Abstract
Vitamin K antagonists, such as warfarin, have a narrow therapeutic window; patients on these therapies therefore require regular international normalized ratio (INR) monitoring to maintain optimal dosing. This involves periodic checks and laboratory testing using venepuncture, which are often perceived as a burden. This study aimed to determine the accuracy and precision of the LumiraDx INR Test, a new point-of-care in vitro diagnostic platform, in an anticoagulation clinic setting. In this observational, cross-sectional study, precision of the LumiraDx INR Test was assessed using paired replicate samples (n = 366) and 3 test strip lots. Accuracy was determined by comparing capillary blood INR, ascertained by the LumiraDx INR Test, with venous plasma INR, measured by the laboratory reference instrument, the IL ACL ELITE Pro. Furthermore, INR was assessed across a range of hematocrit (25%-55%). In addition, feedback was collected from health-care professionals via a self-completed questionnaire. This trial was registered at ClinicalTrials.gov (NCT03682419). The precision (% coefficient of variation) of the LumiraDx INR Test was <4 when samples were applied by direct application or via a capillary transfer pipette, as well as between test strip lots. Accuracy of the LumiraDx INR Test, across the INR range of 0.8 to 7.5, was confirmed by a strong correlation of 0.965 (95% confidence interval: 0.959-0.970) when compared with the IL ACL ELITE Pro, which was maintained across the hematocrit range. Feedback from health-care professionals indicated that the instructions given by the system were easy to follow. In conclusion, the strong agreement between the LumiraDx Platform INR point-of-care test and the IL ACL ELITE Pro laboratory reference system, as well as between the different application methods and test lots, indicates that it can provide a rapid, accurate, and reliable INR analysis.
Keywords
Introduction
Vitamin K antagonists (VKA) oral anticoagulation therapies, such as warfarin, have been used in primary and secondary care for more than 60 years, and both the short-term and long-term effects in patients have been well established. 1 VKAs are guideline-recommended drugs for the treatment of venous thromboembolism (VTE), the prevention of thrombotic events (TEs) in patients with atrial fibrillation (AF), and the management of patients with mechanical heart valves. 2 –4 One of the main disadvantages of VKA therapy is the narrow therapeutic window; the effectiveness of treatment, to prevent bleeding complications or TEs, is highly dependent on dose management. The anticoagulant effect of VKAs must be closely monitored using the international normalized ratio (INR) as standard, to typically maintain patients with VTE and AF within an INR target range of 2.0 to 3.0 and patients with mechanical heart valves within the target range of 2.5 to 3.5. 2,5
INR values are derived from the international sensitivity index, which is calculated as a ratio of the patient’s prothrombin time (PT) to a control PT with the assay method and reagent standardized by the World Health Organization (WHO) standard. 6,7 Dose management of VKA therapy is challenging as an INR can be affected by the patient’s genetic background, comorbid conditions, other drug interactions, and lifestyle. 6,7 To assess the effectiveness of VKA therapy for a given patient, the time in therapeutic range (TTR) has become a standard measure; TTR represents the percent of time the INR remains within target range across time. 8 The prerequisite to optimal VKA therapy using TTR is to ensure the correct assessment of a patient’s INR, which is dependent on high accuracy and precision of the method and device used. 9
There are multiple factors that can affect INR control, such as vitamin K supplementation, drug interactions, and alcohol consumption. 10 Therefore, patients on VKA treatment must undergo periodic checks (in our local anticoagulant service, the average time between testing is 23 days)—these periodic checks and laboratory testing using venipuncture are often perceived as a burden. 3,11 –13 However, the monitoring of patients receiving VKA treatment has been greatly assisted by the introduction of point-of-care (POC) coagulometers for analyzing INR using small volumes of capillary whole blood, at the patient side. 14 The VKA therapy can be managed in different care settings, such as at the general practitioner’s office, hospital outpatient clinics, and specialized anticoagulation clinics, but also in the patient’s home through patient self-testing and patient self-management. 9 The POC coagulometers used in a clinical setting enable rapid clinical decision-making, by providing quicker turnaround time compared with laboratory testing. 14 From a quality control perspective, the comparison of POC coagulometers with a gold-standard laboratory reference method is paramount to assure accuracy and precision and to keep within the narrow therapeutic range of VKA drugs. 9
The LumiraDx Platform is a POC system designed for the measurement of multiple parameters near the patient using small volume blood sampling methods, such as capillary blood from fingersticks. The objective of this study was to evaluate the accuracy and precision of the new LumiraDx Platform INR Test in a POC setting using both direct application and pipette transfer of capillary blood to the test strips for potential inclusion in future routine clinical practice.
Methods
Study Population and Sample Preparation
This study was conducted at 11 anticoagulation clinical sites across the Glasgow and Clyde and Lanarkshire regions (Glasgow Royal Infirmary, Glasgow, United Kingdom; Queen Elizabeth University Hospital, Glasgow, United Kingdom; Golden Jubilee National Hospital, Glasgow, United Kingdom; University Hospital Hairmyres, East Kilbride, United Kingdom; Airdrie Community Health Centre, Airdrie, United Kingdom; Central Health Centre, Cumbernauld, United Kingdom; Central Health Centre, Hamilton, United Kingdom; Kilsyth Community Health Centre, Kilsyth, United Kingdom; Motherwell Health Centre, Motherwell, United Kingdom; Stonehouse Hospital, Stonehouse, United Kingdom; Wishaw Health Centre, Wishaw, United Kingdom). All anticoagulation sites were provided with posters with contact numbers so that patients who wanted to participate in the study could find out further details and sign-up. The poster was also printed and handed out to patients as a flyer at anticoagulation clinics. In addition, several different recruitment methods were used across the sites (ie, patient invitations were sent out, nurses and other health-care providers recruited patients for direct testing and subsequent appointments at anticoagulation clinics, and some patients were prescreened for eligibility based on anticoagulation lists).
Venous whole blood samples were analyzed at the laboratory of LumiraDx UK Ltd in Stirling, United Kingdom. The study protocol complied with the Declaration of Helsinki (2013) and was approved by the East of Scotland Research Ethics Service (REC reference number: 17/ES/0161). All participants gave written informed consent for POC and laboratory testing. This trial was registered at ClinicalTrials.gov (NCT03682419).
A total of 420 participants were enrolled in the study between September and November 2018. Of these, 394 received VKA therapy (warfarin), and a further 26 individuals—who fulfilled the study inclusion criteria but did not receive VKA therapy—were included in order to test the lower limits of detection of the LumiraDx INR Test. All participants were aged older than 18 years and deemed medically appropriate for study participation by the investigator (eg, patients without known inherited [eg, hemophilia or von Willebrand disease] or acquired [eg, liver cirrhosis] conditions that are likely to be associated with a coagulopathy; or patients receiving non-VKA anticoagulant medication). Individuals were excluded if they had previously participated in this study, were within the first 4 weeks of receiving VKA therapy (since most patients are initiated on warfarin using a slow induction protocol and will rarely achieve therapeutic INR levels during the first few weeks of treatment), or had confirmed or suspected pregnancy. A detailed list of eligibility criteria can be found in the Supplementary Material.
Trained clinical staff obtained 2 fingerstick samples of capillary blood from the patients, which were applied directly to test strips for immediate duplicate measurements of INR on the LumiraDx Platform; 2 similar fingerstick measurements were performed using application via capillary transfer pipettes. If the blood sample was inadequate (eg, an insufficient volume of blood), the LumiraDx INR Test showed an error on the screen. During the same study visit, venous whole blood was collected in 2 anticoagulation collection tubes; one whole blood tube was used for 2 measurements of hematocrit on the HemoCue Hb 201+, the second whole blood tube was processed to plasma for subsequent measurement of INR with the IL ACL ELITE Pro (Instrumentation Laboratory, Bedford, MA) laboratory reference system. The IL ACL ELITE Pro used the lyophilized recombinant thromboplastin, HemosIL, ReadiPlasTin (Instrumentation Laboratory, Lexington, MA).
Investigative Device
The LumiraDx INR Test is an in vitro diagnostic medical test that is used to provide INR based on quantitative PT of capillary whole blood. The test is intended for professional POC use for the monitoring of patients on anticoagulation therapy with VKA drugs. The LumiraDx INR Test is a thrombin activation assay in which a quenched substrate is cleaved by thrombin and the emitted fluorescence is detected and quantified. When a blood sample is applied to the test strip, coagulation is activated by the recombinant thromboplastin, HemosIL, ReadiPlasTin (a derivative of RecombiPlastin 2G, containing tissue factor and phospholipids). Coagulation proceeds naturally in the presence of ReadiPlasTin converting prothrombin to thrombin, which subsequently recognizes a peptide sequence on the substrate (Rhodamine). Following cleavage of this peptide sequence, the substrate becomes unquenched and emits a fluorescent signal detectable by the LumiraDx instrument. The amount of signal detected over a specific time is converted by means of an algorithm into INR and the result is displayed on the touch screen. The INR test results are displayed in 60 to 90 seconds. The LumiraDx INR Test was calibrated using the WHO international standard rTF/09 to provide traceability to reference systems and the LumiraDx INR Test. A complete description of the LumiraDx Platform and INR test can be found at the LumiraDx website. 15
Determination of Precision, Accuracy, and Overall Agreement
Precision was measured by correlating duplicate repeated measurements of samples that were either directly applied to the test strips or applied using transfer pipettes. Three test strip lots were used to complete the study. The accuracy of the LumiraDx INR Test using capillary whole blood was assessed by comparing test results with paired venous plasma samples measured using the IL ACL ELITE Pro. The accuracy of the LumiraDx INR Test was assessed across the range of hematocrit 25% to 55% (as measured by HemoCue Hb 201+ System [HemoCue]) using venous whole blood.
Professional User Acceptability Questionnaire
A questionnaire consisting of 29 questions was completed by the health-care professionals who were involved in testing at each of the sites of the study, which was intended to assess the ease of use of the LumiraDx INR Test using a 5-point scale (1 = agree strongly, 2 = agree somewhat, 3 = neither agree nor disagree, 4 = disagree somewhat, and 5 = disagree strongly; Supplementary Material).
Statistical Analysis
Statistical analysis was performed using R version 3.5.1. The precision of the INR measurements performed on the LumiraDx Platform (samples applied directly or via transfer pipettes) was calculated using the mean paired replicate % coefficient of variation (%CV); an analytical variability of ≤5% CV was considered high precision. 16,17 A Passing-Bablok regression analysis was used to compare the INR results with results of the IL ACL ELITE Pro reference instrument to determine the accuracy of the LumiraDx INR Test. The Food and Drug Administration (FDA)–recommended accuracy standards were reviewed at a workshop in March 2016. 18 The FDA recommends that 95% of samples in the INR range from 2.0 to 4.5 should fall within 20% of the reference method and 25% above an INR of 4.5. The accuracy of INR measurements across the range of hematocrit was assessed using linear regression analysis.
Results
A total of 420 patients were enrolled in this study across the research sites. Of these, 54 patients were excluded for the following reasons: transposition error when processing samples (n = 2); being deemed medically inappropriate by the investigator (n = 1); retrospective exclusion by not meeting the inclusion criteria due to a diagnosis of hemophilia A (factor VIII deficiency; n = 1); instrument software update rendering data collected earlier unusable (n = 6); and recalibration of the ACL ELITE Pro reference system meant that paired samples of capillary blood and venous plasma were incomplete and method comparison analysis not possible (n = 44). The INR results from 366 individuals (45% female), with a mean age of 69 years (range, 29-94 years) were included in the analysis, which consisted of a population in receipt of VKA treatment (all on warfarin; n = 340; median INR 2.48, interquartile range [IQR]: 2.07-3.02) and a population without VKA treatment (n = 26; median INR 0.92, IQR: 0.90-0.94). The demographics of the 2 study populations and an analysis of reference INR values are listed in Table 1. Of the 366 patients whose data were included, complete sample sets were not always collected due to patient sampling errors or patients unable to provide all requested blood samples.
Demographic Data Relating to Age and Sex of Recruited Individuals.a

Abbreviations: INR, international normalized ratio; VKA, vitamin K antagonist.
aA subpopulation of 26 individuals not receiving VKA treatment was included in this study in order to test the lower limits of detection of the LumiraDx INR Test.
The precision of the LumiraDx INR Test was calculated using the mean paired replicate %CV. The means of each of the 3 test strip lots demonstrate that the %CV was ≤4 (lot 5000003, n = 167, mean INR 2.42, mean %CV 3.53; lot 5000004, n = 219, mean INR 2.65, mean %CV 3.71; lot 5000006, n = 189, mean INR 2.49, mean %CV 3.52), indicating a high precision of measurements of the different test strip lots (Table 2). Further analysis showed that the analytical variability between different application methods, that is, direct application and via capillary transfer pipette, was ≤4%CV (direct application, n = 284, mean INR 2.54, mean %CV 3.46; transfer pipette, n = 291, mean INR 2.53, mean %CV 3.73). This indicates that the sample application area on the test strip is capable of accepting fingerstick blood samples with high precision directly from a fingertip or using the capillary transfer tube (Table 3).
Precision of INR Measurements Using 3 Test Strip Lots.a

Abbreviations: CV, coefficient of variation; INR, international normalized ratio.
a Capillary whole blood samples were measured in duplicate. Three different test strip lots were used across the study. The data consist of capillary blood applied directly to the test strip or via a transfer pipette.
Precision of Mean INR Measurements of Samples Directly Applied to the Test Strip or by Method Involving a Transfer Pipette.a
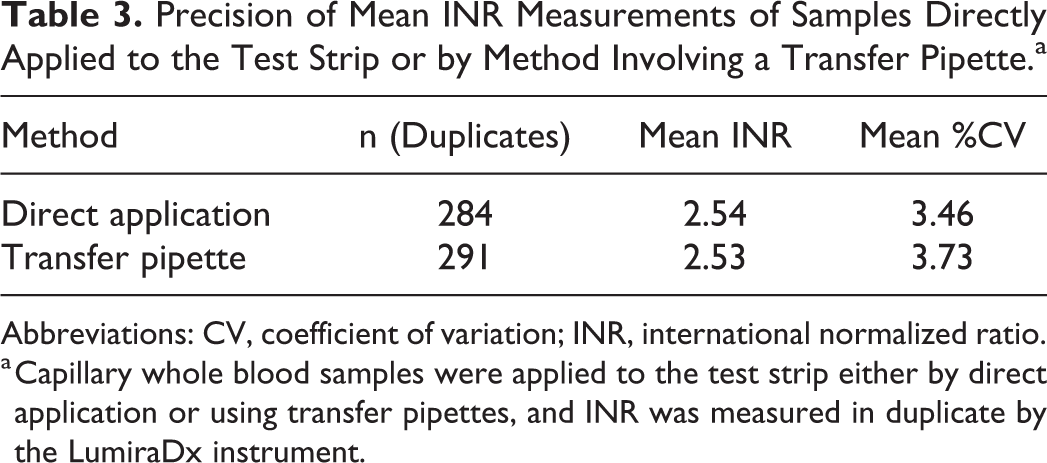
Abbreviations: CV, coefficient of variation; INR, international normalized ratio.
a Capillary whole blood samples were applied to the test strip either by direct application or using transfer pipettes, and INR was measured in duplicate by the LumiraDx instrument.
The accuracy of the LumiraDx INR Test was assessed by correlating the INR measurements of capillary whole blood to the venous plasma samples measured by the IL ACL ELITE Pro reference instrument. The INR results of directly applied samples (n = 596) demonstrated a strong correlation with the IL ACL ELITE Pro across the INR range 0.8 to 7.5 (r = 0.965; 95% confidence interval [CI]: 0.959-0.970; Figure 1). In addition, a strong correlation was measured when using capillary transfer pipettes (n = 598; r = 0.958; 95% CI: 0.950-0.964) for the application of blood samples to the test strip (Table 4). Further analysis was completed according to the FDA-recommended accuracy standards. Results from the LumiraDx INR Test were compared to the IL ACL ELITE Pro and were grouped by INR range. For INR 2.0 to 4.5, 98.6% of LumiraDx direct application samples, and 97.7% of samples using the capillary transfer pipette, were within 20% of the IL ACL ELITE Pro. For INR >4.5, 100% of LumiraDx direct application samples and capillary transfer pipette were within 25% of the IL ACL ELITE Pro. Therefore, results showed good agreement with FDA-proposed accuracy standards.

Scatter plot graph illustrating the correlation of INR measurements between the LumiraDx INR Test and the IL ACL ELITE Pro reference instrument. The INR measurements of directly applied fingerstick capillary whole blood as assessed by the LumiraDx INR Test were correlated with the venous plasma INR assessed by the IL ACL ELITE Pro. Abbreviations: INR, international normalised ratio; Int CI, intersection confidence interval; Slp CI, slope confidence interval.
The Agreement of Individual INR Measurements of Samples Directly Applied to the Test Strip or Using a Transfer Pipette, With the IL ACL ELITE Pro Reference Instrument.a

Abbreviation: INR, international normalized ratio.
a The INR results of each application method were correlated for the LumiraDx INR Test results and the IL ACL ELITE Pro.
The accuracy was further assessed by the relationship of the difference in INR measurements between the LumiraDx INR Test and the IL ACL ELITE Pro, across the range of hematocrit measured by the Hemocue Hb 201+. Results demonstrated that accuracy was maintained across the hematocrit range of 25% to 55% (data not shown).
Overall, feedback from health-care professionals indicated that the instructions on sample collection and application and pretest procedures were easy to follow (mean score of respondents: 1.67 and 1.75, respectively); the display showed clear, easy-to-read results (mean scores of respondents: 1.67); and the system was easy to prepare (mean score of respondents: 1.75) and was simple to clean (mean scores of respondents: 2.08). Furthermore, feedback indicated that additional training was required for the health-care professionals beyond the user manual in the application of samples to the test strips and on how to resolve errors (Figure S1).
Discussion
This observational, cross-sectional study was conducted to evaluate the accuracy and precision of the new LumiraDx Platform INR Test in comparison with a gold-standard laboratory reference instrument, the IL ACL Elite Pro. The INR results of 366 patients, tested in multiple clinical settings, showed that the LumiraDx INR Test correlated well with laboratory testing, as well as between the different application methods and test strip lots. Feedback from health-care professionals indicated that the instructions of the LumiraDx INR Test were easy to follow, results were displayed clearly and were easy to read, and the system was easy to prepare and clean. Overall, based on the tests performed, the LumiraDx Platform INR test provides reliable INR analysis of patients who are on VKA therapy when used by health-care professionals at the POC.
Conventional laboratory methods for testing INR require plasma samples separated from whole blood by centrifugation. This entails venipuncture performed by a phlebotomist, sample transport and handling, and testing performed by a skilled laboratory technician, which adds to turnaround time and workload. 16 The introduction of POC INR tests has offered significant advantages for clinicians and patients. For example, the tests can be performed by non-laboratory-trained clinical staff at the site of the patient, with results available in minutes, which considerably reduces the time from test performance to test result and diagnosis. Patients on VKA anticoagulants require regular monitoring of their INR. The use of POC INR test systems in community-based anticoagulation clinics or in patients’ homes by trained health-care professionals could minimize discomfort by reducing traveling time to a hospital for venous blood draw by a phlebotomist. Furthermore, it can provide direct access to fast results and any required changes to the treatment plan. Moreover, POC testing can measure INR in drops of capillary whole blood obtained by fingerstick (∼10 µL per sample), which is more convenient for the patient. 11,16
Although the introduction of direct oral anticoagulants (DOACs) has meant that clinicians have a wider choice of drugs for the treatment of VTE and the prevention of TEs in patients with AF, there remains a significant group of patients for whom DOACs are unsuitable (eg, mechanical heart valves, renal impairment, interacting drugs). In such patients, VKA treatment is the only alternative and it can be contended that VKA treatment will continue to remain an important option for many patients in the future. Therefore, improving benefit–risk ratios, as well as the technology of POC systems in terms of ease of use, speed, cost, accessibility, and analytical and clinical accuracy, is paramount for improving clinical outcomes. 2
Limitations
This study uses a robust and recognized study design to provide clear results on the objectives. However, we recognize some important limitations. Health-care professionals had different opinions on a number of points in the questionnaire, regarding the simplicity of applying adequate patient samples to the INR test strips, the requirement of additional training, and on how to resolve errors. Following the completion of this study, the manufacturer addressed these points with improved training videos on “sample collection and application” and “how to run a test.” These can be found on the LumiraDx website (https://kc.lumiradx.com/us-en/platform-training/videos) and are also provided to health-care professionals for further training. In addition, further information on test errors has been made available in the user manual. Subsequent evaluations have shown that the application of a sample to the test strip is no longer an issue for users.
A study weakness for ease-of-use evaluation was that health-care professionals in the study were very experienced in a different INR POC method and therefore were more proficient in the method they made the comparison to. Although they perceived that the alternative method was faster, in fact the 2 methods are very similar (CoaguCheck Pro II: INR results in 9.6-96 seconds, and LumiraDx INR Test: INR results in 60-90 seconds). 15,19 To improve the experience of the health-care professionals, additional training has been implemented. Patient experience in this study was based on health-care professional perception and not actual patient feedback; this will be addressed in a future study.
Supplemental Material
Supplemental Material, 2019-09-26_Supplementary_Material_LumiraDx_Questionnaire_v4 - Performance of the LumiraDx Platform INR Test in an Anticoagulation Clinic Point-of-Care Setting Compared With an Established Laboratory Reference Method
Supplemental Material, 2019-09-26_Supplementary_Material_LumiraDx_Questionnaire_v4 for Performance of the LumiraDx Platform INR Test in an Anticoagulation Clinic Point-of-Care Setting Compared With an Established Laboratory Reference Method by Robert Campbell Tait, Annielle Hung and Roy S. Gardner in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Authors’ Note
Ethical approval to report this case was obtained from East of Scotland Research Ethics Service. Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Acknowledgments
The statistical analysis was conducted by Quantics Biostatistics, Edinburgh, United Kingdom. Support with writing was provided by Viola Kooij, integrated medhealth communication (imc), London, United Kingdom.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This study was sponsored by LumiraDx UK Ltd, Dumyat Business Park, Alloa, United Kingdom.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by LumiraDx UK Ltd, Dumyat Business Park, Alloa, United Kingdom.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
