Abstract
Background
Timely and accurate diagnosis of pulmonary thromboembolism (PTE) in the emergency department (ED) is critical, yet remains challenging due to non-specific clinical features. Platelet indices have recently emerged as potential biomarkers, but their diagnostic utility is uncertain.
Methods
In this retrospective single-center study, 1198 adult patients who underwent CT pulmonary angiography for suspected PTE in a tertiary ED were analyzed. Demographic characteristics and a comprehensive panel of hematological, biochemical, and coagulation parameters were compared between PTE (n = 126) and non-PTE (n = 1072) groups. Multivariate logistic regression and ROC curve analyses were performed to assess independent predictors and diagnostic performance.
Results
Platelet indices including platelet count, mean platelet volume, platelet distribution width, plateletcrit, and derived ratios showed no significant differences between groups. However, PTE patients were older, more likely female, and exhibited significantly lower red blood cell count, hemoglobin, hematocrit, and higher neutrophil count (all
Conclusion
Platelet indices do not provide diagnostic value for PTE in the ED. Traditional demographic and hematological markers may aid risk stratification, but clinical integration remains essential. Prospective studies are needed to validate these findings and enhance diagnostic pathways.
Keywords
Introduction
Pulmonary thromboembolism (PTE) is a potentially fatal condition that represents a significant cause of morbidity and mortality among patients presenting to the emergency department (ED).1,2 Its diagnosis remains challenging due to a wide spectrum of clinical presentations, which are often non-specific and can mimic other acute cardiopulmonary disorders.1,3 Early and accurate identification of PTE is critical, as delayed recognition and treatment can rapidly lead to adverse outcomes and increased mortality.2,4
The initial diagnostic approach to suspected PTE commonly relies on clinical risk assessment tools, such as the Wells and Geneva scores, and laboratory markers, especially D-dimer testing.1,3 However, the low specificity of D-dimer, particularly in elderly and comorbid patients, often results in a high rate of unnecessary advanced imaging.1,3 Computed tomography (CT) pulmonary angiography is regarded as the gold standard for definitive diagnosis, but its application is limited by exposure to ionizing radiation, the risk of contrast induced nephropathy or allergic reactions, and variable accessibility across healthcare settings. 1 These limitations underscore the need for supplementary diagnostic tools that are rapid, safe, widely available, and cost effective. 5
In recent years, there has been growing interest in the diagnostic utility of routinely available hematological parameters, notably platelet indices such as mean platelet volume (MPV), platelet distribution width (PDW), and platelet to lymphocyte ratio (PLR).2,5 These parameters can be obtained easily and rapidly from a complete blood count and may reflect thrombosis or inflammatory activity associated with PTE. 5 While some studies have suggested that alterations in platelet indices are associated with PTE or its severity, results across the literature including investigations in other acute conditions such as COVID-19 remain inconsistent, and the clinical utility of these indices as diagnostic biomarkers is still debated.2,6,7
Despite these uncertainties, the pursuit of novel and accessible biomarkers for PTE diagnosis continues to be a priority, especially in the ED where timely decision-making is essential.1,3 Integrating laboratory markers with clinical assessment may enhance diagnostic accuracy, facilitate early risk stratification, and improve the management of patients with suspected PTE. 5 Therefore, further investigation into the role of platelet indices as adjuncts in the diagnostic pathway has the potential to provide valuable insights for clinical practice.
Methods
Study Design and Setting
This retrospective, single-center study was conducted in the ED of Duzce University Faculty of Medicine, a tertiary care center in Turkey. The study protocol was approved by the Non-Interventional Clinical Research Ethics Committee of Duzce University Faculty of Medicine (approval date and number: 21.04.2025-2025/122), and all procedures were performed in accordance with the Declaration of Helsinki and relevant institutional guidelines. The study period extended from April 1, 2022, to April 1, 2025.
Study Population
The study included adult patients who underwent pulmonary arterial phase chest CT angiography in the ED for any clinical indication during the specified period. Patients younger than 18 years of age, those admitted due to trauma, patients presenting in cardiac arrest, and individuals who died before the completion of diagnostic evaluation were not included in the analysis. For patients with multiple ED admissions during the study period, only data from the first admission were considered.
Data Collection and Variables
Data were retrospectively collected through review of electronic medical records and patient files. The variables recorded for each patient comprised age, sex, and a range of laboratory findings. Complete blood count parameters included white blood cell count (WBC), red blood cell count (RBC), hemoglobin (HGB), hematocrit (HCT), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), and red cell distribution width (RDW), as well as lymphocyte, monocyte, neutrophil, eosinophil, and basophil counts and percentages. Platelet indices were recorded, including platelet count (PLT), MPV, PDW, and plateletcrit (PCT). In addition, calculated ratios such as mean platelet volume-to-platelet count (MPV/PLT), mean platelet volume-to-plateletcrit (MPV/PCT), platelet distribution width-to-platelet count (PDW/PLT), and platelet distribution width-to-plateletcrit (PDW/PCT) were assessed. Coagulation parameters included prothrombin time (PT), activated partial thromboplastin time (aPTT), and international normalized ratio (INR), and selected biochemical measures included sodium (Na), potassium (K), calcium (Ca), creatine kinase (CK), and creatine kinase-MB (CK-MB). The diagnosis of PTE was made based on the identification of intraluminal filling defects in the pulmonary arteries on contrast-enhanced thorax CT angiography, as interpreted by an experienced radiologist.
Statistical Analysis
All statistical analyses were performed using SPSS version 23.0 (IBM Corp., Armonk, NY, USA). The distribution of continuous variables was assessed using the Kolmogorov-Smirnov test. As most variables did not follow a normal distribution, continuous data were presented as median and interquartile range (IQR), while categorical variables were expressed as frequencies and percentages. Comparisons between groups were performed using the Mann-Whitney U test for continuous variables and the chi-square test or Fisher's exact test for categorical variables, as appropriate. Variables with
Results
A total of 1198 patients were included in the study, with 126 diagnosed with PTE and 1072 identified as non-PTE. The proportion of female patients was significantly higher in the PTE group compared to the non-PTE group (57.1% vs 45.3%,
In the comparison of laboratory parameters, the PTE group exhibited lower median RBC [3.94 (3.44-4.47) versus 4.19 (3.67-4.69),
Comparison of Demographic and Hematological Parameters According to Pulmonary Thromboembolism (PTE) Status.

Categorical variables were compared using the Chi-square test; continuous variables were compared using the Mann-Whitney U test (because data were not normally distributed). Data are presented as median (25th-75th percentile) for continuous variables and as percentage (number) for categoricalvariables. A
Bold
The comparison of platelet indices between the PTE and non-PTE groups did not reveal any statistically significant differences for any parameter. The median PLT was similar in the PTE and non-PTE groups [257.00 (193.25-346.25) × 109/L versus 251.00 (191.00-318.00) × 109/L,
Comparison of Platelet Indices According to Pulmonary Thromboembolism (PTE) Status.

Continuous variables are presented as median (25th-75th percentile) and were compared between groups using the Mann-Whitney U test (since the data were not normally distributed). A
Among the coagulation parameters, aPTT was significantly shorter in the PTE group [28.40 (24.00-31.90) s] compared to the non-PTE group [29.70 (26.12-33.60) s], with a
Comparison of Coagulation, Electrolyte, and Muscle Enzyme Parameters According to the Presence of Pulmonary Thromboembolism (PTE).

Continuous variables are presented as median (25th-75th percentile) and were compared between groups using the Mann-Whitney U test (since the data were not normally distributed). A
Bold p values represents statistical significance.
In the multivariate logistic regression analysis, several variables were identified as significant predictors of PTE. Female sex was associated with lower odds of PTE (odds ratio [OR]: 0.580, 95% confidence interval [CI]: 0.378-0.892,
Multivariate Logistic Regression Analysis of Predictors for Pulmonary Thromboembolism (PTE).

β = regression coefficient; SE = standard error; OR = odds ratio; CI = confidence interval; RBC = red blood cell count; aPTT = activated partial thromboplastin time. A
The diagnostic performance of the multivariate model was assessed by ROC curve analysis. The area under the curve (AUC) was 0.651, indicating modest discriminative ability for predicting PTE. The ROC curve of the regression model is illustrated in Figure 1.
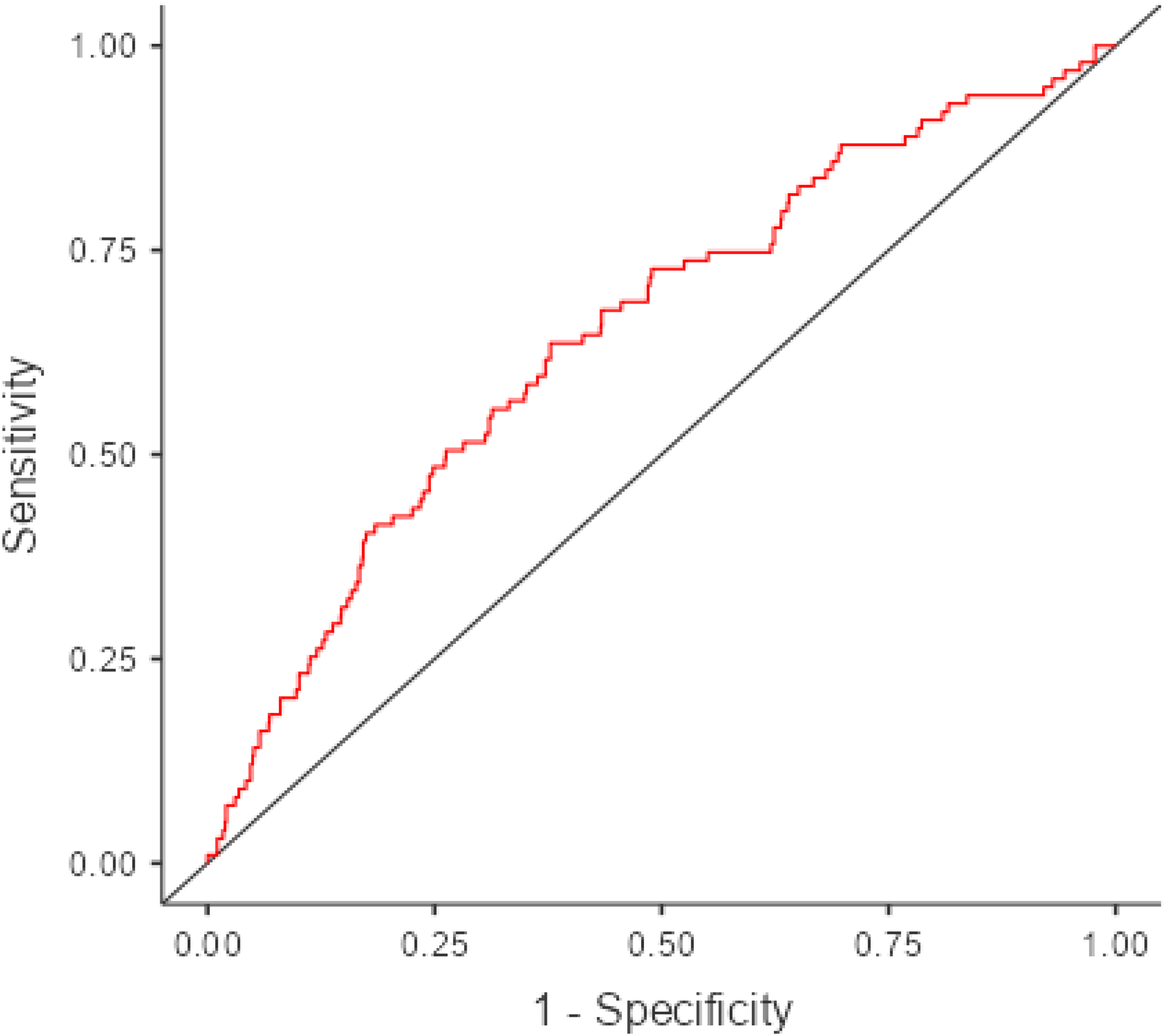
Receiver Operating Characteristic (ROC) Curve of the Multivariate Logistic Regression Model for Predicting Pulmonary Thromboembolism (PTE). The Area Under the Curve (AUC) was 0.651, Indicating Modest Discriminative Ability. The cut-off Value for Classification was Set to 0.5.
Discussion
The principal finding of this study is the lack of a significant association between platelet indices including PLT, MPV, PDW, PCT, and their calculated ratios and the diagnosis of PTE. Despite the growing interest in these readily available hematological markers, our results revealed no significant differences in any platelet index between patients with and without PTE. This observation aligns with the majority of recent studies, which have consistently questioned the diagnostic value of platelet indices for PTE.2,5–10 Although some reports have described possible associations or prognostic roles for certain indices such as MPV or PLR, the collective evidence indicates that these parameters lack sufficient sensitivity and specificity to serve as reliable standalone diagnostic markers in the ED setting.2,6,7 Thus, findings of this study reinforce the notion that while routine hematological indices may offer supplementary information, their utility in the diagnosis of PTE is limited. Notably, other commonly measured hematological parameters such as WBC, MCV, MCH, MCHC, RDW, monocyte, eosinophil, and basophil counts also showed no significant differences between PTE and non-PTE groups in this study.
A noteworthy secondary finding was the significantly higher proportion of female patients in the PTE group. While prior research has produced inconsistent results regarding the relationship between sex and PTE risk, several studies have reported a female predominance, particularly in older populations or those with additional risk factors.11,12 This finding may be partially explained by increased exposure to hormonal risk factors among women, including oral contraceptive use, hormone replacement therapy, and the prothrombotic effects associated with pregnancy and the postpartum period. 12 In addition, differences in healthcare-seeking behavior, sociocultural context, and comorbid conditions may influence the observed sex distribution. Interestingly, in contrast to our findings, a recent study by Alpaslan et al 13 evaluating patients diagnosed with PTE in the ED before and during the COVID-19 pandemic reported that more than 60% of cases were male. This discrepancy may reflect differences in study populations, the unique effects of the COVID-19 pandemic on healthcare utilization, or underlying regional and temporal factors. These conflicting results in the literature highlight the complexity of the relationship between sex and the incidence of PTE and suggest that multifactorial influences warrant further investigation.
Age was also found to be significantly higher in the PTE group compared to the non-PTE group, consistent with previous studies demonstrating an increased incidence of PTE with advancing age.3,14,15 Age-related physiological changes, a higher prevalence of comorbidities such as malignancy and heart failure, and increased periods of immobility all contribute to the elevated risk of thromboembolic events in older adults. The higher median age among PTE patients in our cohort likely reflects the cumulative effect of these risk factors. This finding underscores the importance of considering age-related risk when evaluating suspected PTE, especially in the ED setting where timely and accurate triage is crucial.
In addition to these primary findings, our study also identified lower RBC, HGB, and HCT, as well as higher neutrophil counts in the PTE group. These results may reflect underlying systemic inflammation or comorbid conditions frequently associated with acute thromboembolic events.4,16,17 Multivariate logistic regression analysis confirmed that lower RBC and higher neutrophil count were independent predictors of PTE, while traditional platelet indices were not retained as significant predictors in the model. The diagnostic performance of the multivariate model was assessed by ROC curve analysis. AUC was 0.651, indicating modest discriminative ability. However, from a clinical standpoint, an AUC of 0.651 indicates limited diagnostic accuracy, thus the current regression model alone is inadequate to reliably guide clinical decision-making for suspected PTE cases. Clinicians should integrate these findings cautiously with clinical judgment, and further prospective studies incorporating additional clinical variables are warranted to achieve improved diagnostic performance.
This study also found significant differences in certain coagulation and biochemical parameters, specifically a shorter aPTT and lower Ca levels in the PTE group. The finding of a shorter aPTT in PTE patients may be related to activation of the coagulation cascade and increased thrombin generation, which are hallmarks of acute thromboembolic events.4,16 Previous studies have reported variable findings regarding aPTT in PTE, with some noting shortened or normal values, possibly reflecting a hypercoagulable state at the time of diagnosis. Similarly, lower serum Ca levels in PTE patients have been described in several reports, and hypocalcemia has been linked to worse outcomes in critically ill patients. 17 The mechanisms underlying this association remain unclear but may involve altered calcium homeostasis during acute illness and the role of calcium in platelet activation and coagulation pathways. In contrast, there were no significant differences between the groups in terms of PT, INR, Na, K, CK, or CK-MB. These results suggest that while some coagulation and biochemical parameters may differ in the context of PTE, their clinical utility as diagnostic markers remains limited and should be interpreted with caution.
Although several recent studies have reported that ratios such as neutrophil-to-lymphocyte ratio (NLR) and PLR may have diagnostic or prognostic value in PTE,2,15,17 these indices were not specifically assessed in this study and thus represent a potential area for future research.
Several limitations of this study should be acknowledged. First, the retrospective and single-center design may introduce selection bias and limit the generalizability of our findings. Patient records and laboratory results were reviewed retrospectively, which could result in incomplete or missing data and preclude the identification of all potential confounding factors. Second, we relied on routinely collected data, and certain clinical variables such as the presence of active malignancy, recent surgery, immobilization history, use of oral contraceptives or hormone replacement therapy, and detailed comorbidity profiles were not systematically documented for all patients. This may have limited our ability to fully adjust for known risk factors for PTE.
Third, the diagnostic criteria for PTE were based exclusively on radiological findings from contrast enhanced chest CT angiography. While this modality is the gold standard for PTE diagnosis, interobserver variability in radiology reports and technical limitations of imaging may influence diagnostic accuracy. Fourth, the study population comprised only patients who underwent CT angiography in the ED, potentially excluding individuals with milder symptoms, contraindications to contrast media, or those managed without imaging, thereby introducing spectrum bias.
Fifth, we analyzed laboratory parameters at a single time point upon ED admission; thus, we were unable to evaluate the impact of dynamic changes in hematological or biochemical markers over time or in response to treatment. In addition, as a result of the study's observational design, we could not assess causality between the measured parameters and the occurrence of PTE. Sixth, although our sample size was relatively large, subgroup analyses such as those based on age or sex may have been underpowered to detect significant associations.
Finally, platelet indices and other laboratory markers can be affected by preanalytical variables, such as sample handling, timing of blood collection, and instrument calibration, which were not standardized in our retrospective review. Taken together, these limitations underscore the need for prospective, multicenter studies with comprehensive data collection and standardized protocols to validate and expand upon our findings.
Conclusion
In this retrospective single center study, we found that routinely measured platelet indices including PLT, MPV, PDW, and PCT had no diagnostic value for PTE in ED setting. However, both older age and female sex were more common among patients with PTE, and regression analysis identified lower RBC and higher neutrophil count as independent predictors of PTE. Our findings suggest that platelet indices should not be used as standalone diagnostic markers for PTE, whereas demographic and other hematological parameters may be more relevant in risk assessment. Further prospective studies are needed to confirm these results and to clarify the clinical utility of these parameters in patients with suspected PTE.
Footnotes
Acknowledgements
None.
Ethical Approval
This study was initiated in the emergency department of a tertiary hospital in Düzce, Turkey after ethics committee approval. Ethical approval for the study was obtained from the Düzce University Non-Interventional Clinical Research Ethics Committee (approval date and number: 21.04.2025-2025/122).
Consent to Participate
The need for signed informed consent was waived due to the retrospective design of the study.
Consent for Publication
Not applicable.
Authors Contributions
Concept – HG,EŞ; Supervision – HG,EŞ; Materials – HG,EŞ; Data Collection and Processing – HG,EŞ; Analysis and Interpretation – HG,EŞ; Writing – HG,EŞ.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
