Abstract
Background
Atrial fibrillation (AF) is a frequent and serious cardiovascular complication in oncology patients, driven by shared pathophysiological mechanisms of inflammation, thrombosis, and cardiac remodeling. Cancer and its therapies significantly increase AF risk, impacting survival, quality of life, and management complexity.
Methods
This narrative review synthesizes current evidence on the epidemiology, pathogenesis, and management of AF in cancer, emphasizing inflammatory and prothrombotic pathways, treatment-related cardiotoxicity, and cardio-oncology strategies. Literature was retrieved from PubMed and recent cardio-oncology guidelines to provide an integrated overview of mechanistic insights and therapeutic implications.
Results
Cancer patients exhibit up to a 10-fold higher risk of AF than the general population, particularly with esophageal, lung, and hematologic malignancies. Mechanistically, the NLRP3 inflammasome, neutrophil extracellular traps, and hypercoagulability promote atrial fibrosis and electrical instability. Cancer therapies—including anthracyclines, platinum agents, tyrosine kinase inhibitors, immunotherapies, and thoracic radiation—further potentiate AF. Management is challenging due to drug–drug interactions, bleeding risks, and altered pharmacokinetics. Evidence supports the use of direct oral anticoagulants when feasible and emphasizes multidisciplinary cardio-oncology collaboration. Personalized anticoagulation, vigilant rhythm monitoring, and shared decision-making are key to optimizing outcomes.
Conclusion
AF in cancer arises from a convergence of inflammation, thrombosis, and therapy-induced injury. A structured, multidisciplinary cardio-oncology approach integrating early risk stratification, tailored anticoagulation, and preventive strategies is essential. Future research should focus on oncology-specific risk prediction tools, biomarker-guided management, and randomized trials to refine evidence-based care for this high-risk population.
Introduction
Atrial fibrillation (AF), the most common cardiac arrhythmia, is becoming more widespread. The prevalence was predicted to be 50 million worldwide. This increasing burden emphasizes how crucial it is to treat AF as a serious cardiovascular complication in cancer patients, who are already at higher risk because of their comorbidities and cancer treatments. 1
AF incidence in cancer patients was 17.4 per 1000 person-years, much higher than 3.7 per 1000 person-years in the general population. Notably, this increased risk continued for five years after the diagnosis, highlighting cancer as a separate risk factor for AF. 2 The prevalence of AF in cancer patients was 9.77%, compared with 1.19% in noncancer patients. On the other hand, 13.74% of AF patients also had a cancer diagnosis. The age-stratified random-effects relative risk ratio for AF was 10.45. 3 The implications of AF are significant. AF is strongly associated with increased mortality in newly diagnosed cancer patients. AF after a cancer diagnosis was associated with a 2.55-fold increase in all-cause mortality. 4 Patients with AF and cancer have a significantly worse prognosis than those with AF but without cancer, with a 2-fold higher risk of thromboembolic events and a 6-fold higher chance of heart failure. 5 Approximately 14% of cancer patients hospitalized nationwide experienced atrial fibrillation. AF has the highest incidence and readmission rate in hematological malignancies, while AF-related death was highest in breast cancer. 6
The management of AF in cancer patients follows the same principles as in the general population, but cancer-related variables and treatment-specific risk complicate it. Rate control is still the most prevalent method, rhythm control is considered in symptomatic or difficult-to-control AF, with options including electrical or pharmacologic cardioversion and antiarrhythmic agents; however, structural heart disease, toxicity profiles, and interactions with cancer therapies (eg, taxanes, tyrosine kinase inhibitors, arsenic trioxide, and antiemetics that prolong QT interval) frequently limit their use.7–9
Cardio-oncology is an essential science that studies the relationship between cancer and cardiovascular disease, as many cancer survivors are more likely to develop heart problems because of both treatment toxicity and common risk factors. Cardio-oncology combines early risk assessment, multidisciplinary care, and customized monitoring to help prevent and control cardiotoxicity, ultimately improving survival and quality of life for cancer patients and survivors.10,11 This review summarizes the epidemiological burden, underlying processes, and therapeutic implications of AF in this population, which are critical for improving patient outcomes and guiding future research in cardio-oncology.
Methods
This narrative review was conducted to summarize current evidence on the relationship between cancer, inflammation, thrombosis, and AF. Relevant articles were identified through focused searches of PubMed, Scopus, Web of Science, and Cochrane CENTRAL up to October 2025, using key terms such as “atrial fibrillation,” “cancer,” “inflammation,” and “thrombosis.” Additional references were obtained from review articles and clinical guidelines. Recent studies, meta-analyses, and guideline statements were prioritized, and findings were organized thematically to describe epidemiology, mechanisms, and management approaches.
Epidemiology and Risk Landscape
Cancer patients have a higher incidence of AF than the general population. The total AF incidence rate among cancer patients was 64.29 per 10,000 person-years, significantly higher than that of non-cancer populations. Among cancer subtypes (Table 1), patients with esophageal cancer had the highest risk of AF, 155.94 per 10,000 person-years, while patients with thyroid cancer had the lowest risk, 20.52 per 10,000 person-years. Patients above the age of 65 and males had a greater incidence of AF. Males with lung cancer and females with esophageal cancer had the highest incidence of AF 185.02 and 150.30 per 100,000 person-years, respectively. Esophagus, lung, and gallbladder cancers are the top three malignancies associated with a greater prevalence of AF. 12 Younger individuals and those with hematologic malignancies showed the highest correlations between AF and malignancy. 3
Incidence and Prevalence of Atrial Fibrillation (AF) Across Different Cancer Subtypes.

The timing of cancer diagnosis and therapy has a significant impact on when AF first appears in cancer patients. The relative risk of malignancy was highest in the first three months following the initial AF diagnosis, but remained substantial after one year. This early-phase AF is frequently associated with cardiotoxic medications such as chemotherapy and radiotherapy. 4 For example, radiation-induced AF is increasingly recognized in esophageal cancer; about 21.4% of patients had AF within one year after undergoing thoracic irradiation, with larger left atrial doses considerably boosting AF incidence and death. 13 However, AF identified more than three years after cancer diagnosis does not appear to have the same negative impact on survival, indicating a temporal link between AF development and cancer progression or treatment effects. 14 In addition to treatment exposures, surgical complications are a consideration. Postoperative AF occurs in 4%–37% of patients after radical esophagectomy, usually within the first three days, and is associated with an increased risk of stroke, prolonged hospitalization, and mortality. These perioperative AF episodes emphasize the importance of close surveillance in high-risk malignancies. 15
Several patient-specific factors influence the risk and outcome of AF in cancer patients. Individuals aged 65 and older have a much higher risk of acquiring AF across all cancer types than those under 65. Comorbidities such as hypertension, diabetes mellitus, stroke, peripheral arterial occlusive disease (PAOD), heart failure, myocardial infarction, and end-stage renal disease (ESRD) were discovered before one year of cancer diagnosis. 12 Due to aging, comorbidities, and the effects of treatment, cancer patients are already at risk for frailty, and AF may increase the risk of frailty directly. In the inverse variance weighting analysis, AF was found to be statistically associated with an elevated risk of frailty (odds ratio = 3.017, CI: 1.106-8.232, P = .031). 16 AF and frailty typically coexist in the elderly, and both conditions become more common as individuals age. According to research, the incidence of frailty in AF patients ranges between 4.4% and 75.4%, depending on the assessment method used; however, people with AF appear to be more susceptible to frailty than those without the illness. The relationship between AF and frailty is nuanced and bidirectional: among patients with AF, frailty raises the risk of stroke, death, and longer hospital stays, while AF itself can exacerbate or worsen the fragile condition. 17
Pathophysiologic Mechanism (Table 2)
Inflammatory Pathways
Despite not being fully understood until now, 14 the pathophysiologic pathways of AF development in cancer patients have been linked both to the malignancy itself and to the treatments used. Malignancies and their progression cause profound inflammatory stress (Figure 1, Figure 2), as reflected by elevated levels of inflammatory mediators in cancer patients, including tumor necrosis factor-α, white blood cells, ceruloplasmin, C-reactive protein, and various interleukins. (eg IL-2, IL-6, and IL-8).14,18,19 Siu et al 2005 in their study on colorectal cancer patients found that patients with postoperative high neutrophil counts were more likely to develop AF (odds ratio: 3.2, 95% confidence interval: 1.3-7.8, P = 0.01). 20 These markers are also elevated in AF patients, supporting the mechanism whereby inflammation from these mediators drives electrical, structural, and functional atrial remodeling, ultimately predisposing cancer patients to AF.14,21

Cancer-associated pathways predisposing to AF. Cancer contributes to atrial fibrillation via inflammation/innate immunity (NETs, NLRP3–IL-1β), hypercoagulability (TF, Thrombin–PAR), electrical/structural remodeling (fibrosis), autonomic imbalance, and clinical/therapeutic triggers (perioperative stress, infection, anthracyclines/others), creating an arrhythmogenic substrate. Created in BioRender. Mahafdah, B. (2025) https://BioRender.com/xsaltlt.

Inflammatory triggers of AF in cancer. Cancer and its treatments trigger mitochondrial dysfunction, immune activation, and calcium disturbances that activate the NLRP3 inflammasome. This cascade promotes cytokine release, fibrosis, and electrical remodeling, creating an arrhythmogenic substrate for AF. Created in BioRender. Mahafdah, B. (2025) https://BioRender.com/nhuozf7.
Key Mechanistic and Clinical Studies Linking Cancer and AF.

One key inflammatory pathway is the NLRP3 pathway, a cytosolic protein complex in myocytes that is part of the innate immune system, recognizing abnormal signals and initiating an inflammatory cascade as a protective response.22,23 NLRP3 has also been implicated in the pathogenesis of several human cancers. 22 In their 2020 study, Ju et al reported altered NLRP3 expression in 15 of 24 cancer types. 24 The role of NLRP3 in the development of AF can be briefly summarized that the activation of NLRP3 induces inflammatory signaling, primarily via IL-1β, leading to myocyte fibrosis, gap junction remodeling with conduction defects, and abnormal Ca2+ handling that promotes ectopic activity, together, these processes establish a mechanistic link between NLRP3 and AF pathogenesis, offering a possible explanation for its development in cancer patients.23,25,26
Another important inflammatory pathway involves neutrophil extracellular traps (NETs), which consist of DNA fragments with cytoplasm proteins released from neutrophils. 27 Studies have shown that cancer alters neutrophil defense mechanisms and increases NET formation through tumor cells themselves, platelet interactions, and cytokines such as GM-CSF and IL-17.27,28 NETs also play a bidirectional role, supporting cancer progression by promoting tumor growth, metastasis, and angiogenesis, while also contributing to cancer-associated thrombosis.27,28 Recently, NETs have also been implicated in the development of cardiovascular diseases, including AF. 29 Elevated NET levels have been observed in AF patients. The proposed mechanism involves cardiomyocyte injury through mitochondrial damage, oxidative stress from reactive oxygen species (ROS), atrial fibrosis, and structural remodeling, collectively promoting AF.29–31 He et al (2023) supported this mechanism by showing that using DNase I to degrade NETs in vivo reduced AF-related fibrosis and shortened AF duration in their study. 29 This highlights NETs as a critical inflammatory pathway linking cancer to AF development.
Atrial Remodeling
Atrial remodeling is the main driver for the development of AF in patients with malignancies (Figure 3). It represents the final outcome of the previously mentioned inflammatory pathways in cancer patients, as inflammatory stress and different cytokines contribute to atrial fibrosis due to myocyte damage and collagen deposition. 21 Marrouche et al (2014) found that atrial fibrosis detected by MRI is an independent variable for the development of AF. 32 In addition, fibrosis subsequently leads to disruption of conduction continuity and increases its heterogeneity, thereby enhancing the development of ectopic activity. 21 Another important factor to mention is autonomic dysfunction, which has been observed in some patients with malignancies. 33 Autonomic dysfunction is known to destabilize atrial conduction and lower the threshold for arrhythmias. 34 Together, these structural changes and autonomic alterations are major contributors that potentiate AF in oncology patients.
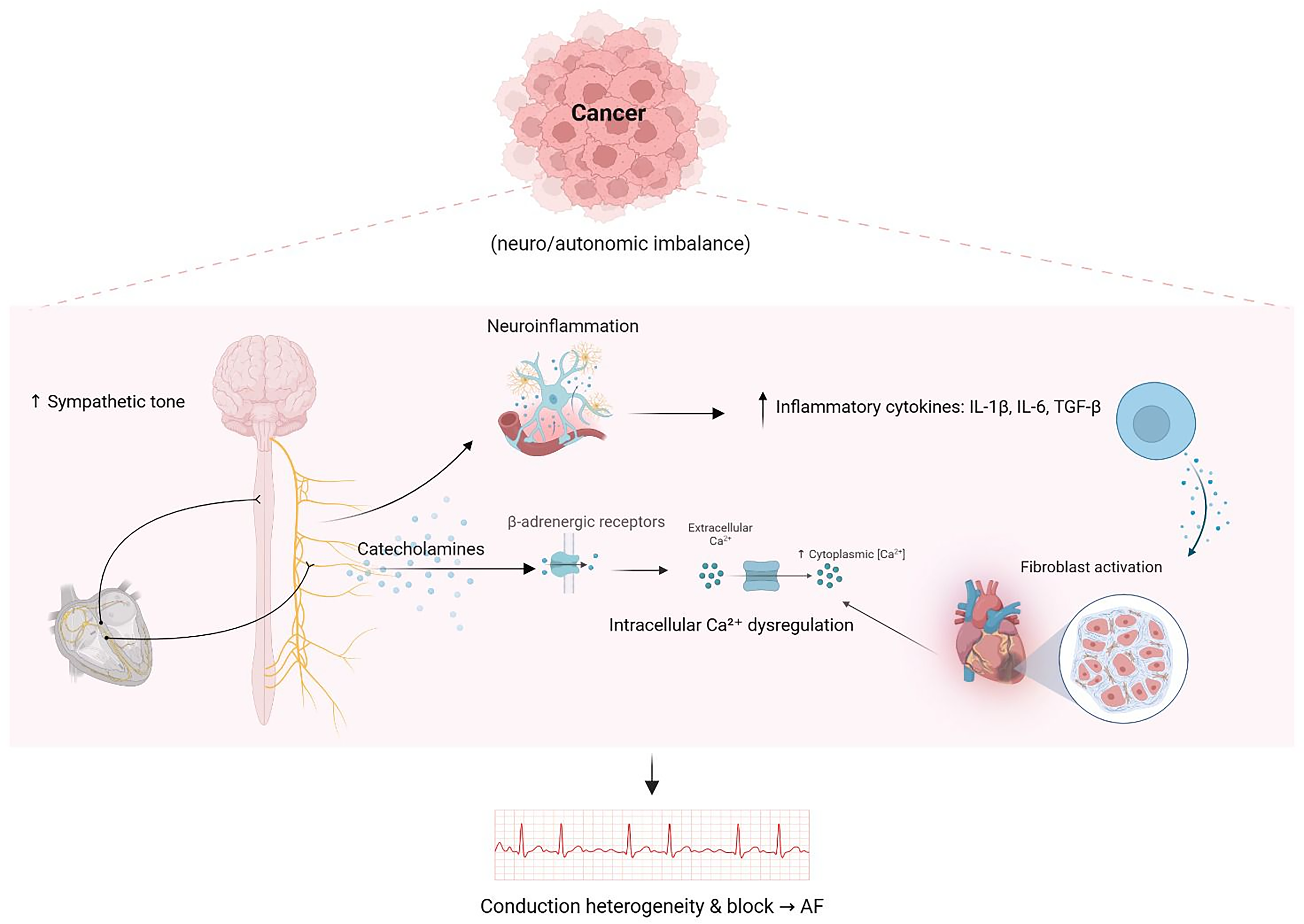
Neuro-autonomic imbalance and atrial remodeling in cancer-associated AF. Cancer-induced sympathetic activation, catecholamine release, and cytokine-driven fibroblast activation promote Ca2+ dysregulation and atrial remodeling, creating conduction heterogeneity that predisposes to AF. Created in BioRender. Mahafdah, B. (2025) https://BioRender.com/sxazect.
Hypercoagulability
Malignancies of various types are well recognized as hypercoagulable conditions (Figure 4). The pathogenesis can be summarized as tumor cells releasing pro-inflammatory cytokines along with procoagulant substances such as tissue factor, cancer procoagulant, microparticles, and NETs. In addition to activation of platelets by tumor cells occurring when the tumor metastasizes hematogenously to distant tissues, and finally direct vascular endothelial injury caused by tumor cells, all of which converge to activate the coagulation cascade and place patients in a hypercoagulable state.27,35 This state not only increases the risk of thromboembolic events in cancer patients but also plays a role in the pathogenesis of AF. 36 In recent studies, it's been shown that coagulation factors such as thrombin activate protease-activated receptors (PARs) on atrial cells, inducing pro-inflammatory and pro-fibrotic responses. This stimulation of atrial fibroblasts promotes fibrosis, disrupts electrical conduction, and increases the risk of developing AF.36,37 And that explains how hypercoagulability augments AF in oncology patients.

Hypercoagulability in cancer as a driver of AF. Tumor-mediated cytokines, procoagulant factors, and coagulation cascade activation promote thrombosis and atrial fibrosis, leading to conduction heterogeneity and AF. Created in BioRender. Mahafdah, B. (2025) https://BioRender.com/sxazect.
Cancer Therapies and AF Risk
Chemotherapies
As previously explained, cancer itself is a risk factor for AF. However, when comparing cancer patients receiving chemotherapy with those who do not, it is evident that chemotherapy independently increases the risk of AF. 38 Although chemotherapy remains the mainstay of treatment for many malignancies, it can also damage non-cancerous tissues, including the heart. 39 Various chemotherapeutic agents, such as anthracyclines, platinum agents, alkylating agents, pentostatin, 5-fluorouracil, docetaxel, capecitabine, and methotrexate, have been reported to induce AF with variable incidence and timing.18,20,39 Anthracyclines, including doxorubicin and idarubicin, are known for dose-dependent cardiac toxicity, 40 with a reported AF incidence of approximately 10% among oncology patients using these agents. 41 AF may occur acutely during infusion or may be delayed for months to years after treatment. 41 Although the exact mechanism of anthracycline-induced AF remains incompletely understood, several theories exist. They may activate the innate immune system via Toll-like receptor 4, trigger cardiac fibroblast activation, and induce a pro-inflammatory response in the heart. Additionally, anthracyclines have been found to increase mitochondrial ROS production and cause mitochondrial dysfunction, both of which can promote cardiac remodeling, providing a substrate for AF.41,42 Platinum agents, such as cisplatin, are associated with a 10% incidence of acute-onset AF in cancer patients, likely due to effects on membrane transporters, oxidative stress, mitochondrial dysfunction, disrupted ionic homeostasis, and inflammation.41,43 Another chemotherapeutic agent linked to AF is melphalan, an alkylating agent commonly used in hematopoietic stem cell transplantation. AF has been observed in 7%–22% of patients treated with melphalan, though the precise mechanism of its cardiotoxicity remains poorly understood.39,41
Targeted Therapy (Tyrosine Kinase Inhibitors)
Targeted therapies have recently emerged as a novel modality in cancer treatment. These agents are designed to inhibit specific molecular targets critical for tumor survival and proliferation and are now commonly used in breast cancer, lung cancer, leukemia, and lymphoma. 44 Many targeted therapies act through kinase inhibition, a key component of B-cell receptor signaling. 41 Tyrosine kinase inhibitors have recently been recognized as significant contributors to AF in oncology patients, as they can cause prolonged QTc, delayed repolarization, and altered ion channel activity, thereby extending the cardiac action potential.39,41,45 Notably, ibrutinib operates via a distinct mechanism: it inhibits PI3K–AKT signaling in cardiomyocytes, promoting sarcoplasmic reticulum Ca2+ leak and triggering arrhythmias without QTc prolongation. AF incidence with ibrutinib ranges from 6% to 38% among treated patients.39,44
Other Cancer Therapies (Immunotherapies and Radiation Therapy)
Other cancer therapies implicated in AF include immunotherapies.18,39 These treatments primarily act as immune modulators, enhancing the immune system's ability to target malignant cells. However, the resulting intense immune activation and T-lymphocyte–mediated inflammation can contribute to AF development.20,39 Examples of immunotherapies associated with AF in cancer patients include immune checkpoint inhibitors, aldesleukin, and chimeric antigen receptor (CAR) T-cell therapy, with reported AF incidences of 4.6%, 4.3%–8%, and 7.5%, respectively.39,46 Radiation therapy is another widely used treatment modality in various cancers, including breast and lung carcinoma. 47 Apte et al (2021) identified radiation therapy as an independent risk factor for AF in cancer patients, with a prevalence of 4.5%, rising to 6.9% when combined with chemotherapy. The prevalence is higher in patients with solid tumors compared with those with hematologic malignancies. 48 Radiation contributes to atrial tissue fibrosis and promotes inflammatory responses in the endothelial compartment, including the coronary vessels, thereby increasing the risk of AF onset. 20
Perioperative and Hospital-Associated AF
Perioperative AF is the most common form of cancer-related AF, 18 with an overall prevalence of 13.5%. 49 Reported incidences include 12.6% after lung cancer surgery, 9.2% after esophageal cancer surgery, and 4.4% after colorectal cancer surgery. 18 Risk factors for perioperative AF include older age, advanced cancer stage, male sex, history of hypertension, history of paroxysmal AF, poor physical status, postoperative tachycardia, longer surgery duration, surgical complications, postoperative blood transfusion, and elevated natriuretic peptides, which may serve as a predictive marker.18,20 Additional hospital-related factors, such as infection and cardiac stress, also contribute to AF development in cancer patients. 50 The mechanisms underlying surgery-related AF include mechanical stimulation of the pericardium, inflammatory damage, effects of anesthetic drugs, and postoperative electrolyte imbalances. 20 The impact of perioperative AF in oncology patients is significant, leading to increased postoperative mortality, longer hospital stays, and higher rates of ICU admission. 51
Therefore, anticoagulation should be considered for AF episodes lasting ≥48 h or in patients with additional stroke risk factors once surgical hemostasis is secured. 52 In cancer patients, conventional risk scores (CHA₂DS₂-VASc and HAS-BLED) are less reliable, and therapy should be guided by an individualized assessment of thrombotic and bleeding risks. 14 Aziz et al 2023, in their retrospective cohort study, showed that perioperative anticoagulation in cancer carries a 30-day arterial thromboembolism risk of 0.7% and a major bleeding rate of 1.7%, highlighting the need for a careful balance between these risks. 52 Temporary prophylactic-dose LMWH or delayed anticoagulation may be appropriate in cases of thrombocytopenia or recent surgery, with multidisciplinary coordination to optimize timing and safety.14,52 Figure 5. Illustrates the major cancer therapies contributing to AF.

Cancer therapies contributing to AF. Multiple treatment modalities, including immunotherapy and radiation, chemotherapy (eg, anthracyclines, cisplatin, melphalan), targeted tyrosine kinase inhibitors (eg, sunitinib, imatinib, sorafenib), and perioperative factors, promote atrial remodeling and conduction heterogeneity, increasing susceptibility to AF. Created in BioRender. Mahafdah, B. (2025) https://BioRender.com/sxazect.
Clinical Evaluation and Risk Stratification
Anemia, deconditioning, dehydration, or cardiotoxic medication may cause dyspnea, tiredness, palpitations, and chest pain in cancer patients. These symptoms might be mistaken for AF and can cause a delay in identifying the danger of an arrhythmia or stroke. 53 Due to common processes (inflammation, hypercoagulability, autonomic dysfunction, and treatment-related damage), the incidence and incident risk of AF are increased in cancer patients who are actively undergoing treatment and in survivors.53,54 AF start and maintenance are also associated with malignancy-related inflammatory processes, such as CRP, interleukin signaling, and platelet-leukocyte crosstalk. 55 According to prospective cohort data, NT-proBNP has a substantial correlation with incident AF, but high-sensitivity troponin and CRP have a less significant correlation. These biomarkers may help with risk estimation in cases when symptoms are vague. 56 Prothrombotic activity that is heightened in cancer is reflected by D-dimer, which is often raised in AF and stays higher even after successful cardioversion. 57
In order to contextualize AF symptoms and direct treatment, baseline transthoracic echocardiography (TTE) is essential for assessing chamber size, function, pericardial status, valvular disease, and left atrial (LA) remodeling. 58 LA size and strain may assist in refining AF risk and help understand dyspnea not explained by LV systolic function. Current cardio-oncology statements advocate TTE (or CMR) before potentially cardiotoxic medications and, where appropriate, during/after therapy. 59 Extended ambulatory monitoring is suitable for cancer patients suspected of having paroxysmal AF if symptoms are sporadic or if biomarker or echo evidence indicates concealed arrhythmia.
In cancer, traditional stroke/bleeding ratings have significant drawbacks. When comparing the prognostic abilities of CHA₂DS₂-VASc and HAS-BLED in AF patients with and without cancer, Ajabnoor et al discovered reduced discrimination in malignancy and advised against relying only on these instruments. 60 A gap in risk categorization was also highlighted by Ullah et al's study that CHA₂DS₂-VASc exhibited low accuracy for ischemic stroke prediction in cancer. 61 Even after controlling for CHA₂DS₂-VASc and HAS-BLED, the registry study reveals that anticoagulant usage differs significantly across oncology groups, indicating physician concerns regarding thrombocytopenia, mucosal illness, procedures, and drug–drug interactions. 62 Instead of using non-oncology ratings alone, risk assessment should practically include tumor type and stage, recent surgery, platelet counts, mucosal involvement, venous thromboembolism (VTE) history, and dynamic therapy phases.
Clinical ratings may be supplemented by biomarker-informed evaluation: increased NT-proBNP and CRP may indicate subclinical myocardial stress or greater AF/stroke risk, while consistently elevated D-dimer may indicate closer monitoring for cardioembolic events or thrombosis in AF patients with cancer. Accurate symptom attribution, personalized risk assessment, and collaborative decision-making regarding anticoagulation and rhythm strategies are supported by a thorough evaluation that includes the patient's history (including the timeline for cancer treatment), exam, lab results (including CBC for cytopenias), biomarkers (NT-proBNP, hs-troponin, CRP, D-dimer), and imaging.56–59
Management Approaches
Managing AF in cancer patients is particularly difficult since treatment choices must take into account the risk of bleeding, stroke prevention, and possible medication interactions with cancer treatments. A personalized, multidisciplinary strategy that incorporates anticoagulation, rate and rhythm management, and procedure alternatives according to the patient's malignancy status and overall prognosis is necessary for optimal tactics. 63 Figure 6 illustrates a summary for stepwise management of AF in cancer patients.

Stepwise management of AF in cancer patients. Management combines tailored anticoagulation, rate and rhythm control, and procedural strategies, with careful consideration of bleeding risk, stroke prevention, and drug–drug interactions. Created in BioRender. Mahafdah, B. (2025) https://BioRender.com/sxazect.
Anticoagulation Strategies:
A rising body of research compares warfarin with direct oral anticoagulants (DOACs) in AF patients with cancer. According to a meta-analysis by Cereda et al, DOACs were just as safe and efficacious as warfarin, if not more so, which supports their usage in situations when gastrointestinal absorption and medication interactions are tolerable. 64 Chai-Adisaksopha et al cautioned against regular long-term LMWH for AF unless temporary bridging is necessary, citing a greater risk of stroke with LMWH/UFH compared to warfarin and equivalent stroke/bleeding with DOACs compared to warfarin in a large observational study. 65 Not all cohorts agree: Khan et al showed that warfarin had a greater overall survival rate than DOACs, which probably reflects indication and cancer type confounding. These inconsistent findings emphasize the need for tailored selection. 66 Platelet counts, tumor location (eg, GI/GU mucosal hemorrhage), renal/hepatic function, procedural requirements, and drug–drug interactions with anticancer treatments should all be taken into account during practical selection. 67
Common limitations include mucosal diseases and cytopenias. Bridging with LMWH may be used temporarily around procedures, but it is not recommended long-term for AF stroke prevention. Expert practice suggests temporarily stopping anticoagulation at platelet counts <25–30 × 10⁹/L and considering a reduced-dose or cautious resumption at 30–50 × 10⁹/L, with escalation as counts recover. Both thrombotic and bleeding events are decreased by multidisciplinary planning centered on radiation, surgery, and chemotherapy regimens.64–67
Rate and Rhythm Control (Interactions and Tolerability)
β-blockers are often the first-line rate-control medications for symptomatic management in cardio-oncology, and they are favored when medicines may impair left ventricular function. 54 There are still choices for digoxin and non-dihydropyridine calcium-channel blockers (diltiazem/verapamil), but these should be used with care in cases of hypotension, extensive AV nodal illness, or medication interactions.67,68 Drug-drug interactions are a defining problem: a lot of supportive medications and anticancer drugs alter CYP3A4, P-glycoprotein, or QT, which interacts with class III drugs, amiodarone, DOACs, and diltiazem/verapamil. 67 When starting or adjusting AF therapy during cancer treatment, Beavers et al's AHA scientific statement offers thorough interaction tables and suggests proactive pharmacist assessment. 67 Amiodarone is frequently chosen for its effectiveness and relative hemodynamic tolerance when rhythm control is important (eg, persistent symptoms despite rate control, tachycardia-mediated cardiomyopathy, or to facilitate cancer therapy). However, clinicians should consider pulmonary, thyroid, hepatic, dermatologic, and other toxicities and interactions; careful monitoring is crucial in the oncology setting.68,69
Procedural Therapies:
A growing number of cancer patients who have symptomatic AF that is not responding to treatment are being evaluated for catheter ablation (CA). Margolis et al conducted a statewide all-comers registry study and discovered that cancer patients receiving AF ablation had more bleeding and infection problems than non-cancer patients, but there was no increase in in-hospital mortality or cardiac/neurological issues. 69 Agarwal et al's Europace data also showed that cancer patients had increased in-hospital mortality and complications after CA, highlighting the need for cautious selection and peri-procedural planning. 70 Although generalizability is restricted, the smaller data collected by Sharma et al indicate that AF recurrence after ablation may be equivalent to non-cancer groups. 71 In general, CA is doable, but it requires consideration of aspects unique to cancer, infection risk, and periprocedural anticoagulation.
For individuals with AF who are not candidates for long-term anticoagulation, left atrial appendage occlusion (LAAO) provides an option. According to a recent US review by Margolis et al, cancer patients receiving LAAO had no increased in-hospital mortality but more in-hospital comorbidities (such as AKI, heart failure, and tamponade). 72 The need for longer-term, cancer-specific outcome data is highlighted by a recent conference report that found similar rates of ischemic stroke, systemic embolization, and severe bleeding in cancer patients after percutaneous LAAO compared to non-cancer patients. 73 In practice, if a shared decision recognizes the paucity of oncology-focused research, LAAO may be appropriate when bleeding risk, thrombocytopenia, or procedure requirements restrict durable anticoagulation.
Integrated oncology–cardiology treatment is essential across all methods. Safety is enhanced and AF management is in line with cancer aims via a systematic route that clarifies the cancer phase and medications, checks for interactions, incorporates biomarkers and echocardiographic results, and dynamically reassesses the risk of stroke and bleeding (Table 3).
Integrated Mechanistic Framework for Cancer-Associated Atrial Fibrillation.

Multidisciplinary and Preventive Strategies
Cardio-Oncology Team-Based Care Models
The most effective and safest way to manage AF in people with cancer is to treat it as a shared clinical problem—oncology, cardiology/electrophysiology, pharmacy, nursing, primary care, in addition to palliative care (when needed), all working in a comprehensive approach from one pathway to achieve the optimal care and management. The 2022 ESC Cardio-Oncology guideline explicitly recommends integrated and structured pathways that begin before treatment (including risk profiling, baseline ECG/echo, and medication reconciliation), continue during therapy (trigger-based rhythm surveillance, biomarker use where appropriate), and also extend after cancer care (survivorship monitoring and hand-offs). 74 These programs explicitly assign responsibilities for rhythm monitoring, drug–drug interaction checks, and anticoagulation decisions, reducing fragmentation and speeding escalation when arrhythmias or toxicity emerge. Building these elements into clinic flow helps catch problems early, facilitates safe anticoagulation around procedures or thrombocytopenia, and ensures smooth transitions between services across the treatment continuum. 75
Recent reviews of cardio-oncology multidisciplinary teams emphasize defined roles for a comprehensive team (oncologist leads cancer therapy decisions; cardiologist/electrophysiologist manages AF strategy; pharmacists run interaction checks and platelet-based anticoagulation plans; nurses coordinate monitoring and education), showing significant improvement in patient-centred, timely care.76,77
For example, for agents with known arrhythmic signals as Bruton tyrosine kinase (BTK) inhibitors, therapy-specific workflows should be applied. Programs should screen for prior AF and cardiovascular disease, plan early rhythm surveillance, and agree in advance on dose adjustments or drug switches.78,79
Preventive and Supportive Care (Risk-Factor Modification and Monitoring)
Most AF risk factors during cancer care are modifiable. Counsel for weight reduction (≥10% weight loss when BMI > 27 kg/m²), regular aerobic activity (210 min/week of moderate–vigorous exercise), tight blood-pressure and glycemic control, alcohol moderation, smoking cessation, and sleep-apnea evaluation were interventions that the 2023 ACC/AHA AF guideline elevates to Class I recommendations for reducing AF burden and recurrence. 1
These statements are established and supported by multiple trials. Structured weight-loss and risk-factor programs (ARREST-AF, LEGACY) reduced AF burden and improved rhythm outcomes,80,81 while a randomized trial showed alcohol abstinence lowers AF recurrences. 82
Build these into chemotherapy teaching and survivorship plans. At the bedside, pair prevention with fit-for-purpose monitoring: start with a baseline ECG, then use interval ECGs or short ambulatory monitors around higher-risk events, such as major surgery, thoracic radiation, and cardio-toxic regimens. The AF guideline supports clinician-interpreted ECG confirmation and notes that consumer single-lead devices can help detect recurrences in selected, engaged patients; ensure someone will review alerts and act on them. 83
On the other hand, follow cardio-oncology guidance for biomarker-guided monitoring where cancer drugs carry cardiac risk: make troponin and natriuretic peptide testing available at baseline and at protocolized intervals, and set clear escalation rules to cardiology for new biomarker rises or arrhythmias. 74
Beyond general lifestyle counseling, adherence to a Mediterranean-style dietary pattern deserves specific emphasis in cancer patients with, or at risk of, AF. This diet, centered on extra-virgin olive oil, vegetables, fruits, whole grains, legumes, nuts, and fish, with limited red/processed meat and refined sugars, has consistently been associated with lower incidence of major chronic diseases and several cancers, likely through anti-inflammatory, antioxidant, antithrombotic, and metabolic effects that directly align with the mechanisms outlined in this review.84–87
Evidence from observational cohorts and mechanistic studies indicates that higher adherence to the Mediterranean diet is linked to reduced cancer burden and improved treatment-related outcomes (eg, better metabolic control, endothelial function, and oxidative stress profiles), and emerging data suggest it may also mitigate AF risk and recurrence by improving adiposity, blood pressure, glycemic control, and atrial substrate stability.88,89
Incorporating structured Mediterranean-style dietary advice into cardio-oncology and survivorship clinics is therefore a pragmatic, low-risk, and biologically coherent strategy that complements weight reduction, exercise, and other guideline-directed measures to reduce AF burden in this high-risk population.
Patient-Centered Decision Making
The 2023 ACC/AHA/ACCP/HRS AF guideline demonstrated shared decision-making (SDM) as an essential part in patient care, naming three recurring decisions that benefit most from SDM: whether and how to anticoagulate, whether to pursue rhythm control (including ablation), and how intensively to monitor for recurrences. In cancer, those choices must be aligned with what the patient is trying to achieve right now (eg, keeping chemotherapy on schedule, minimizing clinic visits, avoiding bleeding) and what they fear most (stroke vs bleeding), while accounting for tumor site, treatment phase, thrombocytopenia, drug–drug interactions, renal/hepatic function, and procedure timing. Using plain-language, individualized risk estimates (annual stroke and bleeding risk) and documenting the plan the team and patient agree on are core elements of best practice. 1
Decision aids can make these conversations faster and clearer. The Anticoagulation Choice tool from Mayo Clinic is designed to be used during the visit. 90 Also, randomized and pragmatic studies show SDM tools reduce decisional conflict, improve knowledge, and support appropriate anticoagulant uptake without increasing adverse events. Moreover, shared decisions improve engagement and adherence and are endorsed in major AF guidance. 91
Research Gaps and Future Directions
Mechanistic and Translational Needs
We still lack definitive explanations for why AF clusters with certain cancers and therapies. Priority work should prospectively map how inflammation, thrombo-inflammation, and atrial remodeling/fibrosis evolve across the cancer pathway (pre-treatment, on therapy, and into survivorship) using linked biospecimens, serial rhythm monitoring, and standardized imaging. 74 Mechanistically, therapy-specific signals deserve focused study, most notably the BTK-inhibitor class, where preclinical and emerging translational data suggest off-target effects that promote atrial fibrosis, calcium dysregulation, and AF susceptibility; defining which targets and which agents carry the greatest risk, and how that risk can be mitigated.92–94
On the measurement side, we need to validate actionable surrogate markers that can trigger prevention or treatment changes. Candidate biomarkers include inflammatory and thrombotic indices (eg, CRP, D-dimer), and myocardial stress/injury markers (NT-proBNP, high-sensitivity troponin). 95 For noninvasive phenotyping of the atrial substrate, left-atrial strain and atrial fibrosis by late-gadolinium–enhanced CMR show promise for risk stratification and tracking disease biology, but require harmonized acquisition/analysis and outcome-linked cut-points in cancer populations. 96
Finally, integrative reviews highlight that AF in cancer likely emerges from shared pathways (systemic inflammation, oxidative stress, prothrombotic states, and traditional cardiometabolic risks), and building and validating those models is an essential next step. 97
Cancer-Specific Risk Scores and Prediction Models
Common tools such as CHA₂DS₂-VASc and HAS-BLED were never designed for the oncology setting. They incompletely capture tumor site and stage, time-varying treatment phases, platelet dynamics, mucosal disease, and complex drug–drug interactions, which all impact thromboembolic and bleeding risk in cancer.98,99
Real-world evaluations are mixed; some analyses suggest CHA₂DS₂-VASc retains some discrimination for ischemic stroke in patients with cancer and AF, whereas others report poor calibration and performance, especially for bleeding risk using HAS-BLED. In particular, newly diagnosed cancer appears to increase arterial thromboembolism even at low CHA₂DS₂-VASc scores, implying that oncology-specific factors add risk not captured by traditional scores. These limitations are acknowledged in contemporary cardio-oncology and AF guidance and motivate the creation of multi-domain, cancer-specific prediction tools. 60 98–101
Next-generation models should integrate tumor characteristics (site, stage, mucosal involvement), treatment exposures (eg, BTK inhibitors, anthracyclines, thoracic radiation), laboratory trajectories (platelet counts, renal/hepatic function), and clinical comorbidity with time-updated signals (eg, current platelet band, active chemotherapy cycle, peri-operative windows), and optional biomarkers/imaging (natriuretic peptides, hs-troponin, left-atrial strain) where available. They should be trained and externally validated across tumor types and treatment eras, report calibration and decision-curve analyses, and output short, clinically actionable horizons (eg, 30-90 day stroke and major-bleeding risk during active therapy).74,102
Guidance documents from ESC Cardio-Oncology and AF societies emphasize precisely these gaps and encourage the development and validation of oncology-aware tools rather than relying on simple transposition of CHA₂DS₂-VASc/HAS-BLED cut-points. Until such scores are available and widely validated, clinicians should pair traditional tools with oncology-specific modifiers (platelets, drug interactions, lesion anatomy) and shared decision-making.74,102
Emerging risk-prediction models. In addition to traditional scoring systems, novel tools such as the C₂HEST score have recently demonstrated significantly improved discrimination for incident atrial fibrillation compared with CHADS₂ and CHA₂DS₂-VASc in non-oncology populations.103,104 The C₂HEST model incorporates variables including chronic obstructive pulmonary disease, hypertension, elderly age, and chronic kidney disease, thereby integrating systemic inflammatory and metabolic factors that parallel mechanisms observed in cancer-associated AF. Despite these advantages, no studies have prospectively validated or calibrated C₂HEST in patients with active malignancy. Future investigations should test the performance of these newer models across cancer types, treatment phases, and clinical outcomes (incident AF, thromboembolism, and bleeding) and compare their discrimination, calibration, and clinical utility against established tools. Such work could help determine whether incorporating oncology-specific modifiers into next-generation algorithms yields more accurate and individualized risk prediction in this high-risk population.
Trials of Anticoagulation and AF Interventions in Oncology
Most guidance for cancer-associated AF still depends on subgroup analyses of general AF trials and observational data, not purpose-built randomized evidence. Pragmatic RCTs are needed that directly compare DOAC strategies versus warfarin across common cancer contexts (solid tumors, hematologic malignancies, active therapy vs remission), embed platelet-informed dosing/interruptions and peri-procedure algorithms, test rhythm-control pathways (including antiarrhythmic drugs and catheter ablation) during active cancer care, and evaluate structured prevention bundles (weight, alcohol, sleep apnea, BP control) delivered inside oncology workflows. Moreover, emphasize cardio-oncology guidance for oncology-specific protocols and provide interim platelet cut-offs and drug–drug interaction frameworks to build into such trials. 105
Registries and claims-based studies should be used to design the trials we need: identify high-risk tumor–therapy niches (eg, GI/GU mucosal tumors, BTK-inhibitor regimens), define realistic platelet bands and interaction checks, and set short, actionable horizons (30-90 days) that match chemotherapy cycles. 7
On the rhythm-control side, early data suggest that AF ablation can be feasible in patients with cancer, but outcomes vary, and complication risks may be higher during active therapy. Therefore, it is another area ripe for pragmatic randomization with cardio-oncology co-management embedded from the start.106,107
Conclusion
This narrative review has set out to integrate current evidence on how cancer, inflammation, thrombosis, and cancer-directed therapies converge to promote atrial fibrillation, and to outline practical implications for clinical evaluation, anticoagulation, rhythm control, and preventive strategies in oncology patients. Taken together, existing data support viewing AF in cancer not as an incidental comorbidity, but as a phenotype of shared inflammatory and prothrombotic pathways, treatment-related injury, and competing risks that demand oncology-specific risk stratification and coordinated cardio-oncology care. However, our conclusions are inherently constrained by the narrative, non-systematic design. Relevant studies may have been missed, selection of evidence may be influenced by availability and reporting bias, and heterogeneity in cancer types, treatments, AF definitions, and follow-up across the included literature limits comparability. Robust oncology-focused risk scores, randomized trials of anticoagulation and rhythm strategies, and prospective mechanistic studies are needed before uniform treatment algorithms can be defined. In the interim, individualized, multidisciplinary decision-making -grounded in current guidelines but adapted to tumour biology, treatment phase, platelet dynamics, and patient priorities- remains the most rational approach for managing AF in this high-risk population.
Footnotes
Abbreviations
Acknowledgments
None.
Ethical Approval
Ethical approval is not required for this review article.
Conflicts of Interest
N/A.
Clinical Trial Number
Not applicable
Authors contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent Statement
Not applicable.
Data Availability Statement
This study did not involve the creation or analysis of any new data. Therefore, data sharing is not applicable.
Declarations of Interest
We affirm that this work is original, has not been published previously, and is not under consideration for publication elsewhere. None of the paper's contents has been previously published. All authors have read and approved the manuscript. We also provide full disclosure of any relationships with industry.
Provenance and Peer Review
Not commissioned, externally peer-reviewed.
