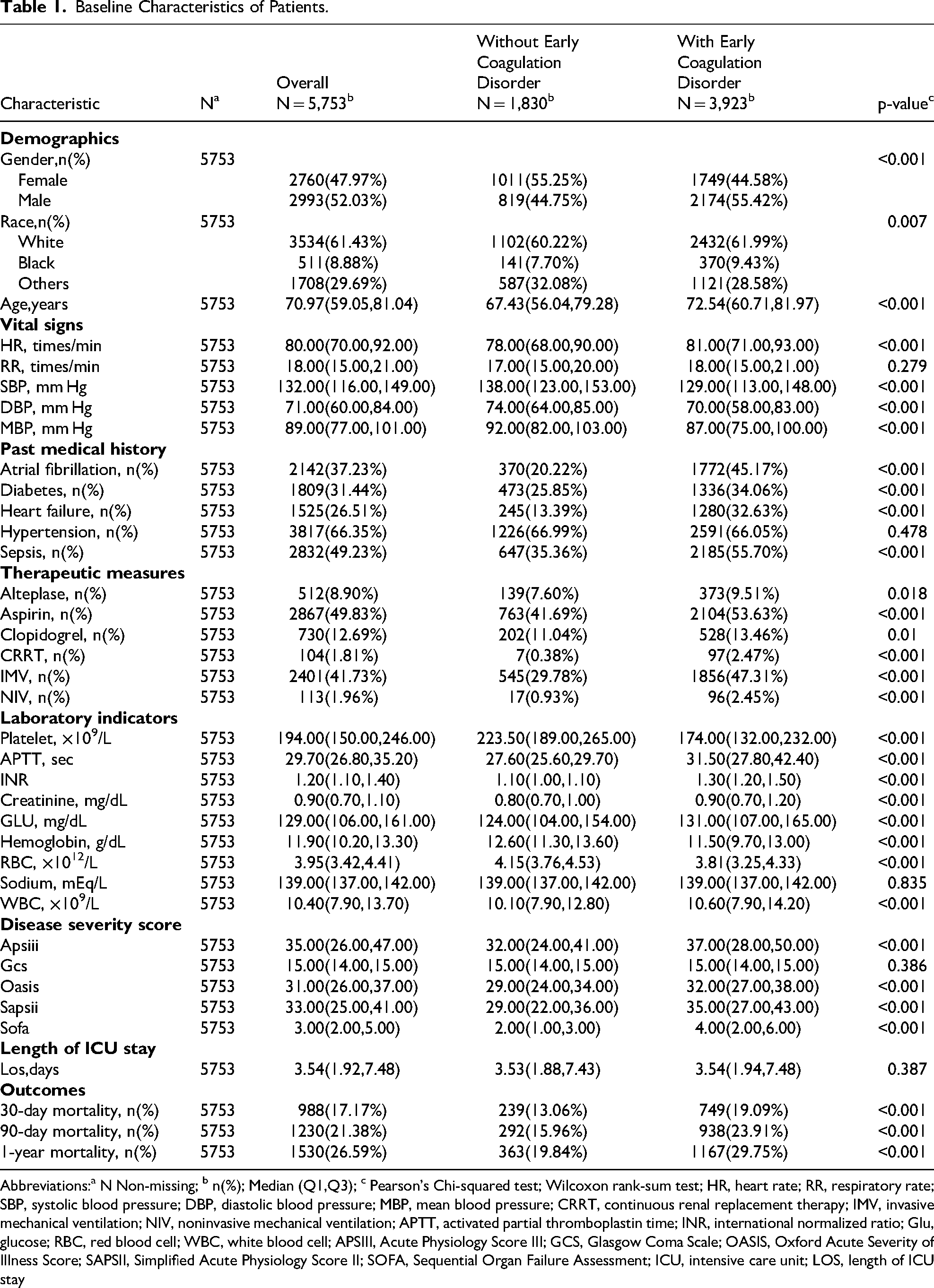
Abbreviations:a N Non-missing; b n(%); Median (Q1,Q3); c Pearson's Chi-squared test; Wilcoxon rank-sum test; HR, heart rate; RR, respiratory rate; SBP, systolic blood pressure; DBP, diastolic blood pressure; MBP, mean blood pressure; CRRT, continuous renal replacement therapy; IMV, invasive mechanical ventilation; NIV, noninvasive mechanical ventilation; APTT, activated partial thromboplastin time; INR, international normalized ratio; Glu, glucose; RBC, red blood cell; WBC, white blood cell; APSIII, Acute Physiology Score III; GCS, Glasgow Coma Scale; OASIS, Oxford Acute Severity of Illness Score; SAPSII, Simplified Acute Physiology Score II; SOFA, Sequential Organ Failure Assessment; ICU, intensive care unit; LOS, length of ICU stay
Study Outcomes
The K-M curves (Figure 2) demonstrate significant distinctions in ACM within 30 days, 90 days, and 1 year between patients with and without ECD. Patients were stratified into four groups based on ECD scores. Marked distinctions in ACM within 30 days, 90 days, and 1 year were also detected among groups. The associations of individual scores of PLT count, APTT, and INR with ACM within 30 days, 90 days, and 1 year are demonstrated in Supplementary Figure S1. Patients with ECD showed considerably elevated ACM within 30 days, 90 days, and 1 year relative to those without ECD (log-rank P < 0.0001). The highest-scoring group had considerably higher ACM within 30 days, 90 days, and 1 year relative to groups with lower scores (log-rank P < 0.0001).
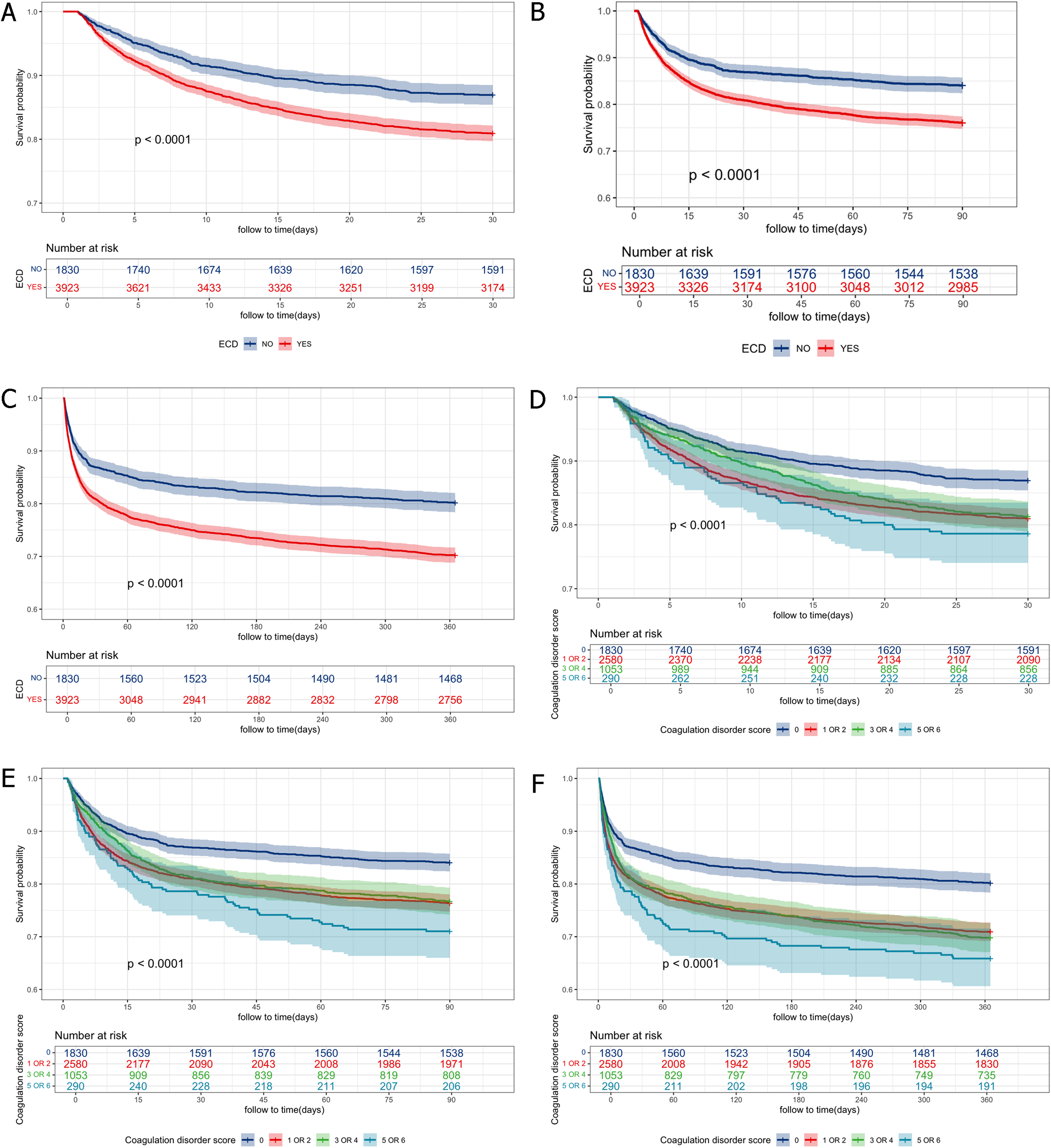
K-M survival analysis curves for ACM. K-M curves of ACM within 30 days (A), 90 days (B), and 1 year (C) stratified by ECD. K-M curves of ACM within 30 days (D), 90 days (E), and 1 year (F) stratified by ECD score. Note: K-M: Kaplan-Meier; ECD: early coagulation disorder; ACM: all-cause mortality.
Relationship Between ECD Score and Clinical Outcomes of Patients with Stroke
The Cox proportional hazards models were leveraged to estimate the association between ECD and adverse outcomes (Table 2). After adjusting for age, sex, and race (Model 2), each 1-point increase in the ECD score (as a continuous variable) was correlated with an 8.2% increase in the risk of ACM within 30 days (HR = 1.082, 95% CI: 1.040-1.125, P < 0.001). When the ECD score was treated as a binary variable, the risk of ACM within 30 days for the group without ECD versus the group with ECD was 1.000 (reference) versus HR = 1.429 (95% CI: 1.233-1.656, P < 0.001). When ECD score was classified as four groups (0, 1 or 2, 3 or 4, 5 or 6), the risks of ACM within 30 days were 1.000 (reference), HR = 1.429 (95% CI: 1.222-1.670, P < 0.001), HR = 1.370 (95% CI: 1.133-1.658, P = 0.001), and HR = 1.656 (95% CI: 1.251-2.193, P < 0.001), respectively. After further adjusting for age, sex, race, HR, RR, SBP, DBP, atrial fibrillation, diabetes, heart failure, hypertension, sepsis, alteplase, aspirin, clopidogrel, CRRT, NIV, creatinine, glucose, RBC, sodium, WBC, GCS, and LOS (Model 3), each 1-point increase in the ECD score (as a continuous variable) was linked to a 6.5% increase in the risk of ACM within 30 days (HR = 1.065, 95% CI: 1.018-1.114, P = 0.006). When ECD score was treated as a binary variable, the risk of ACM within 30 days for the group without ECD versus the group with ECD was 1.000 (reference) versus HR = 1.374 (95% CI: 1.174-1.608, P < 0.001). When ECD score was classified as four groups (0, 1 or 2, 3 or 4, 5 or 6), the risks of ACM within 30 days were 1.000 (reference), HR = 1.366 (95% CI: 1.162-1.607, P < 0.001), HR = 1.403 (95% CI: 1.141-1.725, P = 0.001), and HR = 1.374 (95% CI: 1.011-1.868, P = 0.042), respectively. Similar trends were detected for ACM within 90 days and 1 year.
Cox Proportional Hazard Models for All-Cause Mortality Within 30 Days, 90 Days, and 1 Year.

Abbreviations: CI = Confidence Interval, HR: Hazard Ratio, CI: Confidence Interval
Model1 was adjusted for none.
Model2 was adjusted for Age, Gender, Race.
Model3 was adjusted for Age, Gender, Race, HR, RR, NBPS, NBPD, Atrial fibrillation, Diabetes, Heart failure, Hypertension, Sepsis, Alteplase, Aspirin, Clopidogrel, CRRT, NIV, Creatinine, Glu, RBC, Sodium, WBC, GCS, LOS.
Stratified Analyses
To further ascertain the consistency of the association between ECD and ACM within 30 days, 90 days, and 1 year across subgroups, we implemented subgroup analyses based on sex, age, hypertension, diabetes, heart failure, atrial fibrillation, and sepsis (Figure 3).

Forest plots of stratified analyses of ECD score and all-cause mortality within 30 days (A), 90 days (B) and 1 year (C).
The results showed that within the age subgroup, the significant association between ECD and the risk of ACM within 30 days, 90 days, and one year was observed exclusively in patients aged <70 years. No significant association was found in patients aged ≥70 years. In the sex, hypertension, and diabetes subgroups, ECD demonstrated broadly and consistently significant associations with the risk of ACM within 30 days, 90 days, and one year. Regardless of sex or the presence of hypertension or diabetes, ECD remained a significant risk factor of mortality. In the heart failure and atrial fibrillation subgroups, significant associations between ECD and the ACM within 30 days, 90 days, and one year were observed only in patients without heart failure and atrial fibrillation; no significant associations were found in patients with these comorbidities. In the sepsis subgroup, ECD showed significant associations with the risk of ACM within 30 days, 90 days, and one year among patients with sepsis. Among those without sepsis, the association was significant at 90 days and 1 year, while it approached statistical significance at 30 days.
Interaction analysis showed that age and atrial fibrillation considerably modified the association, with strong interactions observed in ACM within 30 days, 90 days, and 1 year (P for interaction <0.01). Sex also exerted a significant effect, with strong interactions detected in ACM within 30 days and 1 year (P for interaction <0.01). Notably, hypertension showed a significant interaction only for ACM within one year (P for interaction < 0.05), and its clinical implication should be interpreted cautiously. Interactions in the remaining subgroups (diabetes, heart failure, sepsis) were all non-significant (P for interaction > 0.05).
Discussion
This study is pioneering in developing and validating an ECD scoring system for patients with stroke within a large-scale real-world cohort. The results indicate that patients with stroke and ECD had a significantly higher ACM relative to those without ECD. The severity of ECD is positively correlated with the risk of adverse outcomes. When stratified by ECD scores, the low (score 1–2) and the moderate (score 3–4) groups exhibit similar ACM within 90 days and 1 year. However, the high-score group (5–6) has a significantly higher ACM relative to the low-score group (1–2). Subgroup and interaction analyses further confirmed the stability of results. These findings might inform interventions to reduce mortality among individuals with stroke.
The association between the ECD score proposed in this study and adverse outcomes can be explained by pathological mechanisms underlying secondary hemorrhagic predisposition following stroke. In the context of ischemic stroke, while localized arterial thrombosis relies on platelet adhesion and tissue factor (TF)-mediated coagulation cascade activation, 16 persistent ischemic foci and systemic inflammatory responses may lead to consumptive coagulopathy. Subsequent reperfusion injury, inflammatory reactions, and vascular endothelial damage collectively cause disruption of the blood-brain barrier (BBB) and loss of vascular integrity, 17 thereby exacerbating post-stroke hemorrhagic transformation. Similarly, previous reviews have also noted that chronic coagulation factor deficiencies or dysfunctions increase bleeding risk, thereby elevating the incidence of post-stroke HT. This risk might interact with anticoagulation therapy and altered vascular permeability. 18
As an important pathophysiological mechanism in various systemic diseases, ECD has been extensively studied. A retrospective study detailed the mechanism of SIC and its interaction with the coagulation system. 19 During the COVID-19 pandemic, ECD in COVID-19 patients was closely correlated with elevated risk of stroke. 20 Recently, the association between ECDs and adverse clinical outcomes has received growing attention. For example, ECDs are linked to an elevated risk of severe acute kidney injury within 7 days of ICU admission among individuals with traumatic brain injury 21 and correlated with short-term ACM among individuals with congestive heart failure. 22
In this research, when patients were stratified by age (<70 or ≥70) or atrial fibrillation status, significant differences in ACM within 30 days, 90 days, and 1 year were observed (P < 0.05). This suggests that advancing age promotes chronic low-grade inflammation (inflammaging) and immune senescence (immunosenescence), leading to sustained elevation of circulating pro-inflammatory factors like IL-6 and TNF. 23 Concurrently, endothelial cell dysfunction (endothelial senescence) reduces anticoagulant production (eg, NO and prostacyclin), while upregulating vascular coagulation factor vWF and adhesion molecules (eg, ICAM-1, VCAM-1). This promotes rapid PLT adhesion and coagulation cascade activation, resulting in thrombosis and exacerbated brain ischemia. 24 Increased age, combined with BBB decline and microvascular disease burden (white matter lesions), worsens BBB disruption post-cerebral infarction and elevates risks of hemorrhagic complications and infarct expansion. 25 Studies indicate that emboli originating from atrial fibrillation are often rich in NETs or NET-associated components. 26 Neutrophils release NETs composed of DNA, histones, and pro-coagulant/adhesion proteins during the early stages of stroke. These NETs provide a mesh scaffold for fibrin-PLT clots, enhancing thrombus density and inhibiting fibrinolysis. 27 Local fibrinogen rapidly converts into a dense fibrin mesh interwoven with NETs, PLTs, and RBCs, forming fibrin/NETs-enriched emboli. Such emboli exhibit poor responsiveness to tissue plasminogen activator (tPA) and mechanical thrombectomy, resulting in reduced recanalization rates, increased infarct volumes, and worsened prognosis in patients with stroke. 28
In this study, subgroup analysis by sex revealed significant differences in the effect on the risk of ACM within 30 days and 1 year (P < 0.05), whereas no significant differential effect was observed between sexes for the risk of ACM within 90 days (P > 0.05). This phenomenon can be explained by differences in pathophysiological mechanisms and the nature of outcomes. Sex regulates the immune, endothelial, and coagulation systems through sex hormones, leading to systematic differences between males and females in early thrombo-inflammatory responses and PLT activation, 29 as well as differential expression of thrombosis-related biomarkers such as fibrinogen, vWF, PLT activity, and inflammatory cell responses. 30 These differences collectively influence acute-phase outcomes (approximately 30 days), including recanalization rates, early neurological deterioration, and long-term functional recovery. In contrast, ACM within 90 days is a composite endpoint influenced by numerous non-specific factors, such as infections, cardiogenic complications, organ failure, and post-discharge accessibility to medical care or rehabilitation. 31 These factors might mask the sex-specific effects mediated by early coagulation-immune pathways, resulting in a statistically non-significant interaction. 32
Strengths and Limitations
Unlike previous studies primarily focusing on individual coagulation indicators or coagulation disorders in specific conditions (such as sepsis, trauma, or heart failure), this study centers on developing a simple, universal point-of-care scoring system. This system contributes to risk assessment and stratification of patients’ coagulation status, which has potential clinical significance for early identification of patients who may benefit from targeted anticoagulation or hemostatic therapies. This research has several limitations. First, this research is based on a retrospective cohort design utilizing the MIMIC-IV database, which precludes the establishment of clear causal relationships. Although we adjusted for multiple known confounders, unmeasured residual factors (such as subtypes of stroke, severity of neurological deficit (eg, NIHSS score), and specific therapeutic interventions) cannot be completely ruled out. These factors may simultaneously influence coagulation status and outcomes of patients. Future prospective studies should incorporate these variables to further clarify the independent association between the ECD score and clinical outcomes. What's more, owing to the limitations in the database records, several critical indicators (eg, blood lipids, inflammatory factors) are not available, which limits further interpretation of potential pathological pathways. Moreover, the patient population included in the MIMIC-IV database is primarily from the USA, and the generalizability of the findings to other populations is constrained. Lastly, the PLT count, APTT, and INR values reflect the most extreme values within 24 h of ICU admission. While this reduces the influence of treatment and drug interventions to some extent, bias cannot be completely ruled out.
Conclusion
This study demonstrates a significant association between the ECD score and ACM within 30 days, 90 days, and 1 year among critically ill patients with stroke. Higher ECD scores were associated with increased mortality, indicating that coagulation abnormalities may be linked to poorer outcomes in this population. These findings suggest that the ECD score may serve as a clinically relevant marker for evaluating coagulation status and its association with outcomes in critically ill patients with stroke.
Supplemental Material
sj-docx-1-cat-10.1177_10760296261428819 - Supplemental material for Association Between Early Coagulation Disorder and All-Cause Mortality in Critically Ill Stroke Patients: A Retrospective Study Based on the MIMIC-IV Database
Supplemental material, sj-docx-1-cat-10.1177_10760296261428819 for Association Between Early Coagulation Disorder and All-Cause Mortality in Critically Ill Stroke Patients: A Retrospective Study Based on the MIMIC-IV Database by Yezhen Qin, Xiaoqin Han, Min Xia, Yanzi Zhang and Mei Wei in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-2-cat-10.1177_10760296261428819 - Supplemental material for Association Between Early Coagulation Disorder and All-Cause Mortality in Critically Ill Stroke Patients: A Retrospective Study Based on the MIMIC-IV Database
Supplemental material, sj-docx-2-cat-10.1177_10760296261428819 for Association Between Early Coagulation Disorder and All-Cause Mortality in Critically Ill Stroke Patients: A Retrospective Study Based on the MIMIC-IV Database by Yezhen Qin, Xiaoqin Han, Min Xia, Yanzi Zhang and Mei Wei in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgements
Not applicable.
Ethical Approval and Informed Consent Statements
Ethical review and approval were waived for this study, as it was conducted using publicly available data from MIMIC-IV (version 3.1) database. Approval for the research use of these data was obtained from the BIDMC Institutional Review Board.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Author Contributions
QYZ Conceptualization, Methodology, Data Curation, Formal Analysis, Writing – Original Draft. HXQ: Software, Validation, Data Curation, Visualization, Writing – Review & Editing. XM: Investigation, Resources, Writing – Review & Editing. ZYZ: Literature Review, Data Curation, Project Administration. WM: Conceptualization, Supervision, Writing – Review & Editing, Correspondence.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
