Abstract
The use of tranexamic acid (TXA) for reducing blood loss in intertrochanteric fracture (IF) surgery remains controversial. We therefore performed a meta-analysis of randomized controlled trials (RCTs) to evaluate the efficacy and safety of TXA in reducing transfusion requirements and blood loss for IF surgery. Databases, including PubMED, Cochrane, and Embase, were searched for RCTs that were published before February 2018 and that addressed the efficacy and safety of TXA in patients who underwent IF surgery. A total of 746 patients from 7 RCTs were subjected to meta-analysis. The results showed that TXA group had reduced surgical blood loss (weighted mean difference [WMD] = −37.24, 95% confidence interval [CI]: −48.70 to −25.77, P <.00001), reduced total blood loss (WMD = −199.08, 95% CI: −305.16 to −93.01, P = .0002), higher postoperative hemoglobin (WMD = 0.46, 95% CI: 0.12 to 0.79, P = .007), and hematocrit levels (WMD = 1.55, 95% CI: 0.64 to 2.47, P = .008) compared to control group, while no significant differences were found in transfusion rates (relative risk [RR] = 0.75, 95% CI: 0.50 to 1.11, P = .15), postoperative drainage (WMD = −38.82, 95% CI: −86.87 to 9.22, P = .11), and thromboembolic events (RR = 0.94, 95% CI: 0.41 to 2.19, P = .89). In patients undergoing IF surgery, the administration of TXA significantly reduced surgical blood loss and total blood loss, while it had no significant effect on transfusion rate, postoperative drainage, and the risk of thromboembolic events. Nevertheless, due to the variations in the included studies, additional RCTs are required to further validate these conclusions.
Introduction
Intertrochanteric fractures (IFs) are special type of hip fracture that commonly occur in the elderly population with multiple comorbidities. The reported 1-year mortality after sustaining an IF has been estimated to be approximately 25%. 1 Compared to femoral neck fractures, patients with IF incur hidden blood loss and thus more often require blood transfusion. 2,3 In addition, these types of fractures are associated with strong activation of fibrinolytic system which usually continues to increase perioperatively. 2 Moreover, elderly populations with IF are highly susceptible to cardiovascular decompensation in the event of blood loss. 4,5
Tranexamic acid (TXA) is a simple and inexpensive pharmacological agent that interferes with fibrinolysis. The efficacy of TXA is generally accepted in reducing blood loss in elective knee and hip arthroplasty surgery. 6 Nevertheless, the use of TXA in reducing blood loss in IF surgery remains controversial. The aim of this meta-analysis is to evaluate the efficacy and safety of TXA in reducing transfusion requirements and blood loss for IF surgery.
Materials and Methods
This meta-analysis was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. 7
Search Strategies
A systematic electronic search of PubMED, Cochrane, and Embase Database was performed on February 1, 2018, for all published literature. The following key words/phrases were searched: [(Hip Fractures) OR (Fractures, Hip) OR (Trochanteric Fractures) OR (Fractures, Trochanteric) OR (Intertrochanteric Fractures) OR (Fractures, Intertrochanteric) OR (Peritrochanteric Fracture)] AND [Tranexamic Acid]. In addition, we did a hand search of reference lists from all the original articles and identified reviews.
Inclusion and Exclusion Criteria
The inclusion criteria were (1) randomized controlled trials (RCTs); (2) inclusion of adults with IFs for internal fixation; and (3) comparison of the efficacy and safety of TXA. Exclusion criteria were (1) in vitro or animal studies, case reports, reviews, meta-analyses, and letters to editors; (2) inclusion of adults with femoral neck fractures; and (3) not RCTs.
Data Extraction and Quality Assessment
All of the articles were independently reviewed by 2 reviewers (Q-Z.Z., C-X.Y.) according to the inclusion and exclusion criteria. The full texts from all the relevant studies were obtained and reviewed. Any disagreement between the 2 reviewers was settled by another senior reviewer (P.L.).
Each study was evaluated for methodological quality using the Cochrane Collaboration Risk of Bias Tool, which includes random sequence generation, allocation concealment, blinding, incomplete outcome data, selective reporting, and other biases. 8 The quality of evidence of outcomes was judged according to the Grading of Recommendations Assessment, Development and Evaluation (GRADE) 9 criteria. Two authors independently evaluated 5 factors (risk of bias, inconsistency, indirectness, imprecision, and publication bias) that may downgrade the quality level of evidence. The recommendation level of evidence was classified into 4 categories: high, moderate, low, or very low: 9 High quality meant that further research was very unlikely to change the confidence in the estimate of the effect; moderate quality indicated that further research was likely to have an important impact on our confidence in the estimate of the effect and could change the estimate; low quality implied that further research was very likely to have an important impact on our confidence in the estimate of the effect and was likely to change the estimate; and very low quality indicated that we were very uncertain about the estimate.
Outcomes of Interest
The following data were recorded for each study: first author’s name, year of publication, sample size, mean age, sex, fracture type, anesthesia methods, surgical procedure, intervention, control, thromboprophylaxis, transfusion criteria, and follow-up. The following outcomes were sought: transfusion rate, surgical blood loss, total blood loss, postoperative drainage, postoperative hemoglobin (Hgb), postoperative hematocrit, and thromboembolic events (deep vein thrombosis and pulmonary embolism).
Statistical Analysis
The WMD was calculated for continuous outcomes, and the relative risks (RRs) were calculated for dichotomous outcomes; 95% confidence interval (CI) was adopted for both. The heterogeneity was assessed using chi-square (χ2) test and I square (I2) test. When there was no statistical heterogeneity (as judged by χ2 test P >.1 or I2 <50%), a fixed-effect model was adopted; otherwise, a random-effect model was chosen. The reliability of pooled results was tested by sensitivity analyses. All analyses were performed using the software Review Manager 5.3. A P <.05 was considered statistically significant.
Results
Description of Studies
The details of search and exclusion criteria are displayed in the flow diagram (Figure 1). A total of 102 potentially eligible studies were identified by computerized search and reference list hand search. After screening, 7 studies 10 –16 including 746 patients (367 in the TXA group and 379 in the control group) were eligible for inclusion in this meta-analysis. The preoperative baselines were compared for all included trials, and each had similar baseline. Additional characteristics of included studies are shown in Table 1.
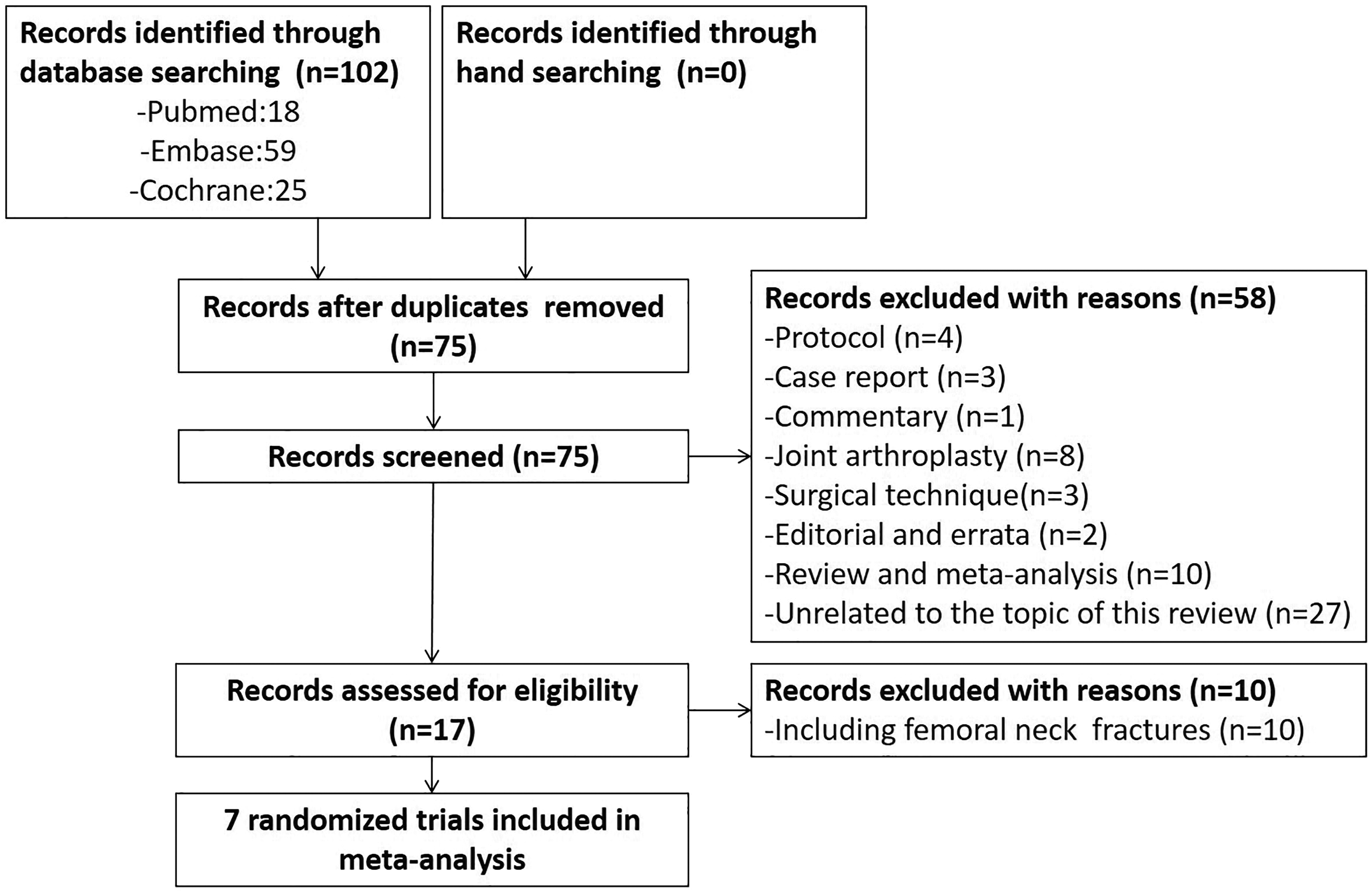
Flowchart of study selection.
Characteristics of Included Studies.
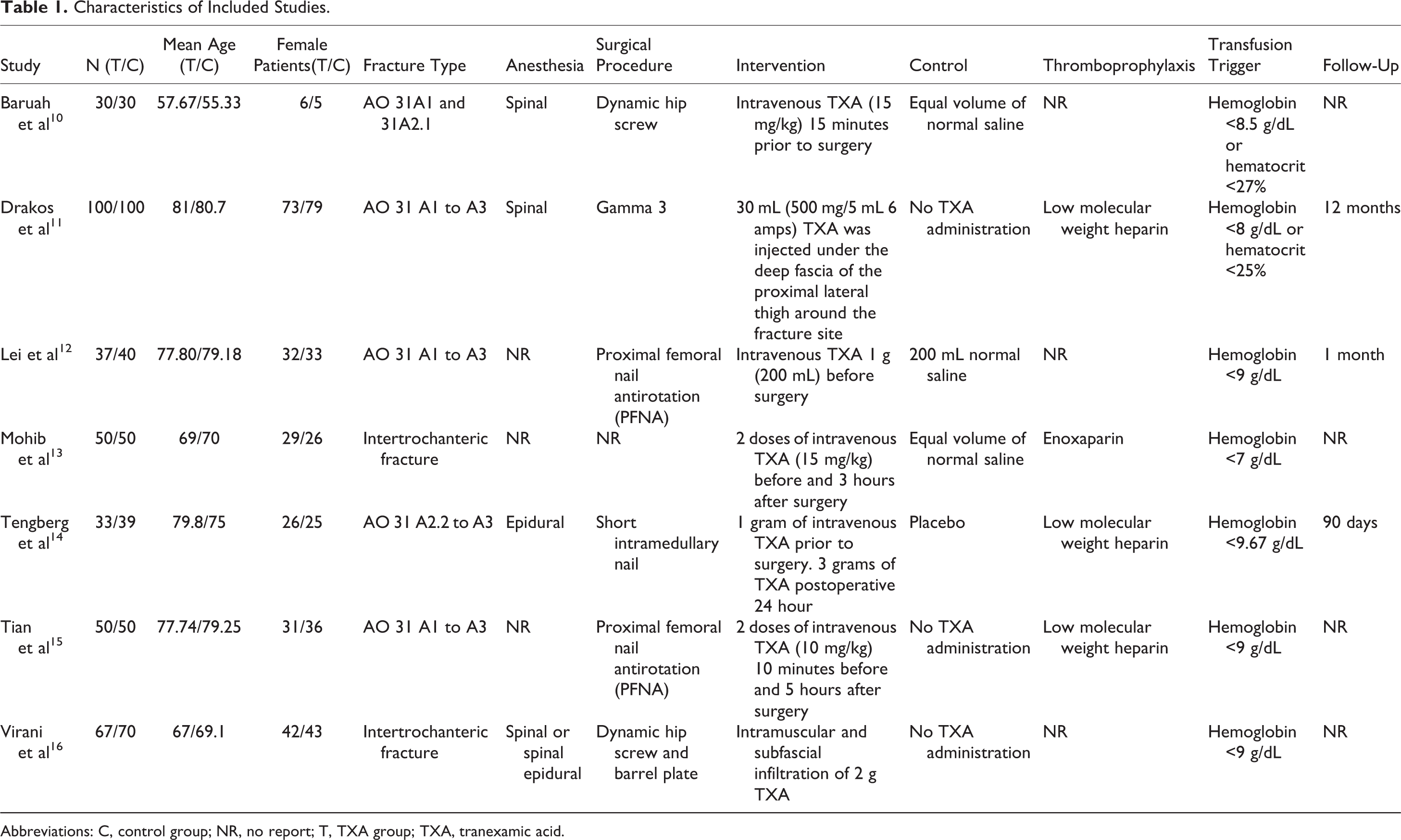
Abbreviations: C, control group; NR, no report; T, TXA group; TXA, tranexamic acid.
Risk of Bias
The Cochrane Handbook for the Systematic Review of Interventions was consulted to assess the risk of bias for the RCTs. Among 7 included trials, there were 5 studies with adequate random sequence generation. In addition, none of the included studies reported adequate concealment of allocation. Two studies applied blinding method for the participants and study personnel, and 3 studies applied the blinding approach for the assessors. Low risk of bias due to incomplete outcome data or selective outcome reporting was detected. The methodological quality of the included studies is presented in Figure 2.
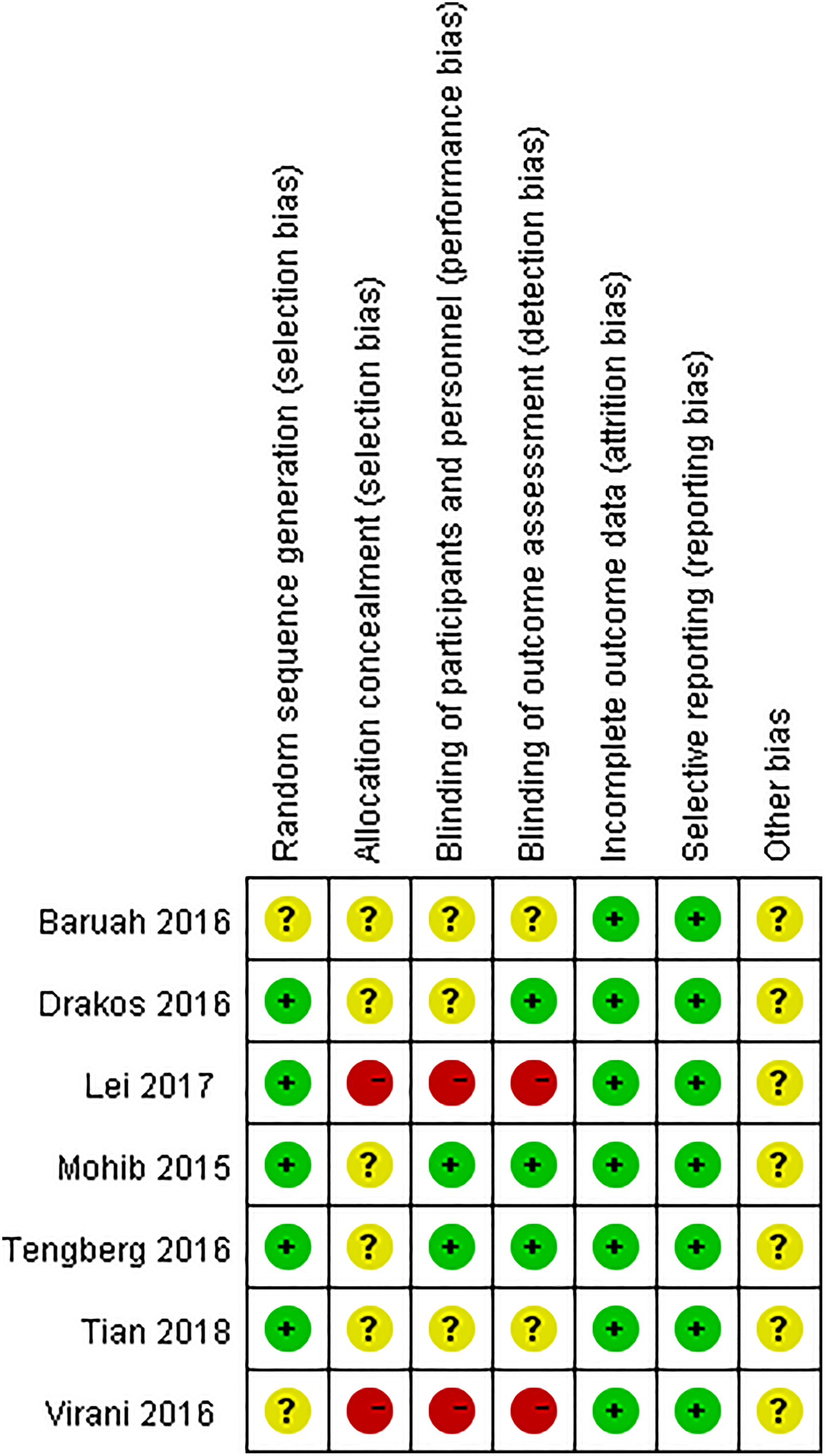
Methodological quality of the randomized controlled trials (RCTs).
Transfusion Rate
Transfusion rate was reported in 7 studies. 10 –16 There was a significant heterogeneity between the studies (χ2 = 72.77, P <.00001, I2 = 92%). Furthermore, there was no significant between-group difference in the transfusion rate (RR = 0.75, 95% CI: 0.50-1.11, P = .15; Figure 3).
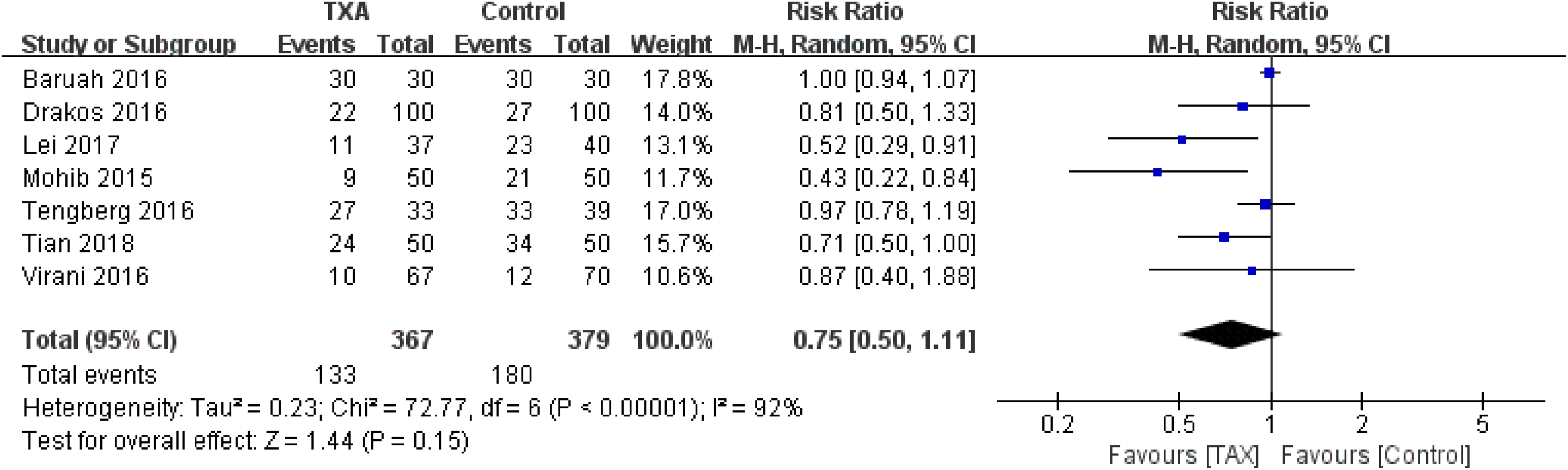
Forest plot for the transfusion rate.
Surgical Blood Loss
Surgical blood loss was compared in 4 studies. 10,12,14,15 Those data were pooled for the analysis. Briefly, no significant heterogeneity was detected between the studies (χ2 = 4.21, P = .24, I2 = 29%); therefore, the fixed-effect model was used. A significant decrease in the surgical blood loss was found in the TXA group compared to the control group (WMD= −37.24, 95% CI: −48.70 to −25.77, P <.00001; Figure 4).
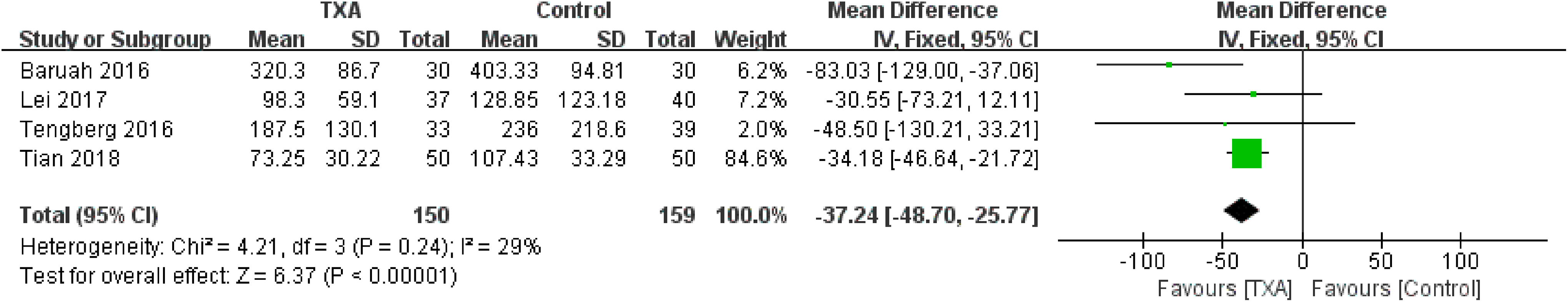
Forest plot for the surgical blood loss.
Total Blood Loss
Total blood loss was reported in 2 studies. No significant heterogeneity was detected between the studies (χ2 = 2.22, P = .14, I2 = 55%); therefore, the fixed-effect model was used for the analysis. Meta-analysis showed that the TXA group was associated with a significantly reduced total blood loss (WMD = −199.08, 95% CI: −305.16 to −93.01, P = .0002; Figure 5).
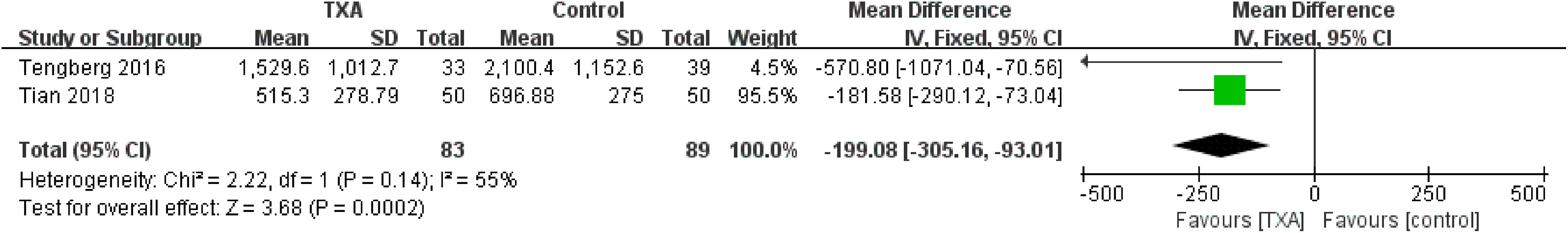
Forest plot for the total blood loss.
Postoperative Drainage
Postoperative drainage was examined in 4 studies. 10,12,15,16 There was a significant heterogeneity between the studies (χ2 = 110.50, P <.00001, I2 = 97%); therefore, the random-effect model was used. Briefly, no significant difference was detected among the studies (WMD= −38.82, 95% CI: −86.87 to 9.22, P = .11; Figure 6).
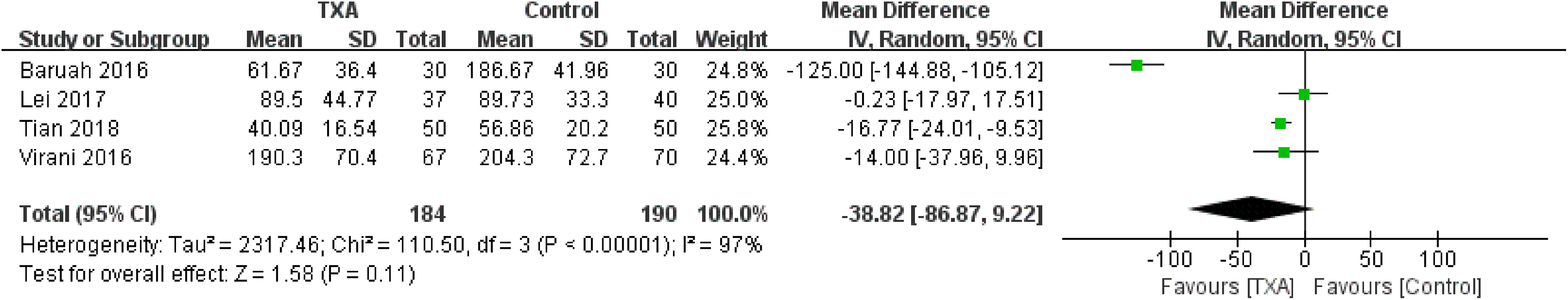
Forest plot for the postoperative drainage.
Postoperative Hgb
Four articles 10,12,13,16 reported the outcomes of postoperative Hgb. No significant heterogeneity was detected between the studies (χ2 = 5.51, P = .14, I2 = 46%); therefore, the fixed-effect model was used. The pooled results demonstrated that the TXA groups had a higher postoperative Hgb compared to the control group (WMD = 0.46, 95% CI: 0.12-0.79, P = .007; Figure 7).
Forest plot for the postoperative hematocrit.
Postoperative Hematocrit
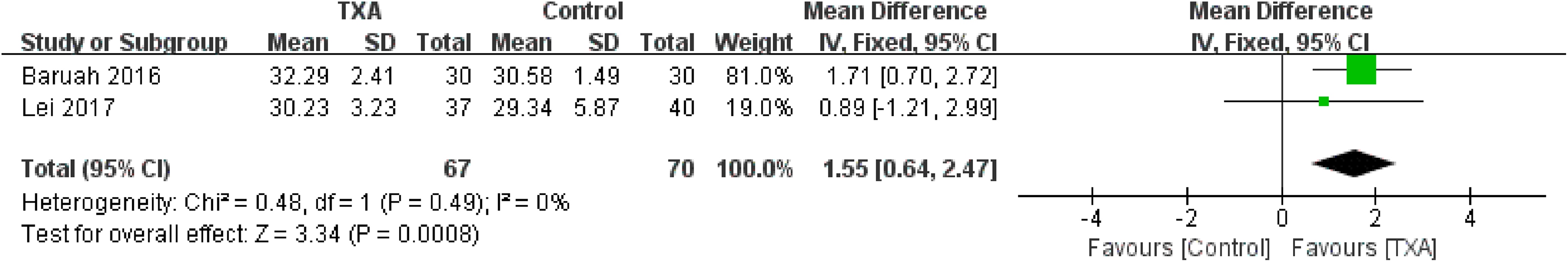
Postoperative hematocrit was reported in 2 studies. 10,12 No significant heterogeneity was detected (χ2 = 0.48, P = .49, I2 = 0%); therefore, the fixed-effect model was used. Meta-analysis showed that the TXA group was associated with a significantly higher postoperative hematocrit (WMD = 1.55, 95% CI: 0.64-2.47, P = .008; Figure 8).
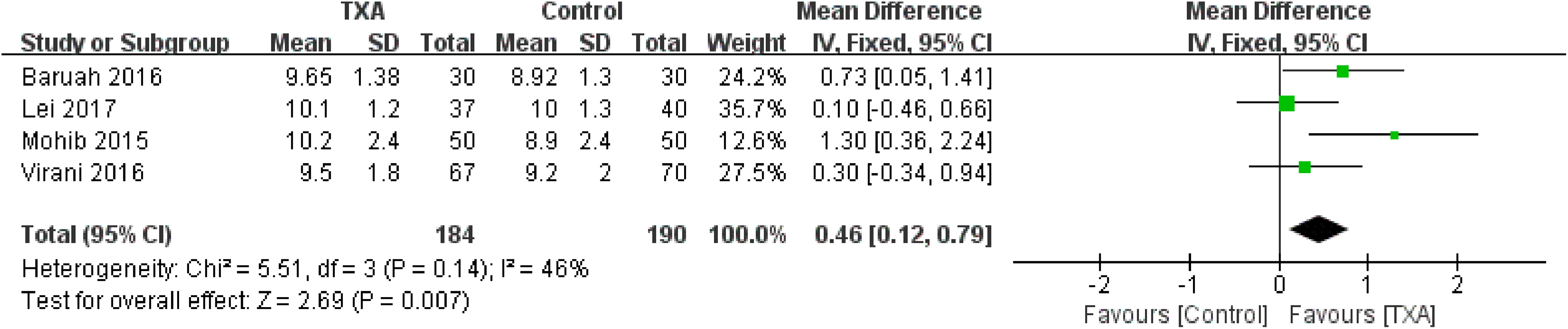
Forest plot for the postoperative hemoglobin.
Thromboembolic Events
Thromboembolic events (deep vein thrombosis or pulmonary embolism) were reported in 6 studies 10,12,14 –16 (statistically homogeneous; χ2 = 1.41, P = .84, I2 = 0%). There was no significant between-group difference in the rate of thromboembolic events (RR = 0.94, 95% CI: 0.41-2.19, P = .89; Figure 9).
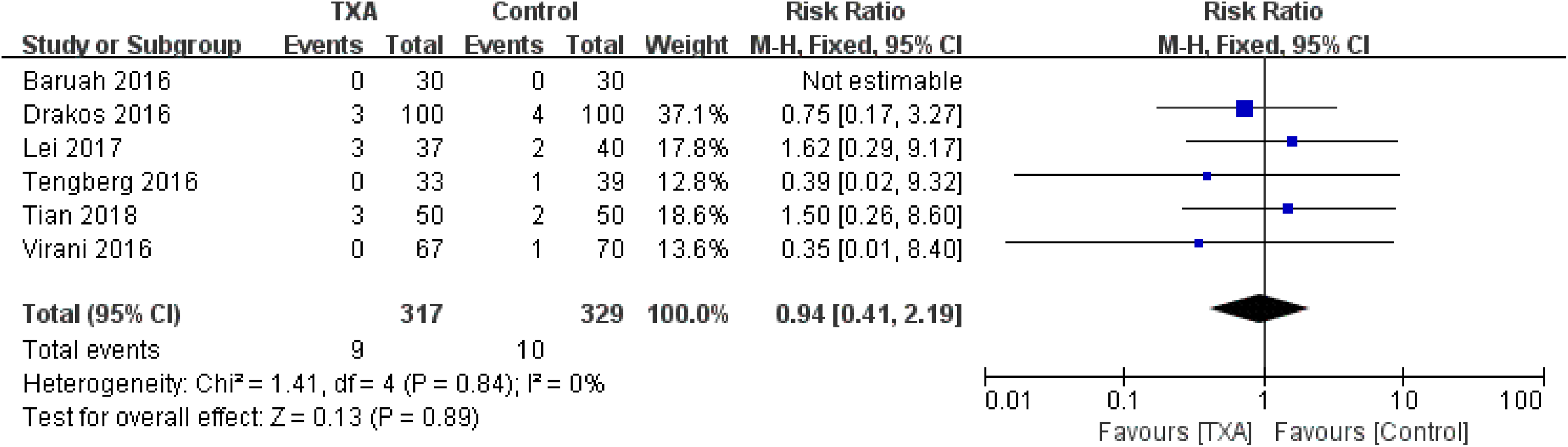
Forest plot for the thromboembolic events.
Subgroup Analysis
Subgroup analysis was conducted based on transfusion rate and thromboembolic events. With reference to the transfusion rate, subgroup analyses of different types of administration, dosage, and time were performed. No significant heterogeneity was detected in the subgroup of local administration at the end of surgery (χ2 = 0.02, P = .89, I2 = 0%). Both preoperative intravenous (IV) 1 dose, and the preoperative IV and postoperative IV 2 doses subgroups showed significant heterogeneity between the studies (χ2 = 50.92, P <.0001, I2 = 98% and χ2 = 9.35, P = .009, I2 = 79%, respectively). In addition, no significant between-group difference among subgroups was detected (Figure 10). We divided the transfusion trigger into 3 subgroups: Hgb < 9.67 g/L, Hgb < 9g/L, and Hgb < 8.5 g/L. In the subgroup of Hgb < 9g/L, there was no significant heterogeneity (χ2 = 1.35, P = .51, I2 = 0%), and the TXA group was associated with a significantly reduced transfusion rate (RR = 0.67, 95% CI: 0.51-0.89, P = .005; Figure 11).With reference to thromboembolic events, there were no significant differences in the rate of thromboembolic events with TXA identified for intravenous administration (RR = 1.26, 95% CI: 0.41-3.82, P = .69) and local administration (RR = 0.64, 95% CI: 0.17-2.41, P = .51).
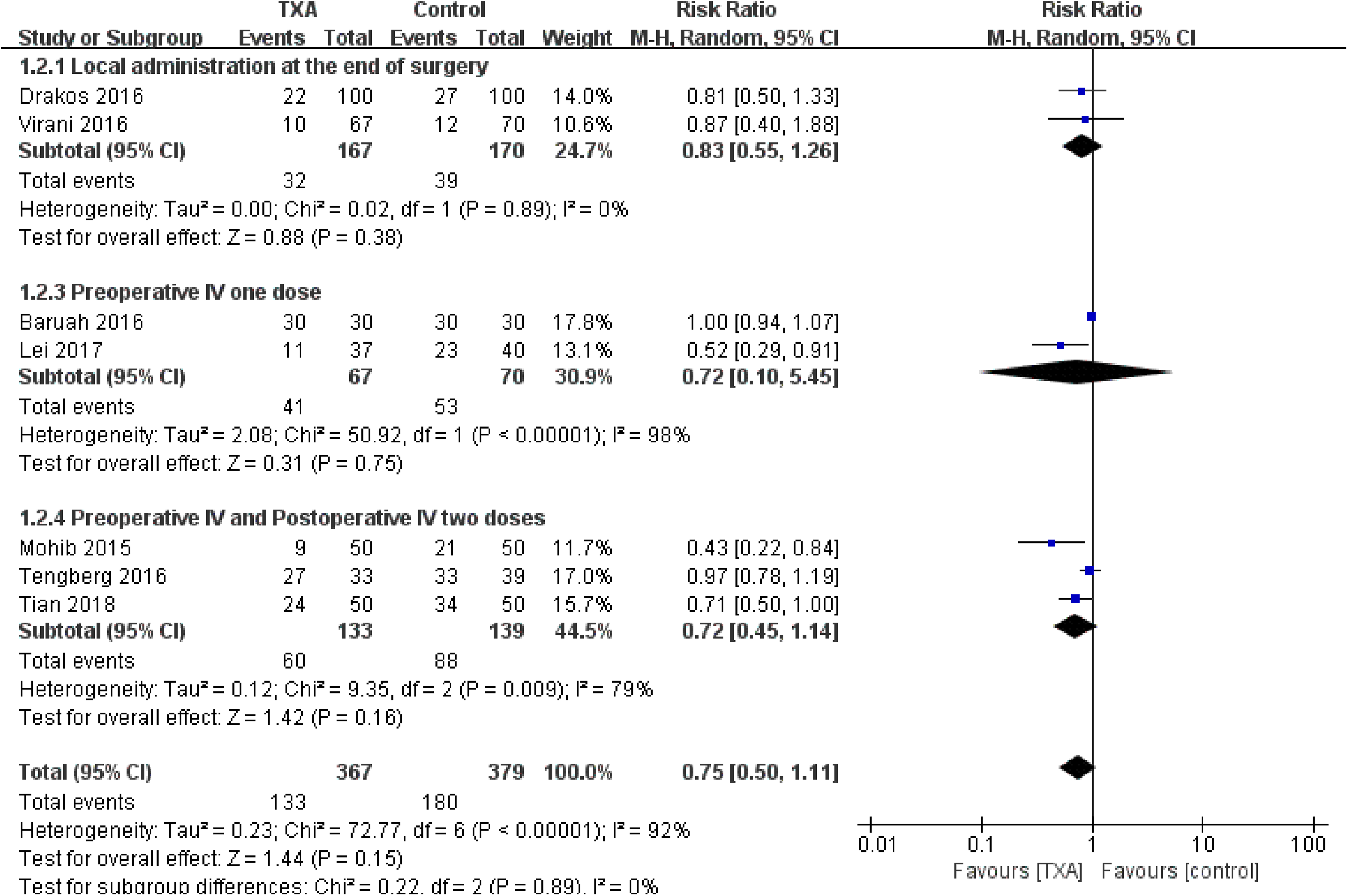
Forest plot for subgroup analysis of transfusion rate for different type of administration, dosage, and time.
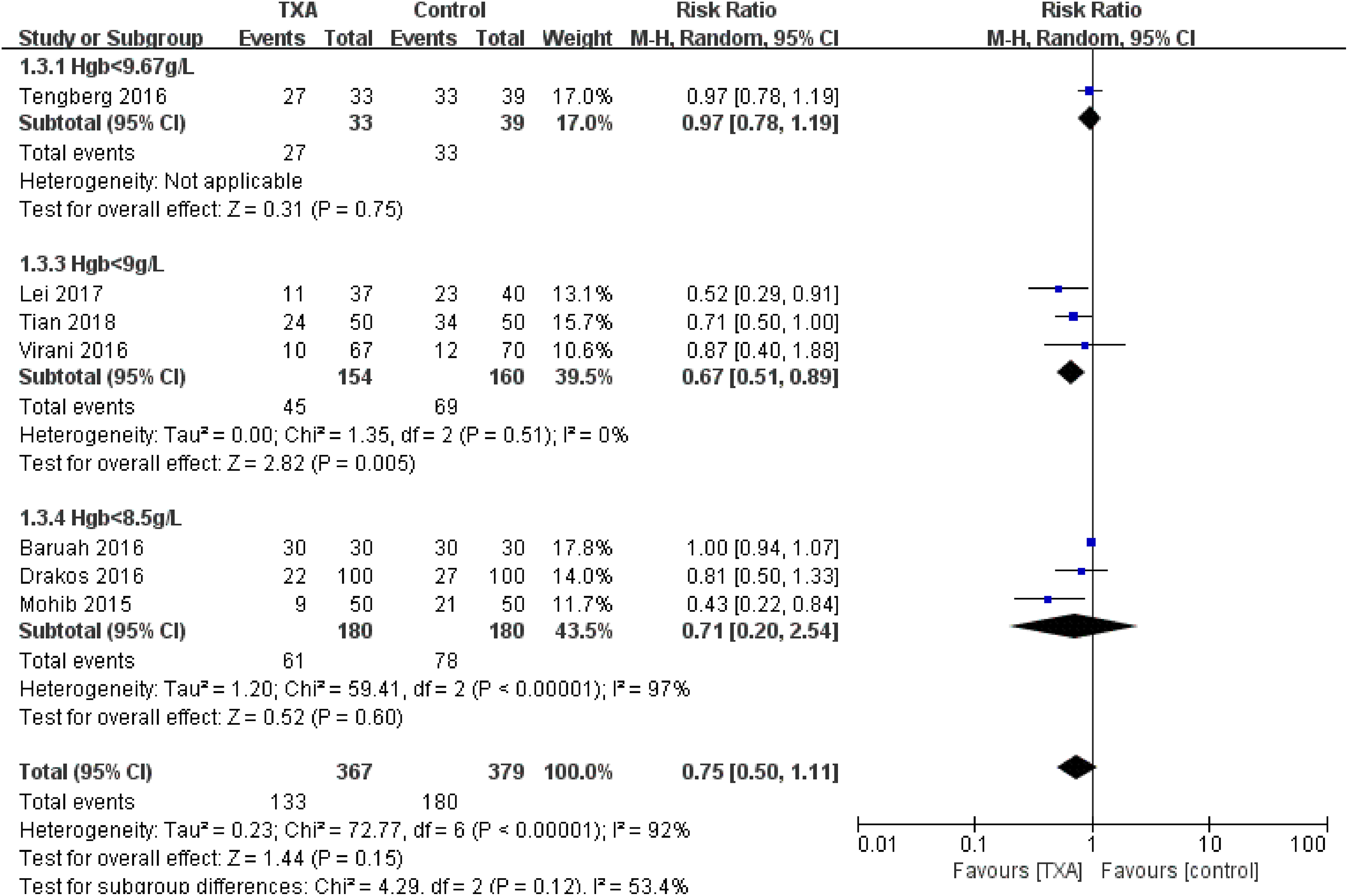
Forest plot for subgroup analysis of transfusion rate for different transfusion trigger.
Sensitivity Analysis
Sensitivity analysis was conducted by deleting 1 study from overall pooled analysis each time so as to check the influence of the removed data to the overall data set. To the transfusion rate, sensitivity analysis excluding Baruah et al 10 resulted in statistical significance (RR = 0.73, 95% CI: 0.55-0.96, P = .02). Moreover, concerning postoperative drainage, sensitivity analysis excluding Baruah 10 resulted in statistical significance (WMD = −12.52, 95% CI: −22.40 to −2.63, P = .01). To the postoperative Hgb, sensitivity analysis excluding Baruah et al 10 or Mohib et al 13 resulted in loss of statistical significance (WMD = 0.37, 95% CI: −0.01 to 0.76, P = .06 or WMD = 0.34, 95% CI: −0.02 to 0.69, P = .06, respectively). Therefore, there was no enough evidence to verify any between-group differences in transfusion rate, postoperative drainage, and postoperative Hgb.
Grading of Recommendations Assessment, Development and Evaluation Analysis
According to the results of the GRADE analysis, the quality of the evidence was high for surgical blood loss, total blood loss, and thromboembolic events. The quality of the evidence was moderate for postoperative Hgb and postoperative hematocrit. The quality of the evidence was low for transfusion rate and postoperative drainage.
Discussion
The results of our meta-analysis suggested that TXA significantly reduces the surgical blood loss and total blood loss and increases postoperative Hgb and hematocrit level, while it has no effect on transfusion rate, postoperative drainage, and thromboembolic events.
Intertrochanteric fracture is a common type of extracapsular hip fracture which compared to femoral neck fractures incurs hidden blood loss. Since there is a major muscles insertion involved around this region, greater bone surface area is available for blood loss in the extracapsular fractures. 17 Patients with IF have larger drop in Hgb that has shown to be associated with the initial trauma rather than with femoral neck fractures. 18 In addition, internal fixation for IF has shown to be associated with increasing perioperative blood loss and increasing requirement for blood transfusions. 3,17 Therefore, it is important to assess the role of TXA in IF surgery separately from femoral neck fracture operation.
Tranexamic acid acts by blocking the lysate binding sites of plasminogen and plasmin, thus inhibiting fibrinolytic and inflammatory effect. 19,20 The administration of TXA has shown to reduce surgically related blood loss after elective total joint replacement. 21 In addition, TXA has shown the ability to reduce blood loss in patients with acute femoral neck fractures undergoing hip arthroplasty. 19 The present meta-analysis indicated that the application of TXA for IF surgery was effective in reducing surgical blood loss and total blood loss and was associated with higher postoperative Hgb and hematocrit level.
Blood transfusion increases the risk of immunological reaction, disease transmission, renal failure, lung injury, coagulopathy, infection, and overall mortality. 22,23 Previous meta-analyses have shown nearly uniform ability of TXA to limit the proportion of transfused patients after hip fractures. 24 –26 Contrarily, in this study, we found no significant difference in the transfusion rate among different groups. Nevertheless, there was a notable heterogeneity in the analysis of transfusion rate, and sensitivity analysis showed insufficient evidence to verify difference. In assessment of potential sources of heterogeneity, subgroup analyses suggested that intravenous administration, dosage, time, and strict transfusion trigger might be the sources of heterogeneity. In addition, we found that patients with hip fracture were usually frail and prone to anemia, which additionally suggested that health status might be a possible source of the variance.
Four studies 10,12,15,16 were involved in the comparison of postoperative drainage. In 2 studies, 10,12 TXA was administered intravenously before surgery, while in 1 study 15 TXA was dosed intravenously 5 hours before and after surgery. In an additional study, 16 intramuscular and subfascial infiltration of TXA was provided before closure. Our meta-analysis showed no significant difference in the postoperative drainage. For both intravenous and intramuscular TXA administration, the elimination half-life was about 2 hours. 27,28 The postoperative blood loss was measured on postoperative days 1 to 3, exceeding the half-life of TXA, which further explained why TXA had no effect on postoperative drainage.
Basic scientific studies have demonstrated that TXA inhibits fibrinolysis only at the site of active thrombogenesis, while it has no effects on systemic blood vessels. 29,30 From a clinical standpoint, Gausden et al 31 have shown TXA has no significant effect on the risk of symptomatic thromboembolic events in patients undergoing orthopedic trauma surgery. Our meta-analysis found high-quality evidence that the use of TXA in IF surgery was not associated with increase in thromboembolic events. Our data further strengthen the support of TXA use in patients with orthopedic trauma.
Local administration of TXA was as effective as intravenous administration after elective total joint replacement and spinal surgery, and the serum levels of TXA was much lower. 32 –34 Local administration could be a better route in patients at risk of thromboembolic complications. Subgroup analysis in our meta-analysis was performed to identify potential benefits of local administration of TXA. Local administration of TXA had no significant effect on transfusion rate and thromboembolic events in IF surgery.
The meta-analysis by Wang and Yu 26 addressing similar questions has been recently published that considers RCT until September 2017. Yet, it had 3 main differences from ours. First, Wang and Yu 26 concluded that “local administration of tranexamic acid is associated with a reduced transfusion requirements in patients with intertrochanteric fractures.” However, our meta-analysis showed no significant between-group difference in the transfusion rate. Second, Wang and Yu 26 concluded that “the evidence quality for each outcome is high.” In our meta-analysis, risk of bias and inconsistency were found to downgrade the quality of evidence. Further research is likely to have an important impact on our confidence in the estimate of effect. Third, our meta-analysis added 3 RCTs, 10,14,15 which provided a more up-to-date source of information.
The present study has some limitations. First, due to lack of guideline for TXA use, the included studies showed variations in type of administration, dosage, and timing. Second, substantial heterogeneity was observed when comparing the TXA with control group in terms of transfusion rate, and the differences in TXA administration and transfusion criteria may contribute to it. Third, sensitivity analysis showed there was insufficient evidence to verify any between-group differences in transfusion rate, postoperative drainage, and postoperative Hgb. Fourth, due to the limited clinical research only investigating the TXA for IF surgery, only 7 studies with 746 patients were included in this review. Finally, the GRADE analysis identified some outcomes as having moderate- or low-quality evidence. The identified results for these outcomes must be interpreted with caution.
Conclusions
In patients undergoing IF surgery, the administration of TXA significantly reduces surgical blood loss and total blood loss and has no significant effect on transfusion rate, postoperative drainage, and the risk of thromboembolic events. Nevertheless, due to the variations among the included studies, more large-sample, unified outcome measures and high-quality RCTs are required to further demonstrate the efficacy and safety of TXA for IF surgery.
Footnotes
Authors’ Note
Our institution does not require ethical approval for reporting meta-analysis.
Acknowledgments
Authors thank MedSci for its linguistic assistance during the preparation of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
