Abstract
Objectives
Platelet-fibrin interactions are critical for hemostasis. Thromboelastography (TEG) assesses these dynamics globally. We investigated the role of fibrinogen on TEG-derived clot strength in pregnant women with and without thrombocytopenia, integrating transcriptomic analysis of public datasets to molecularly corroborate our functional findings.
Methods
A retrospective study was conducted on 140 third-trimester pregnancies (70 with normal platelet counts and 70 with thrombocytopenia) enrolled between 2015 and 2020. TEG parameters, platelet indices, and fibrinogen concentrations were analyzed. In parallel, public transcriptomic datasets were reanalyzed to explore coagulation- and fibrinogen-related pathways during pregnancy.
Results
Mean platelet volume (MPV), platelet count, fibrinogen, and TEG parameters [R-time, K-time, alpha angle, and maximum amplitude (MA)] differed significantly between groups (p < .05). MA positively correlated with platelet count (r = 0.873, p < .001) and fibrinogen concentration (r = 0.410, p < .01). In thrombocytopenic women, MA correlated with platelet count (r = 0.736, p < .0001), MPV (r = 0.447, p < .01), and fibrinogen (r = 0.361, p < .01). Multivariate regression confirmed platelet count and fibrinogen as independent predictors of MA (p < .05). Transcriptomic analysis further demonstrated upregulation of fibrinogen subunits (FGB, F13A1) and enrichment of coagulation pathways in pregnancy, consistent with the functional TEG findings.
Conclusions
Elevated fibrinogen is associated with and may compensate for preserved clot strength in late pregnancy, particularly in thrombocytopenia. Our integrative analysis provides functional and molecular insights into a potential compensatory mechanism, which forms a rationale for future research to evaluate the therapeutic utility of fibrinogen supplementation. These hypothesis-generating findings warrant prospective interventional validation before clinical application.
Introduction
Thrombocytopenia is a common hematologic disorder in women of childbearing age, second only to anemia. ITP has a high incidence and is characterised by a decreased platelet count in the peripheral blood that may present as bleeding in the skin, mucous membranes, or internal organs. To prevent bleeding, most guidelines recommend platelet transfusion when the platelet count falls below 10 × 109/L. 1 However, current constraints in China's blood supply, coupled with the short storage life of platelets, place patients with thrombocytopenia at an elevated risk of bleeding in areas with limited resources to preserve platelet concentrates. In theory, severe bleeding may already be present with platelet count <20 × 109/L, and there may be no or mild bleeding with platelet count >20 × 109/L. Clinical discrepancies between bleeding symptoms and platelet count are common. Using platelet count as the sole basis for judgment in severe bleeding can lead to delayed treatment and serious or even fatal outcomes, while using bleeding symptoms as the sole basis may result in premature and excessive treatment and expose patients to unnecessary medical risks and financial burdens. 2 Platelet destruction increases if platelet transfusion induces the production of antiplatelet antibodies, leading to further thrombocytopenia. Recombinant coagulation factor VII can be used for emergency hemostasis of hemorrhage in patients with thrombocytopenia 3 ; however, it has a short half-life, is expensive, and carries the risk of inducing thrombosis, making it unsuitable for clinical application.
Platelets are only one of several factors affecting clot strength. 4 The levels of prothrombin, coagulation factors, and fibrinogen increase during a normal pregnancy, while anticoagulation and fibrinolytic functions are attenuated, placing the blood in a hypercoagulable state. These physiological changes ensure rapid and effective hemostasis after delivery and may reduce the risk of hemorrhage in pregnant patients with immune thrombocytopenia.
Given the limitations of current therapies and the unique coagulability of pregnancy, we hypothesized that fibrinogen may play a compensatory role in maintaining clot strength despite low platelet counts. To test this, we retrospectively analyzed the relationships between platelet count, fibrinogen levels, and thromboelastography (TEG) parameters in third-trimester pregnancies with and without thrombocytopenia. Furthermore, to molecularly contextualize our findings, we integrated transcriptomic analyses of public pregnancy datasets to investigate coagulation and fibrinogen-related pathways. This combined functional and multi-omics approach aimed to robustly evaluate the biological plausibility of fibrinogen supplementation as a therapeutic strategy in thrombocytopenic pregnancy.
Methods
The clinical data of pregnant women in the third trimester hospitalized at the Union Hospital of Fujian Medical University and Union Hospital of Fujian Medical University Pingtan Branch were collected between 2015 and 2020. None of the patients were taking nonsteroidal anti-inflammatory drugs, and no bleeding was observed in any of them. Complete blood count values, prothrombin time (PT), activated partial thromboplastin time (APTT), fibrinogen, thrombin time, and TEG parameters (R-time, K-time, alpha angle, and maximum amplitude [MA]) were synchronously determined. Patients were categorized into a **normal platelet count group (Group A, PLT ≥ 100 × 10^9/L)** and a **thrombocytopenia group (Group B, PLT < 100 × 10^9/L)**.5,6 Group B patients were clinically diagnosed with immune thrombocytopenia (ITP) by attending physicians, which is a diagnosis of exclusion, excluding HELLP syndrome or other conditions that can cause thrombocytopenia. The final Group B cohort, therefore, consisted of patients in whom no secondary cause for thrombocytopenia was identified, fulfilling the diagnostic criteria for primary ITP.
The Mindray BC-6800 Plus analyzer was used for the complete blood count. Coagulation test parameters (PT, APTT, fibrinogen, and thrombin time) were determined using a Stago STA-R Max (Stago, STA-R Max, France). Plasma fibrinogen concentration was measured using the Clauss method. TEG analysis and measurements were performed on a TEG® 5000 Thrombelastograph® Hemostasis Analyzer system (Haemonetics Corporation).
Ethical Approval and Consent to Participate
The current study was approved by the ethics committee of Union Hospital of Fujian Medical University, Fuzhou, China and conducted in accordance with the principles of the Declaration of Helsinki and its amendment. All patients provided written informed consent prior to treatment, and all the information was anonymized prior to analysis. (Ethics Approval Number: 2024YK289; Date of Ethical Review Approval:10/10/2024).
Bioinformatics Analysis
Pregnancy-related transcriptomic datasets were retrieved from the Gene Expression Omnibus (GEO, https://www.ncbi.nlm.nih.gov/geo/). In particular, dataset GSE154377 was included, which contains cell-free RNA sequencing data from maternal plasma collected across the first, second, and third trimesters, as well as from non-pregnant women. After quality control and normalization, differential expression was assessed using DESeq2 with batch effects included as covariates. Genes with |log2FC| ≥ 0.585 and p < .05 were considered significant. Functional enrichment was performed using Gene Ontology (GO, biological processes) and Kyoto Encyclopedia of Genes and Genomes (KEGG) via the clusterProfiler and org.Hs.egdb packages, with a focus on coagulation- and platelet-related pathways. Gene set enrichment analysis (GSEA) was further applied to hallmark coagulation and complement and coagulation cascades signatures. Differentially expressed genes were mapped onto related pathways (eg, Complement and coagulation cascades, hsa04610) using pathview to generate integrative mechanistic visualizations. To our knowledge, no publicly available transcriptomic dataset directly compares pregnant women with thrombocytopenia to pregnant women with normal platelet counts in late gestation. The dataset we analyzed (GSE154377) instead contrasts pregnant with non-pregnant women. Therefore, our transcriptomic results should be interpreted as external evidence providing mechanistic plausibility for the observed clinical associations, rather than as a direct validation of our cohort findings.
Data Analysis
Statistical analyses were performed using **IBM SPSS Statistics, version 26.0**, and **GraphPad Prism, version 10.0**. Continuous variables are expressed as the mean ± standard deviation. The difference in variables between the thrombocytopenia and control groups was analyzed using t-test. Pearson's and Spearman's correlation coefficients were used to determine the correlation between variables. MA was the dependent variable, and platelet count (PLT), fibrinogen (FIB), and mean platelet volume (MPV) were independent variables in the multivariable linear regression. Differences with p ≤ .05 were considered statistically significant. Normality was assessed by Shapiro–Wilk tests. Between-group comparisons used t-tests or Mann–Whitney U tests as appropriate; correlations used Pearson's (normal) or Spearman's (non-normal). Two-tailed α = 0.05 with Benjamini–Hochberg FDR control was applied to multiple correlations. The MA regression model reported coefficients with 95% CIs and underwent diagnostics (residual plots, Q–Q plots, Cook's distance) and 10-fold cross-validation.
Results
General Characteristics
Seventy healthy third-trimester pregnant women (Group A) and 70 third-trimester pregnant women with thrombocytopenia (Group B) were enrolled in the study. The mean maximum amplitude (MA) in Group A was near the upper limit of the normal range, while that in Group B was near the lower limit. No statistically significant differences were observed in age, white blood cell count, neutrophil count, prothrombin time (PT), or activated partial thromboplastin time (APTT). However, hemoglobin and hematocrit were significantly lower in Group B (both p < .001) (Table 1). Key parameters are visually compared in Figure 1 to illustrate the coagulation profiles between the two groups.

A: Results of R, K, FIB and MPV difference between thrombocytopenia group and normal group; B: Results of angle and MA difference between thrombocytopenia group and normal group. (Note: K, K-time; R, R-time; Angle, alpha angle; FIB, Fibrinogen; MA, maximum amplitude; MPV, mean platelet volume; *, There were significant differences between the two groups.).
General Laboratory Data of the two Groups (Mean ± SD).

Note. WBC, white blood cell; PT, prothrombin time; APTT, activated partial thromboplastin time; R, R-time; K, K-time; Angle, alpha angle; MA, maximum amplitude. Significance codes: * p < .05, ** p < .01, *** p < .001. Reported p-values are two-tailed. Outlier handling and missingness are described in Methods.
Consistent with the data presented in Table 1, significant differences were observed in thromboelastography (TEG) parameters. Compared to Group A, Group B exhibited a significantly shorter R-time (5.94 ± 1.6 vs 7.12 ± 2.1 s, p < .001) and a prolonged K-time (3.4 ± 2.0 vs 2.10 ± 0.7 s, p < .001). The alpha angle was significantly smaller in the thrombocytopenia group (55.27 ± 10.2 vs 60.21 ± 8.4°, p < .01). Most notably, the maximum amplitude (MA), which reflects clot strength, was substantially lower in Group B (47.98 ± 11.5 vs 69.81 ± 5.3 mm, p < .001). These TEG findings confirm that thrombocytopenia significantly alters the coagulation process, leading to impaired clot formation and reduced ultimate clot strength.
Fibrinogen Levels in the two Groups of Pregnant Women
The difference in mean fibrinogen between the two groups was statistically significant (4.57 ± 0.68 vs 4.14 ± 0.89, p = .001). Over half of the pregnant women in both groups had fibrinogen above normal levels (4 g/L), with a higher percentage in group A (84.29% vs 52.86%, p < .01) (Figure 2).
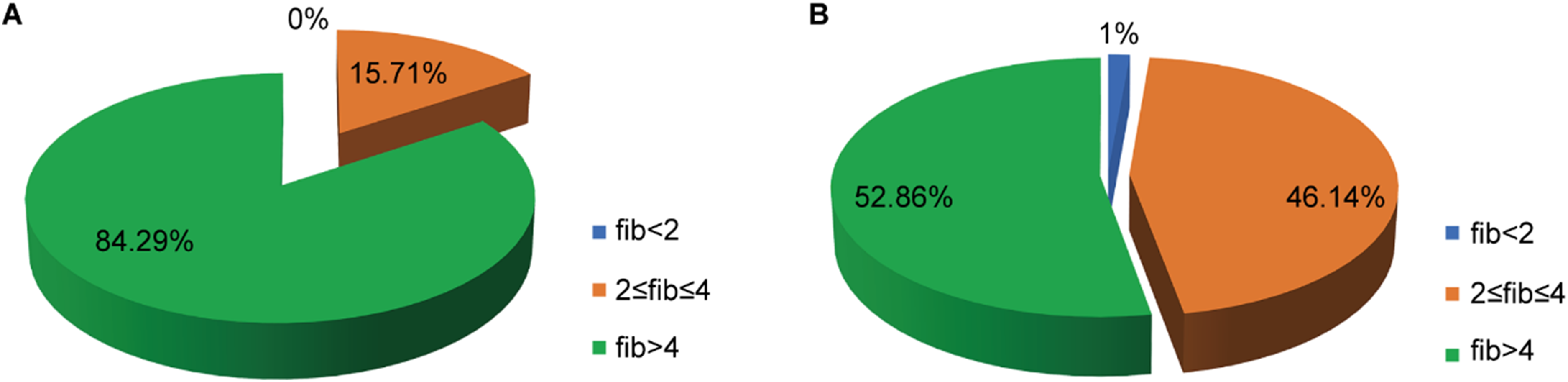
A: Fibrinogen levels of healthy third-trimester pregnant women (Group A); B: Fibrinogen levels of third-trimester pregnant women with thrombocytopenia (Group B). FIB, fibrinogen.
Correlation Between Platelet Count, MPV, Fibrinogen and MA of the Two Groups of Pregnant Women
Clot strength, as measured by maximum amplitude (MA), exhibited a strong positive correlation with platelet counts across the entire cohort (r = 0.873, p < .001; Figure 3). In contrast, no significant correlation was observed between mean platelet volume (MPV) and MA in the overall population (r = –0.242). However, a modest positive correlation was found between fibrinogen levels and MA (r = 0.4102, p < .01). Within the thrombocytopenia group, platelet count remained moderately correlated with MA (r = 0.736, p < .0001; Figure 4A). Additionally, low but significant positive correlations were found between MPV and MA (r = 0.447, p < .01; Figure 4B) and between fibrinogen and MA (r = 0.361, p < .01; Figure 4C).

Fitting curves of PLT counts and MA correlation in the whole group. PLT, platelet.

Linear correlation curves of PLT, MPV, and FIB with MA in the thrombocytopenia group. PLT, platelet count; FIB, fibrinogen; MA, maximum amplitude; MPV, mean platelet volume.
These correlation analyses reveal a hierarchical contribution to clot strength in pregnancy: platelet count is the primary determinant, while fibrinogen serves as a significant secondary modulator. Notably, in the context of thrombocytopenia, the significant positive correlations of both fibrinogen and MPV with MA suggest the emergence of compensatory mechanisms—leveraging both fibrinogen availability and platelet reactivity—to preserve hemostasis. This reinforces the therapeutic rationale for considering fibrinogen supplementation as a means to improve clot strength when platelet counts are deficient.
Regression Analysis of Platelet Count, Fibrinogen, MPV, and MA in Thrombocytopenia Group
MA values remained within the normal range when platelet counts were ≥ 100 × 109/L. Multiple linear regression was performed with maximum amplitude (MA) as the dependent variable and platelet count (PLT), fibrinogen (FIB), and mean platelet volume (MPV) as independent variables (Table 2). The resulting regression model was defined as follows:
Multiple Linear Regression Model Results.

*Dependent variable: MA
Note. FIB, fibrinogen; MPV, mean platelet volume;PLT, platelet count; MA, maximum amplitude; R2, Coefficient of Determination; F, F-statistic; P/p, p value; SE, Standard Error; B/β, regression coefficient; VIF, Variance Inflation Factor; t, t-statistic.
MA = 17.879 + 2.534FIB + 0.764MPV + 0.251PLT
MA = 17.879 + 2.534FIB + 0.764MPV + 0.251PLT
This regression equation describes the association of MA with fibrinogen, platelet count, and MPV within our cohort (R2 = 0.68) and may aid hypothesis generation regarding target ranges. It is not intended to prescribe individual therapy and requires prospective validation.
Multiple linear regression analysis revealed independent associations of platelet count, fibrinogen concentration, and mean platelet volume with MA (all p < .05) (Figure 5).
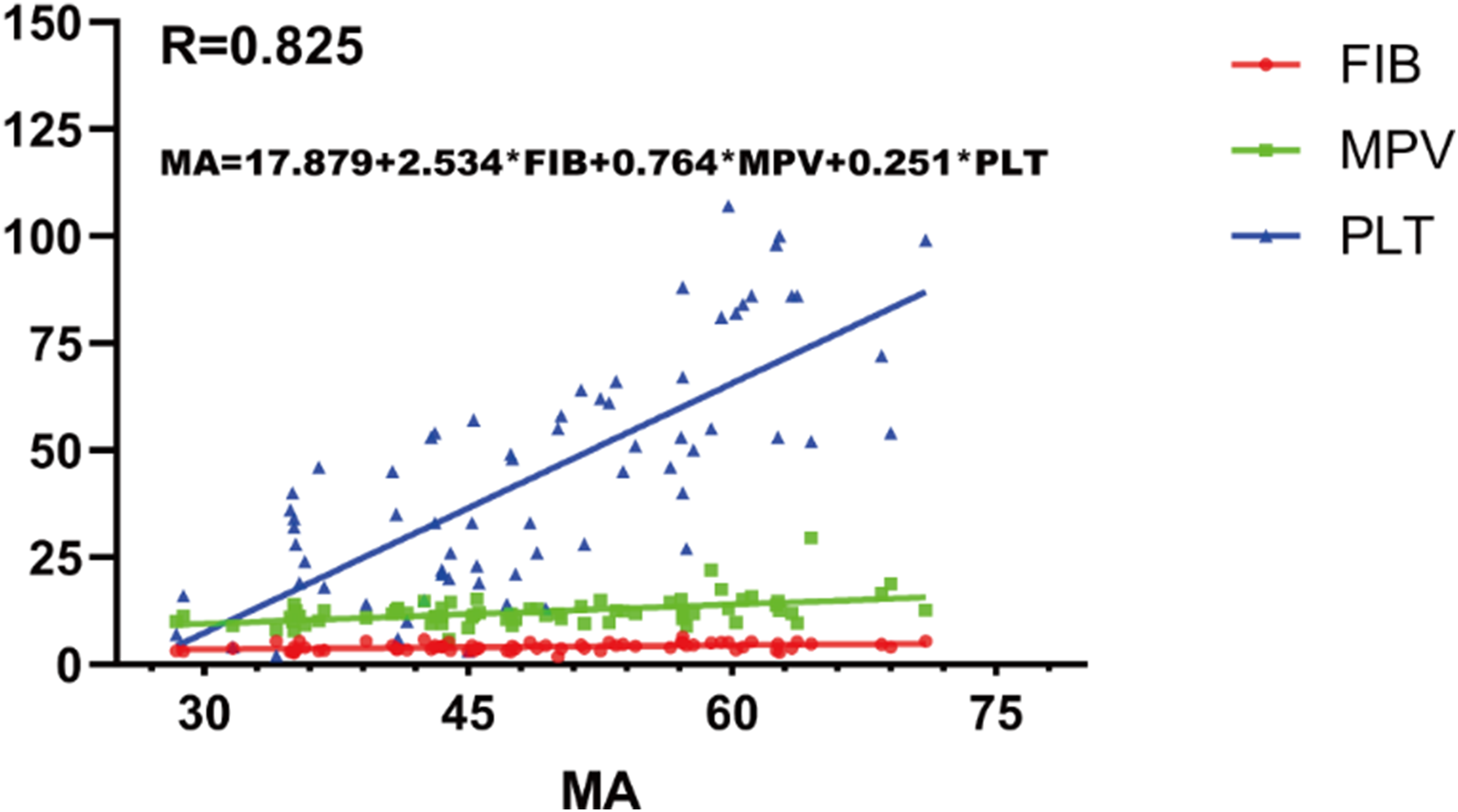
Multiple linear regression analysis results and correlation curves.
Transcriptomic Analysis Supports Coagulation Pathway Upregulation in Pregnancy
To complement the clinical findings, we performed transcriptomic analysis of public pregnancy-related datasets. The volcano plot demonstrated multiple genes significantly altered between pregnant and non-pregnant women, including pregnancy-specific glycoproteins (PSG3, PSG5) and steroid metabolism-related genes (HSD3B1, CCDC27) (Figure 6A). Gene set enrichment analysis (GSEA) confirmed the upregulation of the hallmark “Coagulation” gene set (NES = 1.3, p = .01; Figure 6D). The results of the functional enrichment analysis robustly support the activation of coagulation pathways during pregnancy. Gene Ontology (GO) enrichment analysis revealed significant enrichment for biological processes related to coagulation- and hemostasis-related biological processes in the pregnant cohort compared to non-pregnant controls (Figure 6B). Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis further corroborated these findings, identifying “Complement and coagulation cascades” (hsa04610) as one of the most significantly enriched pathways (Figure 6C). Gene set enrichment analysis (GSEA) confirmed the upregulation of the hallmark “Coagulation” gene set (NES = 1.3, p = .01; Figure 6D).

A: Volcano plot of differentially expressed genes between pregnant and non-pregnant women; B: Gene Ontology (GO) biological process enrichment showing coagulation- and hemostasis-related pathways; C: KEGG pathway enrichment with “Complement and coagulation cascades” among the top pathways; D: Gene set enrichment analysis (GSEA) plot of the Hallmark Coagulation gene set (NES=1.3, P = .01). (Note: GO, Gene Ontology; KEGG, Kyoto Encyclopedia of Genes and Genomes; GSEA, gene set enrichment analysis; NES, normalized enrichment score.).
Pathway-level visualization mapped the differentially expressed genes onto the KEGG “Complement and coagulation cascades” pathway (Figure 7). Notably, fibrinogen subunits (FGB) and factor XIII (F13A1) were upregulated, aligning with our thromboelastography findings that elevated fibrinogen contributes to preserved clot firmness despite reduced platelet counts. These omics results provide independent molecular evidence that supports and extends the functional observations from TEG.
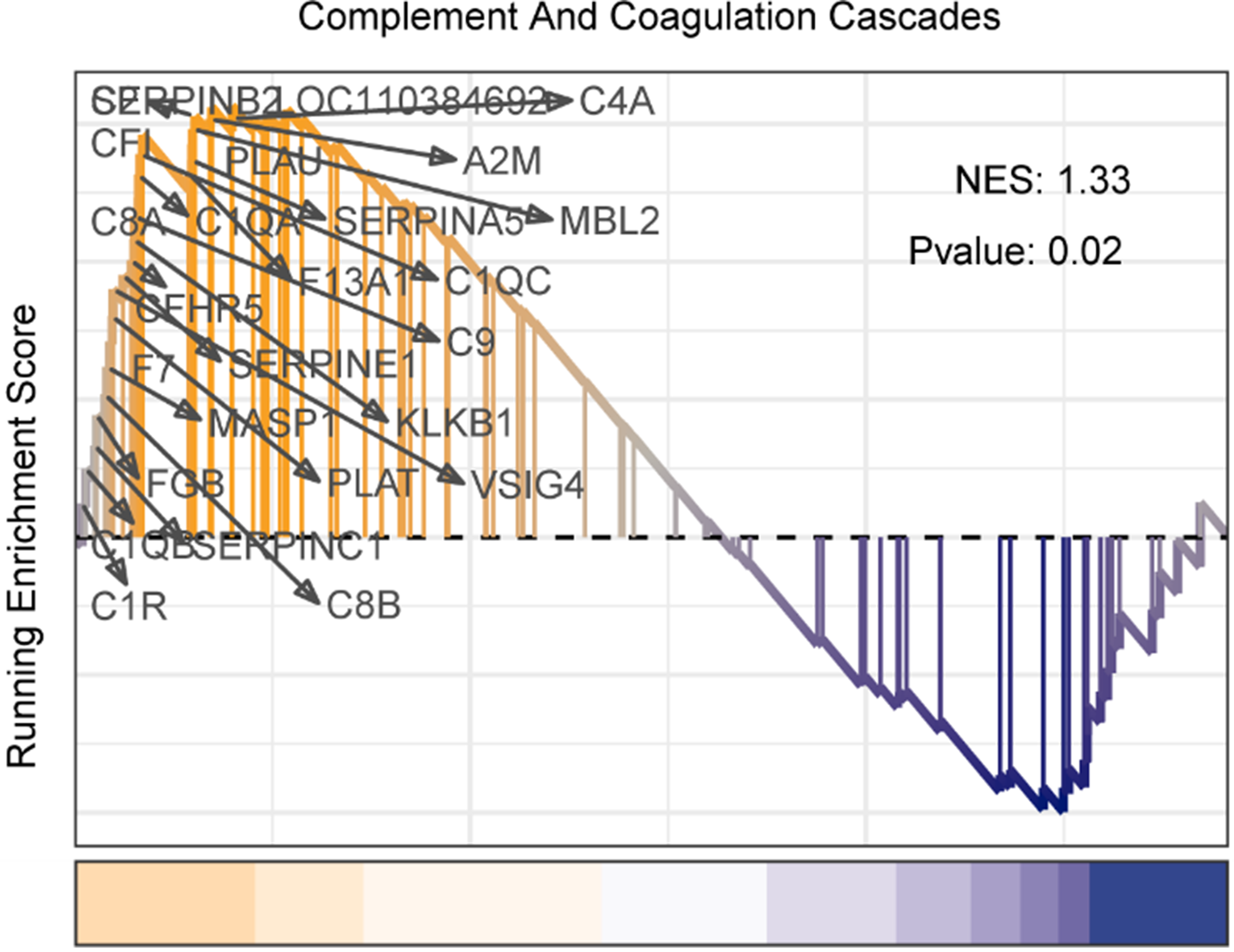
Pathway visualization of the KEGG “Complement and coagulation cascades” (hsa04610), showing upregulated genes (orange) including FGB and F13A1.
Discussion
Results in the Context of Published Literature
TEG is a classic testing method that examines the entire process during which a blood sample in its initial liquid state forms a 20-mm clot and reflects the initiation of coagulation and the dynamic changes in coagulation and fibrinolysis, thereby indicating overall coagulation function. This study confirms that TEG provides a comprehensive assessment of coagulation dynamics in pregnant women with thrombocytopenia. Our key finding is that in this population, MA is independently associated not only with platelet count but also with fibrinogen levels and MPV. This suggests the presence of compensatory mechanisms where larger, potentially more reactive platelets and elevated fibrinogen may help preserve hemostasis when platelet counts are low. Our findings align with the known physiology of pregnancy, where fibrinogen levels rise progressively. An analysis of 797 pregnant women over 9 months of gestation by Simon et al 7 found that fibrinogen levels increase with gestational age; the mean fibrinogen at the first blood draw during gestation was 4.6 g/L, and the mean fibrinogen before delivery was 4.8 g/L. The two sets of data from the present study had values lower than those previously observed, and we believe that the gestational age of our patients during blood collection was lower than that of participants in the Simon study.
This retrospective study further confirms that the use of TEG can be a good predictor of coagulation status. In pregnant women, when the platelet count was ≥ 100 × 109/L, MA was normal, suggesting that cesarean delivery is safe when the platelet count ≥ 50 × 109/L, consistent with previous reports.1,8 Furthermore, MPV significantly correlated with MA in the thrombocytopenia group. This may be because the body should compensate for platelet function during thrombocytopenia by producing larger, denser, and more reactive platelets. In turn, fibrinogen promotes platelet activation, thereby maintaining normal coagulation. Lyu et al 9 also concluded that platelet count in adult patients positively correlated with absolute immature platelet fraction in a study of 271 patients with immune thrombocytopenia. Specifically, absolute immature platelet fraction positively correlated with MA, and the degree of bleeding negatively correlated with absolute immature platelet fraction. This suggests that immature platelets play a role as hemostatic agents in patients with thrombocytopenia and that immature platelets are larger in size and more functionally active than mature platelets. In this study, absolute immature platelet fraction values were not measured during data collection; however, we concluded that MPV is significantly positively correlated with MA. Theoretically, this is consistent with the significant effect of absolute immature platelet fraction on MA. Our future experiments will involve a large-sample prospective study that includes other platelet-related parameters (such as immature and absolute immature platelet fractions). In the current study, regression analysis confirmed that platelet count, fibrinogen, and MPV significantly correlated with MA in patients with thrombocytopenia. A regression equation indicated the target fibrinogen concentration that the patient should achieve. These findings are consistent with the hypothesis that higher fibrinogen may partially compensate for thrombocytopenia-related reductions in clot firmness; however, clinical effectiveness and safety require prospective evaluation.
We also considered the potential confounding influence of anemia, which was more prevalent in the thrombocytopenic group. Dilutional anemia of pregnancy can influence coagulation by reducing blood viscosity, 10 which may theoretically attenuate platelet activation and clot strength, thereby potentially lowering the MA measured by thromboelastography. Had this effect been substantial in our cohort, the lower hemoglobin levels in the thrombocytopenic group should have further reduced their MA values. Nevertheless, we still observed a significant and independent correlation between fibrinogen and MA within this anemic, thrombocytopenic subgroup. This suggests that the compensatory role of fibrinogen in maintaining clot strength is sufficiently robust to be detectable even in the presence of anemia—a factor expected to weaken such associations. Therefore, although a minor modulating effect of anemia cannot be entirely ruled out, its presence would more likely have biased the study against, rather than in favor of, identifying a significant role for fibrinogen.
Lang et al 11 demonstrated that clot strength increased with increasing fibrinogen concentration (concentration-dependence), even when platelet count < 10 × 109/L in an in vitro study using blood samples from healthy volunteers. They also conducted a retrospective analysis of 904 patients with thrombocytopenia and found that clot stiffness decreases when platelet and fibrinogen levels are decreased, and that clot stiffness is restored when thrombocytopenia is accompanied by increased fibrin interactions. This suggests that maintaining fibrinogen concentrations is essential in the context of thrombocytopenia. In vitro studies demonstrate that fibrinogen can substitute for the increased platelets needed for increased clot strength. 12 Additionally, supplementation of fibrinogen to the plasma of patients with hematologic malignancies increases clot stiffness with an efficacy comparable to that of platelet transfusion. 13 The results of these in vitro trials provide further theoretical support that elevated fibrinogen concentrations can compensate for decreased clot strength owing to thrombocytopenia, which motivates future in vivo trials. Increased fibrinogen levels increased clot strength in pigs, even in the presence of moderate to severe thrombocytopenia.14,15 These studies suggest that increasing fibrinogen levels, platelet transfusion, and recombinant coagulation factor VII may be beneficial for hemostasis in emergencies in patients with thrombocytopenia. 3 Finally, fibrinogen may serve as an alternative when platelets are unavailable or platelet transfusion is ineffective.
In addition to these functional findings, our omics analysis provides independent molecular evidence supporting the role of fibrinogen in pregnancy. Transcriptomic profiling revealed upregulation of fibrinogen subunits (FGA, FGB, FGG) and enrichment of coagulation pathways, including “Complement and coagulation cascades.” Pathway-level mapping also confirmed upregulation of factor XIII (F13A1), consistent with the observed preservation of clot firmness in TEG. These results suggest that pregnancy-associated hyperfibrinogenemia is not only a physiological adaptation but also a compensatory mechanism to maintain clot strength in the setting of thrombocytopenia. The convergence of TEG and transcriptomic evidence strengthens the biological rationale for targeting fibrinogen supplementation in thrombocytopenic pregnancy.
Strengths and Weaknesses
The use of actual clinical data in this study confirms that normal clot strength can be achieved by increasing fibrinogen levels corresponding to different degrees of thrombocytopenia, thereby strengthening the clinical relevance of our findings and providing a theoretical basis for future clinical trials. It may be possible to increase clot strength and reduce the risk of bleeding from thrombocytopenia through fibrinogen transfusion in the presence of antiplatelet antibodies (ineffective platelet transfusion) or in remote areas where platelets are unavailable, thereby addressing the problems of limited blood supply and transfusion-related side effects. However, this study had some limitations. First, as a retrospective analysis, this study merely demonstrates the correlation between changes in fibrinogen levels and coagulation intensity, without reflecting the true impact of the treatment on patients with thrombocytopenia. Second, the sample size, while sufficient for initial hypothesis-generating analysis, remains limited and may not fully capture the heterogeneity of the broader patient population. Third, the inherent physiological hypercoagulability of pregnant women may counteract the risk of bleeding associated with thrombocytopenia. Fourth, transcriptomic validation relied on publicly available datasets, which are heterogeneous and not fully matched to our clinical cohort; thus, direct extrapolation should be interpreted with caution. Specifically, no publicly available transcriptomic dataset currently compares thrombocytopenic and non-thrombocytopenic pregnant women in late gestation. The use of GSE154377, which contrasts pregnant with non-pregnant women, provides only external, directional support for our findings. This limitation underscores the importance of future prospective studies integrating TEG and transcriptomic profiling within the same patients. Finally, and importantly, our study demonstrates an association but does not provide direct causal or therapeutic evidence. In the absence of interventional testing, such as the ex vivo or in vivo administration of fibrinogen concentrate to demonstrate a corrective effect on clot strength, our conclusions remain observational. The functional relationship we describe requires validation through studies specifically designed to test the effect of fibrinogen supplementation.
Implications for Practice and Future Research
Moving forward, we aim to conduct a prospective, large-sample clinical trial to evaluate the hemostatic effect of fibrinogen infusion in patients with thrombocytopenia before and after treatment for immune thrombocytopenia, aplastic anemia, and hematologic malignancies and determine whether this increases the risk of thrombosis. Future work should also integrate omics analyses with functional assays in the same patients, enabling a direct link between molecular alterations and coagulation dynamics to refine therapeutic strategies.
Conclusions
Elevated fibrinogen levels were associated with preserved clot firmness in late pregnancy, including in thrombocytopenic women. These observational results, derived from a non-bleeding cohort, should be considered hypothesis-generating and warrant prospective interventional studies before informing clinical practice.
Footnotes
Acknowledgements
We thank all the investigators and patients who participated in the present study.
Consent for Publication
The manuscript has been approved by all authors for publication.
Funding
Startup Fund for scientific research, Fujian Medical University (Grant number: 2020QH1103); National Key Clinical Specialty Discipline Construction Program (2021-76) and Fujian Provincial Clinical Research Center for Hematological Malignancies (2020Y2006).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration of Interest
The authors report that they have no conflicts of interest pertaining to this work.
Availability of Data and Materials
The data generated in this study are available within the article and the Supplemental material files.
