Abstract
Objectives
This study intended to investigate whether neuromuscular electrical stimulation(NMES) effectively prevents deep vein thrombosis(DVT) of lower limbs.
Methods
Major databases encompassing Pubmed, Embase, Web of Science, and Cochrane Library were thoroughly retrieved from their inception to January 1, 2025. Two reviewers separately selected studies, extracted data, and appraised the quality of eligible studies. Statistic analyses were executed leveraging Stata 15.0.
Results
A total of 599 studies were searched from databases. After literature screening,14 studies were included in our analysis. Meta-analysis revealed that NMES significantly increased peak venous velocity (PV) (mean difference [MD]) = 1.18, 95%CI: 0.93-1.43). Subgroup analyses confirmed significant improvements in PV across European and Asian populations, with the Geko device, Duo-STIM stimulator, 1 Hz stimulation, and common peroneal nerve stimulation (all P < 0.05). NMES also significantly enhanced ejected volume (EV) (MD = 0.90, 95%CI: 0.21-1.59) and time-averaged mean velocity (TAMEAN). Notably, the Irish subgroup, 36 Hz/1 Hz stimulation, and higher-intensity parameters (32 V voltage, 27 mA current) demonstrated significant efficacy for TAMEAN (all P < .05).All outcomes were statistically significant (P < .05).
Conclusions
In summary, NMES significantly improves the hemodynamics of the veins of lower limbs, though their effects are parameter-dependent and vary across regions. NMES can effectively improve PV, EV, and TAMEAN by optimizing device selection, targeting the common peroneal nerve, and adjusting stimulation parameters (1-36 Hz frequency, 27-32 V voltage).
Keywords
Introduction
Venous thromboembolism (VTE) includes deep vein thrombosis (DVT) and pulmonary embolism (PE), which are two clinical manifestations of VTE at different anatomical sites and pathological stages. The disease affects nearly 10 million patients worldwide each year and represents a major contributor to the global disease burden. 1 It has been shown that about 1,220,000 new cases of VTE are reported annually in the United States. 2 Approximately 20% of patients die within one year after the diagnosis of VTE, some of whom die directly from VTE, but more individuals often die from serious complications. 3 Post-thrombotic syndrome (PTS) affects 25% to 40% of individuals with DVT and considerably compromises their limb function and quality of life. 4 The risk of VTE elevates exponentially with age. 5 In the context of accelerated population aging, preventing and managing thromboembolic disease has received increasing attention in various fields, especially in surgical care. Therefore, increasing the rate of standardized VTE prevention is one of the national healthcare quality and safety improvement goals.
Currently, VTE prophylaxis is divided into three main categories: basic prophylaxis, pharmacological prophylaxis, and mechanical prophylaxis. 6 However, anticoagulants have contraindications due to bleeding risks, 7 while traditional mechanical prophylaxis is limited by patient tolerance and compliance. Neuromuscular electrical stimulation (NMES), an innovative physical prophylaxis, enhances venous return through simulating muscle contractions, which is non-invasive and adjustable. Several randomized controlled trials (RCTs) in recent years have confirmed that NMES decreases the incidence of VTE in high-risk patients, 8 and NMES alone is not inferior to other mechanical prevention. 9 However, its effectiveness remains controversial, and a systematic review of its applicability to different clinical scenarios is lacking.
Therefore, this study aims to explore the effect of NMES in preventing DVT of the lower extremities. Through rigorous systematic review and meta-analysis, this study not only provides high-quality evidence for the clinical application of NMES, but also provides a more precise evidence-based guidance for the formulation of individualized prevention plans.
Methods
This study was executed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement 10 and has been prospectively registered in the International Prospective Register of Systematic Reviews (PROSPERO, ID: CRD420251079366).
Literature Search
PubMed, Embase, Web of Science, and the Cochrane Library were thoroughly retrieved for RCTs evaluating the efficacy of NMES in preventing DVT of lower limbs, from database inception to January 2025. The search strategy was designed by combining Medical Subject Headings (MeSH) terms and free-text words, including “venous thrombosis”, “deep vein thromboses”, “deep vein thrombosis”, “phlebothrombosis”, “venous thromboses deep”, “electric nerve stimulation”, and “NMES”. Additionally, we manually screened reference lists of eligible studies to determine potentially relevant reports. The specific search strategy is provided in Table S1.
Eligibility Criteria
Inclusion criteria were designed as follows:
(1). Participants: Healthy volunteers or postoperative patients; (2). Interventions: NMES, compared with compression stockings, pneumatic pumps, or no intervention; (3). Outcomes: Peak venous velocity (PV) (cm/s), venous flow volume (VF) (ml/min), time-averaged mean velocity (TAMEAN) (cm/s), ejected volume (EV) (ml), venous ejection volume (VEV) (ml), venous flow velocity (VFV) (cm/s), and venous diameter (VD) (cm); (4). Study design: Randomized controlled trials.
Exclusion criteria were as follows:
(1). Duplicate publications; (2). Non-original research; (3). Patients with preoperative DVT, including those with conditions that predispose them to DVT; (4). Studies deemed irrelevant to the topic, such as reviews, animal experiments, and case reports.
Study Selection and Data Extraction
To ensure accuracy and reliability, two investigators independently screened the literature and extracted data as per predefined eligibility criteria. Dissents were addressed through discussion or by a third researcher. Retrieved records were uploaded into EndNote X9 to delete duplicates. The titles and abstracts were independently reviewed to remove clearly irrelevant studies. Finally, literature that fully met the requirements was identified through careful reading of the full text. Microsoft Excel was used to construct an information extraction table. The extracted information included (1) basic information of the included literature (first author, publication time, country); (2) basic characteristics of the study subjects; (3) specific details of the intervention, including specific exercise type, intensity, frequency, and site; (4) key elements of risk of bias evaluation; and (5) outcome indicators and results measure data.
Risk of Bias Assessment
The quality of eligible RCTs was independently appraised by two investigators utilizing the Cochrane Collabo-ration's Tool for Assessing Risk of Bias (ROB2.0). The risk of bias was evaluated in five categories: (1) randomization process, (2) interventions deviating from expectations, (3) missing outcome data, (4) outcome measurement, and (5) selective result reporting. The risk of bias was categorized as low, unclear, or high. Disagreements between reviewers were addressed through discussion; if consensus was not achieved, a third arbitrator facilitated the final decision.
Statistical Analysis
Meta-analysis was implemented leveraging StataSE 15.0. The efficacy of NMES in preventing DVT of lower limbs was evaluated by calculating the mean difference (MD) with 95% confidence intervals (CI). Heterogeneity was examined via the Q-test and I² statistic. A fixed-effects model was levaraged when no heterogeneity was detected (P ≥ .1 and I² ≤ 50%), whereas a random-effects model was leveraged in cases of heterogeneity (P < .1 and I² > 50%). Sensitivity analysis was implemented by the leave-one-out method to probe into the robustness of the results. Funnel plots and Egger's test were leveraged to ascertain publication bias. A P-value < .05 was indicative of statistical significance.
Results
Study Selection and Characteristics of Included Studies
599 documents were obtained from the initial search, and 578 remained after duplicate removal. Based on title and abstract screening, 527 studies not meeting relevant standards and 29 non-RCTs were deleted. Then, 8 reports were excluded due to missing data or inaccessible full texts. 3 additional studies were sourced elsewhere. Consequently, 14 eligible articles were included.11-24 The study selection flow is illustrated in Figure 1.

Literature Screening Flowchart.
The 14 eligible studies11-24 were from five countries (USA, Japan, Ireland, Turkey, and UK), involving a total of 566 participants aged 18-78 years. Specific characteristics of the included studies are summarized in Table 1.
Specific Characteristics of the Included Studies.

Quality Assessment of Included Studies
Among the eligible studies, 57.1% were rated as low risk, 35.7% as having some concerns, and 7.1% as high risk of bias. One study was judged as high risk due to issues in both the randomization process and deviations from intended interventions (Figure 2).

Methodological Quality Assessment Included in the Study. (A): Risk Bias Maps Included in the Study; (B): Quality Evaluation Chart of Selected Literature.
Meta-analysis Results
PV
A total of 10 studies12-15,18,19,21-24 reported this outcome. Owing to substantial heterogeneity (I2 = 83.7%, P < .001), a random-effects model was leveraged to pool effect sizes. The results indicated that the PV level was significantly elevated in the NMES group in comparison with the control group (MD = 1.18, 95% CI:0.93-1.43,p < .001) (Figure 3A).

The Comprehensive Forest Plot of All Outcome Indicators. (A) PV; (B) EV; (C) TAMAN; (D) VF; (E) Venous Blood Velocityj; (F) Venousdiameter.
Subgroup analyses were performed to identify heterogeneity sources by continent, frequency, stimulation site, and subject type. The results showed that geographically, 9 studies were from Europe and 2 were from Asia. NMES significantly elevated PV levels in both regions (Europe: MD = 1.33, 95% CI:0.56-2.11; Asia: MD = 1.51, 95% CI:0.77-2.24; p < .05 for both). Among the different frequencies of stimulation, encompassing 36 Hz in 2 studies, 1 Hz in 5 studies, and 10 Hz in 1 study, 1 Hz stimulation significantly increased the level of PV (MD = 1.71,95%CI:0.97-2.46). The difference was statistically significant (p < .05). In terms of stimulation sites, 2 studies focused on the flounder muscle, 5 studies on the common peroneal nerve, and 1 study on the calf muscle group. The stimulation of the common peroneal nerves significantly increased the PV level (MD = 1.78, 95% CI:1.01-2.55, p < .05). Subgroup analysis by patient type revealed that NMES significantly increased PV levels in both healthy subjects (7 studies; MD = 1.82, 95% CI:0.87-2.77) and patients on total knee replacement (TKR) (1 study; MD = 1.22, 95% CI:0.43-2.00), with statistical significance (P < 0.05). Substantial heterogeneity was present across all examined subgroups, indicating that country, device type, frequency, site, and patient type were not sources of heterogeneity (Figure 4).

Subgroup Analyses Were Conducted Based on Different Continents, Equipment Types, Frequencies, Stimulation Sites, and Subject Types. (A) Subgroup Analysis of the Impact of Different Continents on PV; (B) Subgroup Analysis of the Influence of Different Device Types on PV; (C)S Ubgroup Analysis of the Influence of Different Frequencies on PV; (D) Subgroup Analysis of the Effects of Different Stimulation Sites on PV; (E) Subgroup Analysis of the Influence of Different Subject Types on PV.
EV
5 studies12-15,17 reported this outcome. Heterogeneity analysis showed a high level of heterogeneity (I2 = 93.5%, p < .001). As such, a random-effects model was leveraged. No statistically significant difference was noted in EV levels (MD = 0.18, 95%CI:-1.19-1.56,p = .793) (Figure 3B)
Subgroup analyses were performed to identify heterogeneity sources by continent, frequency, and stimulation site. Geographically, 4 studies were from Ireland and 1 from the UK. The studies from Ireland demonstrated no significant hemodynamic improvement (MD = −0.01, 95% CI: −1.85-1.83), whereas the study from the UK reported significant improvement (MD = 0.90, 95% CI: 0.21-1.59). The difference was statistically significant (P < .05). Different frequencies of stimulation were utilized, encompassing 36 Hz in 2 studies, 1 Hz in 2 studies, and unspecified frequency in 1 study. The study with unspecified frequency (MD = 0.90, 95% CI: 0.21-1.59) reported statistically significant results (p < .05). In terms of stimulation sites, 2 studies focused on the flounder muscle, 2 studies on the common peroneal nerve, and 1 study did not specify the site. The study of unspecified site showed a significant increase in the PV level (MD = 0.9, 95% CI: 0.21-1.59, p < .05), and the difference was statistically significant. Substantial heterogeneity was present across all examined subgroups, indicating that country, device type, frequency, and site were not sources of heterogeneity (Figure 5).

Subgroup Analyses were Conducted Based on Different Countries, Equipment Types, Frequencies, and Stimulation Sites. (A) Subgroup Analysis of the Impact of Different Countries on EV; (B) Subgroup Analysis of the Impact of Different Device Types on EV; (C) Subgroup Analysis of the Influence of Different Frequencies on EV; (D) Subgroup Analysis of the Effects of Different Stimulation Sites on EV.
TAMEAN
4 studies12,13,18,23 reported this outcome. With substantial heterogeneity (I2 = 95.1%, p < .001), a random-effects model was leveraged to pool effect sizes. The results disclosed no statistically significant difference in TAMEAN levels (MD = 1.27,95%CI:−0.52-3.06, p = .164) (Figure 3C).
Subgroup analyses were performed to identify heterogeneity sources by continent, frequency, and stimulation intensity. Geographically, 2 studies were from Ireland and 2 from the UK. The studies from Ireland reported hemodynamic improvement (MD = 2.20, 95% CI:0.25-4.14, p < .05), whereas the study from the UK demonstrated no significant improvement (MD = 0.34, 95% CI:−1.73-2.41).Different frequencies of stimulation were utilized, encompassing 36 Hz in 1 study, 1 Hz in 2 studies, and unspecified frequency in 1 study. The effect of 36 Hz was statistically significant (MD = 3.18, 95% CI:2.41-3.94), and low heterogeneity was present in the 1 Hz group (MD = 1.30, 95% CI:0.64-1.95) (P < 0.05). These results disclosed that the frequency difference may affect the consistency of the results. Among the different stimulation intensities, 32 V was used in 1 study, 27 mA in 2 studies, and unspecified intensity in 1 study. TAMAN levels were improved in the 32 V group (MD = 3.18, 95% CI:2.41-3.94) and in the 27 mA group (MD = 1.30, 95% CI:0.64-1.95). Substantial heterogeneity was present across all examined subgroups, indicating that country, device type, frequency, and site were not sources of heterogeneity (Figure 6).
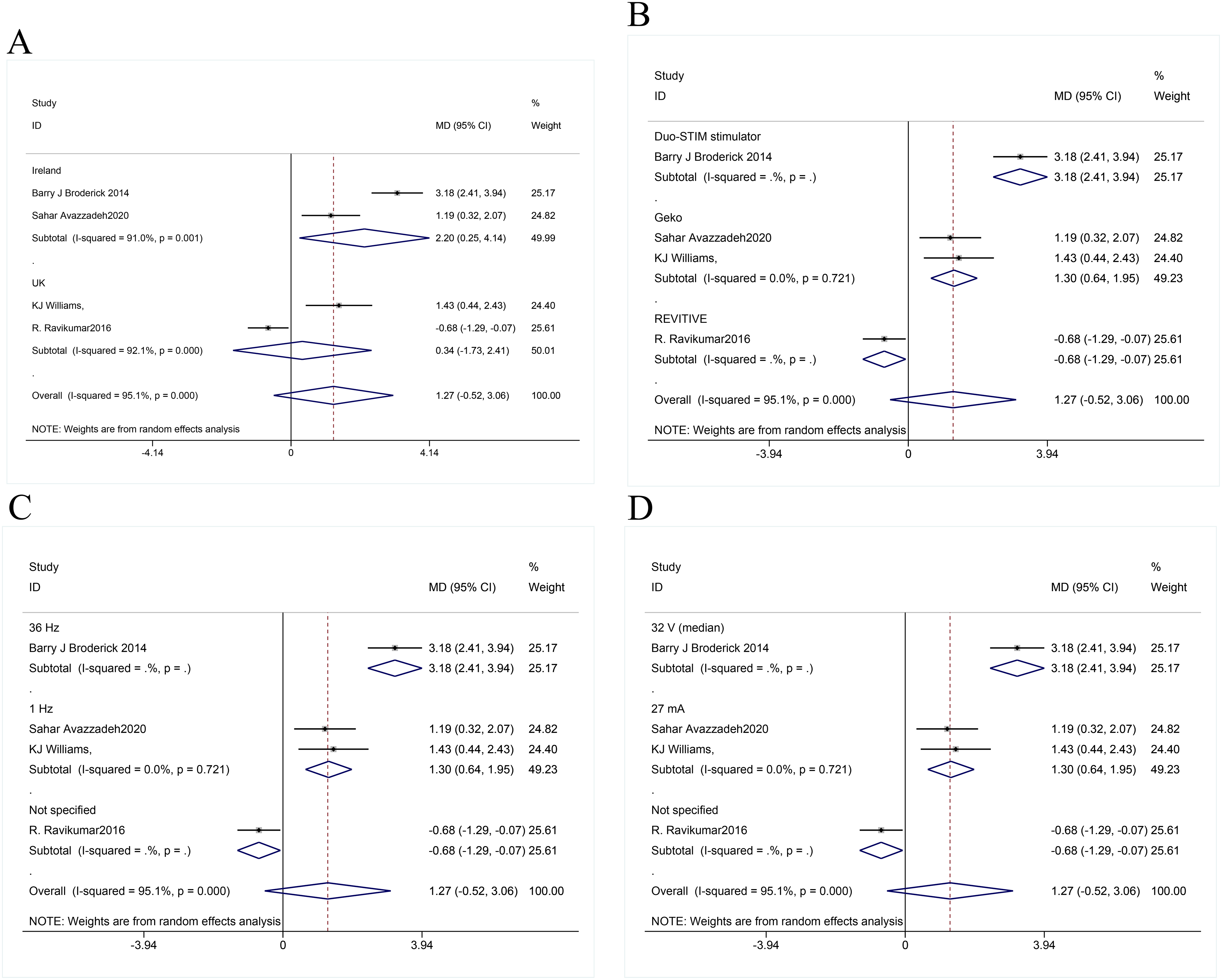
Subgroup Analyses Were Conducted Based on Different Countries, Equipment Types, Frequencies, and Stimulation Intensities. (A) Subgroup Analysis of the Impact of Different Countries on TAMAN; (B) Subgroup Analysis of the Impact of Different Device Types on TAMAN; (C) Subgroup Analysis of the Influence of Different Frequencies on TAMAN; (D) Subgroup Analysis of the Effects of Different Stimulus Intensities on TAMAN.
VF
3 studies15,18,21 reported this outcome. Heterogeneity analysis showed a high level of heterogeneity (I2 = 60.5%, P = .08), and therefore, a random-effects model was utilized. The results disclosed no statistically significant difference in VF levels between the NMES group and the control group (MD = 0.09, 95%CI: −0.56-.75, p = .784) (Figure 3D).
VFV
2 studies11,20 reported this outcome. Heterogeneity analysis showed a high level of heterogeneity (I2 = 95.5%, p < .001). Consequently, a random-effects model was leveraged to pool effect sizes. No statistically significant difference was detected in VFV levels between the two groups (MD = −1.11, 95%CI:−4.35-2.13, p = .503) (Figure 3E).
VD
2 studies20,21 reported this outcome. Owing to substantial heterogeneity (I2 = 0%, p = .429), a random-effects model was leveraged. No statistically significant difference was noted in VD levels (MD = 0.12, 95% CI:−0.36∼0.61, p = .618) (Figure 3F).
Sensitivity Analysis and Publication Bias
To ascertain the robustness of the meta-analysis results, we performed a sensitivity analysis of the included outcome indicators. Changes in effect sizes were assessed by excluding each study individually, and no significant changes were found, suggesting stable results (Figure 6).No publication bias was present for PV, as demonstrated by a symmetric funnel plot (Figure 8) and non-significant Egger's test result (p = .172) (Figure 9). Whereas publication bias was suggested in the EV results, as indicated by an asymmetric funnel plot (Figure 7) and significant Egger's test result (p < .05)(Figure 8).Subsequently, the trim-and-fill analysis was performed. No substantial change was noted in effect estimates after correction, indicating that the observed publication bias, while present, was insufficient to compromise the reliability of the results (Figure 9).

Sensitivity Analysis. (A) PV; (B) EV.

Publication Bias. (A) PV; (B) EV.
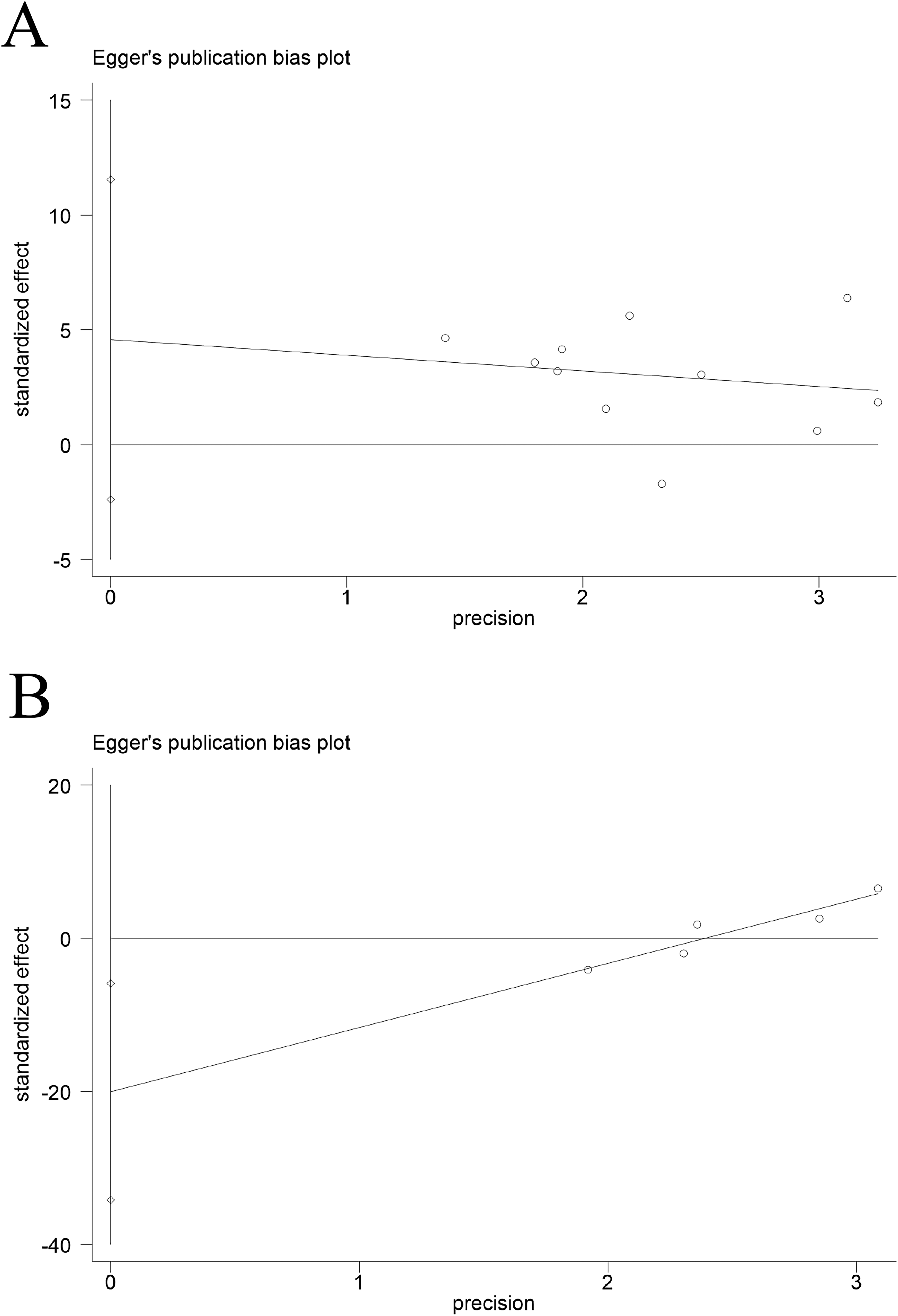
Egger's Test Result. (A) PV; (B) EV.
Discussion
VTE represents a significant global health burden. Hence, evidence support is urgently needed to optimize its preventive strategies.25,26 RCTs have demonstrated the efficacy of NMES in lowering the incidence of postoperative DVT. 9 This study focused on NMES, a non-invasive mechanical prophylaxis modality, to evaluate how device type, stimulation parameters, and target application points influence lower extremity venous hemodynamics. Our meta-analysis integrated data from 14 RCTs and revealed that NMES significantly enhanced peak venous flow rate, ejection volume, time-averaged blood flow velocity, and D-dimer levels. 27 Nevertheless, its efficacy depended on specific parameters and varied across population subgroups. On the other hand, NMES did not demonstrate statistically significant improvements in VF, VFV, or VD. These findings provide critical evidence to guide the clinical application of NMES.
Several studies have demonstrated that NMES significantly enhanced peak VF,28,29 consistent with our findings. This finding further supports that NMES enhances venous return by simulating muscle contractions. Modern NMES devices incorporate adjustable technical parameters, including frequency, pulse width, and stimulation intensity, 30 to accommodate individual patient needs. Subgroup analyses revealed that the superior performance of Geko and Duo-STIM devices may be attributed to their precise current output and efficient muscle activation, aligning with the results reported by Kristen Gelenitis et al. 31 Additionally, the advantage of low-frequency stimulation may be related to physiological muscle contraction rhythms, while the significant effects of high-frequency stimulation on TAMEAN suggest that different stimulation parameters have multidimensional influences on hemodynamics.
For EV, although the overall effect did not reach statistical significance (standardized mean difference [SMD] = 0.18, p = .793), specific devices enhanced EV in UK participants (mean difference [MD] = 0.90, p < .05). This finding partially contradicts Liang et al's review, potentially due to the high heterogeneity among devices in the newly included RCTs, particularly the differences in current output between Geko and Duo-STIM. Additionally, variations in EV measurement methods, for instance, discrepancies in ultrasound techniques, may have further contributed to the inconsistency in results.
Furthermore, TAMEAN levels were elevated in the Irish subgroup and the Duo-STIM device subgroup (MD = 3.18, p < 0.05), consistent with findings from the single-center research by Broderick et al. This suggests that high-intensity stimulation parameters may enhance hemodynamics by augmenting the muscle pump effect, and stimulation intensity parameters may potentially modulate treatment efficacy. However, no significant effect was observed in the UK subgroup, implying that baseline characteristics, such as age, and surgical type, could confound the results. Further, stratified analysis is warranted.
This study systematically evaluated the effects of stimulation parameters and targets on the efficacy of NMES. For instance, the significant effect of common peroneal nerve stimulation (MD = 1.78) underscores that neuro-targeted stimulation is essential in optimizing hemodynamics, potentially through activation of neurogenic antithrombotic pathways (the so-called “fourth thrombogenic factor” 32 ). Our findings demonstrate that parameter-optimized NMES effectively enhances lower extremity venous hemodynamics, offering a safe alternative for high-risk populations such as postoperative or bedbound patients. Additionally, by incorporating RCTs across diverse clinical settings including orthopedics and intensive care, the generalizability of these conclusions has been strengthened.
Study Limitations
Substantial heterogeneity existed in both stimulation parameters (frequency/intensity) and assessment methods (ultrasound localization) across the eligible studies. While subgroup analyses were conducted to investigate potential sources of heterogeneity, they were unable to fully account for all confounding factors. Furthermore, current research has primarily focused on short-term hemodynamic effects, with no long-term follow-up data evaluating the efficacy of NMES in reducing DVT incidence or preventing post-thrombotic syndrome.
Conclusion
In summary, our meta-analysis reveals that NMES significantly improves PV. Among them, 1HZ frequency stimulation and the stimulation of the common peroneal nerve has a significant effect. Therefore, low-frequency stimulation and the stimulation of target points (common peroneal nerve) can be considered in clinical practice in the future, though their effects on other hemodynamic parameters remain statistically insignificant. Nevertheless, as a non-pharmacological prophylaxis, NMES is still a promising technique for clinical application. Future investigations should optimize study design, such as multicenter, double-blind RCTs, and use standardized device parameters and outcome measures to reduce heterogeneity. Furthermore, more studies are warranted to integrate biomarkers (eg, D-dimer, coagulation factors) and imaging technologies to elucidate the influence of NMES on vascular endothelial function and coagulation-fibrinolysis balance. Lastly, studies with long-term follow-up are desired to assess the sustained impacts of NMES on DVT incidence, post-thrombotic syndrome, and quality of life. These advancements will enhance the clinical utility of NMES in DVT prevention.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251366423 - Supplemental material for Effectiveness of Neuromuscular Electrical Stimulation in Preventing Venous Thromboembolism: A Meta-Analysis Based on Randomized Controlled Trials
Supplemental material, sj-docx-1-cat-10.1177_10760296251366423 for Effectiveness of Neuromuscular Electrical Stimulation in Preventing Venous Thromboembolism: A Meta-Analysis Based on Randomized Controlled Trials by Yan Xu, Yingfang Li, Zhihong Chai, Yanfei Tao, Huijiao Lin and Changjun Zheng in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Health Science and Technology Program of Zhejiang Province (2022PY029).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data used to support the findings of this study are included within the article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
