Abstract
Background
Venous thromboembolism (VTE) is frequent in patients in the intensive care unit (ICU). In patients in whom pharmacologic prophylaxis is not applicable, mechanical prophylaxis is indicated, currently with use of pneumatic compressors. In this scenario, PEMF may present as a safe and efficient option.The primary objective of the study is to compare Pulsed Electromagnetic Field Therapy (PEMF) to conventional pharmacological and/or mechanical prophylaxis for the prevention of VTE in patients in the ICU.
Study design
Fifty patients requiring mechanical prophylaxis will be randomized to conventional mechanical VTE prophylaxis or PEMF with a device called Hyperslim®. The primary outcomes are safety, tolerability, and device usability, using the SUS scale, a10-item questionnaire used to measure the perceived usability of a system or product. The exploratory efficacy outcome is a composite of symptomatic and asymptomatic VTE (detected by mandatory Doppler ultrasound 24-48 h after the end of mechanical prophylaxis or PEMF and on day 30 post-randomization) and death due to pulmonary embolism.
Discussion
We expect this study will provide valuable preliminary information on the safety, tolerability, device usability, and efficacy of this new method of mechanical prophylaxis for patients in the ICU who require mechanical VTE prophylaxis.
Keywords
Introduction
Venous thromboembolism (VTE) is the leading cause of preventable death in hospitalized patients in the intensive care unit (ICU). VTE risk should be evaluated, and prophylactic methods should be started as early as possible.1,2 A delay of more than 24 h to start VTE prophylaxis increases mortality in ICU patients3,4 Padua score for medically ill patients and Caprini score for surgical patients are the most used risk assessment models (RAMs) in ICU, and there are prophylactic recommendations from many societies (ACCP, ASH, ESA, and SCCM) for pharmacological and/or mechanical prophylaxis for critical care patients5–7
Pharmacological prophylaxis is indicated in most patients in the ICU, but some patients are inelegible for pharmacological prophylaxis, or require combined mechanical and pharmacological prophylaxis. Patients with active bleeding, hemorrhagic stroke, hematological disorders, and postoperative patients with increased risk of bleeding or VTE are some examples of contraindications for pharmacological prophylaxis and/or indications for combined prophylaxis.2,5
Intermittent compression (IC) is the most used mechanical prophylaxis for patients in the ICU. Mechanical prophylaxis is indicated in 1%-3% of patients, but its efficacy is questionable; its’ use and monitoring are complex and increase costs. Despite the virtually ubiquitous recommendation for mechanical prophylaxis for patients unsuitable for pharmacological prophylaxis, high-quality evidence of its efficacy in ICU patients remains lacking.2,5,7
IC is the most commonly used mechanical prophylaxis. However, its use in the ICU is not free from pitfalls: the necessity of a trained team to operate and monitor the device, patient immobility during use, patient discomfort, and associated costs. 8 Furthermore, continuous use necessitates the allocation of one device per patient and derequires a dedicated team work. Bed restriction is widespread in the ICU; it is a leading risk factor for VTE, and pneumatic compression restricts patients even more in bed. Although modern ICU protocols involve mobilizing patients,; in most cases of mechanical prophylaxis the patients are required to be bedridden for most of the day, and continuous pneumatic compression could complicate this issue even further.9,10
Pulsed Electromagnetic Field Therapy (PEMF) has been demonstrated an efficient tool for muscle recovery in orthopedic patients. Muscle contraction induced by this technology could increase venous blood flow velocity, decrease hypercoagulability and venous stasis, and therefore it presents as a reasonable candidate for reduction of VTE risk. The device is user-friendly and suitable for multiple patients use in the ICU, and its intermittent operation facilitates patient care and mobility.11,12
This study aims to evaluate the safety, usability, and the exploratory efficacy of PEMF compared to conventional intermittent compression in patients in the ICU unsuited for pharmacological prophylaxis or who require combined mechanical and pharmacological prophylaxis.
Methods
Study Design
In this prospective, randomized, open-label, active-controlled study, patients requiring isolated mechanical VTE prophylaxis or combined mechanical and pharmacological prophylaxis in the ICU from two selected sites in Brazil (Vera Cruz Hospital and Felicio Rocho Hospital, both in Belo Horizonte-MG, Brazil) will be enrolled after signing informed consent. If patients are not conscientious, the signing of consent by the patient´s legal representative is necessary. All data will be recorded with a pre-established clinical recording file (CRF - RedCap®) including demographics, risk factors, and clinical aspects of the patient's comorbidities. Previous treatment with anticoagulants will be allowed (Figure 1).
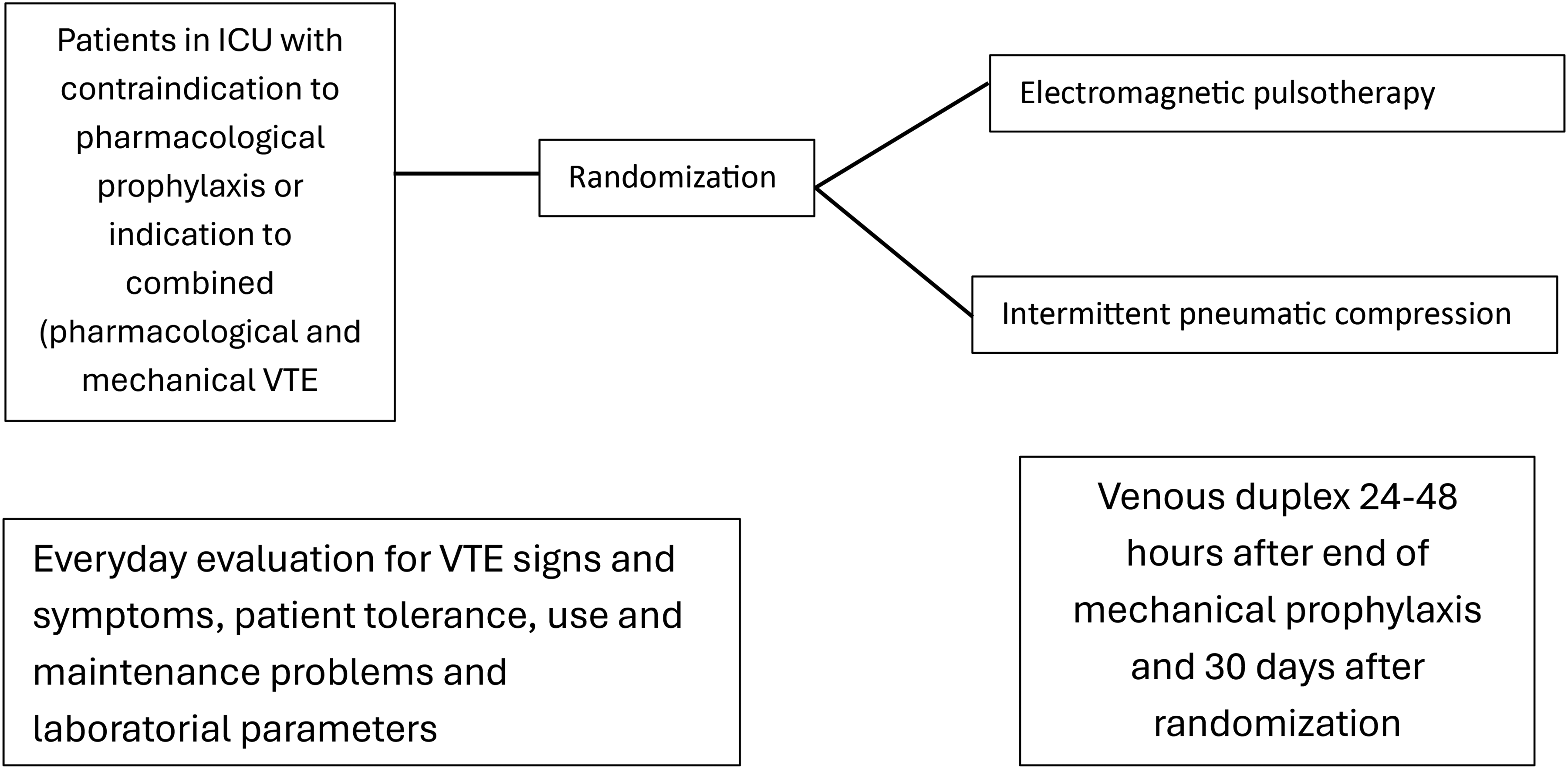
Study Design.
Clinical diagnosis will be performed by a trained team member, with standard clinical evaluations, including imaging when indicated. Patients will be considered eligible if they are >18 years old, have a PADUA score ≥of 4 (medical patients) and/or Caprini ≥3 (surgical patients), and are not suitable for pharmacological prophylaxis or require combined (pharmacological and mechanical VTE prophylaxis). The detailed inclusion and exclusion criteria are listed in Table 1.
Inclusion and Exclusion Criteria.

Ethical Approval and Trial Registration
This study was conducted in compliance with the principles of the Declaration of Helsinki and applicable national regulations. The research protocol was reviewed and approved by the Research Ethics Committee of HAPVIDA Medical Assistance LTDA (CAAE: 86295325.3.1001.0229).
The trial is registered at ClinicalTrials.gov under the identifier NCT06958588.
Randomization and Interventions
After ICU admission, the patient will be interviewed by study investigators or trained physicians or nurses to participate and, after agree, signing an informed consent form, the patients will be screened and included in the study. If patients are not conscientious, the signing of consent by the patient´s legal representative is necessary. Inclusion patients will be randomized to receive either PEMF or intermittent pneumatic compression.
The randomization sequence is generated with a 1:1 allocation using the secure program REDCap® platform (Research Electronic Data Capture, RedCap Consortium, Vanderbilt University Medical Center, Tennessee, USA).
The devices will be placed in the ICUs in accordance with institutional standards and operational procedures. After randomization, trained physiotherapy and nurse teams will place and monitor each device.
For the intermittent pneumatic compression group, the Covidien Kendall SCD 700® (Cardinal Health) or Doctor Life 2600® (Venosan) devices will be used. The devices will be positioned in the lower limbs, following manufacturer's instructions, and will be monitored for 24 h, in accordance with previous institutional protocol. These devices will be kept operational full-time for patients while they are in the ICU.
For the PEMF group, the Hyperslim® device (Figure 2) will be used. The Hyperslim® device is produced by Medical San (Lageado, RS, Brazil). This device is approved by ANVISA, the local regulatory agency of Brazil, for use in orthopedic patients for muscle strengthening.

Hyperslim® Device and its Suggested Use.
Hyperslim® generates electromagnetic pulses through its pads, leading to local muscular supra-maximal contractions sustained for several seconds, increasing stress and workload without muscular adaptation.
The pulsed electromagnetic therapy works using alternate electromagnetic fields, which produce electric currents that depolarize muscle tissue, causing sustained muscle contractions. These muscle contractions provoke an increase in the flow of major veins and small veins, decreasing stasis. This process could also present anti-inflammatory properties. This is the same principle of intermittent pneumatic compression in venous thromboembolism prophylaxis.11–13
The device pads will be placed on the patient's lower limbs for 30 min, three times a day. The critical care team will monitor patients 24 h/day for signs and symptoms of VTE, laboratory changes, and adverse local effects caused by the devices, such as skin lesions, burns, and edema. A tolerability scale will be applied to every awake patient able to answer using a scale: 1 = well tolerated; 2 = tolerated with little discomfort; 3 = tolerated with discomfort; 4 = not tolerated.
Aim and Perspective of the Study
The study aims to compare the safety, ease of use, and exploratory efficacy of PEMF with conventional mechanical prophylaxis in patients admitted to the ICU unsuited for pharmacological prophylaxis or requiring combined mechanical and pharmacological prophylaxis. The perspective is to facilitate ICU teamwork, enhance patient comfort, and reduce costs.
This exploratory use of PEMF is off-label. The manufacturer does not currently indicate the use of PEMF for VTE prophylaxis.
Primary Objective
To explore the safety and efficacy of a new electromagnetic pulse device as VTE prophylaxis in the ICU.
Primary Outcome
The primary outcome is safety, tolerability, and ease of use of the device in the ICU.
Exploratory Efficacy Outcome
The exploratory efficacy outcome is a composite of symptomatic and asymptomatic VTE (detected by mandatory Doppler ultrasound carried out after the end of mechanical prophylaxis or PEMF (that is carried out during ICU stay) and on day 30 post-randomization and death due to pulmonary embolism.
Hypotheses
Muscle contraction induced by PEMF applied to lower limbs could increase venous blood flow, decrease stasis and hypercoagulability, and reduce the risk of thrombotic events.
PEMF is easier to use in the ICU environment
The PEMF group does not demonstrate increased adverse events like skin lesions, muscle pain, injuries, and patient discomfort.
The PEMF group could have a similar incidence of VTE events to the conventional mechanical compression group, as proved by a venous duplex scan done 24-48 h after the end of mechanical prophylaxis in every study patient.
Data Collection
Clinical Data
The following information is recorded from electronic patient records and laboratory databases at study entry: age, sex, admission diagnosis, Padua or Caprini score, latest blood electrolyte status, kidney function, coagulation parameters, CK level, and hemogram.
Device Usability
This evaluation will be conducted by measuring perceived ease of use using the System Usability Scale (SUS), a validated questionnaire. It conducts usability testing, gathering both qualitative (observations, open-ended feedback) and quantitative data (task completion times) on how easily users interact with the device, and analyzes the data against human factors engineering standards (IEC 62366-1) to ensure safety and effectiveness. The System Usability Scale (SUS) is a standardized, 10-item questionnaire used to measure the perceived usability of a system or product, yielding a single score between 0 and 100. Developed by John Brooke in 1986, it utilizes a Likert scale to measure user satisfaction, ease of use, and complexity, offering a quick and cost-effective method for assessing user experience and comparing results against industry benchmarks. The scale alternates between positive and negative statements to prevent response bias, resulting in a quantitative metric that helps designers and developers improve products. 14
At the end of mechanical prophylaxis, patients will be evaluated for thromboembolic events, including diagnosis by imaging, with a mandatory Doppler ultrasound of the lower limbs and a new Doppler ultrasound 30 days post-randomization. Daily evaluation includes major bleeding events, defined as leading to transfusion of 2 or more units of blood or packed red cells, a decrease in hemoglobin of 2 g/dL, bleeding in critical sites, defined as spinal, epidural, intraocular, intracranial, pericardial, or retroperitoneal, local adverse events, including adverse events related to the use of the devices.
Duplex Ultrasound Examinations
A radiologist will perform examinations 24-48 h after the end of mechanical prophylaxis (after ICU stay) and on day 30 after randomization, or immediately if there are signs and symptoms of VTE. The scan is performed on the patient's lower extremities. It includes the femoral common popliteal veins, distal veins, lesser and great saphenous veins, superficial and muscular veins, and symptomatic sites. If thrombosis is diagnosed, the local guidelines recommended management (generally full anticoagulation) will be implemented.
Imaging Studies
Imaging studies will be performed by a radiologist if there is clinical suspicion of pulmonary embolism. Pulmonary computed tomography angiography will be the gold standard, but pulmonary scintigraphy will be used in patients with renal failure (creatinine >2,0 mg%). If pulmonary embolism is diagnosed, the guidelines recommended for management will be followed.
Blinding
The patients and the responsible healthcare staff are not blinded to the intervention. Radiologists performing Doppler ultrasound will be blinded to randomization.
Blood Sampling and Analysis
Samples are drawn from an arterial line if present. Otherwise, they are drawn from a peripheral vein, applying minimal stasis. Samples are analyzed immediately following institutional protocols.
Sample Size
This is a proof-of-concept exploratory study; accordingly, a convenience sample of 50 patients (25 per group) was selected, and no formal sample size calculation was performed.
Statistical Analysis
Given the exploratory nature of this trial and the small sample size, no assumptions of normality will be made.
Continuous variables will be summarised using medians and interquartile ranges, and between-group comparisons will be performed with non-parametric tests (Mann–Whitney U test for independent samples or Wilcoxon signed-rank test for paired data, as applicable).
Categorical variables will be expressed as counts and percentages, and differences between groups will be evaluated using Fisher's exact test or the chi-square test, depending on cell frequencies.
Time-to-event outcomes will be presented descriptively with Kaplan–Meier curves, and comparisons will be made using the log-rank test. All analyses will follow the intention-to-treat principle.
Given the limited sample size, results will be considered hypothesis-generating, and effect sizes with corresponding 95% confidence intervals will be emphasised over p-values.”
Primary Outcomes
Tolerability, safety, and ease of use of PEMF compared to intermittent pneumatic compression using an unpaired t-test.
Exploratory Efficacy Outcomes
Differences in VTE events between the two groups at the end of mechanical prophylaxis and at day 30 will be assessed according to the intention-to-treat principle. Given the limited sample size and exploratory nature of the study, analyses will be primarily descriptive. The cumulative incidence of composite events will be reported, and relative risks with 95% confidence intervals will be estimated. These analyses will be considered hypothesis-generating, and no formal hypothesis testing with fixed error rates will be undertaken
Adverse Events
Skin lesions, burns, muscle pain, and muscle injury will be considered adverse events, and patients will be evaluated and managed. If necessary, the patient will be removed from the study.
Serious Adverse Events
A serious adverse event or reaction (SAE) is any ICU medical occurrence that results in death, is life-threatening, requires prolongation of existing hospitalization, or results in persistent or significant disability. All SAEs are documented on the SAE report form during the treatment period. These assessments are used to monitor SAE survival and the safety of the experimental treatment. Patients who withdraw from the study due to an adverse event or SAE are followed up to 30 days after the event.
Termination of Trial
If there is an excessive frequency (> 20% in the intervention group) of SAEs and VTE it may be necessary to terminate the study. If a VTE is confirmed, the study will terminate for the individual participant.
All participants may leave the study at any time with no warning and no reason given. Furthermore, the participant will leave the study if one of the following occurs:
Adverse events are making it impossible to use the machines Intolerance (scale 4).
Stopping Rules (Device-Related)
Patients will undergo clinical evaluations multiple times throughout the day. Adverse local effects caused by device use, like skin lesions, burns, edema, muscular enzymes elevation more than three times, and intolerance by the patient, will be evaluated by the ICU team. The protocol will be stopped, and necessary management will be started.
The SOPHIE trial comes from the acronym “Pul
Discussion
The present study aims to investigate the possibility of use of PEMF as an efficient and safe device for VTE prophylaxis in critical patients as compared to IC. Although the role of mechanical prophylaxis in ICU patients remains debated, current clinical guidelines consistently recommend its use in individuals with contraindications to pharmacological venous thromboembolism prophylaxis. Moreover, these guidelines favour mechanical prophylaxis over the absence of any prophylactic intervention. Intermittent pneumatic compression is the most commonly used mechanical compression to date. Nevertheless, its use is not devoid of limitations in the ICU: the need for a trained full-time team, the necessitity of patient-dedicated devices, difficulties in placement and maintenance, and patients’ discomfort and immobility.4,9
Caprini and collegaues report frequent misplacement of intermittent pneumatic compression devices, leading to increased risk for patients, even when placed by a trained team. 15
PEMF provokes muscle contraction and increases venous blood flow in lower limbs decreasing hypercoagulability and venous stasis, and possibly reducing the VTE risk. Placing PEMF in ICU patients could prove easier and more manageable when compared to IC. Its intermittent and short use (30 min, three times/day) may facilitate monitoring and patient mobility . We have chosen this interval because most of the studies using PEMF used a 20-30-min interval, in an attempt to maximize time of muscle stimulation decreasing venous stasis. Thirty minutes is well tolerated, and administering three times a day may maximize use and comfort without posing a danger to patients, while reducing venous stasis. We believe this strategy could simulate the regular ambulation for patients in recovery phase of critical conditions. A shorter period of device use could facilitate management of critical patients. 16
In addition, the use of PEMF allows reuse of the same machine for multiple patients, thereby possibly reducing costsand team work.
Although recent and robust studies suggest the use of only symptomatic VTE as outcomes, this is a challenging strategy to implement in ICU patients, in whom signs and symptoms are difficult to detect. In addition, VTE poses a great danger for patients, increasing mortality risk. Therefore we elected to use active search for VTE events, so as to enhance the clinical relevance of the study results. 2
Limitations to this study include possible biases and the impact of not blinding the interventions. This is a recurring limitation for open-label studies. In this study we try to diminish the impact of this possible bias by applying hospital VTE prevention protocol in both groups. The radiologist who will perform the duplex scan after the end of mechanical prophylaxis will be blinded to the intervention of the patient.
Another limitation is the relatively small sample size. This is, nevertheless, a proof-of-concept study in preparation for a larger study, if results are compelling.
In summary, PEMF could prove a safe and efficacious tool for mechanical prophylaxis in ICU patients. It maybe also prove easier for the ICU team to use, more comfortable for patients, and potentially cost-reducing. We expect this proof-of-concept study to generate data to guide the deisgn of more robust studies using PEMF for VTE prophylaxis in critical care patients.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article: Funded by the author with modest support from Science Valley Research Institute. Medical San provided the PEMF device to be used in this study.
Conflicts of Interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ER reports grants and consulting fees from Bayer and Pfizer, the Brazilian Ministry of Science and Technology, and personal fees from Aché Pharma, Novartis, and Daiichi-Sankyo outside the submitted work. EFS reports personal fees from Cardinal Health and Aché. No disclosures were reported for the current study by the other co-authors.
