Abstract
Objective
To explore the factors affecting the Time in Therapeutic Range (TTR) of warfarin anticoagulation therapy and propose a novel TTR prediction model.
Methods
Patients with atrial fibrillation who received warfarin anticoagulation therapy and had complete follow-up data between January 2020 and September 2021 at The First Hospital of Jiujiang City were retrospectively included. All participants were divided into a high quality TTR group and a non high quality TTR group based on whether TTR ≥70%. The predictive value of clinical indicators for TTR was assessed utilizing the area under the receiver operating characteristic curve (AUC).
Results
A total of 136 patients were included in the final analysis. The results showed that PT, RDW, PDW, RAVD, and NXT were significantly associated with TTR. The combination model of PT, RDW, PDW, RAVD, and NXT resulted in the highest AUC of 0.900 (95% CI: 0.848-0.952) with the specificity (0.835) and sensitivity (0.843).
Conclusion
PT, RDW, PDW, RAVD, and NXT were important influencing factors for TTR in warfarin anticoagulation therapy. Combining these clinical indicators can effectively predict the TTR of warfarin anticoagulation therapy.
Introduction
Atrial fibrillation is a prevalent cardiac arrhythmia observed in adults, with a prevalence ranging from 2% to 3% within the populations of Europe and North America. 1 It is also a well-recognized risk factor for the development of thrombus, making anticoagulation therapy essential for patients with atrial fibrillation, 2 moreover, nonvalvular atrial fibrillation (NVAF) is the most common cardiac arrhythmia in association with an increased risk of stroke and systemic thromboembolism. 3 Warfarin, as the cornerstone of anticoagulation therapy, has been proven effective in reducing the incidence of thrombotic events and is widely utilized in various clinical scenarios. It is appropriate for patients with a range of conditions, including atrial fibrillation, cancer, and for those requiring anticoagulation following surgical procedures.4,5 However, despite the extensive clinical application of warfarin, challenges persist in its management, including suboptimal patient adherence 6 and insufficient efficacy in maintaining International Normalized Ratio (INR) control. 7
Guidelines recommend the use of warfarin anticoagulation intensity, measured by the INR, and the stability of anticoagulation, assessed through time in therapeutic range (TTR), as two key aspects for evaluating treatment efficacy.8,9 The INR is routinely monitored in patients undergoing warfarin treatment; however, due to the complex pharmacokinetic and pharmacodynamic characteristics of warfarin, the quality of anticoagulation control necessitates further evaluation using TTR. TTR is defined as the percentage of time a patient's INR remains within the desired treatment range or goal. 10 Nonetheless, the assessment of TTR requires long-term INR monitoring, which limits its clinical applicability and has consequently resulted in a relative paucity of research focused on TTR and its applications, in comparison to studies on anticoagulation intensity and INR. Therefore, the development of an accurate TTR prediction model is of paramount importance.
In recent years, the advancement of molecular biology technology and the diversification of databases, guided by clinical studies, have facilitated the identification of several factors closely associated with the stability of warfarin anticoagulation. These factors include sex, 10 heart structure, 11 hepatorenal dysfunction, 12 red blood cell volume distribution width (RDW) 13 and the utilization of proprietary Chinese medicine. 14 Furthermore, certain indices may serve as predictors for TTR events. Consequently, the primary TTR scoring systems, including the SAMe-TT2R2 score and the CHADS2 scoring system, 15 have integrated various clinical and hematological indicators for TTR prediction; however, their practical application and promotional value remain notably limited. 16 Imaging indicators represent another critical aspect of disease prediction. For instance, Miyoshi et al demonstrated that transthoracic echocardiography (TTE) can estimate the adiposity of the septum in patients with atrial fibrillation, which is significant for prognostic evaluation. 17 Nevertheless, current scoring systems for TTR evaluation do not incorporate imaging parameters, which are readily accessible in clinical settings and vital for assessing cardiac function. Therefore, this study aims to explore the factors affecting warfarin anticoagulation therapy's stability and propose a novel TTR prediction model.
Materials and Methods
Study Design and Patients
Patients with atrial fibrillation who received warfarin anticoagulation therapy at the First Hospital of Jiujiang City between January 2020 and September 2021 were retrospectively included in the study. Inclusion criteria: (1) patients diagnosed with persistent NVAF (PNVAF) defined by the 2023 Chinese Atrial Fibrillation Guideline; 9 (2) the follow-up period was more than 6 months and the medical records were complete; (3) adherence to warfarin during follow-up. Exclusion criteria: (1) patients with malignant tumors, acute cardiac insufficiency, severe liver and kidney dysfunction, diabetes, high-risk of bleeding, abnormal platelet count, pulmonary embolism or vein thrombosis, usage of drugs that could affect the platelet count, history of surgeries on large blood vessels or other operation during follow-up period; (2) accompanied with other diseases during follow-up period (including acute heart failure, liver enzyme elevations exceeding three times the upper limit of the normal reference range, tumors, bleeding events, or underwent interventional diagnostic procedures or treatments) or drugs (including amiodarone, antifungal Drugs, non-steroidal anti-inflammatory drugs) that could affect the anticoagulant effect of warfarin; (3) individuals with a history of psychiatric disorders such as anxiety, depression, or schizophrenia, as well as those taking psychotropic medications during the follow-up period; (4) incomplete examination data and follow-up data; (5) Switching to new oral anticoagulants during the follow-up period.
This study was approved by the Medical Ethics Committee of the First Hospital of Jiujiang City, with the approval number JJSDYRMYY-YXLL-2021-090. Due to the retrospective nature of the study, the Medical Ethics Committee of the First Hospital of Jiujiang City waived the requirement for patient informed consent.
Data Collection
Clinical data were extracted from the institutional medical history system, incorporating variables such as age, gender, and cardiovascular comorbidities, which include hypertension (HBP), coronary heart disease (CHD), and stroke. Additionally, drug usage status was documented, including the use of antiplatelet drugs, ACE inhibitors/angiotensin receptor blockers (ACEI/ARB), beta-blockers, calcium channel blockers (CCB), statins, and Naoxintong (NXT). Laboratory examination results were also recorded, including white blood cell (WBC) count, red blood cell (RBC) count, hemoglobin (Hb) levels, platelet (PLT) count, platelet distribution width (PDW), mean platelet volume (MPV), average red blood cell volume distribution width (RDW), mean corpuscular volume (MCV), triglyceride (TG) levels, total cholesterol (TC) levels, high-density lipoprotein (HDL), low-density lipoprotein (LDL), apolipoprotein A (ApoA), apolipoprotein B (ApoB), lipoprotein(a), free fatty acids (FFA), prothrombin time (PT), activated partial thromboplastin time (APTT), fibrinogen (Fib), thrombin time (TT), D-dimer levels, triiodothyronine (T3), tetraiodothyronine (T4), and thyroid-stimulating hormone (TSH). Furthermore, imaging indicators recorded included left atrial anteroposterior diameter (LAAPD), left atrial left and right diameter (LALRD), left atrial vertical diameter (LAVD), right atrial left and right diameter (RALRD), right atrial vertical diameter (RAVD), left ventricular end-diastolic diameter (LVDD), left ventricular end-systolic diameter (LVESD), left ventricular ejection fraction (LVEF), and stroke volume (SV).
Stability of Warfarin Anticoagulation
In accordance with the recommendations set forth by the Chinese atrial fibrillation guidelines, 8 the assessment of TTR necessitates a follow-up period of no less than six months of INR monitoring (INR = Patient PT/Control PT × ISI, where ISI denotes the International Sensitivity Index). The monitoring of INR was performed based on following criteria: (1) Relatively sufficient data in the first 6 weeks (ie, although it was deleted in the calculation of TTR, it must be available), INR was tested at least once a week with INR values of 5 to 8 times; (2) INR results were fed back every month, and INR data distribution was relatively equal during the 3 and 6 months of follow-up (ie, data in the first 6 weeks and each month were guaranteed).
The process for calculating TTR was as follows: Assuming the INR fluctuated uniformly daily, TTR was computed using the Rosendaal method, which involved: (1) Determining intervals within the therapeutic range of 2.0 to 3.0; (2) Calculating the percentage of these intervals within the range; (3) Estimating days within the range since the last follow-up. To derive the overall percentage of time within the therapeutic range, the total days within the range across each period were aggregated and divided by the total number of treatment days. 18 As recommended by the guidelines, patients were further categorized into a high quality TTR group (TTR ≥ 70%) and a non high quality TTR group (TTR < 70%). The minimum required INR data for this study was 6 months, because TTR can be calculated and changed dynamically, and some case are still been follow up, so there is no maximum follow-up time. Cases with missing TTR data were directly excluded from the study.
Statistical Analysis
Statistical analysis was performed using SPSS22.0 (IBM Corp., Armonk, New York) and GraphPad Prism 8.0.1 (GraphPad Software, San Diego, California, USA). Continuous variables following a normal distribution were expressed as (x ± s) and analyzed using the t-test. Categorical data was expressed as frequency (%) and compared using a chi-square test. Binary Logistic regression analysis was used to explore the related influencing factors of TTR. To strictly exclude colinear features, which could impair the performance of multivariate regression analysis, collinearity between the influencing factors was calculated, and features with a VIF > 2 were excluded for further analysis. The receiver operating characteristic (ROC) curve was drawn to evaluate the predictive value for the prediction of TTR. A two-sided
Results
A total of 236 patients between January 2020 and September 2021 were screened, after exclusion of patients with concomitant admiodarone use (n = 2), malignancy (n = 2), incomplete follow-up data (n = 19), experienced adverse events during follow-up (n = 22), and patients who did not follow treatment plan during follow-up, including non-adherence to Warfarin (n = 26), significant change in concomitant medication (n = 3), heavy alcohol consumption (n = 7), significant change in diet (n = 10), insufficient / missing initial 6 weeks’ INR data (n = 22), and INR data interval > 1 month (n = 26) (Figure 1). Finally, 136 patients with atrial fibrillation receiving warfarin anticoagulation therapy were included for analysis, of which 51 patients (37.5%) were in the high quality TTR group. Compared to the high quality TTR group, the patients in the non high quality TTR group showed lower CHD rate (21.2% vs 54.9%,

Flowchart for participant selection.
Baseline Characteristics of Participants.
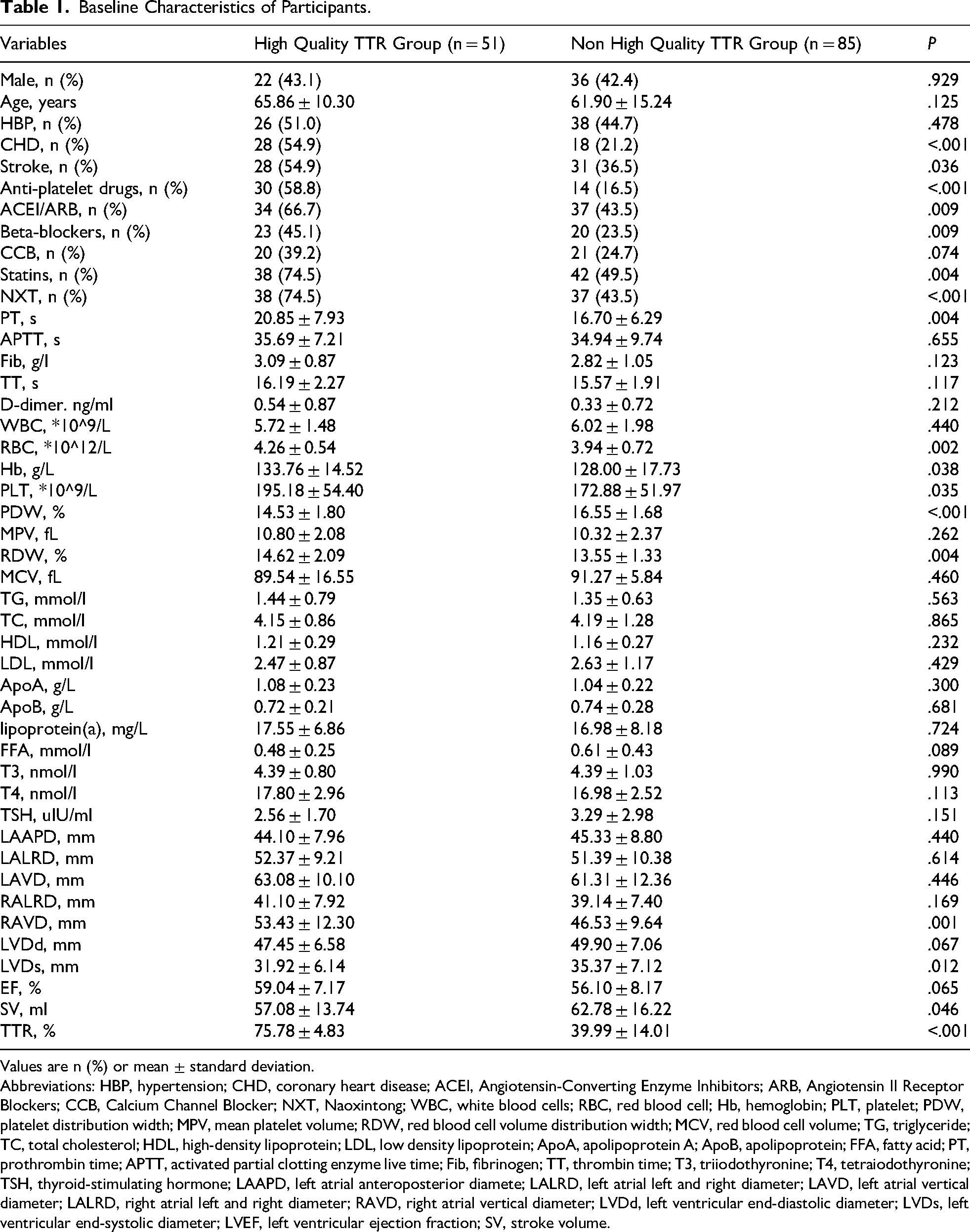
Values are n (%) or mean ± standard deviation.
Abbreviations: HBP, hypertension; CHD, coronary heart disease; ACEI, Angiotensin-Converting Enzyme Inhibitors; ARB, Angiotensin II Receptor Blockers; CCB, Calcium Channel Blocker; NXT, Naoxintong; WBC, white blood cells; RBC, red blood cell; Hb, hemoglobin; PLT, platelet; PDW, platelet distribution width; MPV, mean platelet volume; RDW, red blood cell volume distribution width; MCV, red blood cell volume; TG, triglyceride; TC, total cholesterol; HDL, high-density lipoprotein; LDL, low density lipoprotein; ApoA, apolipoprotein A; ApoB, apolipoprotein; FFA, fatty acid; PT, prothrombin time; APTT, activated partial clotting enzyme live time; Fib, fibrinogen; TT, thrombin time; T3, triiodothyronine; T4, tetraiodothyronine; TSH, thyroid-stimulating hormone; LAAPD, left atrial anteroposterior diamete; LALRD, left atrial left and right diameter; LAVD, left atrial vertical diameter; LALRD, right atrial left and right diameter; RAVD, right atrial vertical diameter; LVDd, left ventricular end-diastolic diameter; LVDs, left ventricular end-systolic diameter; LVEF, left ventricular ejection fraction; SV, stroke volume.
Univariate analysis indicated that the presence of comorbid coronary heart disease, comorbid stroke, and the concurrent administration of anti-platelet drugs, ACEI/ARB, Beta-blockers, statins, and NXT, PT, RBC, Hb, RDW, RAVD, LVDs were potential factors influencing TTR (all
Univariate Analysis of Risk Factors of Poor TTR Status.

Abbreviations: CHD, coronary heart disease; ACEI, Angiotensin-Converting Enzyme Inhibitors; ARB, Angiotensin II Receptor Blockers; NXT, Naoxintong; RBC, red blood cell; PT, prothrombin time; Hb, hemoglobin; PLT, platelet; PDW, platelet distribution width; RDW, red blood cell volume distribution width; RAVD, right atrial vertical diameter; LVDs, left ventricular end-systolic diameter; SV, stroke volume.
To eliminate the potential impact of multicollinearity among exposure factors on the stability and accuracy of the model, factors with a VIF greater than 2, including RBC, Hb, CHD, stroke, antiplatelet drugs, and statins, were excluded for further analysis (Table 3). The multivariable logistic regression analysis revealed that PT [OR: 1.123, 95% CI: 1.044-1.208,
Colinear Diagnosis of Potential Risk Factors.

Abbreviations: CHD, coronary heart disease; ACEI, Angiotensin-Converting Enzyme Inhibitors; ARB, Angiotensin II Receptor Blockers; NXT, Naoxintong; RBC, red blood cell; PT, prothrombin time; Hb, hemoglobin; PLT, platelet; PDW, platelet distribution width; RDW, red blood cell volume distribution width; RAVD, right atrial vertical diameter; LVDs, left ventricular end-systolic diameter.
Multivariate Analysis of the Factors Affecting TTR status.
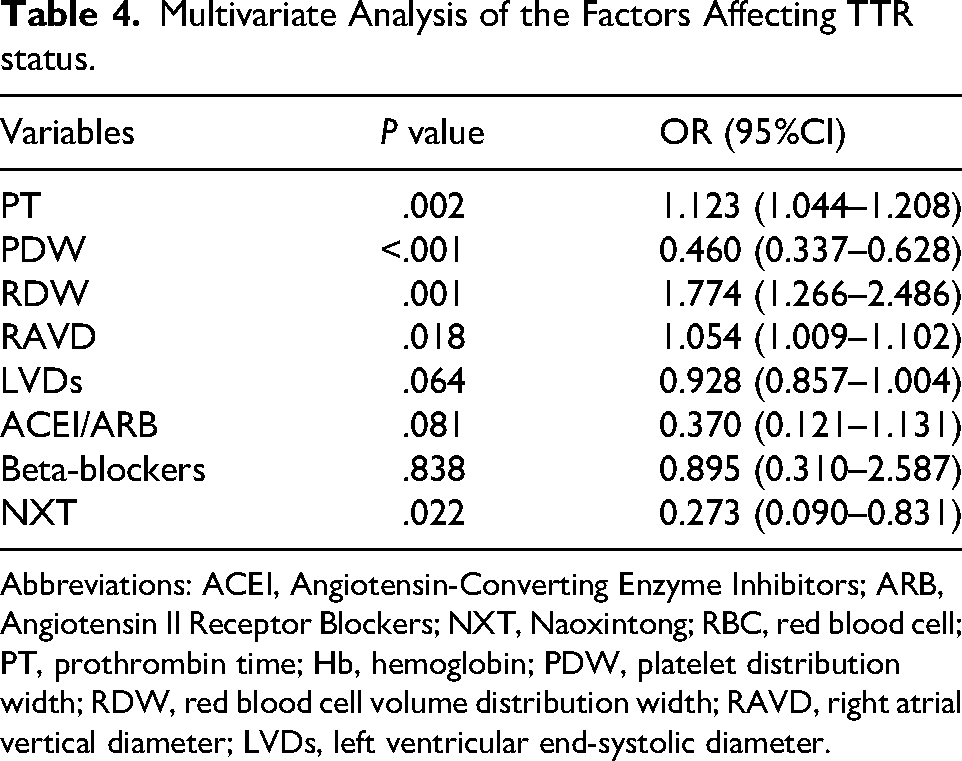
Abbreviations: ACEI, Angiotensin-Converting Enzyme Inhibitors; ARB, Angiotensin II Receptor Blockers; NXT, Naoxintong; RBC, red blood cell; PT, prothrombin time; Hb, hemoglobin; PDW, platelet distribution width; RDW, red blood cell volume distribution width; RAVD, right atrial vertical diameter; LVDs, left ventricular end-systolic diameter.
The predictive value of separate parameter was examined and the results showed that PDW had the highest AUC of 0.797 (95%CI: 0.715 - 0.879), while the AUCs for PT, RDW, RAVD, NXT were 0.637 (95%CI: 0.541 - 0.733), 0.645 (95%CI: 0.549 - 0.742), 0.628 (95%CI: 0.534 - 0.722), and 0.655 (95%CI: 0.561 - 0.749), respectively.
After combination of hematological, and imaging indicators, a combined model was developed and the AUC reached 0.900 (95%CI: 0.848 - 0.952), the specificity (0.835) and sensitivity (0.843) were also the highest among all models (Figure 2 and Table 5).

Receiver operating curves using different parameters for the prediction for high-quality TTR.
AUCs of Different Parameters for Prediction of TTR Status.

Abbreviations: PDW, platelet distribution width; RDW, red blood cell volume distribution width; RAVD, right atrial vertical diameter.
Discussion
This study elucidated that clinical indicators such as PT, PDW, RDW, RAVD, and NXT serve as independent predictors of TTR. The integration of these parameters—PT, PDW, RDW, RAVD, and NXT—demonstrated a robust capacity for predicting TTR in patients undergoing warfarin anticoagulation therapy. This model offered a valuable framework for anticipating anticoagulation outcomes in individuals diagnosed with atrial fibrillation.
Hematological indicators were significant for the evaluation of hemodynamics under various situations. Guan et al pointed out that evaluated the RDW, neutrophil to lymphocyte ratio, and platelet to lymphocyte ratio in patients of AF and revealed that elevated RDW level is associated with AF, moreover, they found RDW > 14.1% is a risk factor for AF, but its diagnostic capacity for AF is not of great value, 19 while Song et al showed that lipoprotein(a) is elevated in high-risk AF patients and lipoprotein(a) could be an emerging biomarker for early-identification of high-risk AF patients, 20 Wang Z pointed out that Hb and RDW can be used to assist in the judgment of blood turbulence and thrombotic events, and this part of the conclusion is consistent with this study to a certain extent. 21 In the present study, PT, RBC, Hb, PLT, PDW, and RDW were significantly different between patients in high quality TTR and non high quality TTR groups, and all of them were potential risk factors, although after multivariate analysis, only PDW, and RDW were independent risk factors. Compared to RDW, PDW was less studied but also an important influencing factor in AF, Lyu showed PDW was an important risk factor for stroke in AF patients, 22 and a meta-analysis performed by Alexander also proved that PDW was different among patients with paroxysmal, persistent and permanent atrial fibrillations. 23 However, the platelet characteristics still require further research.
Cardiac magnetic resonance (CMR) parameters were also important in the assessment of cardiac function and prognosis. Minamisawa M et al also pointed out that different left atrial morphology can have different effects on blood circulation, 24 while Saraf et al pointed out that the probability of thrombosis may increase when the atrial is in the state of like spheroid. 25 This study pointed out that among various morphological parameters, only larger RAVD is independently associated with high quality TTR. However, there is no report on the relationship between right atrial morphology and the anticoagulation effect of warfarin and predicting thrombotic events, one underlying reason could be the technical difficulties in the evaluation of anteroposterior diameter and the upper and lower diameter of the atrium. Based on the data of this study, the authors suggest that medical departments capable of accurate evaluation of arterial parameters should strengthen the measurement and medical exploration of different atrial diameters.
In this study, we observed that patients receiving combined NXT therapy appeared to achieve a more stable anticoagulation effect, as reflected by a higher TTR. And this finding was in alignment with some previous studies. Lu et al previously proposed that NXT has an obvious function in antithrombotic, lipid regulation, and improvement of atherosclerosis, which is expected to become an important role in the prevention and management of cardiovascular and cerebrovascular diseases. 26 Yang W et al pointed out that NXT has a bidirectional regulation effect on the coagulation process. When NXT is used in combination with warfarin, it may affect the metabolic enzymes CYP1A2 and CYP3A4, and then affect the inhibition of the synthesis of coagulation factors by affecting the metabolism of warfarin, and finally participate in the regulation of the coagulation process.27,28 Shen Y et al pointed out that NXT may affect platelet aggregation by regulating the ERK/MAPK signaling pathway to play an antithrombotic role, 29 Luo and colleagues also showed that NXT is involved in the coagulation regulation of warfarin through the MAPK pathway in rats. 30 A clinical observational study by Gao et al showed that in the process of anticoagulant therapy with warfarin, NXT combined with warfarin could reduce the risk of bleeding by reducing the dose of warfarin under the premise of ensuring the INR reached the standard. 31 The study by Huang's team showed that NXT had no significant effect on the INR of patients, but it had a positive significance for INR stability during long-term follow-up. 32 All these studies showed that NXT has potential to be an effective drug for the management of patients who received warfarin treatment, NXT has a relatively obvious antithrombotic effect, and its effect may be bidirectional regulation, but under what circumstances the application of NXT will enhance or weaken the effect of warfarin needs further exploration. Moreover, the results was based on an observational association, and the underlying mechanisms require further investigation and confirmation.
At present, the main calculation methods of TTR include the calculation of days to reach the target and the calculation of times to reach the target. The former is gradually not used in clinical practice due to the complexity of the calculation process, and the latter also has many limitations due to the requirement of “excluding INR data of the first 6 weeks and the calculation cycle is not less than 6 months” in the guidelines. The exclusion of the first 6 weeks of INR data often ignores the most real antithrombotic situation in the initial stage of medication, and the requirement of no less than 6 months of follow-up data undoubtedly reduces the participation and follow-up awareness of patients in the first 6 months of antithrombotic therapy, which undoubtedly has a negative significance for patients taking warfarin.
8
In our previous study, the author found that the “double 6 definition” did not seem to have a significant effect on the calculation of TTR, it is worth thinking whether the calculation method of TTR should be adjusted. The existing TTR evaluation method is mainly the SAMe-TT2R2 scoring system. However, because “
This study still had several limitations, firstly, this was a single-center retrospective study with less than 200 patients, the results of the present study should be further confirmed using a large sample-size prospective cohort. Secondly, our proposed model can only predict the high quality or no high quality TTR, a linear regression model which enables the prediction of exact TTR should be developed in the future for quantitative prediction of TTR. Finally, due to the retrospective nature of this study, some important confounding factors (including diet, alcohol consumption, other lifestyle factors etc) were not obtained and included in the study, in the future prospective study, we will design a questionnaire to include all these important factors.
In conclusion, PT, RDW, PDW, RAVD, and NXT were important influencing factors for TTR in warfarin anticoagulation therapy. Combining these indicators can effectively predict the TTR of warfarin anticoagulation therapy. Integrating clinical indicators such as PT, PDW, RDW, RAVD, and NXT into TTR management has broader applications and impactful potential, promising to become novel biomarkers in the TTR evaluation scoring system.
Footnotes
Ethics Approval and Consent to Participate
I confirm that all methods were performed in accordance with the relevant guidelines. This work has been carried out in accordance with the Declaration of Helsinki (2000) of the World Medical Association. This study was approved by the Medical Ethics Committee of the First Hospital of Jiujiang City, with the approval number JJSDYRMYY-YXLL-2021-090. Due to the retrospective nature of the study, the Medical Ethics Committee of the First Hospital of Jiujiang City waived the requirement for patient informed consent.
Authors’ Contributions
Qibao Zhou and Xiao Luo carried out the studies, participated in collecting data, and drafted the manuscript. Qibao Zhou and Xiao Luo performed the statistical analysis and participated in its design. Qibao Zhou and Xiao Luo participated in acquisition, analysis, or interpretation of data and draft the manuscript. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
All data generated or analyzed during this study are included in this article and supplementary information files.
