Abstract
Fondaparinux sodium is a chemically synthesized selective factor Xa inhibitor approved for the prevention and treatment of venous thromboembolic events, that is, deep vein thrombosis, pulmonary embolism, and superficial vein thrombosis, in acutely ill (including those affected by COVID-19 or cancer patients) and those undergoing surgeries. Since its approval in 2002, the efficacy and safety of fondaparinux is well demonstrated by many clinical studies, establishing the value of fondaparinux in clinical practice. Some of the advantages with fondaparinux are its chemical nature of synthesis, minimal risk of contamination, 100% absolute bioavailability subcutaneously, instant onset of action, a long half-life, direct renal excretion, fewer adverse reactions when compared with direct oral anticoagulants, and being an ideal alternative in conditions where oral anticoagulants are not approved for use or in patients intolerant to low molecular weight heparins (LMWH). In the last decade, the real-world use of fondaparinux has been explored in other conditions such as acute coronary syndromes, bariatric surgery, in patients developing vaccine-induced immune thrombotic thrombocytopenia (VITT) and in pregnant women with heparin-induced thrombocytopenia (HIT), or those intolerant to LMWH. The emerging data from these studies have culminated in recent updates in the guidelines that recommend the use of fondaparinux under various conditions. This paper aims to review the recent data and the subsequent updates in the recommendations of various guidelines on the use of fondaparinux sodium.
Keywords
Introduction
Fondaparinux sodium (Arixtra®) is the first synthetic pentasaccharide and a selective inhibitor of factor Xa approved for the prophylaxis and treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE). It is widely prescribed for the prevention of venous thromboembolic events (VTE) in post-operative adults undergoing major orthopedic surgery of the hip and knee and abdominal surgery. Fondaparinux sodium is also approved for use in patients at a high risk for VTE who are immobilized due to acute illness. It is approved and used for the treatment of acute symptomatic spontaneous superficial vein thrombosis (SVT) of the lower limbs without concomitant DVT. 1,2 Fondaparinux is also used for the treatment of acute coronary syndromes such as unstable angina or myocardial infarction with or without ST-segment elevation.
One of the main advantages of fondaparinux is that it is synthesized chemically, while heparins and heparinoids are prepared from animal extracts. This chemical nature of the synthesis ensures no general supply issues and minimal risk of contamination, and in theory, does not cause heparin-induced thrombocytopenia (HIT). 3 Fondaparinux has 100% absolute bioavailability subcutaneously (SC), instant onset of action, a long half-life (terminal half-life of 17–21 hours), direct renal excretion, and fewer adverse reactions when compared with direct oral anticoagulants. 4 -6 The efficacy and safety of fondaparinux in the prevention of VTE in lower extremities during major orthopedic surgeries and in high-risk abdominal surgery has been demonstrated in many clinical studies and it has been found to be superior or non-inferior to other antithrombotic agents. 7 -13 For the past 2 decades, fondaparinux has been one of the mainstays for VTE prevention in acutely ill adult patients. 14 There are reports suggesting its off-label use under conditions such as HIT in pregnant women. 15 Fondaparinux is also considered for bridging therapy to switch to oral anticoagulants in perioperative anticoagulation management. 16
Emerging data elaborate the advances in the use of fondaparinux, thereby warranting the need to revisit and update the recommendations of various guidelines on the management of thrombosis under different medical conditions and in very obese patients. 17 This article aims to review and present recent data and findings (in the past 5 years) pertaining to the use of fondaparinux sodium as well as recent updates in the recommendations of various guidelines. This would pave the way for generating evidence-based recommendations and facilitate informed clinical decisions.
Fondaparinux—Clinical Development and Previous Studies
Fondaparinux was approved by the United States Food and Drug Administration in the year 2001 and by the European Medicines Agency in 2002 for the post-operative prophylaxis of VTE in patients undergoing hip fracture, hip replacement, and knee replacement surgeries. 18,19
In 2001, fondaparinux sodium was recognized as a new class of anticoagulant that selectively targets factor Xa. 20 Its pharmacokinetic profile (100% bioavailability, longer half-life, and rapid onset of action), ease of dosing (once daily administration), and acceptable tolerability (not affecting prothrombin/activated partial thromboplastin time and platelet function or aggregation, and no in vitro cross-reactivity to heparin antibodies) led to fondaparinux being considered an ideal antithrombotic agent 20 (Figure 1).

Mechanism of the action of fondaparinux. Factor Xa is located at the junction of both extrinsic and intrinsic pathway. 1. Binding of fondaparinux to antithrombin. 2. After binding to fondaparinux, a conformational change occurs in antithrombin. 3. When bound to fondaparinux, antithrombin selectively and rapidly inhibits Factor Xa, thereby blocking thrombin generation and fibrin formation. 4. Fondaparinux is released to interact with other antithrombins. Adapted from Turpie AG. Fondaparinux: a Factor Xa inhibitor for antithrombotic therapy.
Many studies have demonstrated the efficacy and safety of fondaparinux in preventing VTE across a range of medical and surgical settings, for example, major orthopedic surgeries involving lower limbs, abdominal surgeries, and for the treatment of DVT and PE. 21
Fondaparinux was as effective and as safe as unfractionated heparin (UFH) in the initial treatment of hemodynamically stable patients with PE. 22 In the initial treatment of patients with symptomatic DVT, fondaparinux was non-inferior to enoxaparin, and both were well-tolerated. 23 -25
A significant reduction in mortality and reinfarction was observed with fondaparinux (2.5 mg once daily SC) in patients with acute ST-elevation myocardial infarction (STEMI), particularly those not undergoing primary percutaneous coronary intervention without increasing bleeding and strokes. 26 In patients with non-STEMI, fondaparinux (2.5 mg once daily SC) was comparable to enoxaparin (1 mg/kg twice daily; 5.8% vs 5.7%; hazard ratio in the fondaparinux group, 1.01; 95% confidence interval [CI], 0.90–1.13) in reducing the risk of ischemic events (death, myocardial infarction, and refractory ischemia) in patients with acute coronary syndromes at Day 9. Fondaparinux substantially reduced major bleeding in this medical condition, which frequently leads to thrombotic complications and thereby translated to improving long-term mortality and morbidity. 27,28
The efficacy and safety of fondaparinux (2.5 mg once daily SC [vs placebo]) in patients with acute, isolated symptomatic SVT of the legs were evaluated in the CALISTO study, which was the first large randomized controlled study with clinical endpoints. Death from any cause or symptomatic PE, symptomatic DVT, or symptomatic extension to the saphenofemoral junction, or symptomatic recurrence of SVT at Day 47 was the primary efficacy outcome. A significant reduction in the incidence of each component of the primary efficacy outcome was observed in the fondaparinux group when compared with the placebo group, except for the outcome of death (0.1% in both groups). The rate of PE or DVT was 85% lower for fondaparinux versus placebo (0.2% vs 1.3%; 95% CI, 50–95;
Two observational studies, namely POST (2010) and OPTIMEV (2011), included French patients with isolated SVT 30 -32 and assessed the association between SVT, PE, and DVT. In spite of a high rate of initial anticoagulation, patients with SVT incur a significant risk of recurrent VTE and recurrent or extended SVT at 3 months. Hence, a venous ultrasound examination could be helpful in identifying anticoagulation in all patients with suspected SVT. The OPTIMEV study revealed that while PE with DVT has a substantial 3-month mortality, no association was observed with a higher 3-month mortality in PE without DVT. Hence, while diagnosing PE, it is important to consider the associated DVT to optimize appropriate management strategies. 33,34
Fondaparinux will require close monitoring of the anti Xa level when used concomitantly with drugs that affect hemostasis, and will be used with caution in patient weighing <50 kg or with a dose reduced in case of creatinine clearance between 30–50 mL/min. 35 As with most of the other anticoagulants cleared by the kidney, fondaparinux must not be used in patients with severe renal impairment (i.e., with a creatinine clearance <30 mL/min). 35,36 Use in neurosurgery or spinal procedures should be avoided.
Along with the results from the randomized controlled trials (RCTs), the efficacy and safety of fondaparinux was also assessed in real-world studies. RCTs are designed carefully to provide control of bias, meticulous data collection and selection of patient population, and adherence to treatment protocol in order to reduce variation which could influence the study results. However, they are limited in terms of the patient population enrolled in the study, who match the inclusion/exclusion criteria and may not be fully representative of the patients observed in routine clinical practice. Hence, real-world studies are important to evaluate the efficacy and safety of the drug among diverse patient populations with varying health status and comorbidities and inform outcomes with long-term therapy. Thus, the results of both RCTs and real-world studies complement each other and help the clinician in making a treatment decision for the patient. 37,38
Recent Advances in the Use of Fondaparinux Sodium
This section describes the recent advances in the use of fondaparinux (both in approved indications and off-label use) in the past 5 years, as reported in the literature. In the last decade, the real-world use of fondaparinux has been explored under conditions such as acute coronary syndromes, bariatric surgery, in patients developing HIT-like syndromes post vaccination and in pregnant women with HIT and the results obtained in the past 5 years are summarized in this section 39 -42 (Table 1).
Recent Advances in the Use of Fondaparinux Sodium.
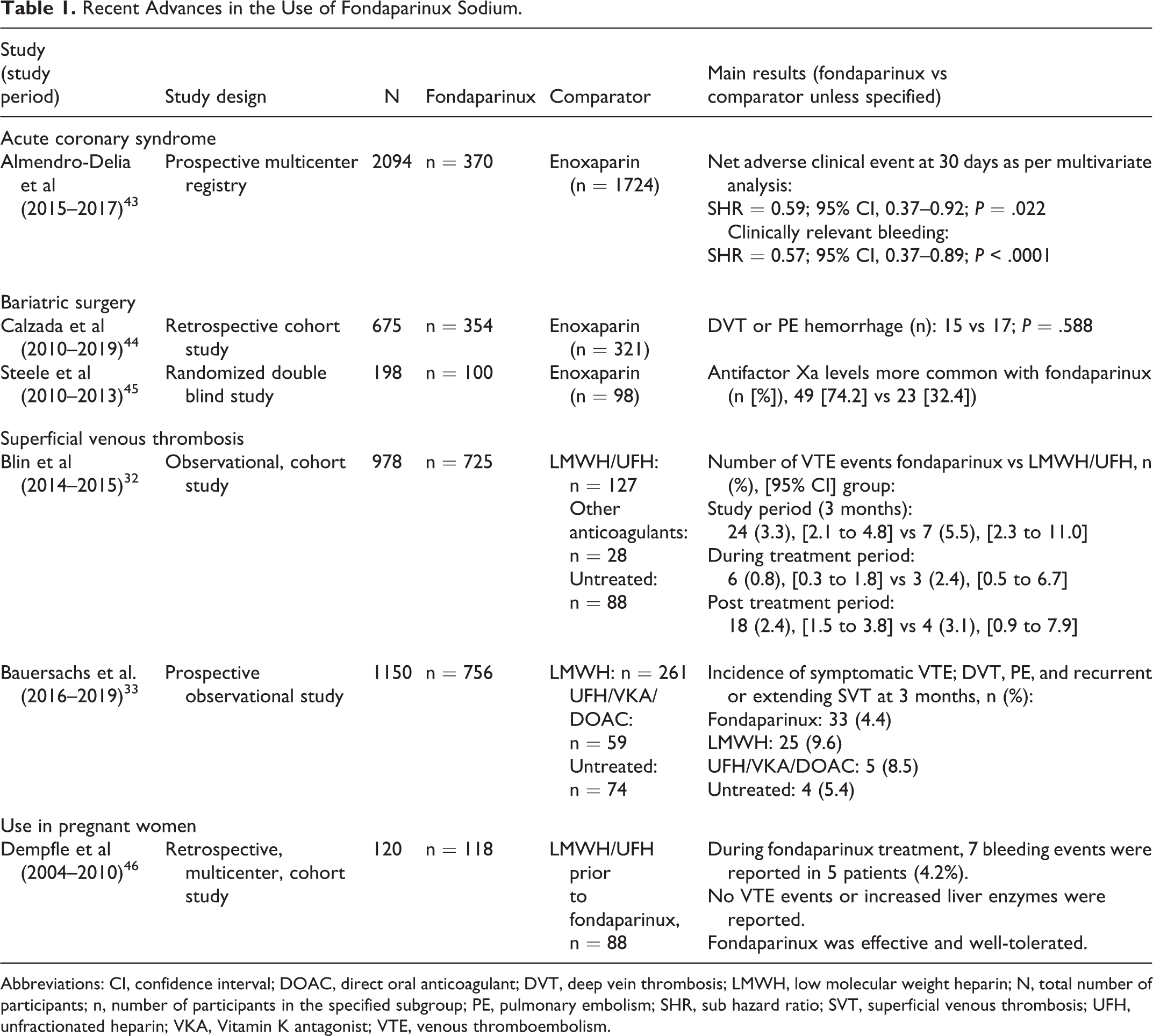
Abbreviations: CI, confidence interval; DOAC, direct oral anticoagulant; DVT, deep vein thrombosis; LMWH, low molecular weight heparin; N, total number of participants; n, number of participants in the specified subgroup; PE, pulmonary embolism; SHR, sub hazard ratio; SVT, superficial venous thrombosis; UFH, unfractionated heparin; VKA, Vitamin K antagonist; VTE, venous thromboembolism.
Acute Coronary Syndromes
The prospective data of non-ST-elevation acute coronary syndrome patients who received fondaparinux or enoxaparin from February 2015 to December 2017 were analyzed from the multicenter ARIAM Andalucia registry. The composites of clinically relevant bleedings and major adverse cardiovascular events were assessed for 30 days using survival models within a competing risks framework including site-specific random effects. A net clinical benefit was seen in the results from the multivariate analysis which was in favor of fondaparinux versus enoxaparin (sub hazard ratio [SHR] 0.59; 95% CI, 0.37–0.92), with a reduction in bleeding (SHR 0.57; 95% CI, 0.37–0.89). There was a significant 43% reduction in the adjusted risk of bleeding in the fondaparinux group with fewer clinically relevant major and non-major “actionable” bleeding events versus the enoxaparin group.
43
Furthermore, greater reductions in bleeding were observed in the exploratory analyses with fondaparinux among patients undergoing a transradial approach. The study demonstrated a significant interaction between treatment and vascular access on the multiplicative scale (
Bariatric Surgery
A retrospective cohort study (2010–2019) assessed the thrombotic and hemorrhagic risk in bariatric surgery with multimodal rehabilitation programs in vertical gastrectomy or gastric bypass surgeries (N = 675). Patients with similar baseline characteristics were treated with fondaparinux (fixed dose of 2.5 mg/day, n = 354) or enoxaparin (single daily dose adjusted to BMI: 40 mg/day for a BMI of 35–40 and 60 mg/day for a BMI 40–60, n = 321). The results showed no DVT or PE and 32 incidences of hemorrhage (4.7%), 15 in the fondaparinux group and 17 in the enoxaparin group (
Steele et al compared the prophylactic effect of fondaparinux (5 mg once daily) versus enoxaparin (40 mg twice daily) for routine VTE prophylaxis in patients undergoing bariatric surgery. This dosage of fondaparinux was higher than that proposed in the product information leaflet (2.5 mg); studies have shown that with a 2.5 mg dose, 53% of morbidly obese patients in the study population reached the target antifactor Xa levels, while with a 5 mg dose, the morbidly obese patients achieved target antifactor Xa concentrations more consistently. Fondaparinux was more commonly associated with adequate antifactor Xa levels (74.2%) than with enoxaparin (32.4%). However, both regimens were equally effective in reducing the risk of DVT. 45 A recent statement by the International Society on Thrombosis Scientific and Standardization Committee suggested to use a parenteral anticoagulant such as LMWH or fondaparinux in patients undergoing bariatric surgery and to prescribe direct oral anticoagulants in the post-acute phase following bariatric surgery. 17 Since novel oral anticoagulants cannot be used in this acute setting, fondaparinux provides a safer and more consistent anticoagulant activity in such challenging high-risk patients.
Superficial Vein Thrombosis
The PERSEUS study, which was a post-marketing French cohort study, was conducted in adults with acute symptomatic spontaneous SVT of the lower limbs without concomitant DVT who received anti-thrombotic agents (fondaparinux, LMWH/UFH, aspirin, clopidogrel, rivaroxaban, or vitamin-K antagonist) and who received no treatment other than for SVT. This study was designed to include 1,000 adults (recruited prospectively and consecutively by physicians [up to 10 adults per health care practitioner over a 6-month period]) with a diagnosis of acute symptomatic spontaneous isolated SVT of the lower limbs who were followed up for 3 months. The main outcomes studied were symptomatic venous thromboembolism (VTE: DVT, PE, and SVT extension or recurrence) and major bleeding through review of compression ultrasonography, hospital discharge summary, and other pertinent medical reports. The overall incidence of VTE events during the treatment and post-treatment period of the PERSEUS study was numerically lower with fondaparinux versus the LMWH/UFH group (3.3% [95% CI = 2.1–4.8] vs 5.5% [95% CI = 2.3–11.0], respectively). The results showed that the overall rate of symptomatic recurrent VTE was low with fondaparinux treatment. During the treatment period, the incidence of VTE was lower with fondaparinux versus LMWH/UFH (0.8% vs 2.4%). The incidence of VTE was 2.4% and 3.1% for patients during the post-treatment period, initially treated with fondaparinux and LMWH/UFH, respectively. 32 These results demonstrated that fondaparinux (prescribed in 75% of the patients) was the first choice of vascular physicians in France for the treatment of SVT. 48 These real-world clinical practice outcomes confirm the benefits of fondaparinux, as was shown in the CALISTO study. 29
Another prospective, multicenter, observational study, the Investigating SIGnificant Health TrendS in the management of Superficial Vein Thrombosis (INSIGHTS-SVT), was conducted in Germany to collect representative data on patient characteristics, diagnosis, management, and outcomes of isolated SVT under real-world conditions.
33
A composite of symptomatic DVT, PE, and extension or recurrence of SVT at 3 months was the primary outcome assessed in the study. Clinically relevant bleeding was the primary safety outcome. After adjusting for the propensity score and duration of drug treatment, there was a 5.2% absolute reduction in the probability of the primary VTE outcome in patients treated with fondaparinux versus those treated with LMWH (4.4% vs 9.6%; 95% CI, 0.30–0.88; hazard ratio [HR] 0.51;
The Surprise study was a prospective, randomized, open-label, blinded adjudication trial conducted in patients with superficial phlebitis treated for 45 days with rivaroxaban versus fondaparinux which aimed to demonstrate non-inferiority of rivaroxaban with fondaparinux in preventing the combined efficacy endpoint of thrombus progression, SVT recurrence, DVT, PE, and death. This study was conducted after the placebo-controlled CALISTO trial and allowed patients with a high risk for thromboembolic complications to be included. Though the study met the noninferiority criterion, the clinically relevant non-major bleeding and more severe treatment-emergent adverse events were numerically higher in the rivaroxaban group than in the fondaparinux group. Hence, the study results revealed that patients with SVT at high-risk require at least 45 days anticoagulation and a prophylactic dose of 2.5 mg of fondaparinux (over rivaroxaban) was sufficient for the high-risk patients with a low rate of recurrence during treatment. 49
Pregnancy
During pregnancy, women at risk of VTE who receive LMWH can develop HIT. LMWH is the preferred option for the prevention and treatment of VTE during pregnancy, as it does not cross the placenta and is non-teratogenic in nature. The approved treatment options argatroban and danaparoid are expensive and also associated with some limitations. Argotraban is correlated with an increased risk of bleeding and requires monitoring during intravenous infusion. Danaparoid is administered intravenously (IV) or through multiple SC injections and is not available in some countries. In such scenarios, where approved treatments cannot be used, fondaparinux is considered as the next choice. 50 Although there are reports of its off-label use in HIT, no clinical study has been conducted yet; hence, there is no clear posology.
To expand the evidence available in literature for this condition, a systematic literature study reported that fondaparinux is an effective and safe anticoagulant for the treatment of acute HIT despite the absence of randomized trials. However, it should be used cautiously in patients with renal insufficiency. 51
Dempfle et al conducted a retrospective, multicenter, cohort study in Germany including women who received fondaparinux pre-, peri-, and/or post-partum for ≥7 days for prophylaxis/VTE. All participants received LMWH as the first-line therapy and unfractionated heparin. The treatment was changed to fondaparinux, owing to heparin allergy (41.7%) or HIT (10.0%). During the treatment, there were no observations of VTE events or increased liver enzymes. The study results showed that fondaparinux was well-tolerated by those pregnant women. 46 Fondaparinux is an available treatment option in the pregnant women when LMHWs are contraindicated or not well tolerated.
Recent Recommendations From Guidelines
Fondaparinux is still recommended as one of the treatment options for the prevention and treatment of VTE by various guidelines in acutely ill (including those affected by COVID-19), cancer patients and those undergoing surgeries. The emerging data have culminated in recent updates in the guidelines that recommend the use of fondaparinux under various conditions (Table 2).
Recently, there were reports of thrombotic events associated with the administration of COVID-19 vaccines, a condition called vaccine-induced immune thrombotic thrombocytopenia (VITT). Non-heparin anticoagulants including direct oral anticoagulants, direct thrombin inhibitors, and fondaparinux are recommended for the treatment of VITT. 59
Recommendations From Various Recent Guidelines for the Use of Fondaparinux.

Abbreviations: ASH, American Society of Hematology; COVID, corona virus disease; LMWH, low molecular weight heparin; PCI, percutaneous coronary intervention; SC, subcutaneous; SVT, superficial venous thrombosis; UFH, unfractionated heparin; VTE, venous thromboembolism.
Future Directions
Fondaparinux has been approved for the prevention and treatment of VTE in 2001 and is being recommended for use by various guidelines. The evidence and scientific knowledge available in the literature demonstrates the efficacy and safety of fondaparinux sodium. Future research can be aimed at generating additional real-world data on the use of fondaparinux in bariatric surgery. Further studies may provide more insights regarding the use of fondaparinux in HIT, particularly in pregnant women, and it might rationalize the current off-label use to being approved for this indication. Apart from the evidence demonstrated by the clinical trials, the prescribing decisions are also influenced by other factors such as clinical experience, positive outcomes, patient demographics, guideline recommendations, health needs, preferences, cost, treatment access, and insurance coverage (more common in North America than in Europe). 60 Fondaparinux is cost-effective compared with LMWH by preventing the incidence of VTE, especially when being considered for long-term prophylaxis, 61 -63 which supports its wider patient access and hence good compliance. 64 Real-world evidence studies in the future should aim to include long-term efficacy, safety, cost-effectiveness, and compliance to generate evidence that would be helpful in clinical- and policy-decision making.
Conclusions
Fondaparinux is a unique synthetic injectable pentasaccharide, whose efficacy as an ideal anti-thrombotic agent has been demonstrated by many studies. With its well-established efficacy and safety, recent updates in guidelines recommend its use in medical and surgical settings to prevent and treat VTE. Recent real-world data of fondaparinux support the results observed in the clinical trials. Fondaparinux plays a critical role in the prevention of VTE in acutely ill patients including COVID-19 patients. Reports suggest that fondaparinux can be a therapeutic option in pregnant women with HIT or those not tolerating LMWH. However, further research is warranted to provide more insights on the benefits that fondaparinux could offer in these settings.
Footnotes
Authors’ Note
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Acknowledgments
Medical writing assistance was provided by Dr. Shridevi Venkataramani, and editorial assistance was provided by Mr. Gaurav Anand (both employed by Tata Consultancy Services, India) under the direction of the author. The author thanks Dr. Jean-Philippe Verbist from Viatris for his continuous scientific advice during the development of the draft.
Author Contributions
The author contributed to data interpretation, development, and review of this manuscript. The author confirms that he has read the journal’s position on issues involved in ethical publication and affirm that this report is consistent with those guidelines. The sponsor also provided a formal review of this manuscript. The author meets the ICMJE criteria and had access to the study data, made the final decision about where to publish these data and approved submission to this journal.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Prof. Bauersachs worked as a paid consultant and received speaker fees from Bayer AG, Bristol-Myers Squibb, LEO Pharma, Pfizer Inc., and Viatris Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the medical writing assistance was provided by Viatris Inc.
