Abstract
Background
This systematic review aimed to evaluate the comparative effectiveness and safety of direct oral anticoagulants (DOACs) compared to traditional anticoagulation (vitamin K antagonists or low-molecular-weight heparins) in cirrhotic patients with portal vein thrombosis (PVT).
Methods
We conducted a literature search in PubMed and Embase databases up to May 2024. Studies were selected according to the PICOS criteria, focusing on cirrhotic patients with PVT treated with DOACs (dabigatran, rivaroxaban, apixaban, or edoxaban) compared to traditional anticoagulation.
Results
Our systematic review included four observational studies conducted in Japan, China, and the United States, involving a total of 223 patients with cirrhosis and PVT. The included studies collectively suggested that anticoagulation therapy, including DOACs, was associated with improved recanalization rates and reduced progression of PVT in patients with liver cirrhosis, without a significant increase in bleeding complications. Specifically, edoxaban demonstrated superior effectiveness in reducing PVT volume compared to traditional anticoagulation, while maintaining a favorable safety profile.
Conclusion
DOACs may provide a promising therapeutic option for PVT in cirrhotic patients. Further research is needed to confirm the potential benefits and risks of DOACs in this population.
Keywords
Introduction
Splanchnic vein thrombosis (SVT) is a specific blood clotting disorder affecting the splanchnic venous system, which includes key vessels such as the portal, mesenteric, and splenic veins. Historically considered rare, the incidence of SVT has been increased, a trend attributed to improved diagnostic techniques. SVT is frequently associated with conditions such as liver cirrhosis, malignancies, post-surgical states, thrombophilia, and myeloproliferative disorders. The heterogeneity of SVT etiologies complicates the selection of the most effective anticoagulant therapy. Among these, portal vein thrombosis (PVT) in cirrhotic patients is relatively common.
Managing PVT involves the use of anticoagulants that are tailored to each patient's clinical context. Potential options include vitamin K antagonists (VKAs), low-molecular-weight heparins (LMWHs) and direct oral anticoagulants (DOACs).1,2 Traditional VKAs such as warfarin, while effective, have certain limitations compared to DOACs, such as the need for dosage adjustments, susceptibility to drug-drug interactions, and dietary restrictions. Previous studies have shown that DOACs provide superior net clinical benefits compared to VKAs, including a reduced risk of stroke, lower bleeding rates, and improved patient compliance. Therefore, DOACs have become the preferred treatment for atrial fibrillation (AF)-related thromboembolic events3,4 and venous thromboembolism (VTE). 5 Unlike AF or VTE,6,7 for which DOACs are supported by large-scale randomized clinical trials (RCTs), no RCTs exist for PVT in cirrhosis. However, emerging guidelines offer a framework for the use of anticoagulants in this challenging population. 8
Previous systematic reviews and meta-analyses have sought to evaluate the effects of DOACs compared to traditional anticoagulation (VKAs or LMWHs) in patients with PVT.9–11 However, the reliability of these studies was compromised by the inclusion of a retracted RCT 12 and the variability in the studied population. 9 In this updated review, we excluded the retracted RCT 12 and included more recent publications,13,14 focusing on assessing the comparative effectiveness and safety of DOACs compared to traditional anticoagulation exclusively in cirrhotic patients with PVT.
Methods
Eligibility Criteria
Our inclusion criteria were based on the PICOS framework: (1) Population: cirrhotic patients diagnosed with PVT. (2) Intervention: treatment with DOACs, specifically dabigatran, rivaroxaban, apixaban, or edoxaban. (3) Comparison: traditional anticoagulation, including VKAs or LMWHs. (4) Outcomes: Effectiveness outcomes included complete or partial recanalization of affected blood vessels, PVT extension, and all-cause mortality, while safety outcomes included bleeding events such as major bleeding, any bleeding, intracranial hemorrhage, and gastrointestinal bleeding. (5) Study design: RCTs and observational studies. Single-arm studies focusing on DOACs without providing comparative analysis to VKAs were excluded. In cases of duplicate studies, we prioritized those with longer follow-up periods or larger sample sizes.
Literature Search
The literature search was conducted in PubMed and Embase databases until May 2024, utilizing keywords as follows: (i) “non-vitamin K antagonist oral anticoagulants” OR “direct oral anticoagulants” OR “dabigatran” OR “rivaroxaban” OR “apixaban” OR “edoxaban”; (ii) “vitamin K antagonists” OR “warfarin” OR “low-molecular-weight heparins”; (iii) “splanchnic vein thrombosis” OR “portal vein thrombosis”; (iv) “cirrhosis” OR “cirrhotic”. Additionally, a manual review of references from prior reviews9–11 was conducted to identify any overlooked studies.
Study Selection and Data Collection
After the literature search, two independent reviewers screened the titles and abstracts of identified studies, applying our exclusion criteria to exclude irrelevant studies such as animal experiments, case reports, reviews, meta-analyses, editorials, and meeting abstracts. The full-text screening phase followed to ascertain which studies met our criteria. A third reviewer was engaged in cases of any disagreements. For each study, we extracted the following data: authors’ names, publication years, sample size, age, sex, study design, data sources, enrollment period, assessment methods, DOAC and traditional anticoagulation types used, assessed outcomes, and follow-up duration.
Quality Assessment
The Newcastle-Ottawa Scale (NOS) was used to evaluate the quality of observational studies, focusing on the selection of study groups, comparability in design or analysis, and ascertainment of exposure or outcomes. Studies with an NOS score ≥6 were considered to be of moderate to high quality.15,16
Systematic Review
This study adhered to the guidelines outlined in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement. Due to the heterogeneity of study populations and endpoints, along with the limited number of included studies, a meta-analysis was not performed to avoid misleading results. The narrative synthesis allows a more contextual interpretation of the potential clinical benefits of DOACs in the treatment of PVT in cirrhotic patients.
Results
Study Selection
The flow diagram in Figure 1 outlines the process for the selection of studies in our systematic review. Our search strategy initially identified 174 studies. After screening the titles and abstracts, we excluded 159 irrelevant studies. The remaining 15 studies underwent full-text evaluation for potential inclusion. Among these, 11 studies were excluded for the following reasons: 1) they were single-arm studies, which did not offer comparative data (n = 2),17,18 2) the reference anticoagulation therapy was not VKAs or LMWHs (n = 2),19,20 3) the study did not provide separate analysis for PVT patients or the cirrhotic population (n = 5),21–26 and 4) the retracted RCT (n = 1). 12 Finally, 4 observational studies13,14,27,28 met our inclusion criteria and were incorporated into our review.
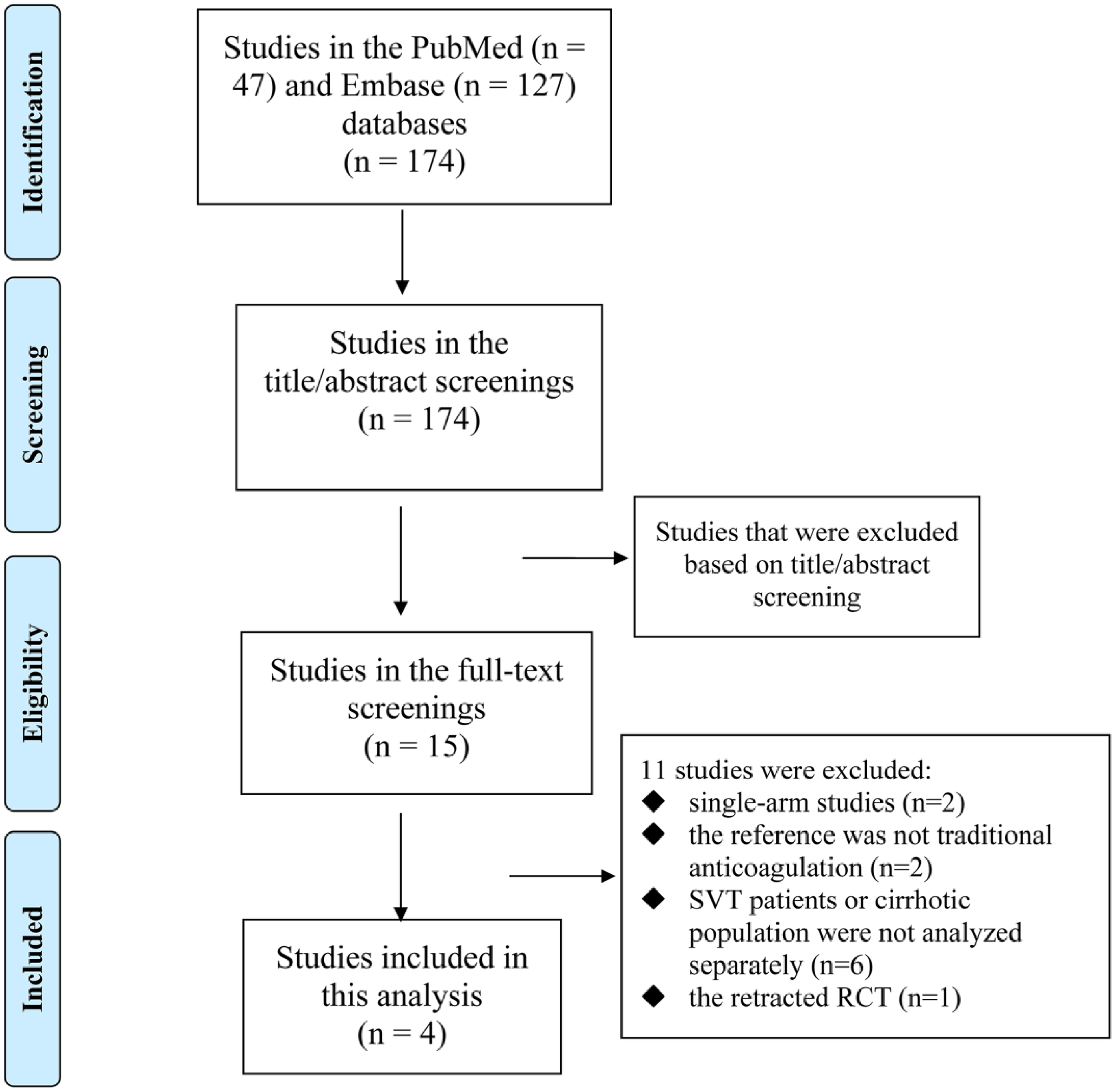
The PRISMA flow diagram of study selection in our systematic review.
Table 1 summarizes the baseline characteristics of studies included in our review. The studies were conducted in Japan,13,28 China, 14 and the United States, 27 with sample sizes ranging from 27 to 86 patients and follow-up durations varying from 6 months to 5 years. The quality of the studies was assessed using the NOS, with scores ranging from 6 to 7. All included studies scored 6 or above, indicating moderate to high quality.
Baseline Characteristics of Included Studies.

DOACs, direct oral anticoagulants; LMWH, low-molecular-weight heparin; VKA, vitamin K antagonist; PVT, portal vein thrombosis; CDFI, color Doppler flow imaging; US, Ultrasound; CT, computed tomography; MRI, magnetic resonance imaging; NOS = Newcastle-Ottawa Scale.
Baseline Characteristics Within the Included Studies
The included studies reported both differences and similarities in the baseline characteristics of the patient populations. Nagaoki et al found no significant differences in the clinical characteristics between the edoxaban and warfarin groups. Similarly, Naymagon et al found no differences in age, sex, etiology of cirrhosis, Child-Pugh Class, MELD score, presence of varices, or baseline laboratory variables between the anticoagulated and non-anticoagulated groups. In contrast, Tadokoro et al did not explicitly comment on baseline differences between treatment groups. However, Zhang et al found differences in baseline characteristics, such as history of splenectomy and platelet count, that were significant between the anticoagulant and non-anticoagulant groups. These differences may affect the interpretation of study results, as they could introduce confounding factors influencing treatment outcomes. Despite these variations, the overall similarity in patient demographics and disease severity across studies provides a consistent basis for comparing the effects of various anticoagulation strategies. Furthermore, the methodological rigor in accounting for these differences through statistical adjustments helps ensure that observed outcomes are more likely due to treatment interventions rather than pre-existing patient characteristics.
DOACs in Cirrhotic Patients with PVT
The study by Nagaoki et al 28 from Japan investigated the effectiveness and safety of edoxaban compared to VKAs for the treatment of PVT in patients with liver cirrhosis. This retrospective cohort study included 50 cirrhotic patients with PVT who were initially treated with danaparoid sodium for 2 weeks and then switched to either edoxaban (n = 20) or VKAs (n = 30) for up to 6 months. The primary effectiveness endpoint was the change in PVT volume measured by dynamic CT scans at baseline and at 2 weeks, 1 month, 3 months, and 6 months. The study found that edoxaban treatment significantly reduced PVT volume from 1.42 cm³ at 2 weeks to 0.42 cm³ at 6 months, preventing exacerbation of PVT (P = 0.016). In contrast, VKAs treatment resulted in an increase in PVT volume from 1.73 cm³ at 2 weeks to 2.85 cm³ at 6 months, even when the International Normalized Ratio (INR) was controlled in 57% of patients. Multivariate regression analysis identified edoxaban therapy as the only significant independent determinant of PVT reduction at 6 months. In terms of safety, there were no significant differences in adverse events between the two groups ( Supplemental Table 1 ). Thus, edoxaban following danaparoid sodium was an effective anticoagulant and could be considered a treatment option for PVT in cirrhotic patients.
The study by Tadokoro et al 13 from Japan retrospectively assessed the outcomes of edoxaban or VKAs among 61 patients with PVT from 2012 to 2021. The median overall survival time was 4.2 years, with a 1-year survival rate of 70.7% and a 5-year survival rate of 47.9%. The primary effectiveness endpoint was the overall response rate for thrombolysis, which was significantly higher in the edoxaban group (76.7%) than in the VKA group (29.4%). Edoxaban also demonstrated long-term improvement of PVT, while VKAs were only temporarily effective. In terms of safety, there were no deaths due to adverse events associated with either edoxaban or VKAs. Tadokoro et al highlighted that edoxaban, as a single agent, could achieve long-term recanalization without compromising hepatic reserves and was easy to initiate, even in an outpatient setting.
Naymagon et al 27 conducted a retrospective analysis of cirrhotic patients with PVT comparing outcomes in those who received anticoagulation treatment (n = 86) to those who did not (n = 128). This study included 214 patients from the United States (January 2000 to February 2019), with a median follow-up of 27 months. The main findings indicated that anticoagulation was associated with a significantly higher rate of complete radiographic resolution of PVT (48% vs 27%, p = 0.0007). This study also included a comparative analysis of DOACs (dabigatran, apixaban, rivaroxaban) versus VKAs and LMWHs. Among the 86 patients who received anticoagulation, 18 were treated with DOACs. Naymagon et al found that patients treated with DOACs had a complete resolution rate of 55.6% and a major bleeding rate of 5.6%. These outcomes were favorable when compared to those treated with VKAs or LMWHs. Specifically, for LMWHs, the complete resolution rate was 38.1% with a major bleeding rate of 21.4%, while for VKAs, the complete resolution rate was 57.7% with a major bleeding rate of 19.2%. Although this study could not conduct a rigorous statistical comparison due to the limited number of patients in the DOAC group, the results suggest that DOACs may offer a safety and effectiveness profile at least as favorable as traditional anticoagulants, with potentially better outcomes regarding PVT extension and major bleeding events.
In the retrospective cohort study by Zhang et al 14 from China, the effectiveness and safety of anticoagulant therapy in patients with cirrhotic PVT were evaluated. Zhang et al included 77 patients, with 27 in the anticoagulant group and 50 in the non-anticoagulant group. The median follow-up time was 26 months. The primary effectiveness endpoint was the PVT recanalization rate, which was significantly higher in the anticoagulant group (44.4%) than in the non-anticoagulant group (20%), with a log-rank P-value of 0.016. Secondary outcomes included bleeding rate, liver function, and mortality. The total bleeding rate was not significantly different between the two groups (14.8% vs 24%, P = 0.343), nor was there a significant difference in major bleeding (3.7% vs 6%, P = 0.665) or variceal bleeding (3.7% vs 16%, P = 0.109). The safety and effectiveness of different anticoagulants, including VKAs, LMWHs, and DOACs such as rivaroxaban and edoxaban, were found to be similar ( Supplemental Table 1 ). Notably, Zhang et al found no significant difference in the rate of PVT recanalization among the different anticoagulants used. They concluded that anticoagulants could increase the PVT recanalization rate and reduce the PVT progression rate without increasing bleeding rates in patients with cirrhotic PVT. However, this study also highlighted the need for further research to optimize the use of anticoagulants in this patient population.
Discussion
Although several prior meta-analyses9–11,29–31 have investigated the impact of DOACs in cirrhotic patients with PVT, these studies had several limitations. Firstly, most of these meta-analyses included a retracted RCT. 12 Secondly, while these studies examined the use of DOACs in patients with cirrhosis, some of them did not analyze the data specifically for PVT and also included other indications for anticoagulant therapy, such as AF and VTE.23,24 Thirdly, some studies included patients with Budd-Chiari Syndrome,9,32 a condition related yet distinct from PVT. Lastly, some studies relied on single-arm articles for analysis, focusing primarily on the occurrence of events in the DOACs group without comparing it to traditional anticoagulation.17,18,29 Thus, we updated systematic review by removing some studies12,20,23,24,32 that should be not included and incorporating the most recent published studies.13,14
Our systematic review investigated the use of anticoagulants, particularly DOACs, in the treatment of PVT in patients with liver cirrhosis. The included studies suggested that anticoagulation therapy, including DOACs, was associated with higher rates of PVT recanalization and resolution without significantly increasing the risk of major bleeding events. Specifically, compared to VKAs, edoxaban demonstrated a significant reduction in PVT volume, higher complete response rates, and was associated with long-term improvement of PVT. Safety profiles of DOACs were found to be similar or better than VKAs, with no significant difference in gastrointestinal bleeding or other adverse effects. While preliminary data from the studies suggest potential advantages of DOACs over warfarin in terms of bleeding risk, the underdosing of warfarin may complicate direct comparisons. More rigorous randomized studies are warranted to draw clearer conclusions regarding the relative efficacy of DOACs.
DOACs were associated with a significant reduction in PVT volume, a higher thrombolysis response rate, and improved long-term survival outcomes compared to traditional anticoagulation. These findings were particularly noteworthy given the historically limited treatment options for PVT in cirrhotic patients, who often face challenges such as drug-drug interactions and dietary restrictions when treated with VKAs. Our findings align with recent reviews by Hu et al 33 and Valeriani et al, 34 which explore the use of DOACs in cirrhotic patients with thrombotic disorders, emphasizing the need for further research in this area.
Given the bleeding predisposition in cirrhotic patients, the risk of intracranial, major, and fatal bleeding must be carefully weighed. Strategies to mitigate these risks are paramount, especially when initiating anticoagulation in patients with advanced liver disease. In terms of safety, DOACs demonstrated a comparable safety profile to traditional anticoagulation, with no significant difference in bleeding rates. This is a critical consideration given the increased risk of bleeding complications in cirrhotic patients due to impaired liver function and coagulopathy. In the context of cirrhosis and impaired hepatic function, monitoring plasma levels of DOACs may be considered to optimize therapy and mitigate risks.
Previous concerns have been raised regarding the metabolism of DOACs, such as rivaroxaban and apixaban, through the hepatic CYP3A4 pathway, which may lead to drug-drug interactions and potentially increase bleeding rates. Notably, the dosing strategies for DOACs in the included studies were carefully adjusted for hepatic impairment, which may have influenced the observed outcomes. In addition, the differences in efficacy may, in part, result from the cautious dosing of warfarin, given its narrow therapeutic index and the risk of severe bleeding complications. However, recent evidence indicates that DOACs may present a lower bleeding risk compared to LMWH or warfarin in patients with liver cirrhosis, which supports our findings and warrants further investigation.
Managing bleeding complications in patients on anticoagulant therapy requires careful consideration of reversal strategies. Traditional VKAs like warfarin can be reversed with vitamin K, prothrombin complex concentrates, or fresh frozen plasma, though these options carry risks such as volume overload and thrombotic complications. In contrast, DOACs have more specific reversal agents: idarucizumab for dabigatran and andexanet alfa for rivaroxaban and apixaban, offering targeted management for bleeding events. Clinicians should weigh the benefits of anticoagulation against bleeding risks, particularly in patients with cirrhosis and PVT.
Limitations
We acknowledged limitations including a retrospective design, small sample sizes, and potential selection bias that could limit the generalizability of our findings. Further studies are needed to determine the optimal duration of anticoagulant therapy and to assess its long-term effects. The pharmacokinetics of DOACs differ significantly, with varying degrees of hepatic metabolism. This variability necessitates a personalized approach to anticoagulation, especially in cirrhotic patients. In addition, the studies included in this review utilized varying doses of DOACs, including both full and reduced doses, which could affect both the effectiveness and safety outcomes. The INR targets for VKA patients varied across studies, which could affect the comparability of the results. Future prospective studies should confirm the benefits and safety of DOACs for treating PVT in cirrhotic patients.
Conclusion
DOACs appear to be a promising therapeutic option for PVT in cirrhotic patients. Current data should be regarded as preliminary and require further validation through larger, well-designed studies.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241303758 - Supplemental material for Direct Oral Anticoagulants Versus Traditional Anticoagulation in Cirrhotic Patients with Portal Vein Thrombosis: Updated Systematic Review
Supplemental material, sj-docx-1-cat-10.1177_10760296241303758 for Direct Oral Anticoagulants Versus Traditional Anticoagulation in Cirrhotic Patients with Portal Vein Thrombosis: Updated Systematic Review by Xiulin Xiao, Wengen Zhu and Qixin Dai in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Author Contributions
Qixin Dai and Wengen Zhu designed this review. Xiulin Xiao and Qixin Dai contributed to the data interpretation and checked the data to ensure accuracy. Xiulin Xiao finished the first draft, whereas Qixin Dai revised the manuscript. Wengen Zhu edited the manuscript before submission to ensure standard English grammar.
Availability of Data and Materials
All relevant data and materials are presented in the paper.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
