Abstract
Time in therapeutic range (TTR) with intravenous unfractionated heparin (UFH) and its association with outcomes is poorly explored. UFH can be monitored with anti-Xa or aPTT, however, evidence indicates anti-Xa monitoring of UFH improves time to therapeutic anticoagulation. The objective of this study was to determine if TTR correlates with thrombotic and safety outcomes for patients receiving UFH, and if differences exist between these methods of monitoring. This retrospective, single center, cohort study stratified patient admissions based on the laboratory monitoring technique used (aPTT vs anti-Xa). The primary outcome was the presence of a new thrombotic event (TE) during the index visit. The secondary outcome was major bleeding during the index visit. Clinical outcomes were each assessed for their association with TTR. TTR was defined as the percentage of time in therapeutic range for each patient, as determined via the linear interpolation method. Of 4773 patient admissions (aPTT, n = 2,939, anti-Xa, n = 1834), TE occurred in 6.5% and 4.6% of visits in the aPTT and the anti-Xa cohort, respectively (p = .006). Bleeding events occurred in 33.3% and 31.6% of visits in the aPTT and the anti-Xa cohort, respectively (p = .204). TTR was 41.6% [IQR 23.3-57.7%] in the aPTT cohort compared to 56.1% [IQR 33.5-75.2%] in the anti-Xa cohort (p < .001). The monitoring of IV UFH with anti-Xa was associated with significantly fewer TEs and greater overall TTR when compared to those monitored with aPTT.
Introduction
Therapeutic drug monitoring using activated partial thromboplastin time (aPTT) or anti-factor Xa (anti-Xa) are commonly utilized for patients receiving unfractionated heparin (UFH) continuous intravenous (IV) infusions.1,2 Anti-Xa concentrations directly measures the activity of heparin on factor Xa, whereas aPTT is a functional assay that reflects the intrinsic and common pathways of the coagulation cascade. 3 The anti-Xa assay is affected by few pre-analytical variables and has increasingly replaced aPTT-based monitoring as the preferred method for monitoring UFH in clinical practice.4–7 These include more preanalytical and biologic variables for aPTT, including diurnal variations, factor XII deficiency, increased fibrinogen, factor VIII, impaired renal function etc Fewer variables impact anti-Xa monitoring, such as recent Xa inhibitor use, hypertriglyceridemia, hyperbilirubinemia etc Both are impacted by laboratory markers such as choice of reagent, delayed sample analysis, degree of centrifugation and underfilled sample tubes.4–7 Previous literature has compared the rates of venous thromboembolic events (VTE) and bleeding and found no difference between anti-Xa and aPTT-based monitoring in clinical outcomes. 8 Time in therapeutic range (TTR) has been used as an indicator of quality anticoagulation control with warfarin.9,10 However, there is no evidence evaluating percentage TTR-related outcomes (thrombosis and bleeding) for UFH. 1 Therefore, the purpose of this study was to evaluate the association of TTR with clinical outcomes for patients administered IV UFH monitored through aPTT and anti-Xa concentrations. We hypothesize that anti-Xa monitoring will lead to a greater TTR and could reflect in improved outcomes.
Methods
Design and Setting
This study was a retrospective, single center, cohort study for adult patients who were treated with IV UFH. The collection of de-identified data from the medical record was approved by the Institutional Review Board at the University of Pittsburgh. Patient index admissions were included for those who were 18 years or older and received IV UFH for greater than one day at UPMC Presbyterian-Shadyside Hospital with subsequent aPTT monitoring between January 1 to December 31, 2014, or anti-Xa monitoring from January 1 to December 31, 2016. An early time period for the anti-Xa group was chosen to reflect similar equipment, nomograms and practice styles with the aPTT group. The activated partial thromboplastin time and the anti-Xa measurements in our institution were each based on methods developed by Diagnostica Stago, Inc. 11 Our institution transitioned from aPTT to anti-Xa monitoring in 2015, therefore, we executed a pre/post study design to minimize confounding between these methods of laboratory monitoring. The employed nomograms are available in supplemental eTable S1 and S2. Choice of which nomogram to follow and any patient specific adjustments to the dosing nomogram were per clinician discretion. Patient admissions were excluded if their home medication list contained a direct oral anticoagulant (except dabigatran) to avoid heparin anti-Xa concentration interference.12,13 Additional exclusions were lack of availability of home medication information, concurrent home use of enoxaparin, dalteparin or fondaparinux, a diagnosis of heparin-induced thrombocytopenia at their visit, or repeat visits in the same pre/post period. Additional data collected included: demographic information at the index visit, ICD-9 and ICD-10 diagnosis and procedure codes, inpatient heparin charges, home medication lists, anti-Xa and aPTT laboratory results, hemoglobin levels, blood transfusion, and imaging reports (chest computed tomography [CT] scans, chest CT angiograms, venous duplex ultrasonography, and ventilation-perfusion scans). Patient admissions were stratified based on the laboratory monitoring technique used (anti-Xa vs aPTT).
Outcomes
The primary outcome was the presence of a new venous thrombotic event during the index visit per documented imaging reports, from the time of UFH initiation until its discontinuation. The secondary outcome was the incidence of major bleeding during the index visit. A major bleeding event was defined as any patient admission requiring a transfusion with ≥ 2 units of blood, or a ≥ 2 g/dL decrease in hemoglobin from the baseline value. 12 TTR was defined as the percentage of time in therapeutic range for each patient, as determined via the linear interpolation method. 9 Goal aPTT therapeutic range was defined as between 68 to 106 s as determined by our institutional laboratory, while goal anti-Xa therapeutic range was between 0.3-0.7 units/mL. 1 Given bleeding or thrombosis can occur within or outside the proposed therapeutic ranges, we also investigated the percentage of time and the incidence of these outcomes, above and below the therapeutic range.
Statistical Analysis
A modified Rosendaal (linear interpolation) equation was used to calculate percentage TTR. 5 This differs from laboratory values in therapeutic range, which uses a percentage of instances in which the laboratory value collected was within a specific range. TTR uses linear interpolation to map out trends between two subsequent laboratory values, allowing a more accurate projection in the rise and fall of laboratory values between two known data points. When applied to a time range, this process allows the visualization of the total percentage of time a laboratory value would stay within (or stray above or below) a desired range. TTR is expressed as a percentage of time within the given therapeutic range and is provided as a median [interquartile range] or mean [standard deviation]. Mann Whitney-U tests and the Pearson chi-square tests were used for statistical analysis of categorical variables. The Kolmogorov-Smirnov and Shapiro-Wilk tests were used to determine the normal distribution of our data. Nonparametric, independent-samples median tests were used for continuous variables. Log regressions were used to correlate percentage TTR with the odds of having a TE or bleeding event.
Results
Baseline Characteristics
A total of 4773 patient visits were included in this retrospective study, with 2939 patient visits in the aPTT group and 1834 patient visits in the anti-Xa group, which may reflect institutional practices such as low-molecular-weight-heparin (LMWH) use, which is not routinely monitored. No differences in baseline demographic characteristics were observed between cohorts as shown in Table 1. Regarding comorbidities, no differences were observed except for myocardial infarction, atrial fibrillation and thrombophilia / coagulopathy which were more common in the anti-Xa group. Home medications of interest were similar between groups. (Table 1)
Baseline Characteristics.

Continuous data shown as median (IQR) and categorical data shown as n (%).
Time in Therapeutic Range
Among the aPTT cohort, the median TTR was 41.6% [IQR 23.3-57.7%] compared to 56.1% [IQR 33.5-75.2%] in the anti-Xa cohort (p < .001). For the median time above therapeutic range (TaTR), the aPTT cohort had a median TaTR of 4.1% [IQR 0-14.9%] compared to 0% [IQR 0-8.2%] in the anti-Xa cohort (p < .001). For the median time below therapeutic range (TbTR), the aPTT cohort had a median TbTR of 45.4% [IQR 27.4-68.1%] compared to 31.2% [IQR 12.1-58.3%] in the anti-Xa cohort (p < .001).
Thrombotic Outcomes
VTEs occurred in 190 (6.5%) of visits in the aPTT cohort and 84 (4.6%) of visits in the anti-Xa cohort (p = .006). Patients in the aPTT cohort were on UFH infusions for a median of 52 h [IQR 22.1-206.5 h], compared to patients in the anti-Xa cohort that were on UFH infusions for a median of 140.6 h [IQR 66.6-268.8 h] (p < .001) prior to the VTE.
Bleeding Outcomes
A major bleeding event defined by a hemoglobin drop of ≥ 2 g/dL occurred in 940 (32%) patient visits in the aPTT cohort, compared to 557 (30.3%) of patient visits in the anti-Xa cohort. Similarly, major bleeding events defined by a transfusion requirement of ≥ 2 units of blood occurred in 272 (9.3%) of patient visits in the aPTT cohort, compared to 173 (9.4%) of patient visits in the anti-Xa cohort. In the aPTT cohort, 232 (7.9%) of patient visits met both bleed criteria (hemoglobin drop and need for blood transfusion), whereas 151 (8.2%) of patient visits observed in the anti-Xa cohort met both bleed criteria. Overall, 980 (33.3%) of patient visits in the aPTT cohort had a major bleeding event compared to 579 (31.6%) visits in the anti-Xa cohort (p = .204).
Univariable Regressions
There was no statistically significant difference in the incidence of either thrombotic or major bleeding events when compared with TTR in either group (Table 2). To estimate cut-offs for clinically appropriate TTR, percentage TTR was divided into quartiles (TTR < 25%, 25%–50%, 50%–75%, ≥ 75%), with TTR < 25% as the comparator for univariable logistic regressions for the outcomes of interest (Table 3). There were no statistically significant differences between TTR quartiles and a thrombotic or major bleeding event in the aPTT cohort. In the anti-Xa cohort, TTR ≥ 75% significantly reduced the incidence of thrombotic and major bleeding events. Likewise, lesser time in therapeutic range (TTR < 75%) had a significantly higher incidence of a major bleeding event (Table 3).
Univariable Logistic Regressions for Thrombotic and Bleeding Outcomes for Heparin Anti-Xa and aPTT Time in Therapeutic Range.

Univariable Logistic Regressions for Thrombotic and Bleeding Outcomes for Heparin Anti-Xa and aPTT Quartiles of Time in Therapeutic Range.
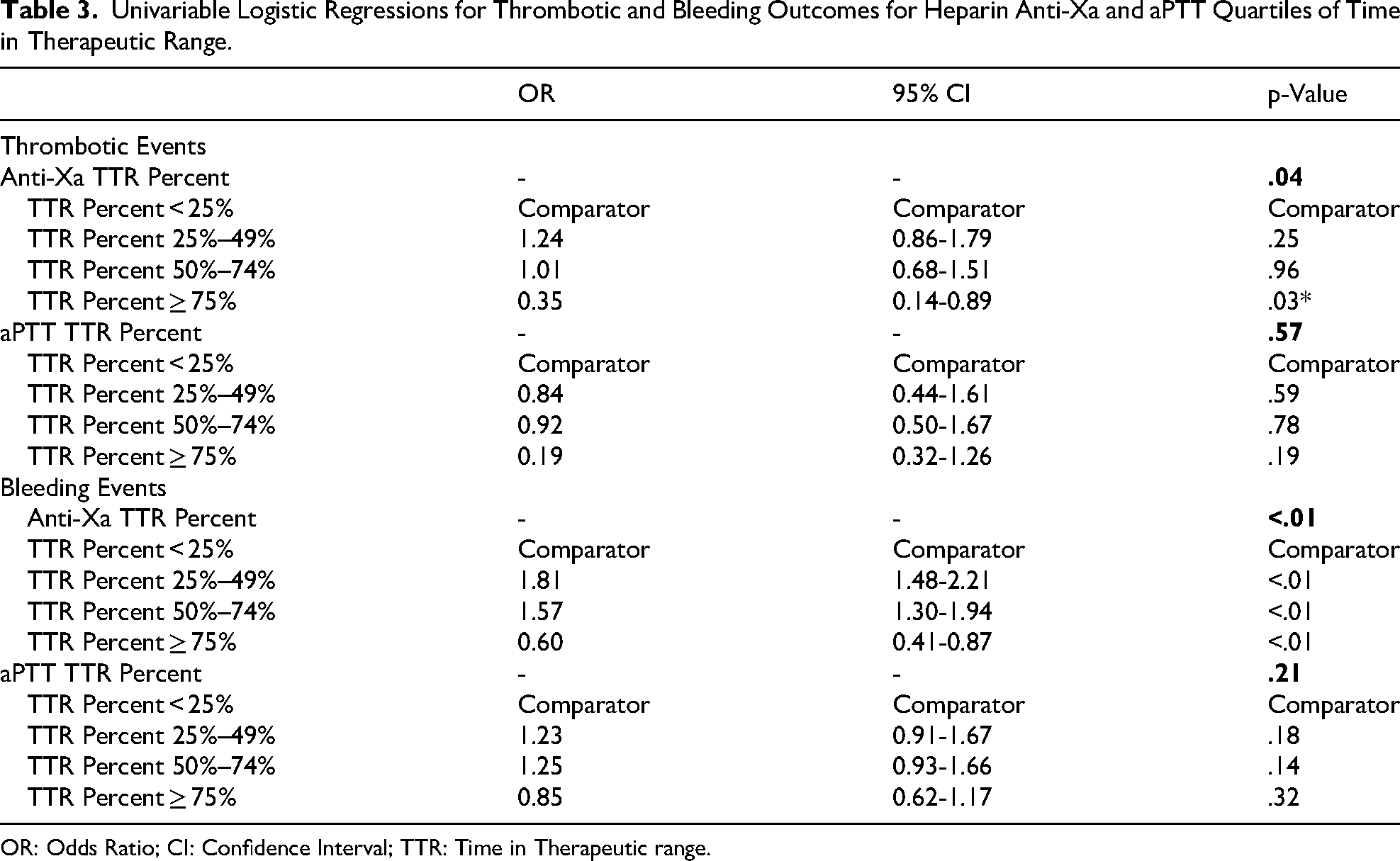
OR: Odds Ratio; CI: Confidence Interval; TTR: Time in Therapeutic range.
Discussion
This study was the first to report TTR for IV UFH and correlated clinical outcomes in a cohort of nearly 5000 patients. We observed a TTR of 42% and 56% for aPTT and anti-Xa monitoring, respectively (p < .001). All patient visits in our study received IV UFH for at least 24 h and had monitoring in accordance with institutional nomograms. The time above and below therapeutic range were significantly more likely with aPTT monitoring. TbTR was seen in approximately 30% to 45% of patient visits overall, whereas TaTR was relatively uncommon (0% to 4%), which may be attributed to cautious clinical dose adjustments or patient specific factors such as anemia, surgeries and bleed risk. We also observed significantly higher rates of VTEs with aPTT compared to anti-Xa monitoring, which is consistent with the TbTR observations. Given the median time to TEs were significantly earlier in the aPTT group, it is possible that this reflects the variability in achieving and maintaining initial therapeutic goals when using aPTT.14–16 However, no correlation was found in the univariable regression model comparing percentage TTR and VTEs. In contrast, there was no difference in bleeding events between groups which aligned with the findings that few patients overall had TaTR. When evaluating the most appropriate TTR regarding our effectiveness and safety outcomes, TTR ≥ 75% demonstrated the most favorable target but only in the anti-Xa group. TTR < 75% demonstrated greater bleeding risk in the anti-Xa group, despite very low TaTR in this group, which likely represents variable interpatient bleed risk (such as higher coagulopathy) within the larger safety range of 0.3-0.7 for anti-Xa and is perhaps better represented using individual patient anti-Xa goal ranges and nomograms – which we could not collect retrospectively for all patients.
Despite UFH being the most commonly used parenteral anticoagulant, its TTR has not been well-documented. 15 In contrast, multiple studies have reported the TTR with warfarin using the Rosendaal method and associated this with clinical outcomes.9,17–19 One study of UFH with aPTT monitoring determined TTR using a modification of the Rosendaal method. Their results showed a TTR rate of approximately 44% which is similar to our finding of 42%. However, correlation with clinical outcomes was not conducted. 20 Our study adds to this literature by documenting TTR and correlating findings with anti-Xa and aPTT target ranges and outcomes. Our study also builds upon previous reports which compared methods of monitoring using aPTT and anti-Xa concentrations. Several studies have shown that anti-Xa monitoring was associated with faster time to therapeutic anticoagulation and fewer dose adjustments, despite no apparent difference in clinical outcomes compared to aPTT. These studies did not explore the relationships between TTR and outcomes.8,21–23 Our study provides the largest evaluation of TTR for UFH-treated patients to date and showed a significant difference in thrombotic outcomes with correlation to TTR.
The limitations of our study included the retrospective, single center, design and the potential for missing or incomplete data that were not documented in the electronic health record. While our study data were collected in 2014 and 2016, these years reflect appropriate timings given institutional transition from aPTT to anti-Xa and overall heparin nomograms have not changed since that time. The diagnosis of venous thrombosis was at the discretion of the interpreter of venous duplex ultrasound or CT scan. Because new venous thrombosis was our primary outcome of interest, we did not capture arterial thrombotic events. Thrombotic and bleeding outcomes were collected at any time during or after discontinuation of IV UFH during the index hospital visit. At our institution, medication charges are formed upon the administration of an agent, so UFH use was determined based on charge data and so could not account for temporary interruptions in therapy. Additionally, due to limitations in accurately pulling medication indications retrospectively, we did not evaluate UFH indication and nomogram selection or adherence. The decision to use a certain nomogram was based on provider discretion, though it is generally aligned with treatment indication and clinical assessment of thromboembolic and bleed risks. Therefore, the potential for unmeasured clinical confounders remains. For anti-Xa monitoring, we considered 0.3 to 0.7 IU/mL to be therapeutic as that is consistent with national recommendations for VTE treatment efficacy.1,2 This range also includes the lower and upper ends across our local nomograms and was a conservative way of reporting TTR, TaTR and TbTR. For aPTT, we used the corresponding range to anti-Xa as determined by our institution's laboratory which was 68-106 s. We did not modify analysis for UFH indication or comorbidities since the hospital nomograms offered dose adjustments for UFH to achieve the therapeutic goal. Additionally, while hospital ICD codes for VTEs are more likely to represent an acute presentation as opposed to a secondary diagnosis, the low positive predictive values of 17%–65% greatly reduces the utility of relying on these codes as the primary indications of UFH therapy in this cohort.24,25
The strengths of our study included the large cohort and the ability to independently assess both aPTT and anti-Xa monitoring for TTR and outcomes. We had a unique opportunity to assess each method of monitoring as our institution converted from aPTT to anti-Xa in 2016. 22 The use of the modified Rosendaal linear interpolation method allowed us to project trends in aPTT and anti-Xa values between 2 data points regardless of the frequency of labs collected, which is a major advantage compared to calculating percentage of values in therapeutic range since it can adjust to the frequency to lab collections per hospital protocol (eg every 6 vs 12 vs 24 h). 5 Thrombotic outcomes were assessed through annotation of imaging reports (chest computed tomography [CT] scans, chest CT angiograms, venous duplex ultrasonography, and ventilation-perfusion scans), whereas bleeding events were harmonized using the International Society on Thrombosis and Haemostasis (ISTH) definition that included hemoglobin drop and transfusion requirement. 26 We captured a variety of important demographic, clinical, and medication variables known to potentially affect outcomes and observed no clinically relevant differences in baseline characteristics between groups. We purposely excluded patients who received a selective factor Xa inhibitor prior to admission to mitigate the potential for interference with anti-Xa monitoring.12,13
Conclusion
Our results demonstrated that anti-Xa monitoring led to significantly greater TTR. The TTR had no correlation with outcomes when using aPTT, which had a higher incidence of thrombosis and more variable time above and below therapeutic range. Achieving a TTR ≥ 75% with anti-Xa monitoring led to significant reductions in thrombosis and major bleeding.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251371003 - Supplemental material for Outcomes Associated with Time in Therapeutic Range for Patients Receiving Intravenous Unfractionated Heparin
Supplemental material, sj-docx-1-cat-10.1177_10760296251371003 for Outcomes Associated with Time in Therapeutic Range for Patients Receiving Intravenous Unfractionated Heparin by Mujtaba Mahmud, Maia Klimatcheva, Angela Tsang, Andrew La, Victoria Miklus, Jack Becker, Benjamin Herrmann, Amanda Cremeans, Neal J Benedict, Carlo J Iasella, Roy E Smith and James C Coons in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgements
The authors would like to thank Melissa Saul, MS for her expertise and contributions to data de-identification and management.
Ethics Approval Statement
All patient information was de-identified prior to conducting the study. The collection of de-identified data from the medical record was approved by the Institutional Review Board at the University of Pittsburgh.
Patient Consent Statement
This study was approved as exempt (no human subjects research) by the University of Pittsburgh Institutional Review Board. Therefore, patient consent was not necessary.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Supplemental materials and additional data for the study are available from the corresponding author upon request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
