Abstract
Background
Venous thromboembolism (VTE) often necessitates extended anticoagulation to reduce the risk of recurrence. While direct oral anticoagulants (DOACs) have largely replaced vitamin K antagonists due to their improved safety and convenience, the optimal long-term dosing strategy (reduced vs full dose) remains uncertain. This systematic review assesses the efficacy and safety outcomes of reduced-dose DOACs compared with full-dose therapy for extended VTE treatment.
Methods
We conducted a systematic review of randomized controlled trials (RCTs) comparing reduced-dose and full-dose DOACs in patients with VTE. Due to heterogeneity in study designs and outcomes, a narrative synthesis was performed.
Results
Three RCTs were included. In AMPLIFY-EXT, recurrent VTE or death occurred in 1.7% of patients receiving apixaban (both 2.5 mg and 5 mg) versus 8.8% with placebo. In EINSTEIN CHOICE, recurrent VTE occurred in 1.2% and 1.5% of patients receiving rivaroxaban 10 mg and 20 mg, respectively, compared with 4.4% with aspirin. The safety outcomes were comparable between the two DOAC doses in AMPLIFY-EXT and EINSTEIN CHOICE. RENOVE reported 5-year cumulative incidences of recurrent VTE of 2.2% with reduced-dose and 1.8% with full-dose DOACs (hazard ratio 1.32, 95% confidence interval [CI]: 0.67-2.60; P = .23 for non-inferiority). Major or clinically relevant non-major bleeding events occurred in 9.9% patients in the reduced-dose group and 15.2% patients in the full-dose group (HR 0.61; 95% CI 0.48-0.79).
Conclusion
Current evidence suggest that reduced-dose DOACs provide efficacy comparable to full-dose regimens without a corresponding increase in bleeding events in the extended treatment of VTE.
Introduction
Venous thromboembolism (VTE), encompassing deep vein thrombosis (DVT) and pulmonary embolism (PE), represents a major global health burden associated with significant morbidity and mortality.1,2 Patients with unprovoked VTE or persistent risk factors are at particularly high risk of recurrence, often necessitating extended anticoagulation therapy.3,4 Direct oral anticoagulants (DOACs), including dabigatran, apixaban, rivaroxaban, and edoxaban, have largely superseded vitamin K antagonists (VKAs) due to their more favorable safety profile, predictable pharmacokinetics, and elimination of the need for routine monitoring.5,6 Nevertheless, determining the optimal long-term dosing strategy to balance efficacy in preventing recurrent VTE against the risk of bleeding remains a clinical challenge. 7
Recent randomized controlled trials (RCTs) have investigated the efficacy and safety of reduced-dose versus full-dose DOAC regimens for extended VTE treatment. The AMPLIFY-EXT trial compared apixaban (2.5 mg or 5 mg twice daily) with placebo 8 ; the EINSTEIN CHOICE trial evaluated rivaroxaban (20 mg or 10 mg once daily) against aspirin 9 ; and the RENOVE trial directly compared reduced-dose (apixaban 2.5 mg or rivaroxaban 10 mg) with full-dose (apixaban 5 mg or rivaroxaban 20 mg) DOACs. 10 While some trials suggested comparable efficacy between dose regimens, others indicated that although reduced-dose DOACs did not meet non-inferiority criteria compared to full-dose therapy, they were associated with a significantly lower incidence of bleeding.8–10 This systematic review synthesizes current evidence from clinical trials assessing the efficacy and safety outcomes of reduced-dose DOACs compared with full-dose therapy for extended VTE treatment.
Methodology
Literature Search
A comprehensive literature search was conducted using the PubMed database up to October 2025 to identify relevant studies. The literature search used the following Medical Subject Headings (MeSH) and free-text terms: (“Venous Thromboembolism”[MeSH] OR “Deep Vein Thrombosis”[MeSH] OR “Pulmonary Embolism”[MeSH]) AND (“Direct Oral Anticoagulants”[MeSH] OR apixaban OR rivaroxaban OR dabigatran OR edoxaban) AND (“extended treatment” OR “long-term anticoagulation”) (Supplementary Table S1). Additionally, the reference lists of included studies and prior reviews11,12 were manually reviewed to identify any additional publications. No language restrictions were applied to maximize retrieval of relevant publications.
Eligibility Criteria
Study inclusion criteria were defined according to the PICOS framework: (1) Population: Adult patients (aged ≥18 years) diagnosed with VTE; (2) Intervention: Extended treatment (defined as anticoagulation beyond the initial 6 to 12 months following acute VTE) with a DOAC; (3) Comparison: Reduced-dose versus full-dose DOAC; (4) Outcomes: Efficacy outcomes (eg, recurrent VTE, death related to VTE) and safety outcomes (eg, major bleeding events, major or clinically relevant non-major bleeding events) as defined by the original studies; (5) Study design: RCTs.
Studies were excluded if they were conference abstracts (due to inadequate methodological and outcome reporting), animal studies, case reports, review articles, meta-analyses, editorials, and observational studies. RCTs conducted in specific VTE subpopulations, such as patients with cancer-associated VTE were also excluded.
Study Selection and Data Extraction
Two independent reviewers screened the titles and abstracts of identified records against the eligibility criteria. Full-text articles were retrieved for potentially eligible studies, and any discrepancies between reviewers were resolved through consensus or by consultation with a third reviewer.
Data were systematically extracted from each included study. The extracted information included: (1) Study characteristics: first author, year of publication, study design, trial acronym, study locations, sample size, and duration of follow-up; (2) Participant characteristics: demographics, creatinine clearance distribution, comorbidities, and prior anticoagulant treatment; (3) Intervention details: type and dosage of DOAC used; and (4) Outcomes: event rates for primary efficacy and safety outcomes.
Risk of Bias Assessment
The methodological quality and risk of bias of the included RCTs were assessed using the Cochrane Risk of Bias 2 (RoB 2) tool. 13 This tool evaluates five domains: (1) bias arising from the randomization process, (2) bias due to deviations from intended interventions, (3) bias due to missing outcome data, (4) bias in measurement of the outcome, and (5) bias in selection of the reported result. Each domain was judged as conferring “low risk,” “some concerns,” or “high risk” of bias. 14
Data Synthesis
Given the heterogeneity in study designs, patient populations, interventions, and outcome definitions, coupled with the limited number of eligible trials, a meta-analysis was deemed inappropriate to avoid potentially misleading conclusions. Consequently, a narrative synthesis was performed to comprehensively summarize and interpret the findings regarding the efficacy and safety profiles of reduced-dose versus full-dose DOACs in patients with VTE.
Results
Study Selection
The initial PubMed search yielded 15 records. After screening titles and abstracts, 6 studies were excluded due to irrelevance. The remaining 9 studies underwent full-text review, and 6 of them were excluded due to their failure to meet inclusion criteria.15–20 Finally, 3 RCTs (AMPLIFY-EXT, 8 EINSTEIN CHOICE, 9 and RENOVE 10 ) were deemed eligible and included in the narrative synthesis.
Baseline Characteristics of Included Studies
Table 1 summarizes the baseline characteristics of the three included RCTs. The AMPLIFY-EXT trial enrolled 2486 patients who had completed 6 to 12 months of anticoagulation and randomized them to receive apixaban (2.5 mg or 5 mg twice daily) or placebo. 8 The EINSTEIN CHOICE trial, a phase 3 study, randomized 3396 patients who had completed 6 to 12 months of anticoagulation to receive rivaroxaban (10 mg or 20 mg once daily) or aspirin (100 mg once daily). 9 The RENOVE trial included 2768 patients deemed to require extended anticoagulation after 6 to 24 months of initial therapy and randomized them to receive either a reduced-dose (apixaban 2.5 mg twice daily or rivaroxaban 10 mg once daily) or a full-dose (apixaban 5 mg twice daily or rivaroxaban 20 mg once daily) DOAC regimen. 10
Baseline Characteristics of Included Studies.
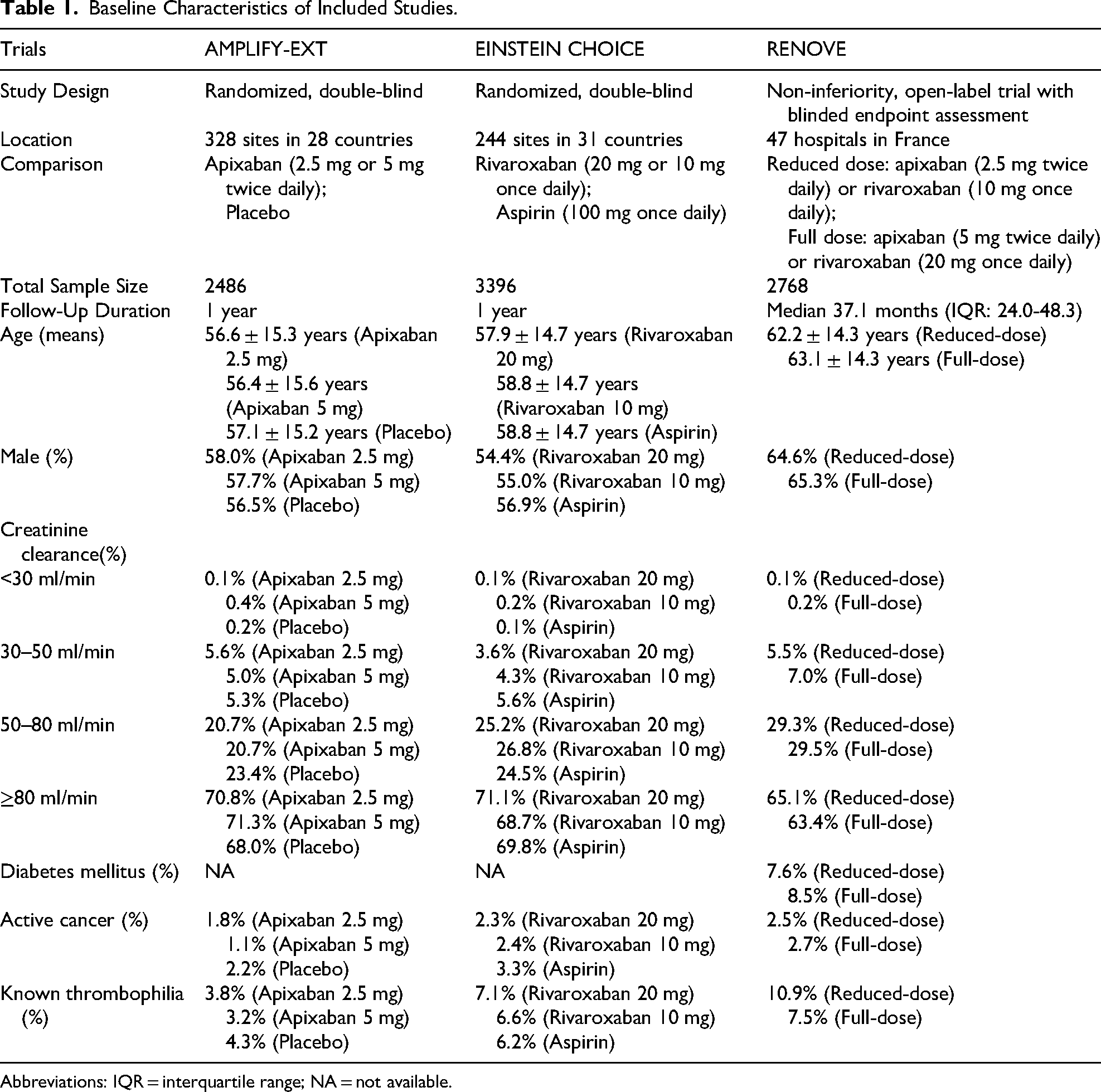
Abbreviations: IQR = interquartile range; NA = not available.
Risk of Bias Assessment
Assessment using the RoB 2 tool indicated a low risk of bias for three included trials.8–10 The randomization processes were adequately described and resulted in balanced groups. Interventions were implemented as intended, with appropriate blinding procedures in AMPLIFY-EXT and EINSTEIN CHOICE. Rates of missing outcome data were low. Outcome measurement was performed independently by blinded adjudication committees. There was no evidence of selective outcome reporting.
Efficacy and Safety Outcomes Between Different Doses of DOACs
The AMPLIFY-EXT trial, a randomized double-blind study, aimed to determine the efficacy of lower-dose apixaban (2.5 mg twice daily) and its association with reduced bleeding compared to the higher dose (5 mg twice daily). 8 Symptomatic recurrent VTE or VTE-related death occurred in 8.8% of patients receiving placebo, compared to 1.7% in both the 2.5 mg and 5 mg apixaban groups. The relative risk in symptomatic recurrent VTE or VTE-related death between the two apixaban doses (2.5 mg vs 5 mg) was 0.97 (95% confidence interval [CI], 0.46 to 2.02), indicating no significant difference in efficacy. Major bleeding events occurred in 0.5% of the placebo group, 0.2% in the 2.5 mg apixaban group, and 0.1% in the 5 mg apixaban group. The relative risk in major bleeding events between the 2.5 mg and 5 mg doses was 1.93 (95% CI 0.18-21.25), which was not statistically significant. 8 There were no differences in major or clinically relevant nonmajor bleeding events between groups (relative risk 0.74; 95% CI 0.46-1.22).
The EINSTEIN CHOICE trial, a phase 3 randomized double-blind study, compared the efficacy and safety of two rivaroxaban doses (10 mg and 20 mg once daily) with aspirin (100 mg once daily). 9 Both rivaroxaban doses were superior to aspirin for preventing recurrent VTE (rates: aspirin 4.4%, rivaroxaban 20 mg 1.5%, rivaroxaban 10 mg 1.2%; P < .001 for both comparisons). The similar efficacy outcome of recurrent VTE between the two rivaroxaban doses (20 mg vs 10 mg: hazard ratio[HR] 1.34; 95% CI 0.65-2.75) suggests the reduced dose of rivaroxaban is a potential option. Major bleeding event rates were low and comparable: 0.5% (20 mg), 0.4% (10 mg), and 0.3% (aspirin). 9 There were no differences in major bleeding events (HR 1.23; 95% CI 0.37-4.03) and major or clinically relevant nonmajor bleeding events (HR 1.37; 95% CI 0.83-2.26) between the two rivaroxaban doses.
The RENOVE trial was a randomized, open-label, blinded-endpoint, non-inferiority study designed to assess whether reduced-dose DOACs were non-inferior to full-dose DOACs in terms of efficacy and superior in terms of safety among patients at high risk of recurrent VTE. 10 The primary efficacy outcome (symptomatic recurrent fatal or non-fatal VTE) occurred in 19/1383 patients (5-year cumulative incidence 2.2%; 95% CI 1.1-3.3) in the reduced-dose group and 15/1385 patients (1.8%; 95% CI 0.8-2.7) in the full-dose group (adjusted hazard ratio[HR] 1.32; 95% CI 0.67-2.60; absolute difference 0.40%; 95% CI −1.05 to 1.85; p = .23 for non-inferiority). In addition, major or clinically relevant non-major bleeding events occurred in 96 patients (9.9%; 95% CI 7.7-12.1) in the reduced-dose group and 154 patients (15.2%; 95% CI 12.8-17.6) in the full-dose group (adjusted HR 0.61; 95% CI 0.48-0.79), demonstrating a significant reduction in bleeding with the reduced dose. 10
Discussion
This systematic review synthesizes evidence from three pivotal RCTs (AMPLIFY-EXT, EINSTEIN-CHOICE, and RENOVE) evaluating reduced-dose versus full-dose DOACs for extended VTE treatment. Collectively, these trials indicate that reduced-dose DOAC regimens offer comparable efficacy to full-dose therapy in preventing recurrent VTE without increased bleeding risk events, although important uncertainties remain regarding their applicability across all patient subgroups.
Both the RENOVE and AMPLIFY-EXT trials reported notably low cumulative incidence rates of recurrent VTE with extended anticoagulation, with no statistically significant differences observed between reduced-dose and full-dose groups.8,10 This suggests that dose reduction may not substantially compromise thromboprophylactic efficacy, even in high-risk populations such as those with unprovoked VTE or severe initial presentations.21,22 However, the RENOVE trial did not formally establish the non-inferiority of the reduced-dose regimen, 10 leaving open the question of whether certain patient subgroups might still derive greater benefit from full-dose therapy. In addition, the EINSTEIN-CHOICE trial provided indirect support for dose reduction by demonstrating that a prophylactic dose of rivaroxaban (10 mg daily) was significantly more effective than aspirin and non-inferior to the treatment dose (20 mg daily) in preventing VTE recurrence, with comparable major bleeding event rates. 9
The RENOVE trial demonstrated a 39% relative risk reduction in major or clinically relevant non-major bleeding events with reduced-dose therapy, translating to a 33% reduction in net adverse clinical events. Similarly, the AMPLIFY-EXT trial suggested a high number needed to harm for major bleeding events, reinforcing the safety advantage of lower doses. These findings underscore the potential for personalized anticoagulation strategies, especially for patients at heightened risk of bleeding.22,23 It is important to note differences in the studied populations: AMPLIFY-EXT and EINSTEIN CHOICE included patients for whom there was clinical equipoise regarding continued anticoagulation, whereas RENOVE specifically enrolled patients with a clear indication for extended therapy due to high recurrence risk. This distinction may explain the lower attrition rates and the higher observed bleeding incidence in the full-dose group of RENOVE compared to the shorter-term bleeding rates in AMPLIFY-EXT, highlighting the critical role of patient selection in interpreting these outcomes.
While reduced-dose DOACs appear effective for preventing recurrent VTE with minimal efficacy loss compared to full-dose regimens, the failure to confirm non-inferiority in RENOVE and the underrepresentation of specific high-risk subgroups (eg, severe renal impairment, active cancer) in these trials caution against overgeneralization. Future research should prioritize identifying patient subgroups that derive the greatest benefit from reduced-dose therapy (eg, the elderly, those with high bleeding risk scores) versus those who may require full-dose anticoagulation (eg, patients with severe thrombophilia, recurrent VTE on prophylaxis, or antiphospholipid syndrome). Furthermore, long-term studies exceeding 3 to 5 years are needed to assess the durability of the efficacy and safety benefits of reduced-dose DOACs, given that VTE often necessitates lifelong management. Head-to-head comparisons between different DOAC agents at reduced doses would also provide valuable evidence for guiding optimal drug and dose selection.
Limitations
A recent meta-analysis by Akhtar et al synthesized four RCTs (n = 8421) comparing reduced-dose versus full-dose DOACs for extended VTE. 11 However, this study included the API-CAT trial, 16 which exclusively enrolled patients with cancer, a population with distinct thrombotic and bleeding risks. 24 In contrast, our systematic review focused solely on three pivotal RCTs (AMPLIFY-EXT, EINSTEIN CHOICE and RENOVE) that investigated extended anticoagulation in unselected VTE populations. This design choice ensured a more homogeneous evidence base and improved clinical applicability to the broader VTE population.
Our current review has several limitations. First, the control groups in AMPLIFY-EXT (placebo) and EINSTEIN CHOICE (aspirin) do not represent the current standard of care for patients with a definite indication for extended anticoagulation, potentially limiting the direct applicability of those comparisons to clinical practice where a DOAC is the preferred agent. Second, the open-label design of the RENOVE trial, though mitigated by blinded endpoint adjudication, may have introduced potential bias. Third, the follow-up duration of 12 months in AMPLIFY-EXT and EINSTEIN CHOICE is insufficient to evaluate long-term risks and benefits, and even the median 3-year follow-up in RENOVE remains shorter than the often lifelong treatment duration required in practice. Finally, the underrepresentation of elderly patients, those with significant renal impairment, and patients with active cancer-associated VTE in these trials limits the generalizability of the findings to these important subgroups.
Conclusion
Current evidence suggest that reduced-dose DOACs provide efficacy comparable to full-dose regimens without a corresponding increase in bleeding events in the extended treatment of VTE. Although the RENOVE trial did not formally confirm the non-inferiority of reduced-dose DOACs for efficacy, its findings, combined with those from AMPLIFY-EXT and EINSTEIN CHOICE, suggest that reduced-dose regimens represent a valuable and often preferable therapeutic option, particularly for patients at increased risk of bleeding.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251408061 - Supplemental material for Reduced-Dose Versus Full-Dose Direct Oral Anticoagulants for Extended Treatment of Venous Thromboembolism
Supplemental material, sj-docx-1-cat-10.1177_10760296251408061 for Reduced-Dose Versus Full-Dose Direct Oral Anticoagulants for Extended Treatment of Venous Thromboembolism by Deming Deng, Zixuan Xu and Wengen Zhu in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Ethical Approval
Not required.
Author Contributions
DM-D and ZX-X were responsible for the study design, literature search, systematic review, data collection, interpretation of the results, and manuscript drafting. WG-Z., as the principal investigator, contributed to the study design, evaluated all results, and revised the manuscript for typographical and grammatical accuracy.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
All relevant data and materials are presented in the paper.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
