Abstract
Objective
This study aimed to develop and validate a predictive model for venous thromboembolism (VTE) risk in high-altitude hospitalized patients by combining the Caprini score with D-dimer levels and assessing the impact of platelet count in a unique cohort.
Methods
We enrolled 1002 hospitalized patients at Lhasa People's Hospital (3650 m). Clinical data, including Caprini score, D-dimer, and platelet count, were collected at admission. Logistic regression identified predictors of VTE, and model performance was evaluated using ROC curves, with sensitivity analyses excluding patients on thromboprophylaxis. External validation was performed in 400 patients from the Tibet Military Region General Hospital using the same protocol.
Results
Among the 1002 hospitalized patients, 135 (13.5%) developed venous thromboembolism (VTE), consisting of 61 cases of deep vein thrombosis (DVT), 42 cases of pulmonary embolism (PE), and 32 cases of both DVT and PE. Multivariate analysis identified Caprini score, D-dimer levels, platelet count, and C-reactive protein (CRP) as independent predictors of VTE. However, platelet count and CRP were excluded from the final predictive model due to their limited incremental predictive value. The combined Caprini–D-dimer model demonstrated superior predictive accuracy with an area under the curve (AUC) of 0.89, compared to 0.84 for Caprini score and 0.81 for D-dimer alone. Sensitivity analysis excluding patients who received thromboprophylaxis improved the AUC to 0.904. External validation in a cohort of 400 patients confirmed the model's robustness with an AUC of 0.86.
Conclusion
The combined Caprini score and D-dimer model provides a more accurate tool for predicting VTE risk in high-altitude hospitalized patients compared to individual predictors. This model offers enhanced sensitivity and specificity, making it a valuable tool for clinical decision-making in high-altitude settings. The model maintained high predictive performance after excluding patients receiving prophylactic anticoagulation, suggesting robustness across different clinical scenarios.
Introduction
Venous thromboembolism (VTE) is a major cause of morbidity and mortality in hospitalized patients. 1 It is estimated that VTE affects millions of people worldwide each year, with significant consequences if left untreated.2,3 In high-altitude regions, such as Tibet, the physiological stressors of hypoxia significantly increase the risk of thromboembolic events.4,5 Hypoxia can lead to a hypercoagulable state, endothelial dysfunction, and increased blood viscosity, all of which contribute to the heightened risk of VTE in these environments.6-8 Among the latest guidelines-recommended risk assessment tools, although the Padua Prediction Score (PPS) is widely used for VTE risk assessment in medical inpatients, recent studies indicate that it may not be fully applicable to certain populations, such as those with acute respiratory conditions, as it tends to underestimate the VTE risk in these patients.9,10 Other risk-prediction tools, such as the Caprini score and D-dimer levels, have also been widely employed for VTE risk prediction in medical inpatients and may offer better accuracy in these contexts.11,12 The Caprini score has been validated across diverse patient populations for its effectiveness in stratifying VTE risk based on clinical factors. 13 However, these models may not adequately address the distinct challenges of high-altitude environments, where the interaction between hypoxia and thrombosis presents added complexity.14,15
This study aims first to validate the predictive capability of the existing Caprini score in high-altitude hospitalized patients and, if necessary, seek ways to improve its accuracy. Based on initial findings, we will explore the addition of D-dimer levels to enhance the model's performance. We hypothesize that the combination of these two predictors will offer a more precise and tailored approach to assessing VTE risk in this unique population, outperforming the individual components when used alone.
Materials and Methods
Study Design and Population
This prospective cohort study was conducted at Lhasa People's Hospital, located at an altitude of approximately 3650 meters above sea level. A total of 1002 patients were included in the study, who were admitted to the Department of Pulmonary and Critical Care Medicine, Lhasa People's Hospital, from July 2022 to July 2024 consecutively. Eligible participants were adults (≥18 years), hospitalized for at least 3 days, and had complete clinical and laboratory data available. Patients with VTE events confirmed within the first 24 h of hospitalization were excluded. Other exclusion criteria included active bleeding, severe liver or renal dysfunction, known coagulation disorders, anticoagulant use, previous VTE, or surgery lasting more than 45 min during hospitalization. Patients with a life expectancy of less than 3 months were also excluded.16,17
The derivation cohort sample size was calculated using the 10 Events Per Predictor (EPP) guideline to ensure sufficient statistical power for the development of a multivariable predictive model. To account for the planned inclusion of up to 10 predictors in the model, a minimum of 100 VTE events was necessary. Preliminary data estimated the incidence of VTE among hospitalized internal medicine patients in the Tibetan region at 13.97%. The sample size estimation was based on the following formula: n = 100/0.1397≈715. This calculation demonstrated that a sample size of at least 715 patients was required to secure the desired number of VTE events for robust predictive model development.
Data Collection
Data were systematically collected for each patient upon admission and during their hospital stay. Key variables included demographic information such as age, sex, ethnicity, smoking and drinking history, as well as clinical details like pulmonary infections, lower limb swelling, and immobility status. Laboratory parameters were recorded within 24 h of admission, including white blood cell count, platelet count, prothrombin time, international normalized ratio, C-reactive protein levels, and D-dimer levels, among other relevant biomarkers. The primary outcome was the occurrence of venous thromboembolism (VTE), which includes deep vein thrombosis (DVT) and pulmonary embolism (PE). The diagnosis of DVT was confirmed through compression ultrasonography of the lower limbs, following established clinical guidelines, 10 while duplex ultrasonography was used in uncertain cases to assess venous compression and blood flow. 18 Pulmonary embolism was diagnosed using computed tomography pulmonary angiography (CTPA), the gold standard for detecting thrombi in the pulmonary arteries. 19 In cases where CTPA was contraindicated or unavailable, a ventilation-perfusion (V/Q) scan was employed. 20 Additionally, patients with elevated D-dimer levels, defined as three times the upper limit of normal, underwent further imaging regardless of symptoms to rule out asymptomatic VTE. 21 Diagnostic risk stratification tools, such as the Wells and Geneva scores, were applied to guide the diagnosis of PE in symptomatic patients.22,23 Importantly, patients with VTE confirmed within the first 24 h of admission were excluded from the study to ensure that pre-existing cases were not included in the analysis. Furthermore, detailed information on thromboprophylaxis strategies (including pharmacological agents, physical measures, and patient education) was recorded for all patients to enable planned subgroup analyses evaluating the impact of prophylactic anticoagulation on model performance.
Data collection continued throughout the hospital stay, with updates on laboratory results, clinical assessments, and imaging studies. Patient confidentiality was maintained, and all procedures were conducted in accordance with institutional ethical guidelines. 24
Statistical Analysis
All statistical analyses were conducted using SPSS software, version 26.0 (IBM Corp.). Descriptive statistics were used to summarize baseline characteristics, with continuous variables presented as mean ± standard deviation (SD) or median and interquartile range (IQR) for non-normally distributed data, and categorical variables expressed as frequencies and percentages. Univariate logistic regression was performed to identify significant predictors of VTE, including variables such as the Caprini score, D-dimer levels, platelet count (PLT), and C-reactive protein (CRP). Variables with a P-value < .05 in the univariate analysis were subsequently included in a multivariate logistic regression model to determine independent risk factors for VTE. 25
A multivariable logistic regression model was developed using significant predictors identified through univariate analysis. The final model for predicting VTE risk can be represented by the following equation: logit(p) = −4.563 + 0.629 × Caprini score + 2.182 × lgD2, where ppp represents the probability of developing VTE. The logistic regression coefficients were derived from a dataset comprising variables such as Caprini score and D-dimer (lgD2).
To further assess the performance of the model, sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were calculated at different cut-off points to determine the optimal threshold for VTE risk prediction. Calibration was evaluated using the Hosmer-Lemeshow goodness-of-fit test, confirming the model's reliability in predicting outcomes. All statistical tests were two-sided, with a P-value < .05 considered statistically significant.
To evaluate the generalizability of the model in different high-altitude hospital settings, an external validation was conducted at the Tibet Military Region General Hospital. A total of 400 hospitalized patients meeting the same inclusion and exclusion criteria as the primary study were included. Data collection followed the same protocol as the primary study, including demographic information, laboratory parameters, and clinical assessments. The Caprini score and D-dimer levels were calculated using the same methods, and the occurrence of VTE was confirmed through compression ultrasonography and computed tomography pulmonary angiography (CTPA) when necessary. The predictive performance of the combined model, as well as individual predictors, was assessed using ROC curve analysis, with AUC values compared to the original study cohort.
Ethical Considerations
This study was conducted in accordance with the principles of the Declaration of Helsinki. Ethical approval was obtained from the Lhasa People's Hospital Ethics Committee (approval number: SYLL2224079) and the Beijing Chao-Yang Hospital Ethics Committee (approval number: 2024-ke-808). All patient data were anonymized to maintain confidentiality, and the need for informed consent was waived due to the retrospective nature of the study.
Results
Patient Disposition
See Figure 1.
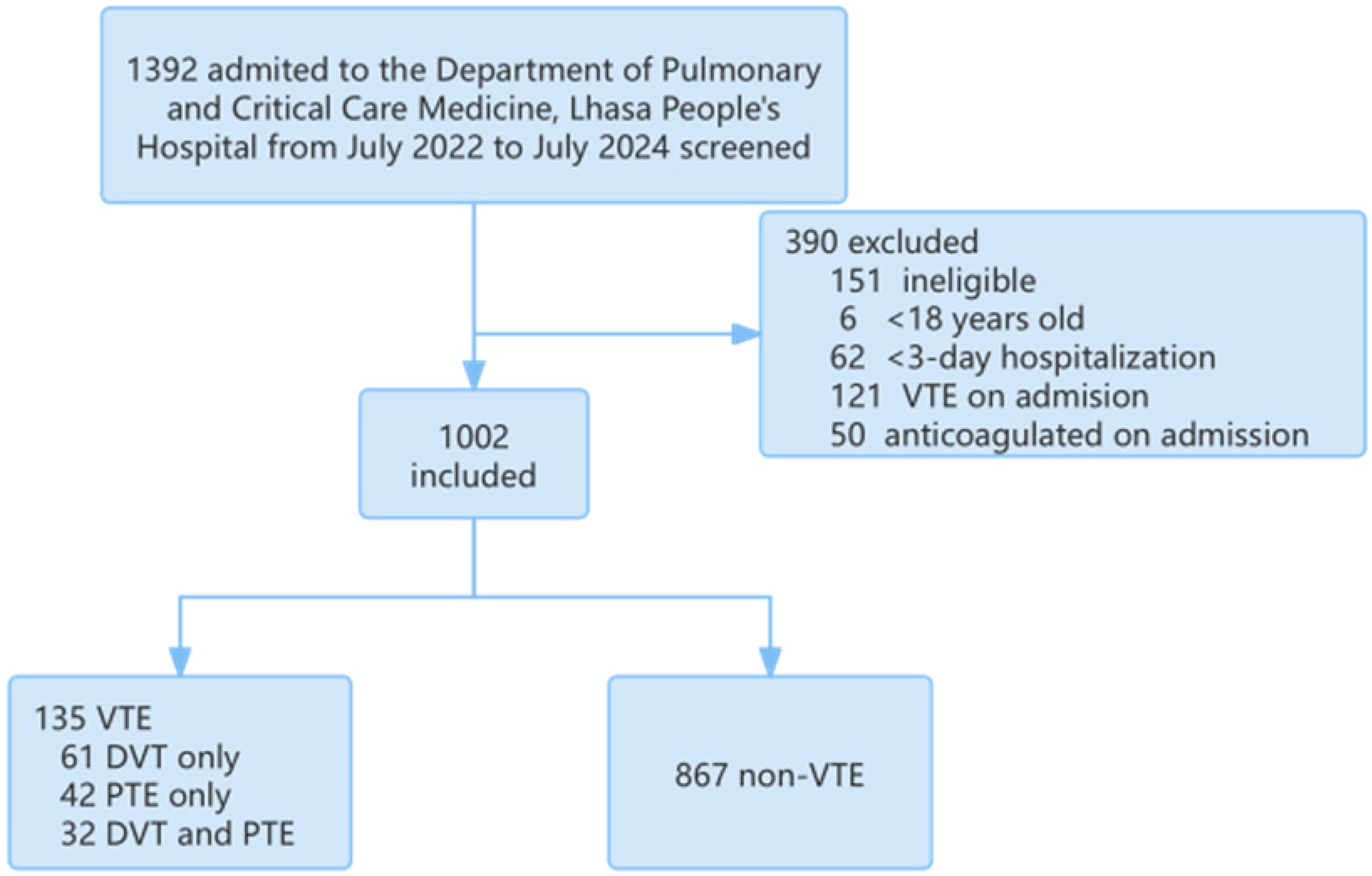
Patient Disposition.
Patient Enrollment and Endpoint Events
A total of 1002 patients were consecutively enrolled in this study, all admitted to Lhasa People's Hospital between July 2022 and July 2024. These patients met the inclusion criteria, having been hospitalized for at least three days with complete clinical and laboratory data available. During the course of hospitalization, 135 patients (13.5%) developed venous thromboembolism (VTE), which included 61 cases of deep vein thrombosis (DVT) only, 42 cases of pulmonary embolism (PE) only, and 32 cases of both DVT and PE. The diagnosis of VTE was confirmed through compression ultrasonography for DVT and computed tomography pulmonary angiography (CTPA) for PE. Importantly, no patients with VTE identified within the first 24 h of admission were included in the study, as these cases were excluded to focus on VTE events occurring during hospitalization. The median time to VTE diagnosis was 7 days (interquartile range: 5-10 days) from admission. Patients who developed VTE demonstrated significantly higher risk factors, including elevated Caprini scores and D-dimer levels, compared to those who did not experience VTE. No major bleeding events or fatal outcomes related to anticoagulant therapy were observed. These results underscore the significance of VTE as a common complication in hospitalized patients at high altitudes and highlight the need for accurate risk prediction and timely prophylaxis.
Thromboprophylaxis usage varied across the study population. A total of 1002 patients were included in the analysis, and the thromboprophylaxis strategies employed included a range of treatments. Some patients received only patient education or no intervention, while others were given physical measures plus education. A portion of patients received pharmacological interventions, such as dalteparin (5000U-10000U, subcutaneous injection, once daily), low-molecular-weight heparin (5000U-10000U, subcutaneous injection, once daily), and enoxaparin (4000U-8000U, subcutaneous injection, once daily). Additionally, a small group of patients were given rivaroxaban (10-20 mg, oral administration, once daily) and edoxaban (30 mg, oral administration, once daily). The diversity of thromboprophylaxis usage reflects the variety of clinical approaches employed in preventing VTE in hospitalized patients. The inclusion of these different prevention strategies underscores the importance of timely prophylaxis in reducing the risk of VTE, particularly in the high-altitude environment of Lhasa.
Analysis of Baseline Data
The baseline characteristics of the study population, including gender, age, body mass index (BMI), pulse rate, respiration rate, and blood pressure, were analyzed and compared between the VTE group and the non-VTE group. Upon examination within 24 h of admission, several significant differences were identified between the two groups. Patients in the VTE group were generally older, with a mean age of 69.56 ± 11.15 years, compared to 58.21 ± 16.25 years in the non-VTE group (p < .001). Additionally, pulse rate and respiration rate were significantly higher in the VTE group, with a mean pulse of 87.89 ± 18.76 beats per minute compared to 83.25 ± 15.15 beats per minute in the non-VTE group (p < .001). No statistically significant differences were observed in gender distribution, systolic blood pressure, or diastolic blood pressure between the two groups. These findings suggest that age, elevated pulse, and increased respiration rate are associated with a higher risk of VTE in this patient cohort, highlighting their potential value as predictive factors in the development of venous thromboembolism (Table 1).
Analysis of Baseline Data Between Two Groups.

The results of comparing the complete blood count, coagulation profile, liver and kidney function, lipid profile, Caprini score, and D-DI/PLT within 24 h of admission between the two groups revealed significant differences in white blood cell count, hematocrit, platelet count, prothrombin time, D-dimer, total bilirubin, blood urea nitrogen, uric acid, Caprini score, and D-DI/PLT (p < .05) (Table 2).
Comparison of Examination and Other Indicators Within 24 Hours of Admission Between the Two Groups.

Abbreviations: WBC, white blood cell count; RBC, red blood cell count; HGB, hemoglobin; HCT, hematocrit; PLT, platelet count; PT, prothrombin time; APTT, activated partial thromboplastin time; INR, international normalized ratio; FIB, fibrinogen; D-DI, D-dimer; ALT, alanine aminotransferase; AST, aspartate aminotransferase; TBIL, total bilirubin; Cr, creatinine; BUN, blood urea nitrogen; UA, uric acid; TC, total cholesterol; TG, triglycerides. *p < .05.
Univariate and Multivariate Analysis
Univariate logistic regression was performed to identify significant predictors of VTE, including the Caprini score, D-dimer levels, pulmonary infection, lower limb swelling, pulse rate, platelet count (PLT), prothrombin time (PT), and C-reactive protein (CRP). Significant variables from the univariate analysis (p < .05) were then included in a multivariate logistic regression model to assess their independent association with VTE.
In the univariate analysis, factors such as Caprini score (OR: 0.67, 95% CI: 0.53-0.81, p < .001), D-dimer (OR: 0.22, 95% CI: 0.14-0.29, p < .001), lower limb swelling (OR: 1.38, 95% CI: 0.94-1.82, p < .001), pulse rate (OR: 0.02, 95% CI: 0.00-0.03, p = .015), PLT (OR: −0.01, 95% CI: −0.01-0.00, p < .001), PT (OR: 0.16, 95% CI: 0.06-0.26, p = .002), and CRP (OR: 0.01, 95% CI: 0.00-0.01, p < .001) were all significantly associated with VTE risk.
In the multivariate analysis, Caprini score (OR: 0.58, 95% CI: 0.43-0.74, p < .001), D-dimer (OR: 0.16, 95% CI: 0.08-0.23, p < .001), PLT (OR: −0.00, 95% CI: −0.01-0.00, p = .017), and CRP (OR: 0.01, 95% CI: 0.00-0.01, p = .009) remained significant independent predictors of VTE. Other variables, such as pulmonary infection, lower limb swelling, pulse rate, and PT, did not retain statistical significance in the multivariate model. The results of both the univariate and multivariate analyses are summarized in Table 3.
Univariate and Multivariate Logistic Regression Analysis of VTE Risk Factors.

Abbreviations: PLT, platelet count; PT, prothrombin time; CRP, C-reactive protein; OR, odds ratio; CI, confidence interval. *p < .05.
ROC Curve Analysis
The predictive performance of several factors, including platelet count (PLT), C-reactive protein (CRP), the Caprini score, and D-dimer levels, was evaluated using receiver operating characteristic (ROC) curve analysis to determine their ability to predict VTE risk. The results demonstrated that the Caprini score and D-dimer levels were significantly better at predicting VTE risk compared to other factors. Specifically, the AUC for D-dimer was 0.81, and the AUC for the Caprini score was 0.84, both of which indicate good predictive accuracy.
In contrast, PLT and CRP had lower predictive capabilities, with AUC values of 0.33 and 0.62, respectively, indicating poor discrimination between VTE and non-VTE cases. These lower AUC values suggest that PLT and CRP are not reliable predictors of VTE when compared to the Caprini score and D-dimer. As a result, PLT and CRP were excluded from the combined prediction model.
The performance of the model was evaluated using receiver operating characteristic (ROC) curves, with the area under the curve (AUC) serving as the primary measure of predictive accuracy. The combined Caprini–D-dimer model demonstrated superior predictive accuracy, yielding an AUC of 0.89, compared to the Caprini score alone (AUC = 0.84) and D-dimer alone (AUC = 0.81).26,27 This indicates that the combined model offers enhanced predictive capacity for VTE risk in high-altitude hospitalized patients.
The final model, the predictive accuracy of the logistic regression model integrating the Caprini score and D-dimer levels was assessed through ROC curve analysis. The logistic regression model yielded the following equation: logit(p) = −4.563 + 0.629 × Caprini score + 2.182 × lgD2, which integrates the Caprini score and D-dimer levels, demonstrated superior predictive power for VTE risk, further supported by its AUC of 0.89. This combined model outperformed the individual factors, providing more accurate and reliable predictions of VTE risk in hospitalized patients. The comparative ROC curves are presented in Figure 2, showing the clear advantage of the Caprini score and D-dimer over PLT and CRP in terms of predictive accuracy.
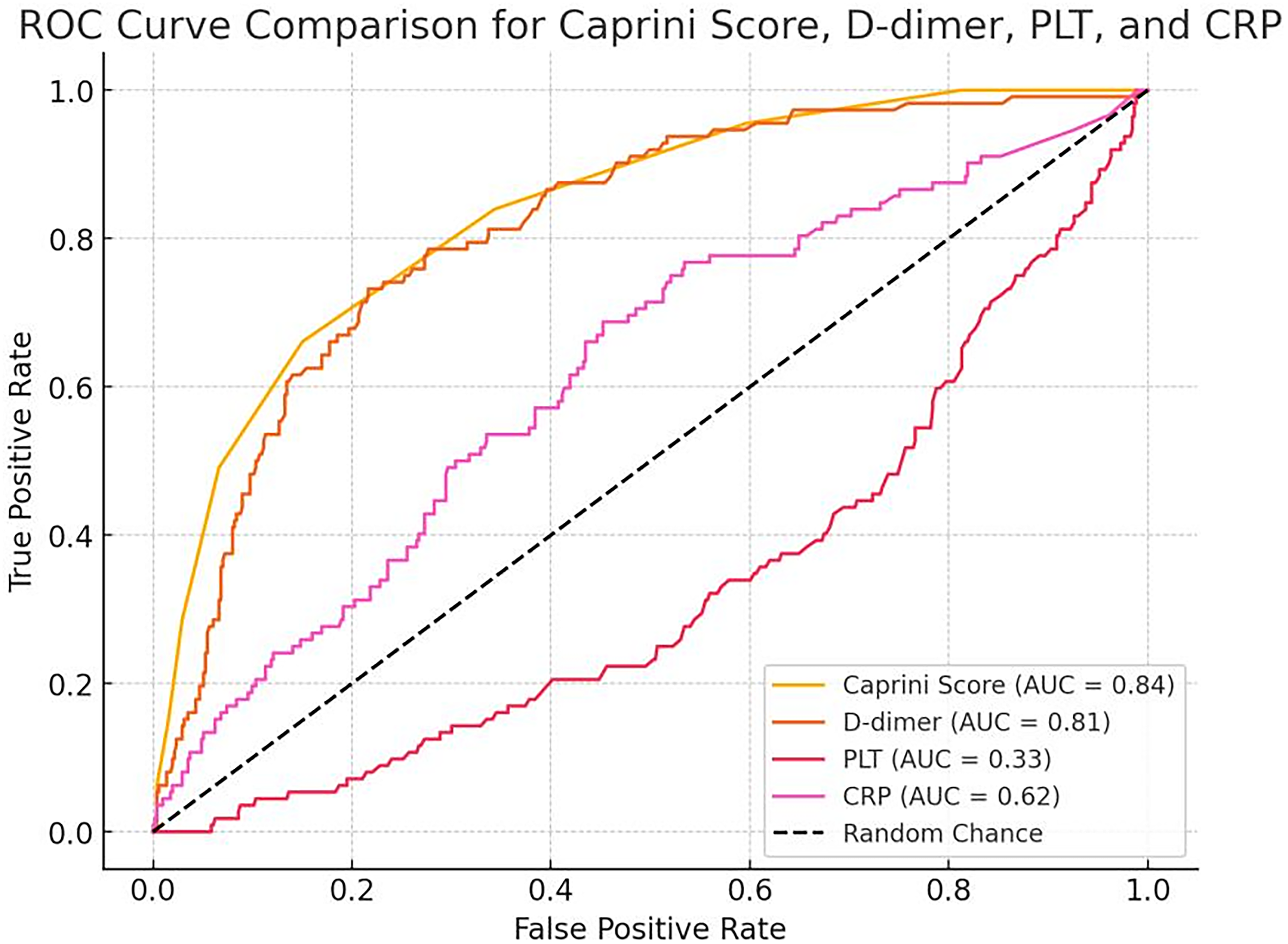
ROC Curve Comparison.
The ROC curves for the Caprini score, D-dimer, and the combined model are shown in Figure 3. The combined model demonstrated the highest AUC (0.89), indicating superior predictive performance compared to the Caprini score (AUC = 0.84) and D-dimer (AUC = 0.81).

ROC Curve Comparison for VTE Prediction Models.
In a prespecified sensitivity analysis excluding all patients who had received any form of thromboprophylaxis prior to VTE diagnosis in the primary cohort, the AUC of the combined model increased from 0.890 to 0.904, indicating slightly improved discriminative performance in this subgroup. The ROC curve for this sensitivity analysis is shown in Figure 4.
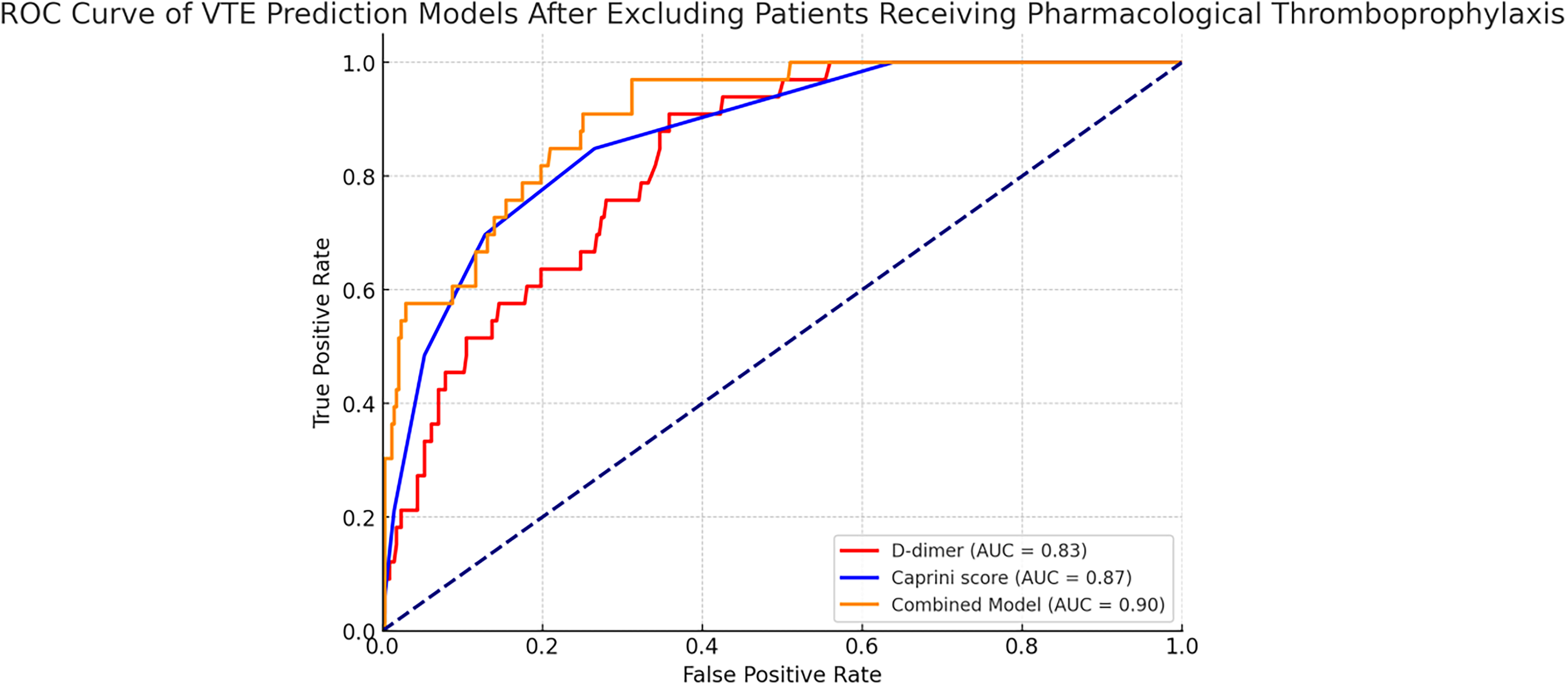
ROC Curve of VTE Prediction Models After Excluding Patients Receiving Pharmacological Thromboprophylaxis.
A total of 400 patients were included in the external validation cohort, recruited from Tibet Military Region General Hospital between July 2024 and March 2025. Among them, 54 patients (13.5%) developed VTE during hospitalization. ROC curve analysis demonstrated that the AUC for the combined Caprini–D-dimer model in the external validation cohort was 0.86, slightly lower than the 0.89 observed in the primary cohort. The AUC values for the Caprini score and D-dimer alone were 0.72 and 0.83, respectively, compared to 0.84 and 0.81 in the primary cohort. These results indicate that the combined model maintains strong and consistent predictive performance across different hospital settings, with only a modest decline in discrimination. The ROC curves for the validation cohort are presented in Figure 5.

ROC Curves for External Validation.
Discussion
Previous studies have demonstrated the widespread utility of the Caprini score in predicting venous thromboembolism (VTE) risk, particularly in surgical populations and hospitalized patients. 28 The Caprini score is known for its inclusion of a comprehensive range of risk factors, including patient age, previous VTE, immobilization, and comorbidities. However, while it has proven effective in many low-altitude settings, its applicability in high-altitude environments remains underexplored. 29 High-altitude conditions, characterized by chronic hypoxia and increased blood viscosity, introduce additional physiological stressors that may increase the risk of thromboembolic events. 30 These factors are not fully accounted for by the Caprini score alone.
Our study sought to address this gap by evaluating the predictive accuracy of a combined Caprini score and D-dimer-based model for VTE risk in hospitalized patients residing at high altitudes. We found that integrating D-dimer levels significantly improved the predictive power of the Caprini score, with the combined model achieving an AUC of 0.89. This is in contrast to the AUC of 0.84 for the Caprini score alone, indicating that the addition of D-dimer enhanced the model's sensitivity and specificity in this unique patient population. 31 To further validate the model, we conducted external validation at the Tibet Military Region General Hospital, located in the same high-altitude environment as the primary study site. The external validation results demonstrated a strong predictive performance of the combined model (AUC = 0.86), with only a slight reduction from the primary study (AUC = 0.89). The Caprini score alone showed a decrease in predictive performance (AUC = 0.72 vs 0.84 in the primary study), while the AUC for D-dimer alone slightly increased (0.83 vs 0.81). These findings suggest that variations in model performance are more likely due to differences in patient management practices and clinical decision-making rather than altitude-related factors. D-dimer, a well-established biomarker for fibrin degradation, is particularly useful for identifying thrombotic activity,21,32 and its inclusion in the model appears to compensate for the limitations of the Caprini score in accounting for high-altitude-induced hypercoagulability. 33
Unlike previous studies conducted in low-altitude settings, where the Caprini score has been validated without modification, 28 our findings highlight the importance of adjusting predictive models to account for environmental factors specific to high altitudes.29,30 The chronic hypoxic conditions experienced by patients in regions such as Tibet lead to increased blood viscosity and endothelial dysfunction, factors that significantly contribute to heightened VTE risk. 30 Our external validation results reinforce the applicability of the combined model across different hospitals in Lhasa, supporting its clinical relevance in this population. However, the slight variations observed in AUC values indicate that institutional differences, such as thromboprophylaxis strategies and diagnostic thresholds, may influence the predictive performance of the model. In our prespecified sensitivity analysis excluding all patients who had received any form of thromboprophylaxis prior to VTE diagnosis, the AUC of the combined model increased from 0.890 to 0.904 in the primary cohort, suggesting that prophylactic anticoagulation may modestly attenuate the model's apparent discriminative ability. This finding underscores the robustness of the combined model even in patients without prior preventive interventions, and highlights its potential utility as a pre-intervention risk stratification tool in high-altitude clinical settings.
However, despite the robust performance of the combined Caprini score and D-dimer model, there is still room for improvement, particularly in considering genetic predispositions to thrombosis. Variants in genes such as EPAS1 and EGLN1, which are known to play key roles in hypoxia adaptation, could have a significant impact on thrombotic risk, especially in high-altitude populations. These genetic factors have been shown to affect blood coagulation and thrombosis risk, and could provide an additional layer of stratification in predicting VTE risk in these patients. While our study did not include genetic data, future research could integrate genetic analysis to assess how specific genetic variations interact with the environmental stressors of high altitude, such as chronic hypoxia, to influence VTE outcomes. Investigating these genetic markers in future cohorts, particularly in Tibetan or other high-altitude populations, could further refine the predictive accuracy of the model.
Incorporating genetic factors into predictive models may significantly enhance their capacity to identify high-risk individuals, particularly those with genetic markers for thrombophilia. We suggest that future studies should explore genetic screening as part of the VTE risk assessment process in high-altitude regions, which could potentially provide a more comprehensive and personalized approach to thrombosis prevention.
The discrepancy between our results and those of studies conducted at lower altitudes can be attributed to these physiological differences, suggesting that the Caprini score alone may not be sufficient for accurate VTE risk assessment in high-altitude settings. Although the Caprini score is widely used in clinical practice, it has not been specifically validated for Tibetan populations or other high-altitude groups. The patients in our study were hospitalized in Tibet, a region where chronic hypoxia poses unique challenges for VTE risk prediction. 30 Therefore, while the Caprini score offers a solid foundation for risk assessment, our results indicate that further validation and adjustment of this model are necessary to ensure its accuracy for use in high-altitude populations. Importantly, future research should also investigate how different thromboprophylaxis strategies interact with predictive models, potentially refining risk estimation by accounting for the modifying effects of preventive interventions.
In conclusion, our study demonstrates that combining the Caprini score with D-dimer levels significantly improves VTE risk prediction for high-altitude hospitalized patients. 21 This enhanced model better accounts for the unique physiological stresses induced by high altitude, including hypoxia and increased blood viscosity.29,30 The model's stability across external validation and its maintained or improved discrimination after excluding patients receiving prophylactic anticoagulation further support its robustness.However, further studies are needed to validate this combined model in broader high-altitude populations and to explore additional environmental and genetic factors that may influence VTE risk in these regions. Tailoring VTE risk prediction models for high-altitude environments will allow for more precise risk stratification and improve clinical outcomes through earlier and more targeted prophylactic interventions.34,35
Conclusion
The combined Caprini score and D-dimer model demonstrated enhanced predictive accuracy for assessing venous thromboembolism (VTE) risk in high-altitude hospitalized patients compared to using either predictor alone. This model represents a significant improvement in risk stratification by integrating the Caprini score's comprehensive risk factors with D-dimer's sensitivity to thrombotic activity, thus addressing the unique physiological challenges imposed by high-altitude environments, such as hypoxia and increased blood viscosity. By enabling the earlier identification of high-risk patients, this model has the potential to guide more timely and effective preventive interventions, ultimately reducing the incidence of VTE and improving clinical outcomes in high-altitude settings. Furthermore, this study underscores the importance of modifying conventional VTE risk assessment models to better reflect the environmental and physiological stressors faced by patients at high altitudes. In addition to the demonstrated effectiveness of the combined model, external validation conducted at another high-altitude hospital confirmed its robustness and clinical applicability across different institutions. While the Caprini score alone showed some variability in predictive performance, D-dimer remained a strong independent predictor, suggesting that institutional differences in patient management and thromboprophylaxis strategies may influence model performance beyond altitude-related physiological effects. Importantly, in a prespecified sensitivity analysis excluding patients who had received any form of thromboprophylaxis prior to VTE diagnosis, the AUC of the combined model increased from 0.890 to 0.904, further demonstrating its robustness and potential utility for risk assessment in patients without prior preventive interventions. The study highlights the broader need for tailored VTE risk prediction models in high-altitude environments. Standard models such as the Caprini score may underestimate the risk of VTE in these settings due to the unique complications of altitude, and thus, clinicians working in high-altitude hospitals are encouraged to incorporate the combined model into their practice to ensure more accurate risk assessments. Moving forward, further research should focus on validating this model across larger and more diverse high-altitude populations to ensure its generalizability and effectiveness. Moreover, additional studies should explore the inclusion of other biomarkers and clinical factors that could further enhance the model's predictive capabilities. Ultimately, refining risk prediction models for high-altitude environments will enable clinicians to implement more personalized and effective preventive strategies, reducing the burden of VTE in these populations and improving patient care in challenging clinical settings.
Footnotes
Ethics Approval
Ethical approval to report this case series was obtained from Lhasa People's Hospital Ethics Committee (Approval Number: SYLL2224079) and Beijing Chao-Yang Hospital Ethics Committee (Approval Number: 2024-ke-808).
Informed Consent
Informed consent for patient information to be published in this article was not obtained because the retrospective nature of this study using anonymized clinical data met criteria for waiver of informed consent under institutional ethical guidelines.
Author Contribution Statement
Xue Shi: Study design, data collection, data analysis, manuscript writing. Li Yang: Data collection, data analysis, literature review, manuscript writing. Na Cui: Data collection, statistical analysis, manuscript editing. Yuntao Zhang: Study design, manuscript review, funding support. Suqiao Yang: Study design, data analysis, manuscript writing, funding support.
Funding
S. Yang is supported by the National Natural Science Foundation of China (62476179, 81900047), Noncommunicable Chronic Diseases-National Science and Technology Major Project (2025ZD0551906), Key Project of Natural Science Foundation of Tibet Autonomous Region (XZ202301ZR0052G), and the National Research Program for Prevention and Management of Pulmonary Embolism and Deep Venous Thrombosis (Y096). X. Shi is supported by the Graduate High-Level Talent Training Program of Xizang University, “Construction and Validation of a Risk Prediction Model for Venous Thromboembolism in Inpatients in Plateau Internal Medicine” (Project No. 2022-GSP-S118).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
