Abstract
The management of pregnant women at increased risk of thromboembolic/other vascular events is still a matter of debate. In a single-center, retrospective, observational trial, we analyzed the safety and efficacy of prophylactic anticoagulation with certoparin in pregnant women at intermediate- or high-risk by EThIG criteria of thromboembolic/other vascular events. Subcutaneous certoparin 8,000 IU once daily was administered immediately after pregnancy confirmation and continued for 6 weeks postpartum. We investigated 74 pregnancies (49 women; mean age 31.8 years; weight 77.3 kg). Most prevalent risk factors were factor V Leiden mutation (40.5%), thrombogenic factor II mutation (12.2%) and protein S deficiency (8.1%). In 76 control pregnancies prior to registry inclusion/without anticoagulation there were 14 cases [18.4%] of venous thromboembolism (between week 7 gestation and week 8 postpartum); 63.2% pregnancies resulted in abortion (median week 8.6 gestation). With certoparin anticoagulation, thromboembolism was 1.4%, exclusively non-major bleeding was 4.1% and abortion was 10.8%. One case of pre-eclampsia necessitating obstetric intervention occurred. Prophylactic anticoagulation with intermediate-dose certoparin throughout pregnancies at increased venous vascular risk was safe and effective.
Keywords
Introduction
There is age-old knowledge regarding the relationship between pregnancy and post-thrombotic syndrome (previously referred to as “kindsfoot”). 1 The discovery of highly prevalent prothrombotic coagulation disorders 2,3 and the development of low-molecular-weight heparins (LMWH) soon led to the first studies investigating prophylactic anticoagulation (PA) with LMWH in pregnancies at increased risk of venous thromboembolism (VTE). 4,5 Systematic research revealed an association of thrombogenic coagulation disorders not only with VTE, but also with other obstetric events, like miscarriage, intrauterine growth restriction and pre-eclampsia. 6 In contrast to earlier promising investigations, trials showed no benefit of PA in women with recurrent miscarriage in the absence of a thrombophilic phenotype. 7,8 Current guidelines, therefore, recommend PA for women with hormone-related or idiopathic VTE and severe thrombophilia, but not for sole recurrent miscarriage. 9 –12
Although the indication for PA in venous vascular risk pregnancies has been established, the optimal dosage of LMWH still needs to be defined. Early trials aimed at the maintenance of certain anti-factor Xa activity levels showed higher efficacy of higher-than-standard prophylaxis dosages 4,13 and provided high success rates with maintained safety. 14 A recent cohort study investigating low-dose LMWH revealed a high rate of VTE despite PA, thereby questioning the efficacy of PA with low-dose LMWH. 15
Based on published data and positive, previous experience, 16 a protocol of PA using a uniform, intermediate dose of the LMWH certoparin for pregnancies at intermediate- or high vascular-risk was established. Certoparin was prescribed at the dose of 8,000 IU once daily (od) administered subcutaneously. Treatment was initiated upon pregnancy confirmation and was continued throughout the entire pregnancy, paused shortly during labour and continued for 6 weeks postpartum. Clinical course, coagulation and safety parameters were followed with scheduled visits around the 10th, 20th and 30th weeks of gestation by a small and constant team. A retrospective analysis was conducted to evaluate the safety and effectiveness outcomes of this approach.
Methods
Study Design and Population
Women with alleged or known coagulation disorders were referred from surrounding hospitals, general practitioners and other medical specialists to the Coagulation Clinic at Heidenheim hospital. Treatment data from the Coagulation Clinic was used to conduct a retrospective, single-centre study. The computerised medical files of all pregnancies registered in the Coagulation Clinic were searched using the terms “thrombosis” or “embolism” between 2009 and 2019. Only patients which received a dose of 8,000 IU subcutaneously once daily were selected. The same cohort of patients used in this registry was also assessed for prior pregnancy outcomes by way of comparison. The data analysis protocol was approved by the local ethics committee (University of Ulm, 28/18) and is listed in the German clinical trials registry (DRKS00018099).
Anticoagulation
Pregnant women received PA according to the local hospital protocol if they were at high to very risk of VTE because of their thrombophilic profile and history of VTE following the EThIG criteria. 17 We considered a further subcategorization based on type of risk but refrained from it due to power considerations and to maintain clinically applicable selection criteria. The local hospital protocol initiated PA treatment with immediate effect upon pregnancy confirmation (around the 6th week of gestation). Patients with a prior need for anticoagulation were initiated on certoparin when they desired to become pregnant.
The LMWH certoparin was administered at a dose of 8,000 IU subcutaneously once daily for all risk groups, irrespective of bodyweight. This dose was chosen based on the low to intermediate dose of either enoxaparin or dalteparin used in the current HighLow study (clinicaltrials.gov NCT01828697). Patients with antiphospholipid antibody syndrome received additional aspirin. Regular control visits were conducted at the Coagulation Clinic around gestational week 10, 20 and 30. In consultation with anaesthesiologists (for caesarean sections) and gynaecologists (for induced births), a short-term pause (<36 hours) of PA was allowed once the subject was admitted during labour and for spinal anaesthesia. Postpartum PA was continued for 6 weeks. Women with contraindications to anticoagulant treatment, known intolerance to certoparin, women already receiving LMWHs other than certoparin and women receiving doses of certoparin other than an intermediate dose were excluded from this analysis.
Definitions
The diagnosis of protein-C or protein S deficiency was based on personal and family history, repeated functional assays and was confirmed by DNA testing. Factor II mutations were based on DNA analysis of a potential G2010A prothrombin mutation and Factor V Leiden mutations were based on DNA analysis of a potential G1691A Factor V gene mutation (Xpert® FII and FV; Cepheid). Antiphospholipid syndrome was diagnosed using either liquid-phase coagulation assays to detect lupus anticoagulant or solid phase ELISA assays to detect anti-cardiolipin or anti-beta-2-glycoprotein I antibodies according to the International Society on Thrombosis and Haemostasis (ISTH) criteria. 18 Bleeding severity was also assessed using the International Society on Thrombosis and Haemostasis (ISTH) criteria. 19 –21
Research Questions
The principal research question was the rate of pregnancy-related symptomatic thromboembolism. Thromboembolism was considered pregnancy-related if it occurred during pregnancy or within 12 weeks of delivery. Venous thrombosis was assumed when a new or extended area of a non-compressible deep venous segment was seen on compression ultrasound; a diagnosis of pulmonary embolism was made by computed tomography (CT) scan.
According to standard chart review protocols, clinical status and laboratory values were recorded at gestational weeks 10, 20 and 30. During these visits, women were investigated for bleeding symptoms, obstetrical endpoints, symptomatic vascular or other treatment-related events following a checklist. In addition, laboratory control values, full blood count, coagulation and clinical chemistry markers were determined.
Statistical Analysis
Results are presented as mean ± standard deviation if the data were normally distributed. Non-normally distributed data are presented as medians with ranges. Kaplan–Meier survival analysis was used to visualise the timing of pregnancy-related thromboembolism and time of abortion or birth. The statistical analysis was performed using GraphPad Prism, version 8 (GraphPad Software, San Diego, CA, USA).
Results
Demographics, Medical History and Risk Factors
We investigated 74 pregnancies (including 1 twin pregnancy) in 49 women (Table 1). The mean age at pregnancy was 31.8 years; in 31.1% of all pregnancies women were older than 35 years. Patient weight was determined at the beginning of pregnancy, usually at the beginning of prophylaxis (approximately 6-8 weeks gestation). The mean patient weight was 77.3 kg, mean height was 167.7 cm; in 32.4% of all pregnancies, women were at least overweight (BMI >25 kg/m2).
Patient Characteristics.

Abbreviation: BMI, body mass index.
aProtein C and S deficiency were initially diagnosed based on activity and confirmed with molecular genetics.
bFactor V Leiden mutations were based on DNA analysis of a potential G1691A Factor V gene mutation (Cepheid test system).
cAll heterozygous.
Patients had a known thyroid disorder in 33.8% of pregnancies and an autoimmune disorder in 6.8% of pregnancies. Factor V Leiden mutation was the most prevalent risk factor (40.5%, the majority being heterozygous [n = 27]), followed by thrombogenic factor II mutation (12.2%, all heterozygous), and protein S (8.1%) or protein C deficiency (4.1%).
A history of thromboembolism in first-degree relatives was known in 12.2% (9/74) cases. Prior VTE was reported in 52.7% (39/74) of all pregnancies, of which 71% were singular and 29% were recurrent. Isolated deep-vein thrombosis (DVT) was the major manifestation (82.1%), 10.3% had combined DVT plus pulmonary embolism (PE) and 7.7% isolated PE (Table 2). Vascular events were pregnancy-associated in 35.9% cases, hormone treatment-associated in 43.6% cases, associated to other risk factors (e.g., operation) in 12.8% cases and idiopathic in only 7.7% cases.
Type of Prior Venous Thromboembolism Versus Risk Factors (n = 39).
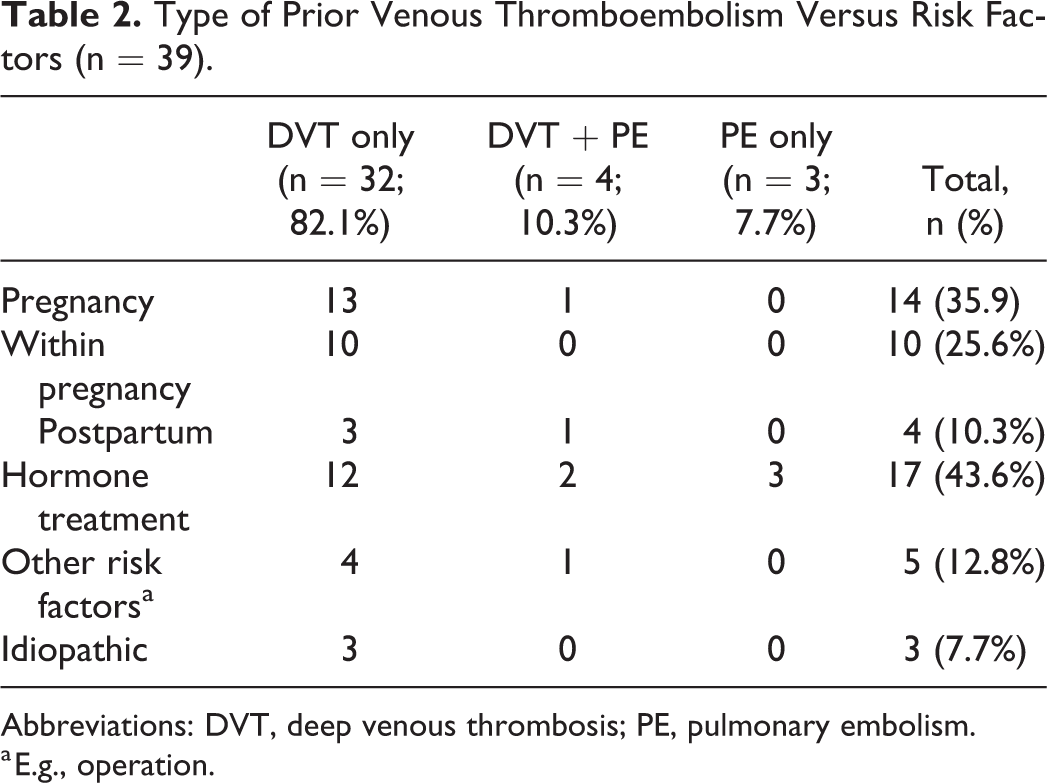
Abbreviations: DVT, deep venous thrombosis; PE, pulmonary embolism.
a E.g., operation.
Prior Pregnancies
During 76 pregnancies of the same women prior to inclusion in the registry, 14 cases of venous thromboses occurred between the 7th week of gestation and the 8th week postpartum resulting in a rate of pregnancy-associated thrombosis of 18.4% (Figure 1A). It was difficult to obtain reliable data on other vascular events that occurred during prior pregnancies; no overt haemolysis, elevated liver enzyme levels and low platelet levels (HELLP) syndrome and no other severe forms of pre-eclampsia, necessitating urgent birth were documented. Of these pregnancies, 63.2% (48/76) resulted in abortion (median 8.6 week of gestation), 36.8% (28/76) resulted in live births (median 40.0 week of gestation) (Figure 1B).

Pregnancies: Prior to Registry (A, B) and during the Registry (C, D).
Prophylactic Anticoagulation (PA)
Prior to becoming pregnant, 17.6% of all women (n = 13) were on long-term anticoagulation (either vitamin K antagonists [VKA] or non-VKA oral anticoagulants [NOACs], no cases of long-term LMWH treatment). To be eligible for PA, women had to have at least an intermediate-risk corresponding to the EThIG classification. 17 The cumulative number of pro-thrombotic risk factors was 1 in 33.8% (25/74), 2 in 35.1% (26/74) and 3 or more in 33.8% (25/74) of pregnancies.
In the registry pregnancies, PA was started on average (median) in the sixth week of gestation. There was no case of premature termination of PA.
Outcomes of Registry Pregnancies
Of the 74 pregnancies included in the registry, 10.8% (8/74) resulted in abortion (median 10.5 week [interquartile range (IQR) 10-12] of gestation) and 89.2% (66/74) resulted in live births (including 1 healthy twin birth; median 40 week [IQR 39-40] of gestation for all pregnancies) (Figure 1D). Deliveries were spontaneous in 69.7% cases, a caesarean section was performed in 30.3% cases. Overall, 49.2% were male babies and 50.8% female babies; mean birth weight was 3,475 ± 599 g and mean height was 51.6 ± 2.7 cm.
Clinically manifesting VTE occurred in 1 pregnancy (1.4%; 1/74) (Figure 1C) in a women with prior extensive, proximal DVT of the right leg 10 weeks after delivery of their first child. The rate of VTE in patients with prior VTE therefore was 2.56% (1/39). The present VTE presented as a calf vein thrombosis in the 8th week of gestation. A large, retroplacental haematoma was diagnosed simultaneously with the calf vein thrombosis. As a result, aspirin (100 mg od) was administered in addition to the PA already administered. Repeat control examinations showed regression of both the haematoma and the DVT; the remainder of the pregnancy was uneventful, and the woman delivered a healthy girl on time. In addition, there was 1 case of minor thrombophlebitis of the calf in the 9th week of gestation; the thrombophlebitis was treated locally and PA was continued unchanged. Fondaparinux was considered but disregarded because the expansion was low (<5 cm). Symptoms of pre-eclampsia developed in 1 pregnancy in the 35th week of gestation; symptoms resolved after urgent caesarean section. A retroplacental haematoma was diagnosed during 2 pregnancies (in the 11th and 13th week of gestation, respectively); PA was continued under close clinical surveillance and both pregnancies remained uneventful. As the cause of retroplacental haematoma was thought to be regional, placental infarction, these events were not recorded as bleeding events. 22,23
Small injection site haematoma (diameter <2 cm) occurred occasionally in approximately two-thirds of pregnancies; no change to PA or other intervention was necessary in these patients. Clinically relevant bleeding events were reported in 3 pregnancies, 1 occurring during the 12th week of gestation and 2 occurring in the 1st week postpartum. The latter 2 bleeding events were observed in a women with caesarean section which was diffuse and stopped spontaneously and the other after normal birth based on placental residues. None of these bleeding events necessitated specific hospital treatment, no blood transfusions were administered and, therefore, the bleedings were classified as clinically relevant non-major bleedings (3/74; 4.1%). 19
Results of Clinical and Laboratory Monitoring
Hepatic function tests within the first, second and third trimester were comparable (Figure 2). Mean haemoglobin concentrations decreased from 13.4 ± 0.9 to 12.4 ± 0.9 and 12.1 ± 0.9 g/dL with the differences vs. baseline being significant (P < 0.0001). Mean platelet counts remained stable.

Results of clinical and laboratory monitoring. ALT indicates alanine aminotransferase; AST, aspartate aminotransferase; ns, non-significant; trim, trimester. *P < 0.05; ****P < 0.0001.
Overall, we had 60 samples at weeks 10, 20 and 30 for D-dimer, antithrombin, haemoglobin, platelets and fibrinogen; approximately 45 samples each for protein S, protein C, aXa, ALT and AST; and 20 samples each for factor VIII (which became a routine test later than the other aforementioned ones).
Mean D-dimer concentrations were 0.7 ± 0.8, 1.0 ± 0.8 and 1.2 ± 0.6 mg/L over the course of gestation; the increases from week 10 to 20 and from week 10 to 30 were significant (P = 0.0176 and P = 0.0012, respectively). Mean factor VIII activities increased from 153% ± 30% to 188% ± 39% and 229% ± 46%; the increase from the 10th to the 30th week was significant (P = 0.0002). Mean fibrinogen activities (Clauss method) increased from 3.4 ± 0.8 to 3.9 ± 0.7 and 4.3 ± 0.7 g/L with the respective increases being significant (P < 0.0001) (Figure 3).

Markers of procoagulant activity. ns indicates non-significant; trim, trimester. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
Mean antithrombin activities remained stable at 94% ± 11%, 92% ± 12% and 95% ± 13% at weeks 10, 20 and 30, respectively. Mean protein C activities increased from 104% ± 25% to 112% ± 26% and 127% ± 23%; the increases were significant (P < 0.001) from 10th to 20th week and P < 0.0001 from 10th to 30th week). Mean protein S activities were already decreased to 57 (±20)% by the 10th week of gestation and decreased further to 50 ± 21% (P = 0.01) and 49% ± 17% (P = 0.004) by the 20th and 30th week of gestation, respectively (Figure 4). No symptomatic skeletal complications were recorded, and no HIT2-syndrome was diagnosed.
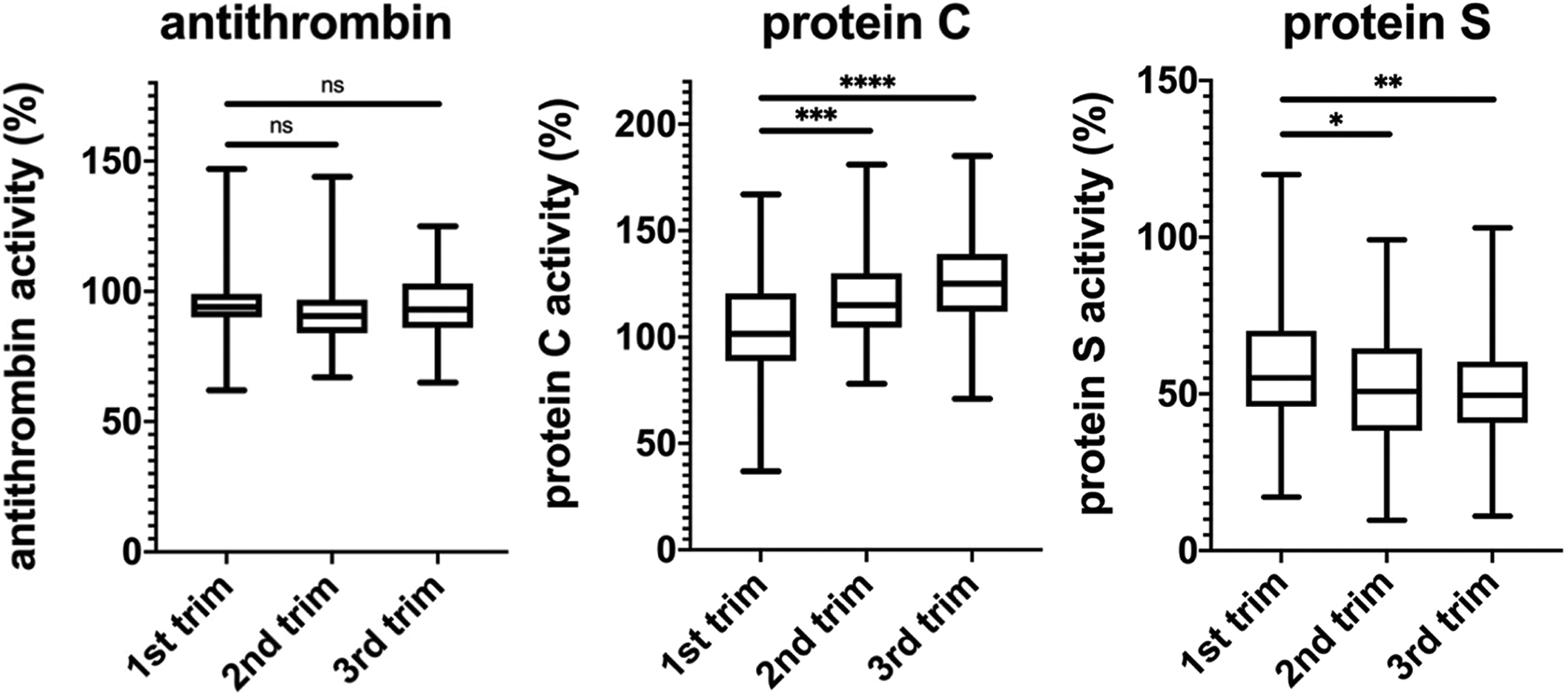
Markers of anticoagulant activity. trim indicates trimester. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
Anticoagulant Activities of Intermediate-Dose Certoparin During Pregnancies
The anticoagulant activity of PA with intermediate-dose certoparin, administered once daily, was determined with anti-factor Xa activity (aXa) measurements to assess product accumulation during long-term therapy. In women injecting their dose in the morning, aXa measurements took place <6 hours after injection, in women injecting their dose in the afternoon, aXa measurements took place >12 hours after injection. Mean aXa activities were 0.22 ± 0.12, 0.25 ± 0.11 and 0.26 ± 0.07 U/mL in the 10th, 20th and 30th week of gestation, respectively, in women injecting in the mornings (depicted in red in Figure 5) and 0.07 ± 0.07, 0.12 ± 0.12 and 0.12 ± 0.12 U/mL in the 10th, 20th and 30th week of gestation, respectively, in women injecting in the afternoon (depicted in blue in Figure 5). None of the differences within 1 application group reached the level of statistical significance.

Anticoagulant activities of certoparin. ns indicates non-significant; trim, trimester.
Discussion
Women are at increased risk of both venous and arterial thromboembolism during pregnancy, and the postpartum risk of VTE is even higher in the first 6 weeks. 24,25 Early reports already suggested benefits of PA in pregnancies at high-risk of thrombosis, but these did not remain undisputed. 5,26 In our large, single-centre series of 74 pregnancies in 49 women, we found that anticoagulation using 8,000 IU certoparin once daily was safe and effective in preventing thromboembolism and reducing spontaneous abortion rates. The rate of clinically manifesting thromboembolism was 1.4%, (exclusively non-major) bleeding was 4.1% and abortion was 10.8%. These results were particularly encouraging against the background of 76 pregnancies documented in the same cohort of women without anticoagulation, where 14 cases of venous thromboses occurred (18.4%) and 48 pregnancies resulted in abortion (63.2%). Furthermore, the bleeding rate of patients receiving certoparin in our study is comparable to previously reported bleeding rates for certoparin in previous studies. 27,28
Who to Treat and Who Not to Treat With PA
The question of who not to treat with PA during pregnancy is largely settled, and misconception and pregnancy loss of unknown cause are not established indications for PA. 29 –31 The question of who to treat, namely women at distinctly increased risk of vascular complications, also seems to be resolved. However, regulatory guidance is somewhat vague. For example, dose recommendations (“less treatment for lower risks, more treatment for higher risks”) are often difficult to translate into clinical practice. Moreover, the level of risk may change throughout pregnancies and the practice of dose adjustment following changes in weight, D-dimer levels, anti-Xa target levels or other parameters is cumbersome and only poorly sustained by scientific evidence. 32,33 Furthermore, there is evidence that underdosing PA may be detrimental. 15 For all these reasons we chose a “one indication—one dosage” approach with certoparin using an intermediate (half therapeutic) dose for PA in our institution. Certoparin was the anticoagulant of choice because of its universal bodyweight-independent dosing and its safety record. 34 –37
The Optimal Timeframe of PA During Gestation
Determining the most appropriate time to initiate PA and when, in the postnatal period, treatment should be stopped is still a matter of debate. In 2014, a Cochrane review of prophylaxis for VTE in pregnancy and the early postnatal period analysed 43 reports, but only 6 of these had investigated antenatal PA. They concluded that there is insufficient evidence on which to base recommendations for thromboprophylaxis during pregnancy and the early postnatal period. 38 In the TIPPS trial, PA was initiated at only 11.9 weeks (mean) of gestation. 39 Of note, in this PACER-VARP registry, the only vascular event under PA occurred in the 8th week of gestation and spontaneous abortions occurred at a median of 10.5 weeks under PA and at 8.6 weeks during prior pregnancies without PA. Very early initiation of PA, therefore, is advisable and initiating treatment promptly after confirmation of pregnancy is recommended.
According to our protocol, PA was terminated 6 weeks after birth. Few studies have investigated the optimal time to stop PA. Thromboembolic risk is highest during the first 3 weeks and remains elevated throughout the first 12 weeks postpartum. 40 More recent data suggest that the thromboembolic risk could remain elevated even beyond 175 days after birth. 41 However, current guidelines usually recommend a postpartum prophylaxis for 6 weeks postpartum. 9 –12 In this PACER-VARP registry, 36% (5/14) of all VTE that occurred in prior pregnancies before inclusion in the registry (and without PA), occurred postpartum; in contrast no VTE were observed until 12 weeks postpartum in our patients treated according to protocol (including PA until 6 weeks after birth). This observation strongly supports the current practise of postpartum administration of PA for 6 weeks.
Establishing a Suitable Dose
Establishing and monitoring an appropriate LMWH dose during pregnancy is challenging. 42 Previous studies have shown that the dose of the LMWH enoxaparin and dalteparin should be increased as the pregnancy progresses to maintain a constant dose. 32,33,43,44 To overcome this, we chose a fixed, intermediate-dose of certoparin (8,000 IU od) because of its universal bodyweight-independent dosing. Mean aXa activities met or slightly exceeded the recommended target range of 0.23 U/mL 45 with determined values of 0.22, 0.25 and 0.26 U/mL in the 10th, 20th and 30th week of gestation. Our data shows this dose is safe and effective over the course of the pregnancy.
Recurrent Pregnancy Loss (RPL)
Over the last few decades, a link has been established between a hypercoagulable state (a contributing factor to VTE in pregnancy) and adverse pregnancy outcome, in particular recurrent pregnancy loss. 46 Although guidelines do not generally recommend PA for sole recurrent miscarriage, a number of studies have shown, that PA, mostly LMWH combined with aspirin, reduces the risk of recurrent pregnancy loss in the subset of women with persistent antiphospholipid antibodies. 47 Yet, in women without known 7 or with a low incidence of thrombophilia, 48 PA does not increase the life birth rate in women with a history of RPL. 31 It was assumed however, that a small subset of women might benefit from PA for the prevention of RPL under certain conditions. 49 Our retrospective analysis supports these findings; we found a benefit not only for women with persistent antiphospholipid antibodies, but also for other forms of high-risk thrombophilia, suggesting that PA may reduce the likelihood of RPL in women also at high-risk of VTE. Of note, our treatment protocol combines rigorous admonition for a very early start of PA with intermediate LMWH dosage, in this differing from any other trial thus far. Our life-birth rate of 88% compares favourably with life-birth rates of around 65%-77% reported from earlier trials. 7,29,48,50 The still ongoing ALIFE2 trial is an instructive example of how difficult it is to conduct successful research on this topic 51 ; it remains to be seen whether this study can conclusively answer the question of who will benefit of PA for RPL.
Challenges With Clinical Trials in Pregnant Women
While there is a clear need for additional information on the use of PA during pregnancy, clinical trials in this patient group are notoriously difficult. In the mid-1980s, a number of case series and trials investigating PA in venous vascular risk pregnancies were published. Patient selection and therapeutic approaches have been markedly variable, rendering conclusive statements difficult. Attempts to perform randomised trials on the topic failed repeatedly, due to extremely low recruitment. 52,53 For example, the TIPPS trial, which was published in 2014 as the first, randomised trial on PA, took in excess of 12 years to recruit the planned patient numbers. 39 With this in mind, retrospective registry analyses appear to present a way forward to gleaning information on the effectiveness of LMWH in patients at high risk of VTE during pregnancy.
Potential Limitations
The main limitation of this analysis is its retrospective nature, which makes it subject to potential selection bias. All women who met the criteria for treatment according to our protocol were sequentially included in our registry. Women with more than 1 pregnancy were included in the analysis for each pregnancy, but increasing age and every new pregnancy represents a new thrombosis risk. Furthermore, we compared the data from registered pregnancies (with PA) with 76 pregnancies of the same cohort that took place prior to establishing the registry (without PA). Due to the retrospective nature of the study the datasets may be incomplete for some patients; for example, bleeding is notoriously difficult to determine particularly primary and secondary postpartum haemorrhage and prolonged lochia. In addition, some data may have been lost or missed during referral to the coagulation clinic. Another potential limitation is that the study assessed only 1 anticoagulant, certoparin, and 1 dose (8,000 IU od). This does, however, make it easier to compare the results of the PA protocol across all patients in the registry. In an ideal scenario, women on long-term anticoagulation would have been excluded from the study, but the registry-style nature of this study means that was not possible. However, inclusion of these patients reflects a more realistic representation of a population of pregnant women. The final limitation of this study is that it was a single-centre study, which limits the generalisability of the results. On the other hand, the treatment modalities within a single centre are handled very homogenously so that practitioner-dependent differences do not play a role.
Conclusions
The data from the PACER-VARP registry presented here verify our assumption, that a 1 indication—1 dosage approach using intermediate-dose certoparin for PA in women at distinctly increased venous vascular risk during pregnancies is effective and safe. No major bleedings were recorded in our registry. Only 1 symptomatic, vascular event occurred, which was an isolated calf vein thrombosis recurrence in the 8th week of gestation in a patient with a pre-existing post-thrombotic syndrome due to an extensive, proximal DVT 10 weeks after delivery form her first child. Adopting this treatment approach has the potential to reduce VTE events and adverse pregnancy outcomes in pregnant women at high risk of thromboembolic and other vascular events.
Footnotes
Authors’ Note
Martin Grünewald (MG), Esther Häge, Stephanie Lehnert and Martina Güth conceived and conducted the study. AS and CM contributed to the conception of the study. MG performed the explorative analysis and wrote the first draft of the manuscript. PB and CM revised the manuscript for important intellectual content. All authors approved the final version of the manuscript. The data analysis protocol was approved by the local ethics committee (University of Ulm, 28/18) and is listed in the German clinical trials registry (DRKS00018099). Patient-informed consent was not required due to the retrospective nature of the study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Martin Grünewald received lecture fees from Aspen and other manufacturers of various anticoagulants. Peter Bramlage (PB) is a consultant to Aspen and has worked with other makers of low-molecular-weight heparins as well (Sanofi, Novartis). Christiane Maier (CM) and Alexandra Schimke (AS) are employees of Aspen.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors report funding from Aspen, Germany, supporting their clinical and statistical evaluation.
