Abstract
Objective
To derive and validate a D-dimer cutoff for ruling out pulmonary embolism (PE) in COVID-19 patients presenting to the emergency department (ED).
Methods
A retrospective cohort study was performed in an integrated healthcare system including 22 adult ED's between March 1, 2020, and January 31, 2021. Results were validated among patients enrolled in the RECOVER Registry, representing data from 154 ED's from 26 US states. Consecutive ED patients with laboratory confirmed COVID-19, a D-dimer performed within 48 h of ED arrival, and with objectively confirmed PE were compared to those without PE. After identifying a D-dimer threshold at which the 95% confidence lower bound of the negative predictive value for PE was higher than 98% in the derivation cohort, it was validated using RECOVER registry data.
Results
Among 3978 patients with a D-dimer result, 3583 with confirmed COVID-19 infection were included in the derivation cohort. Overall, PE incidence was 4.1% and a D-dimer cutoff of
Conclusion
A D-dimer cutoff of
Keywords
Introduction
The SARS-CoV-2 virus discovered in Wuhan, China in December 2019 has since spread across the globe, infecting more than 146 million people and leading to more than 3.8 million COVID-19 deaths worldwide.1,2 Severe COVID-19 has been associated with an increased risk of venous thromboembolism (VTE),3–6 but the symptoms of pulmonary embolism superimposed on this infection may be difficult to distinguish from the symptoms of the primary viral pneumonia. Pulmonary embolism (PE) is therefore a common concern for patients presenting to the emergency department (ED) with COVID-19.
D-dimer is among the inflammatory markers that are elevated in the setting of COVID-19 infection,7–9 and is associated with an increased risk for venous thromboembolic (VTE) disease.10–18 Unfortunately, increases in the D-dimer associated with the infection itself impede the usual application of this sensitive VTE marker as part of pre-test probability pathways designed to rule out VTE and avoid unnecessary radiation, intravenous contrast exposure, and costs associated with diagnostic imaging.3,19–30 Since application of the conventional D-dimer thresholds produces a high false positive rate, we hypothesized that an alternative D-dimer threshold could be derived and validated to safely refute PE among ED patients with acute COVID-19 infection with negative predictive value similar to threshold values widely accepted for application in non-COVID-19 patients.
Methods
We performed a derivation and validation study to report the operating characteristics of the serum d-dimer for patients found to have radiographically confirmed pulmonary embolism. D-dimer values are reported as fibrinogen equivalent units in both the derivation and validation centers. The Stago STA-LIATEST(R) D-DI assay was used for all tests in the derivation cohort. The specific D-dimer assay is unknown for each of the centers in the validation cohort. All D-dimer units were converted to μ/ml for ease of reporting. Retrospective analysis of data from the Intermountain Healthcare Prospective Observational COVID-19 (IPOC) Registry was determined to be exempt from review by the Intermountain Healthcare institutional review board. Use of data from the registry of suspected COVID-19 in emergency care (RECOVER) network collaborative registry was approved by individual site IRBs, without requirement of individual patient consent. 19
Derivation Cohort
Patients presenting to one of 22 hospital emergency departments within an integrated healthcare system in Utah and Idaho between March 23, 2020, and February 31, 2021 were eligible for inclusion in the derivation cohort if they had a positive PCR or antigen test for COVID-19 during or within the 14 days preceding ED visit and if a serum D-dimer value was measured within 48 h of ED arrival. PE was identified by using the natural language processing methodology that we previously described applied to chest CT, pulmonary perfusion, or pulmonary ventilation/perfusion scans that were conducted within 48 h of ED arrival. 20 We excluded 28 patients with DVT and an absence of PE from analysis. Patient demographics and clinical characteristics were extracted from the electronic data warehouse using a validated data set that had been adjudicated through manual review by one of the study's investigators (IDP).
Validation Cohort
The RECOVER Network collaborative registry database served as the validation cohort for this study. RECOVER enrolled patients presenting to one of the 154 community and academic centers and undergoing testing for SARS-CoV-2 during the ED encounter or within the preceding 14 days for suspected symptomatic COVID-19. 31 Patients who had a positive COVID-19 test and had a D-dimer value recorded during the index ED encounter were eligible for inclusion in the validation cohort. Patients enrolled a derivation cohort clinical site were censored from the validation cohort prior to analysis. Patients with DVT in the absence of PE were excluded from analysis. Presence of PE within 30 days was ascertained through a combination of electronic health record query and manual chart review per previously reported methods. 19
Exposure and Outcome
The primary exposure was the first-available D-dimer within 48 h of ED arrival. The primary outcome was the occurrence of pulmonary embolism identified per clinical routine on contrast-enhanced chest CT or either pulmonary ventilation/perfusion or pulmonary perfusion nuclear medicine testing within 48 h of ED arrival for the derivation cohort and within 30 days for the validation cohort. The decision to obtain imaging was at the discretion of the treating ED clinician. Patients in whom DVT was diagnosed in the absence of PE were excluded from the analysis.
Statistical Analysis
The predictive performance (sensitivity, specificity, negative predictive value [NPV] and positive predictive value [PPV]) for PE across a range of D-dimer values was calculated in the derivation cohort. The study design defined the derivation cohort D-dimer threshold result that would yield in a negative predictive value for PE greater than 98% with 95% confidence interval that also had a lower bound greater than 98%. These criteria aimed to minimize the false positive rate while providing a negative predictive value of at least 98% (ie, a false negative rate of less than 2%), equivalent to negative predictive values for D-dimer thresholds in widely used PE pretest probability algorithms.25–27,29 For each D-dimer, the 95% equal tail confidence intervals for test performance characteristic were computed using a Bayesian posterior based on the binomial likelihood and a uniform prior distribution. The performance of the candidate D-dimer threshold for ruling out PE identified in the derivation cohort was then tested in the validation cohort. Further logistic regression models were used to explore a possible relationship with age to find an age adjusted cut-off value in COVID-19 patients. Analysis was performed using R Core Team 2020 (Vienna, Austria). 21
Results
In the derivation cohort among the 3978 patients with a D-dimer, 3583 had laboratory confirmed COVID-19 and a D-dimer that was performed within 48 h of the index ED encounter. Patients with PE were older (65.0 vs 57.1), more likely to be male (53.4% vs 48.0%) and had a prior history of VTE (37.8% vs 7.9%) (Table 1). Among patients with COVID-19, 53.6% (1920/3583) were admitted to the hospital on index encounter. CTPA was performed in 3583 patients and PE was present in 148, for an overall incidence of 4.1%. The median D-dimer among COVID-19 positive patients with PE was 3.74 μ/ml (IQR 1.71−9.62), compared to 0.73 μ/ml (IQR 0.39−1.48) in those without PE present (Table 1.).
Patient Characteristics Derivation and Validation Cohorts.

(--) indicades data not available, Obesity definition: Overweight: Body Mass Index (BMI) 25-29.9, Obese: BMI 30-34.99, Morbidly obese: BMI >34.99,
A D-dimer cutoff of

Distribution of D-dimer values stratified by age in the derivation cohort. Blue dot = PE absent, Red dot = PE present, D-dimer reported as μ/ml.

D-dimer value stratified by presence or absence of PE in the derivation cohort with COVID-19. Green bars represent D-dimer thresholds of 0.5, 1, and 2mcg/ml from left to right on a logarithmic scale.
D-Dimer Test Characteristics in Derivation Cohort Patients with COVID-19.
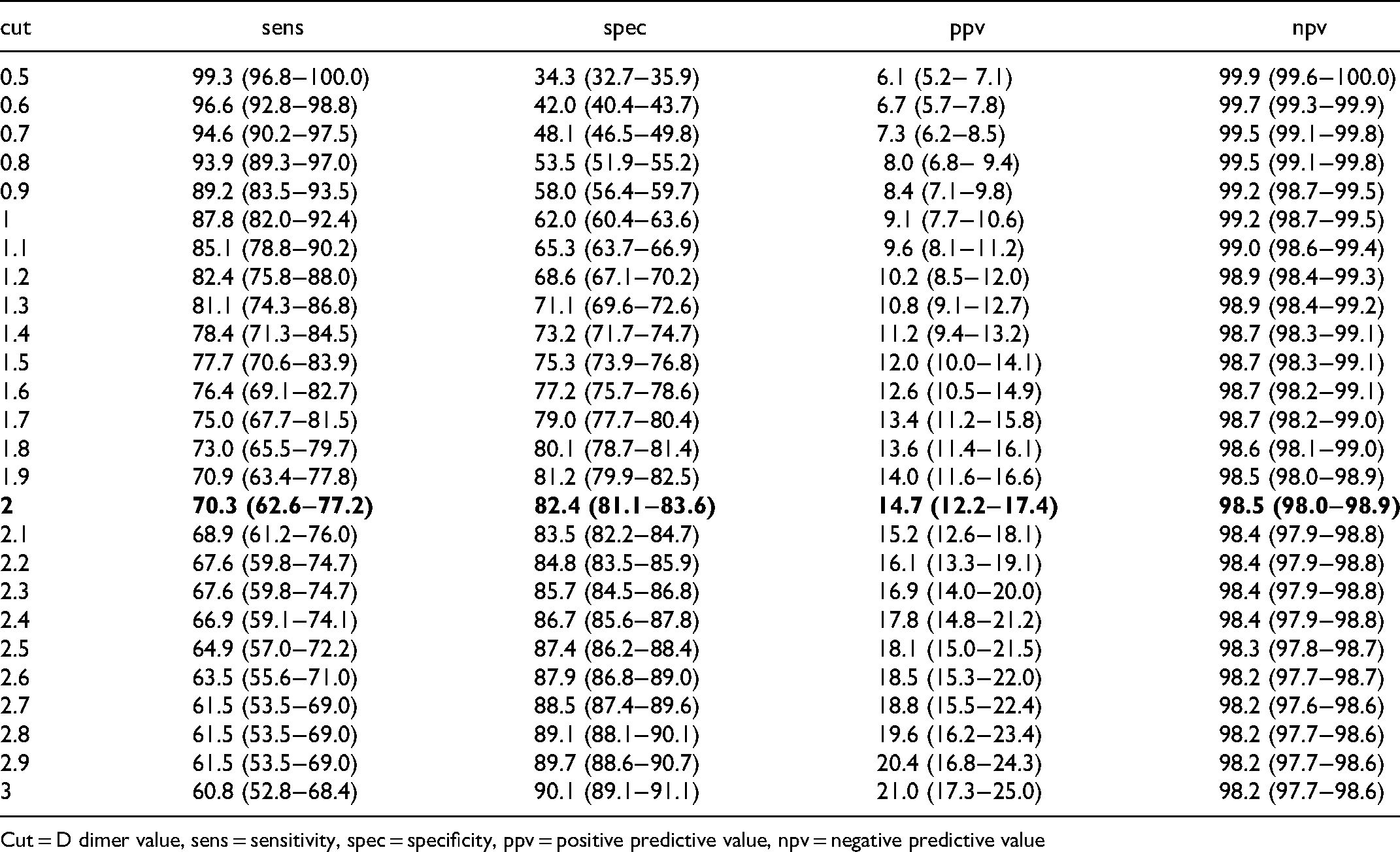
Cut = D dimer value, sens = sensitivity, spec = specificity, ppv = positive predictive value, npv = negative predictive value
Patient characteristics in the validation cohort were similar to the derivation cohort. (Table 1). In the validation cohort of 13,091 with D-dimer values available 7748 patients had confirmed COVID-19 infection, of those 85.7% (6640/7748) were admitted to the hospital on index encounter, and PE was diagnosed in 88 of the 7748 that yielded an incidence of 1.14%. For the outcome of PE the D-dimer cutoff of
COVID-19 Adjusted D-Dimer Threshold Test Characteristics.

Discussion
Among ED patients with COVID-19, we determined that a D-dimer threshold of 2 μ/ml provided a high NPV
22
while minimizing the false positive rate. This value is 4 times higher than the traditional cutoff of <0.5 μ/ml, the upper limit of normal for this assay, commonly used to justify avoidance of diagnostic imaging in patients without COVID-19. However, the sensitivity for PE associated with the D-dimer threshold of
VTE is a common concern among patients presenting to the ED with hypoxia or dyspnea. This is especially true for patients with COVID-19, who often present with these symptoms due to the viral pneumonia but for whom clinicians also recognize an increased risk of VTE. Unfortunately, D-dimer elevations observed with this disease have rendered uncertain the role for D-dimer and pretest probability-based care pathways in COVID-19 patients that were formerly proven to safely refute suspected PE and reduce the use CT pulmonary angiogram in general ED populations. Our interest was in assessing for an alternative COVID-adjusted D-dimer threshold could account for the hyperinflammatory state attributed to COVID-19 to explore the hypothesis that a higher than usual D-dimer threshold might be proposed as safe to refute PE among COVID-19 patients thereby avoiding the cost and risk associated with unnecessary CTPA for a select group of these patients. It has been suggested that clinical pretest probability may be effective in safely electing variable D-dimer thresholds for refuting PE among non-COVID-19 patients. However we did not collect pretest probability as part of this study, 24 and information surrounding pretest probability and VTE is limited. 25
Our observations were consistent with former publications reporting that D-dimer elevations above traditional thresholds occur in COVID-19 patients.6,18 In our study cohort, D-dimer values above cutoffs conventionally used to guide PE diagnostic evaluation were common and occurred in 66.8% of the derivation cohort and in 82.7% of the validation cohort. Elevated D-dimer among patients with PE seen in our study were similar to previous studies reporting among patients with COVID-19 infection.14,18,26–28 Diaz et al reported a sensitivity of 81% using a cutoff of 2903 ng/ml (2.9 μ/ml), in patients hospitalized with COVID-19, however further report that the sensitivity can be improved to 91-94% when using a lower D-dimer cutoff of 1221−1721ng/mL in their population. Choi found that progressively increasing D-dimer thresholds increased the likelihood of PE being present among hospitalized adults, yet cautioned that a post-test probability of PE of 3% existed among patients with a D-dimer less than 1000 ng/mL. 29 Recently however D-dimer assays have been shown to yield a high sensitivity of D-dimer among COVID-19 patients suspected of PE, 30 and higher thresholds for positivity have been suggested. 32 However concern exists surrounding adjusting D-dimer among COVID-19 patients suspected of PE with sensitivity being lost. 31 Our study, is to our knowledge the first to focus on D-dimer thresholds among the population of emergency department patients in the US with suspected PE in the setting of COVID-19 infection. The ED is an environment where evaluation of patients in various stages of COVID-19 infection from very mild to severe disease occurs and represents a setting where diagnostic tests for suspected PE are most commonly performed in the US.
Strengths and Limitations
Strengths of the present study include derivation of the D-dimer threshold in a large multi-hospital integrated healthcare system and validation in a nationally-representive and generalizable COVID-19 cohort. Our selecting RECOVER as the validation cohort permits simulation of the impact of applying the suggested threshold in real-world settings. Limitations of our study include the retrospective data ascertainment. Also, pre-test probability assessment was not available for these patients. Therefore, we are unable to assess the effect of pretest probability on our results, and with rare exception33,34 consensus exists that pre-test probability assessment is necessary for VTE risk assessment. We were unable to assess for missed diagnoses of PE at 90 days. The PE diagnosis in the validation cohort was reported up to 30 days following the ED encounter in which the D-dimer was obtained. This may explain the missed PE rate that was higher than what we anticipated for both the standard cutoff and and COVID adjusted cutoff D-dimer values (Table 3). In the setting of COVID, longer term follow-up for venous thromboembolic events may have captured PE not present on the initial ED encounter. We also assumed that D-dimer orders indicated evaluation for suspected PE, but some laboratory testing may have been obtained for COVID-19 prognostication or evaluation of other suspected processes (eg, disseminated intravascular coagulation). The fact that 96% of patients in the derivation cohort and 100% in the validation cohort received a CTPA following a D-dimer test supports our hypothesis that any inclusion of patients whose D-dimer was obtained solely for COVID-19 prognosis would not significantly alter the results of the study. However, this is one potential reason that the incidence of PE was lower among patients in the validation cohort than the derivation cohort, and also lower than the PE incidence in reports of unselected ED patients with COVID-19.28,35 Variability in PE outcome ascertainment methods across the RECOVER Network may also have led to incomplete PE detection at some study sites. The fact that diagnostic testing for PE was obtained at clinicians’ discretion rather than systematically may mean some PE events went undetected in both cohorts.
The low incidence of PE in both cohorts, but in particular the validation cohort, contributes to the high negative predictive value for the D-dimer threshold that we report but with a comparatively low D-dimer sensitivity (Table 3). This finding is likely multi-factorial, but could include false positive CTPA results given that performing CTPA on low-risk patients increases the likelihood of a false-positive CTPA up to 67%. 36 In addition the prevalence of PE may be misrepresented due to our cohort selection excluding patients with PE diagnosed without the presence of a D-dimer test. Our results highlight the clinical limitations of interpreting D-dimer among COVID-19 patients and our findings are hypothesis-generating only. Prospective evaluation with protocol mandated D-dimer assessment is needed before alternative D-dimer thresholds may be proposed that could be used clinically to safely refute PE among COVID-19 patients. Future studies should also include standardized PE pretest probability assessment including whether either objective or gestalt pretest probability adjustment or additional biometric data might be useful in reducing the use of CTPA without increasing the rate of missed PE among COVID-19 patients.
Conclusion
A COVID-19 adjusted D-dimer cutoff of
Footnotes
Author Contributions
Dr Bledsoe takes full responsibility for the integrity of the manuscript in its entirety. Concept and design: Bledsoe, Woller, Knox, Peltan. Analysis, Acquisition or interpretation of data: Bledsoe, Woller, Knox, Peltan, Lloyd, Horne, Snow, Connors, Kline. Drafting of the manuscript: Bledsoe, Woller, Critical revision of the manuscript: Bledsoe, Woller, Knox, Peltan, Lloyd, Horne, Snow, Connors, Kline.
Financial Disclosure
See COI forms
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
