Abstract
Objective:
We sought to compare the length of stay (LOS) and total costs for patients with pulmonary embolism (PE) treated with either rivaroxaban or parenterally bridged warfarin.
Methods:
This retrospective claims analysis was performed in the Premier Database from November 2012 to March 2015. Adult patients were included if they had a hospital encounter for PE (an International Classification of Diseases, Ninth Revision code = 415.1×) in the primary position, a claim for ≥1 diagnostic test for PE on day 0 to 2, and initiated rivaroxaban or parenteral anticoagulation/warfarin. Rivaroxaban users (allowing ≤2 days of prior parenteral therapy) were 1:1 propensity score matched to patients receiving parenterally bridged warfarin. Length of stay, total costs, and readmission for venous thromboembolism (VTE) or major bleeding during the same or subsequent 2 months following the index event were compared between cohorts. Analysis restricted to patients with low-risk PE was also performed.
Results:
Characteristics of the matched PE cohorts (n = 3466 per treatment) were well balanced. Rivaroxaban use was associated with a 1.36-day shorter LOS and $2304 reduction in total costs compared to parenterally bridged warfarin (P < .001 for both). Rates of readmission for VTE were similar between cohorts (1.7% vs 1.6%; P = .64). No difference was observed between treatments for readmission for major bleeding (0.2% vs 0.2%; P > .99). In analyses restricted to low-risk patients (n = 1551 per treatment), rivaroxaban was associated with a 1.01-day and a $1855 reduction in LOS and costs, respectively (P < .001 for both). Rates of readmission were again similar between treatments (P > .56 for all).
Conclusion:
Rivaroxaban significantly reduced hospital LOS and costs compared to parenterally bridged warfarin, without increasing the risk of readmission.
Introduction
Traditional long-term oral anticoagulation therapy for patients presenting with acute symptomatic pulmonary embolism (PE) consisted of warfarin. 1 However, because warfarin requires several days to become therapeutic, “bridging” therapy (transition from an immediate acting agent to warfarin) is required. During this time period, patients receive both agents and often remain in the hospital for close anticoagulation monitoring.
Rivaroxaban is a direct, oral factor Xa inhibitor approved to treat and reduce the risk of recurrent venous thromboembolism (VTE) and does not require bridging therapy or anticoagulation monitoring. In the North American patient subset of the pivotal EINSTEIN randomized clinical trial (RCT), rivaroxaban significantly reduced index hospitalization length of stay (LOS) in PE patients by a mean of 1.7 days (P = .0002). 2 To date, no real-world study has confirmed these clinical trial results.
The primary objective of this study was to determine whether rivaroxaban was associated with a reduced hospital LOS compared to parenteral bridging to warfarin in patients with PE treated outside of a clinical trial setting. Secondary objectives included total hospital costs and readmission for recurrent VTE or major bleeding.
Methods
This retrospective claims study used Premier Perspective Comparative Hospital Database data from November 2012 to March 2015. The Premier database captures ∼20% of all acute care hospital discharges in the United States. This database was chosen as it provides highly granular data regarding the hospital encounters including a date-stamped listing of all charged items at the individual patient level including medications, laboratory and diagnostic tests, therapeutic services, discharge status, and subsequent hospital encounters or readmissions over time. Data in this database are deidentified and fully compliant with all Health Insurance Portability and Accountability Act (HIPAA) privacy and security requirements to protect participant anonymity and confidentiality.
We included adult patients (≥18 years of age) if they had a hospital encounter for PE (including those restricted to the emergency department [ED], coded as an observation stay, or that resulted in an admission to an inpatient bed), an International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) diagnosis code for PE (ICD-9-CM: 415.1x) in the primary position, ≥1 claim for a diagnostic test for PE (computed tomography, ventilation-perfusion scan, or pulmonary angiography) on day 0 to 2 and treated with either rivaroxaban or parenteral anticoagulation (unfractionated [UFH], low-molecular-weight heparin, or fondaparinux) and warfarin. Consistent with the EINSTEIN PE RCT, 3 rivaroxaban patients were allowed to receive <2 days of prior parenteral therapy and still be included. We excluded patients transferred from another health-care facility.
The primary end point for this study was hospital LOS for the index PE hospital encounter. Secondary end points included total hospital costs and readmission rates for VTE or major bleeding in the same or 2 subsequent months following the index PE. Patients with an estimated in-hospital mortality risk ≤1.5% according to the multivariable In-Hospital Mortality for Pulmonary Embolism Using Claims Data (IMPACT) prediction rule and who did not require thrombolysis or pulmonary embolectomy on days 0 to 2 were classified as low risk. 4 Analysis restricted to patients with low-risk PE was also performed.
As this was an observational study, the investigators had no control over which patients received rivaroxaban and which received parenteral bringing to warfarin. As a result, substantial differences were likely to occur in important observed covariates, which could lead to a biased estimate of treatment effect. To reduce bias due to confounding factors and minimize differences between patients receiving rivaroxaban or parenteral bridging to warfarin, rivaroxaban users were 1:1 propensity score matched to patients receiving parenteral bridging to warfarin using a greedy matching algorithm with calipers set at 1/2 the standard deviation of the propensity score. 5 Propensity scores were calculated using a multivariable logistic regression model incorporating age (as a categorical variable), gender, race, marital status, primary payer, attending physician specialty (family practice/hospitalist/internal medicine, cardiology, emergency medicine, pulmonary medicine, and other), year of index encounter, geographic region, hospital characteristics (rural vs urban) ≥500 beds, teaching vs nonteaching, Agency for Healthcare Research and Quality (AHRQ)-29 comorbidity status 6 and other comorbidities not included in AHRQ-29 (atrial fibrillation, myocardial infarction, stroke, prior major bleeding, and cognitive dysfunction), and patient admission severity (minor, moderate, major, or extreme). Propensity score matching using these same covariates was performed separately on the low-risk PE cohort.
Mean and standard deviations (SDs) were used to describe continuous variables, while frequency counts and percentages were used to summarize the categorical variables. The adequacy of the matching procedures was assessed by comparing differences in baseline characteristics between the two anticoagulation cohorts using standardized differences (difference <10% considered well balanced). 5 Differences in LOS and total costs were compared between groups using a generalized linear model with a γ-distributed error and log link. The proportion of patients readmitted during the same or subsequent 2 months following the index PE (method of reporting readmission in the Premier database) were compared between cohorts via chi-square analysis. Similar analyses were performed on the population of patients deemed to be at low risk for in-hospital post-PE mortality. In all cases, a P value < .05 was considered statistically significant. SAS version 9.3 (SAS Institute Inc, Cary, North Carolina) and IBM SPSS version 22.0 (IBM Corp, Armonk, New York) were used to perform all database management and statistical analysis.
Results
We identified 46 435 hospital encounters (ED visit, observation stay, or inpatient admission) with a primary ICD-9-CM code for PE accompanied by ≥1 diagnostic test for PE in the Premier database between November 2012 and March 2015. Of these, 34 774 were treated with either rivaroxaban (allowing ≤2 days of prior parenteral therapy) or parenteral bridging to warfarin therapy. Following 1:1 propensity score matching, 6932 patients were included in the overall analysis cohort. Of the 3466 rivaroxaban users, 56.6% did not receive rivaroxaban until the second day of their admission. Approximately 36% of patients in the overall analysis cohort were deemed low risk for early mortality, resulting in 3102 patients (1551 per treatment) being included in the low-risk PE analysis cohort. Demographic, clinical, and hospital characteristics of patients included in the overall cohort and the low-risk cohort analyses are depicted in Tables 1 and 2. The rivaroxaban and parenteral bridging to warfarin users were well matched in both analysis sets, with no characteristic exhibiting a standardized difference >10%.
Patient Demographics, Clinical, and Hospital Characteristics of Matched Rivaroxaban and Parenteral Bridging to Warfarin Users.
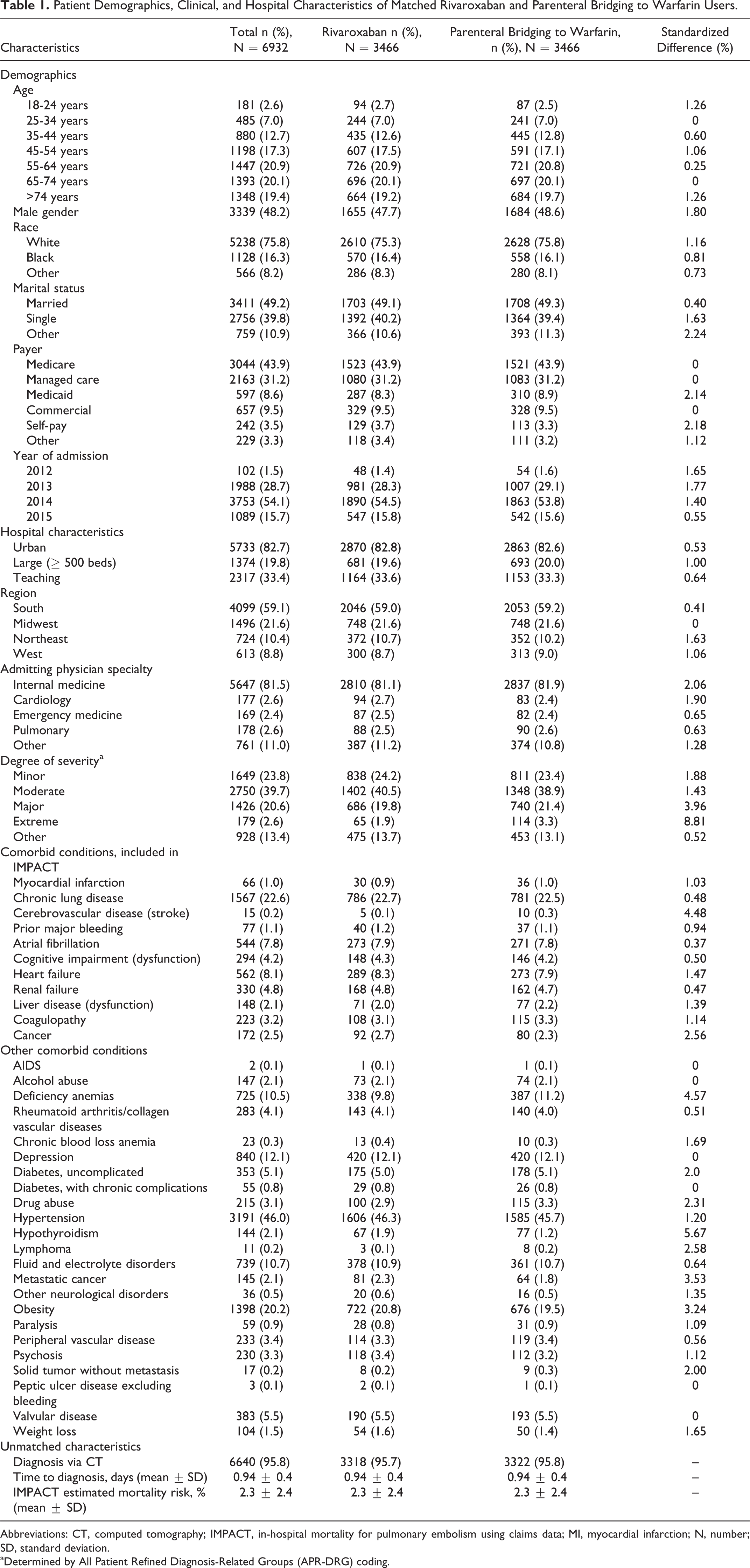
Abbreviations: CT, computed tomography; IMPACT, in-hospital mortality for pulmonary embolism using claims data; MI, myocardial infarction; N, number; SD, standard deviation.
aDetermined by All Patient Refined Diagnosis-Related Groups (APR-DRG) coding.
Patient Demographics, Clinical, and Hospital Characteristics of Low-Risk Matched Rivaroxaban and Parenteral Bridging to Warfarin Users.
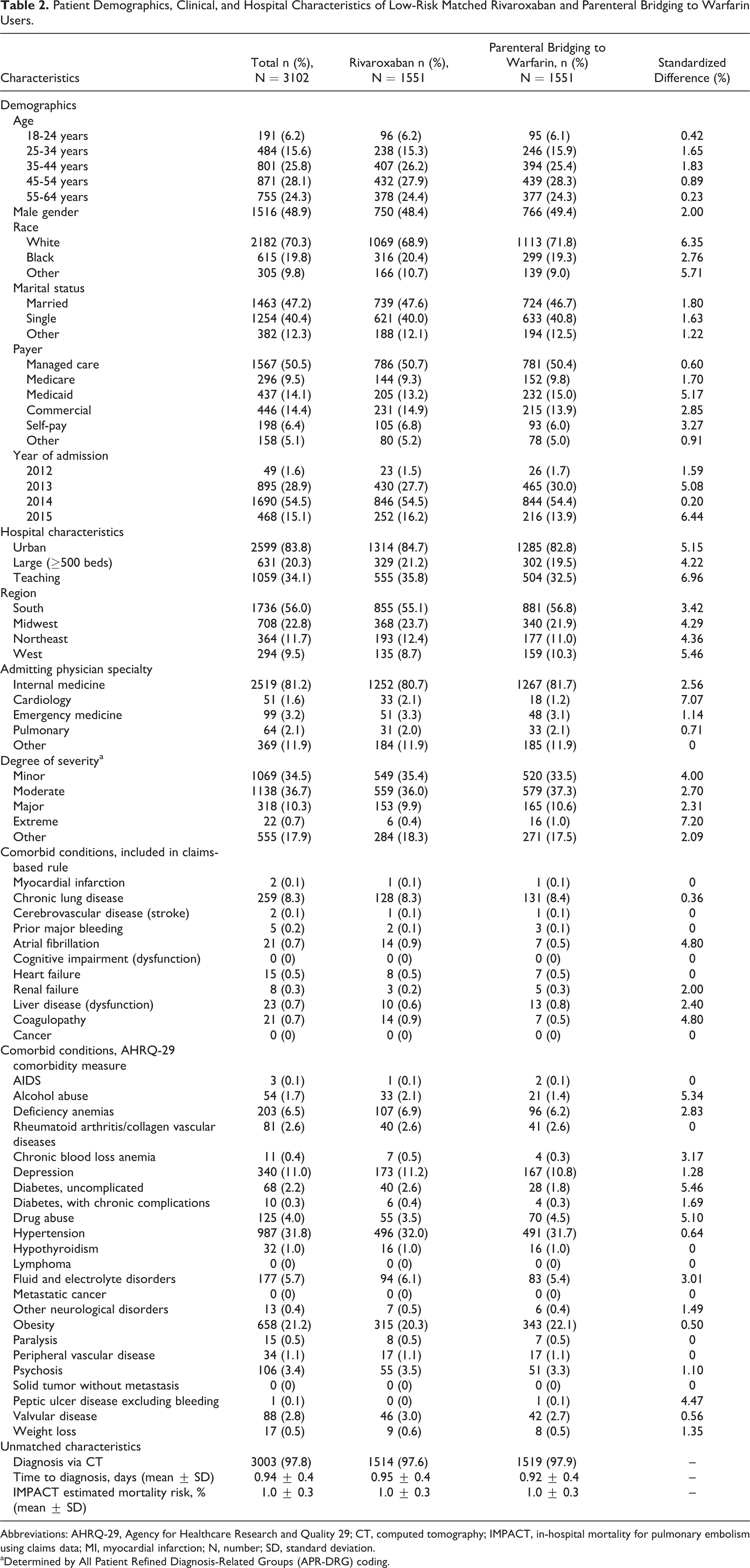
Abbreviations: AHRQ-29, Agency for Healthcare Research and Quality 29; CT, computed tomography; IMPACT, in-hospital mortality for pulmonary embolism using claims data; MI, myocardial infarction; N, number; SD, standard deviation.
aDetermined by All Patient Refined Diagnosis-Related Groups (APR-DRG) coding.
In the full PE analysis cohort, rivaroxaban use was associated with a 1.36-day shorter LOS (P < .001) and US$2304 reduction in total costs (P < .001) compared to parenteral bridging to warfarin (Table 3). Rates of readmission for VTE were similar between cohorts (P = .64) as were rates of readmission due to major bleeding (P > .99).
Outcomes in the Overall and Low-Risk Pulmonary Embolism–Matched Rivaroxaban and Parenteral Bridging to Warfarin Cohorts.
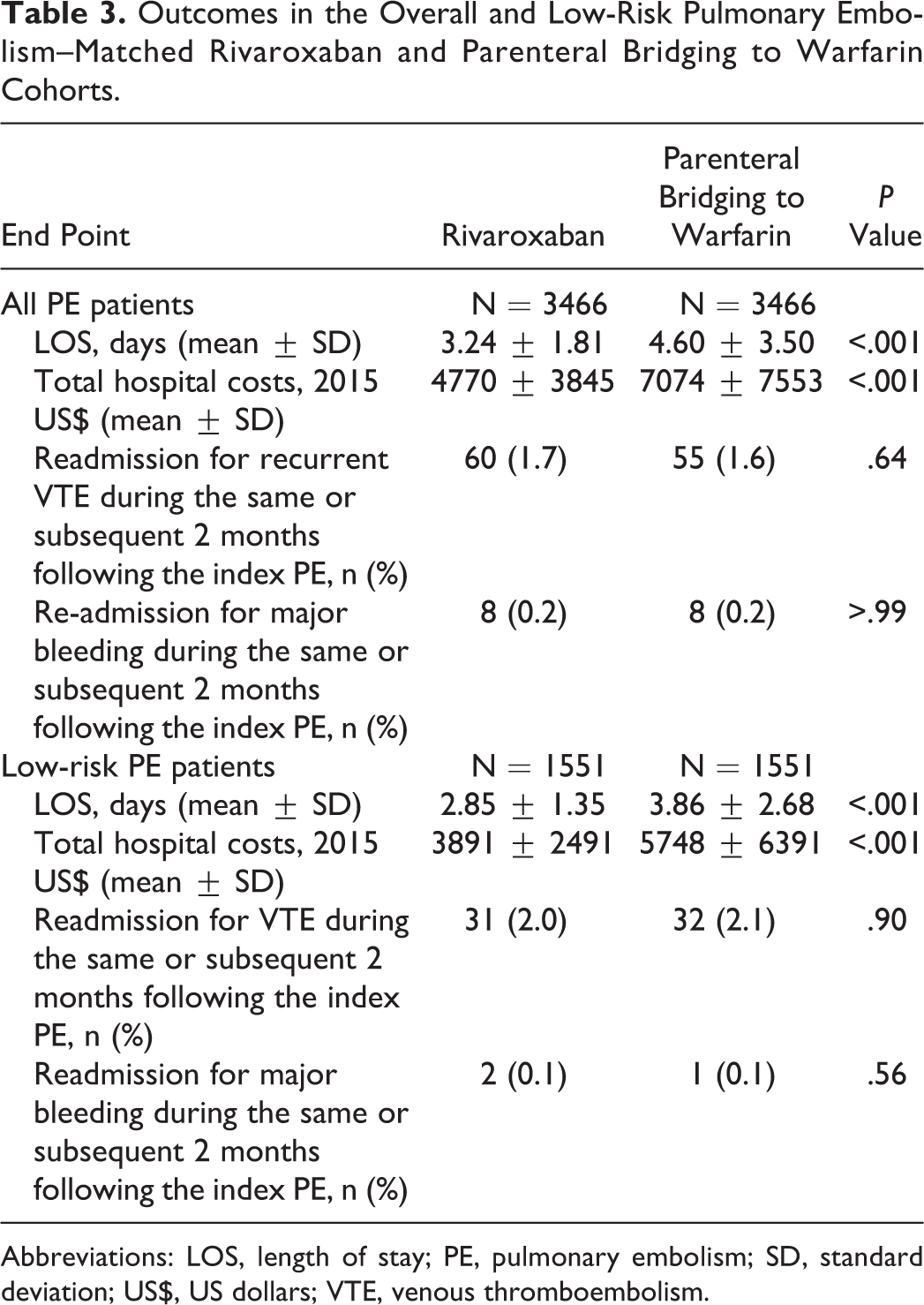
Abbreviations: LOS, length of stay; PE, pulmonary embolism; SD, standard deviation; US$, US dollars; VTE, venous thromboembolism.
The mean hospital LOS for the low-risk cohort was lower than that of the overall cohort (3.35 ± 2.2 vs 3.92 ± 2.87); however, the association between rivaroxaban use and LOS and costs remained similar. Rivaroxaban users had a 1.01-day and US$1857 reduction in LOS and total costs, respectively (P < .001 for both). The rates of readmission for VTE or major bleeding were similar between rivaroxaban users and those bridged to warfarin (P = .90 for VTE and P = .56 for major bleeding).
Discussion
This large claims database analysis of nearly 7000 patients with acute PE suggests rivaroxaban use was associated with significantly reduced length of hospital stay and total costs compared to parenteral bridging to warfarin. Moreover, the reductions in LOS and costs were achieved without increasing the short-term risk of adverse events including in readmission for VTE or major bleeding. The benefits of rivaroxaban remained even when administered to PE patients classified as “low risk,” and by default, reduced LOS and treatment costs.
The reductions in LOS seen with rivaroxaban versus parenteral bridging to warfarin in our study are consistent with prior research. 2,3,7 van Bellen and colleagues 7 evaluated the differences in LOS in the intention-to-treat population of EINSTEIN PE (n = 4821 patients with acute symptomatic PE with or without deep vein thrombosis) randomized to receive either rivaroxaban (n = 2419) or enoxaparin bridging to a vitamin K antagonist (VKA; n = 2413). That analysis demonstrated LOS was significantly shorter in patients who received rivaroxaban than in those who received enoxaparin/VKA (exponentiated least square mean of log-transformed LOS = 6.6 vs 7.5 days, respectively; difference = 0.9 days; P < .0001). Bookhart and colleagues 2 evaluated the impact of rivaroxaban on LOS among 321 hospitalized acute symptomatic PE patients recruited into EINSTEIN PE in North American sites. In these patients with PE, rivaroxaban use resulted in a 1.7-day mean reduction in LOS compared with enoxaparin/VKA (4.5 vs 6.2 days; P = .0002). The researcher’s further projected rivaroxaban use (vs enoxaparin/VKA) would reduce total hospital treatment costs by ∼$3000 per patient stay (based on estimated AHRQ Healthcare Cost and Utilization Project data suggesting the cost of an average hospital day to treat PE was $1842).
Since RCTs are designed to maximize internal validity, the applicability of their results to real-life practice is often reduced. For example, the rigorous study protocols used in RCTs can themselves inadvertently drive health-care utilization (including LOS). 8 This may partially explain why the duration of stay in our analysis (3.24 for rivaroxaban and 4.60 days parenteral bridging/warfarin) was somewhat shorter than observed in the overall and North American subset analyses of the EINSTEIN PE trial. 2,7 Another explanation for the shorter LOSs seen in our study is the multinational nature of the EINSTEIN clinical trial program. In EINSTEIN PE, substantial variations in mean LOS were observed across subject recruitment regions. 7 For these reasons, our real-world verification of rivaroxaban’s impact on LOS within a US population provides additional useful information.
There are some limitations to this analysis that need to be considered. First, Premier contains data on only a subset of US hospitalizations and is not a random sampling of all encounters. Nonetheless, encounters contained in the Premier databases are generally considered representative of those for the United States as a whole. Second, billing inaccuracies and missing data may occur in claims database analyses. Third, despite propensity score matching based on a large number of covariates, the presence of residual confounding cannot be excluded. Finally, our identification of patients with low-risk PE was not based on a common clinical tool such as the PE Severity Index (PESI) simplified PESI or Hestia. 9 These tools require access to vital sign and laboratory data not available in Premier. This being said, the multivariable IMPACT prediction rule used to identify patients with low-risk PE has been validated in several claims database studies. 4,10
In summary, rivaroxaban significantly reduced hospital LOS and costs compared to parenteral bridging to warfarin, without increasing the short-term risk of adverse events. Our real-world results confirm those observed as part of the randomized pivotal EINSTEIN clinical trial program. 2,3,7
Conclusion
Rivaroxaban significantly reduced hospital LOS and costs compared to parenteral bridging to warfarin, without increasing the short-term risk of readmission.
Footnotes
Author Contribution
CIC, GJF, PW, VA, CC, JRS, and WFP were involved in study concept and design. CIC, VA, CC, and JRS were involved in acquisition of data. CIC, GJF, PSW, VA, CC, TJB, PW, JRS, and WFP were involved in analysis and interpretation of data. CIC and ERW were involved in drafting the manuscript. CIC, GJF, PSW, VA, CC, TJB, PW, JRS, and WFP were involved in critical revision of the manuscript for important intellectual content. CIC, ERW, TJB, and VA were involved in administrative, technical, or material support. CIC was the study supervisor. CIC and TJB had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. All authors read and approved the final manuscript. The authors meet criteria for authorship as recommended by the International Committee of Medical Journal Editors (ICJME) and were fully responsible for all content and editorial decisions and were involved in all stages of manuscript development.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Coleman has received grant funding and consultancy fees from Janssen Pharmaceuticals; Bayer Pharma AG and Boehringer-Ingelheim Pharmaceuticals, Inc. Mrs Ashton and Drs Crivera, Schein, and Wildgoose are employees of Janssen Scientific Affairs LLC. Dr Peacock has received grant funding and consultancy fees from Janssen Pharmaceuticals and Portola. Dr Fermann has received grant funding for Pfizer and is on the advisory board and speaker’s bureau for Janssen Pharmaceuticals. Dr Wells has received grant funding from Bristol Myers Squib and Pfizer, is on the advisory board, has received speaker’s fees from Bayer Healthcare, has received consultancy fees from Janssen Pharmaceuticals, and served on a writing committee with Itreas. Drs Weeda and Bunz have no conflict of interest germane to this manuscript to report.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: funded by Janssen Scientific Affairs, LLC, Raritan, New Jersey, USA.
