Abstract
Objectives
This study aimed to evaluate the incidence trend and related factors for hospital-acquired upper extremity deep vein thrombosis (HA-UEDVT) in inpatients of a large medical center in China over 13 years, and to summarize and analyze the complications and preventive treatments of HA-UEDVT.
Methods
We conducted a retrospective analysis on the epidemiological characteristics, related factors, and use of different prophylactic anticoagulation methods in patients with HA-UEDVT at Peking University People's Hospital from January 1, 2009, to December 31, 2021. The data of all patients were obtained through the hospital's medical record system.
Results
Over the 13 years, there has been 239 cases of HA-UEDVT, with a much lower incidence rate compared to HA-LEDVT (0.31 vs 2.28 per 1000 admissions). HA-UEDVT incidence peaked in 2015 and 2016 (both 0.44 per 1000 admissions). The highest HA-UEDVT incidence rates were in the respiratory and gastroenterology departments, with 1.13 and 1.22 per 1000 admissions, respectively. The top three related factors of HA-UEDVT were having a CVC, concurrent of breast cancer, and concurrent of HA-LEDVT, with the β values of 4.81, 4.68, and 3.11, respectively. Thrombosis-related preventive measures were implemented in 30.1% of patients, significantly less frequently than HA-LEDVT (30.1% vs 82.4%, p < .001). The low molecular weight heparin (LMWH) subcutaneous injection being the main thromboprophylaxis used before HA-UEDVT diagnosis.
Conclusions
HA-UEDVT mainly occurred in medicine departments and has shown a decreasing trend in recent years. Having CVC, breast cancer, and HA-LEDVT are highly associated with HA-UEDVT occurrence. There was no statistical difference in the incidence of pulmonary embolism between CVC-related HA-UEDVT and HA-LEDVT groups. More high-quality prospective studies are needed to guide HA-UEDVT prevention.
Introduction
Upper extremity deep vein thrombosis (UEDVT) is defined as thrombosis of the deep venous system, including subclavian, axillary, brachial, ulnar, and radial veins. It can be caused by thoracic outlet anatomic obstruction (primary) 1 or by central intravenous catheters (secondary). 2 UEDVT may present clinically as asymptomatic, or the clinical symptoms may include acute severe pain and swelling. 1 With the advancement of imaging technology, duplex ultrasound is first used to confirm the diagnosis in clinically suspected patients. 3 UEDVT is much less common than lower extremity deep vein thrombosis (LEDVT), registry data currently estimate that UEDVT represents about 6% of DVT cases. 2 A recent prospective epidemiological study in France found the annual incidence of UEDVT to be 0.98 per 10,000 inhabitants. 4 However, recent studies have reported significant complications of hospital acquired UEDVT (HA-UEDVT), including pulmonary embolism (PE), loss of vascular access, superior vena cava syndrome, and post-thrombotic syndrome. 5 Additionally, the incidence of HA-UEDVT may increase due to the widespread use of central venous catheters and the rising number of cancer patients, but there are few studies on HA-UEDVT incidence trends. It is widely believed that the two major related factors for HA-UEDVT are the placement of deep venous catheters and malignancy,6,7 but there is still a lack of sufficient data to support this. To improve the understanding of HA-UEDVT and its differences of incidences, related factors and anticoagulation treatments toward hospital acquired LEVDT (HA-LEDVT), we conducted a retrospective study of patients with HA-UEDVT and HA-LEDVT in Peking University People's Hospital from January 1, 2009, to December 31, 2021.
Methods
Ethical Approval
The informed consent in this study was exempted and the study was approved by the Ethics Committee of the Peking University People's Hospital (approval number 2024PHB321-001), in accordance with the Declaration of Helsinki.
Study Population and Data Collection
We conducted a retrospective analysis of discharged patients from Peking University People's Hospital between January 1, 2009, and December 31, 2021. The study covered 28 clinical departments, including 13 medicine departments (department of respiratory, gastroenterology, cardiology, hepatology, hematology, neurology, geriatrics, endocrinology, nephrology, rheumatology & immunology, traditional Chinese medicine, pain medicine, emergency medicine), 13 surgical departments (orthopedics & traumatology, arthritis clinic & research center, musculoskeletal tumor, gastrointestinal surgery, breast disease surgery, hepatobiliary surgery, urology, thoracic surgery, neurosurgery, cardiac surgery, vascular surgery, otolaryngology, gynecology and obstetrics).
The diagnosis of upper and lower extremity DVT in all inpatient departments from January 1, 2007, and December 31, 2021 was determined by an experienced vascular surgeon based on the results of venous ultrasound and reviewed by another vascular surgeon following inclusion criteria: (1) age >16 years; (2) acute upper or lower extremity DVT confirmed by venous ultrasound; (3) from the beginning of admission, the patient's hospital stay exceeds 48 h. Exclusion criteria were: (1) patients hospitalized within 90 days before admission; (2) acute venous thromboembolism (VTE) event diagnosed upon admission; (3) acute VTE event diagnosed within 48 h of admission. HA-UEDVT patients who received a central venous catheter (CVC) within 30 days before the DVT diagnosis were classified as CVC-associated HA-UEDVT. The remaining HA-UEDVT patients were classified as non-CVC-associated HA-UEDVT. PE was diagnosed by positive contrast-enhanced spiral chest CT, enhanced magnetic resonance angiography, or conventional pulmonary angiography.
We collected baseline characteristic data of these patients from medical records, including demographic characteristics, medical history (including history of hypertension, diabetes, malignancy or chemotherapy, surgical history and medicine treatments), laboratory results, examinations, and anticoagulant treatment information. The data were verified and proofread by more than two researchers to ensure accuracy. For detailed processes see Figure 1.

Flow Diagram. Abbreviations: DVT: Deep venous thrombosis; UEDVT: Upper extremity deep venous thrombosis; LEDVT: Lower extremity deep venous thrombosis; HA-UEDVT: Hospital-acquired upper extremity deep venous thrombosis; HA-LEDVT: Hospital-acquired lower extremity deep venous thrombosis.
Statistical Analysis
The total, annual and departmental incidence rates of HA-UEDVT and HA-LEDVT were calculated according to the equations below respectively:
Total incidence rate = (Number of new DVT cases diagnosed after 48 h of hospitalization over 13 years ÷ Total number of eligible hospitalizations over 13 years) × 1000. Annual incidence rate = (Number of new DVT cases diagnosed after 48 h of hospitalizations of surgical/medicine departments, and total in each year ÷ Number of eligible hospitalizations of surgical/medicine departments, and total in the corresponding year) × 1000. Departmental incidence rate = (Number of new DVT cases diagnosed after 48 h of hospitalization in each department over 13 years ÷ Total number of eligible hospitalizations in the corresponding department over 13 years) × 1000.
Categorical variables were presented as counts and percentages, while continuous variables were reported as mean with standard deviation (SD) or median (range). Categorical variables are presented as frequency and percentage. Normality tests were performed, and normally distributed continuous data were compared using t-tests or analysis of variance, while non-normally distributed continuous data were compared using rank-sum tests. Poisson regression was used to analysis the differences in patient-hospitalization day groups between surgical and medicine departments. Additionally, the chi-square statistic was used to assess differences in proportions between the HA-LEDVT group and the two HA-UEDVT groups. When these tests were significant (2-tailed α<0.025, Bonferroni correction), pairwise comparisons were made between the two HA-UEDVT groups and between the non-CVC-associated HA-UEDVT group and the HA-LEDVT group (continuous variables using t-tests and dichotomous variables using chi-square tests). We did not compare HA-LEDVT patients with the CVC-associated HA-UEDVT patients because any differences between these two groups could reasonably be explained by differences between CVC patients and HA-LEDVT patients.
Since the use of CVC was not randomly assigned in this patient population, potential confusion and selection bias were addressed via propensity score matching 8 : HA-UEDVT patients were matched with non-UEDVT patients from the total eligible hospitalization population according to similar baseline characteristics in a 1:4 ratio, and matching covariates were sexes, age, length of hospital stay (from admission to discharge), time of diagnosis (from admission to diagnosis of DVT), height, and weight, prophylactic treatment, and activities of daily living (ADL) score. Finally, 28 variables were included to create a Lasso regression model to study independent related factors for HA-UEDVT development. All statistical comparisons were performed using SPSS 27 and R version 4.4.0.
Results
Demographic Characteristics and Incidence Trends
Over 13 years, the center had a total of 779,848 inpatient admissions, including 2345 (0.30%) patients with ultrasound-confirmed DVT, of which 239 (0.031%) were HA-UEDVT cases. Among these, there were 114 (47.7%) males, 9 cases (3.8%) were complicated by PE. Among the 239 patients, 156 (65.3%) were from medicine departments, which was significantly higher than those from surgical departments: 83 (34.7%) (p < .001), and the median ages were 53 years (44-63 years) in surgical departments while 55 years (37-67 years) in medicine departments. The overall incidence rate of HA-UEDVT was higher in medicine than in surgical departments (0.54 vs 0.17 per 1000 inpatients). The cases of HA- UEDVT patients with malignancy in medicine departments were significantly higher than those in surgical departments (p = .002). The diagnosis and hospital stay times were longer in medicine (time for HA-UEDVT diagnosis: 8 days [3-15 days]; hospitalization time: 26.5 days [16-40 days]) than in surgical departments (time for HA-UEDVT diagnosis: 3 days [2-7 days]; hospitalization time: 16 days [12-26 days]) (both p < .001). here was a significant difference in patient-hospitalization day (≥ 22 days group) between surgical and medicine departments (0.021 patient vs 0.024 patient per 1000 days, IRR = 0.31, p = .044) (Tables 1 and S1). There was no significant difference in the mean BMI and four BMI groups between HA-UEDVT patients from medicine and surgical departments, HA-UEDVT was most common in patients aged 60-69 years, with the lowest incidence in those aged 30-39 years, and the incidence decreased progressively with age after 70 years. Detailed data can be found in Tables 1, S2 and Figure 2.
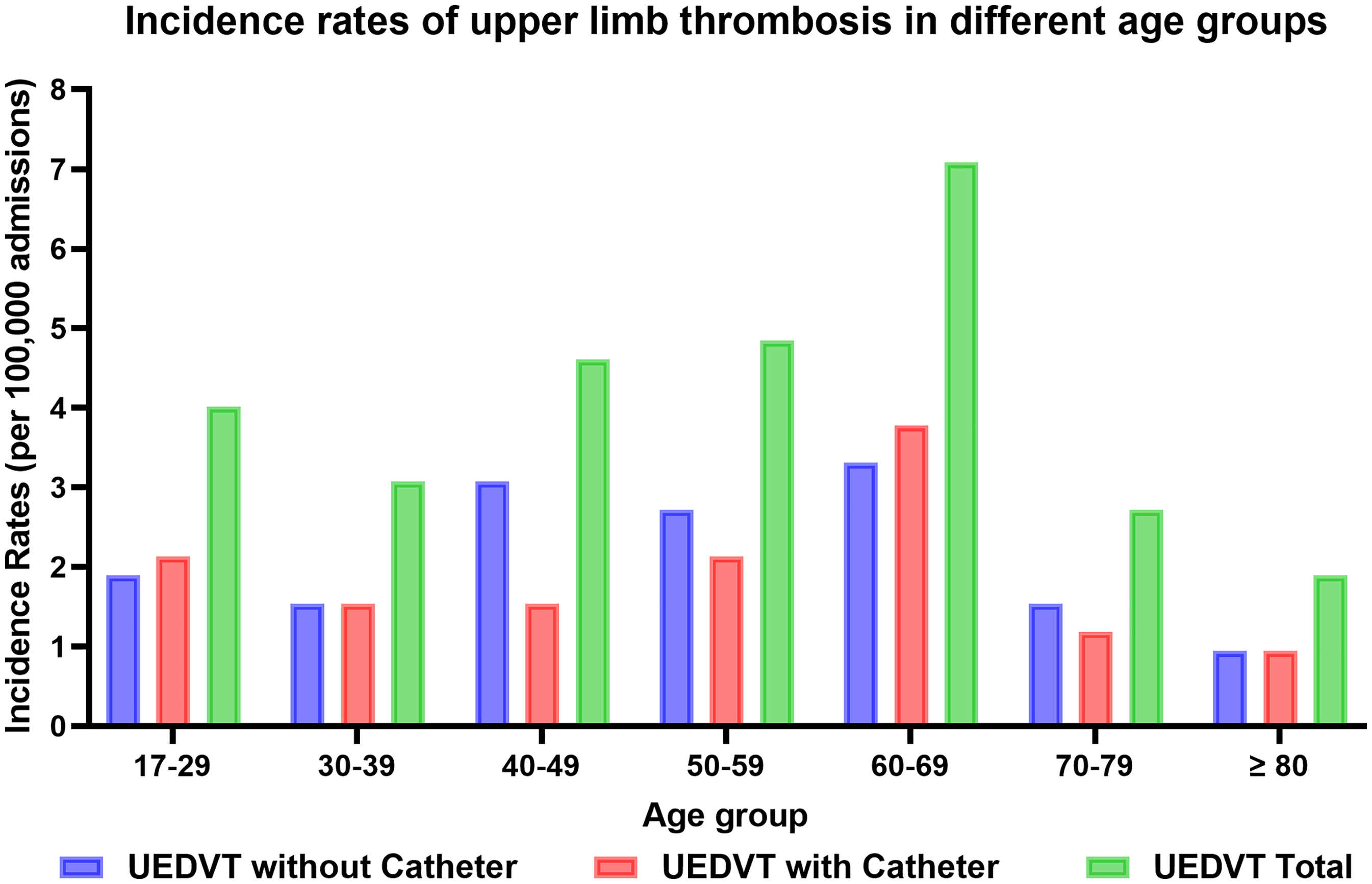
The incidence Rates of HA-UEDVT in Different Age Groups. Abbreviations: HA-UEDVT: Hospital-acquired upper extremity deep venous thrombosis; IR: Incidence rate.
Demographic and Clinical Characteristics of HA-UEDVT Population.

NOTE: Data are presented as n (%), mean ± SD or median (range).
Comparison was between surgical and medical patients. p values are calculated by chi-square test.
Comparison was between surgical and medical patients. p values are calculated by rank-sum test.
Time for diagnosis: days from admission to HA-UEDVT diagnosis.
Time for hospitalization: days from admission to discharge.
Abbreviations: HA-UEDVT, Hospital-acquired upper-extremity deep venous thrombosis; BMI, Body mass index; SD, Standard deviation; PE, Pulmonary embolism; n, Number of participants.
From 2009 to 2021, the cumulative incidence rates for HA-UEDVT and HA-LEDVT are shown in Figure 3. The 13-year cumulative incidence rate of HA-UEDVT was much lower than that of HA-LEDVT (0.31 vs 2.28 per 1000 inpatients). The incidence rate of HA-UEDVT peaking in 2015 and 2016; the incidence rates significantly decreased in 2019 and 2021. Detailed incidence trends are shown in Table S2.

The Trends of Annual Incidence Rates of HA-UEDVT and HA-LEDVT. Abbreviations: HA-UEDVT: Hospital-acquired upper extremity deep venous thrombosis; HA-LEDVT, Hospital-acquired lower extremity deep venous thrombosis; Sp: Surgical patient; Mp: Medicine patient; Tp: Total patient.
We collected HA-UEDVT events from 28 major clinical departments. As shown in Figure 4, the departments with the highest incidence rates were the departments of respiratory and gastroenterology, with 1.13 and 1.22 per 1000 inpatients, respectively, while HA-LEDVT incidence was the highest in the emergency medicine and geriatrics, with 15.72 and 11.97 per 1000 inpatients, respectively. No HA-UEDVT events were observed in the hepatology, endocrinology, traditional Chinese medicine, pain medicine, arthritis clinic & research center, spinal surgery, neurosurgery, obstetrics, and otolaryngology. Interestingly, the incidence of HA-UEDVT was high in departments where the incidence of HA-LEDVT was very low, such as hematology, breast disease surgery, and thoracic surgery, with these three departments accounting for 51% of all HA-UEDVT cases. Detailed data are provided in Table S3.

The Incidence Rates of HA-UEDVT and HA-LEDVT in Each Department Over 15 years. Abbreviations: HA-UEDVT: Hospital-acquired upper extremity deep venous thrombosis; HA-LEDVT, Hospital-acquired lower extremity deep venous thrombosis; IR: Incidence rate.
Related Factor Analysis for HA-UEDVT
Related Factors for HA-UEDVT
In Figure 5, Lasso regression results indicated that having a CVC and breast cancer were the strongest predictive factors of HA-UEDVT, with β values of 4.81 and 4.68, respectively, while concurrent HA-LEDVT had a β value of 3.11.

The Results of Lasso Regression Analysis for Related Factors of HA-UEDVT. (A) Cross-Validation Curve; (B) Variation Characteristics of the Coefficient of Variables; (C) Dependent Variable β Value Plot. Abbreviations: d: Days; HA-UEDVT: Hospital-acquired upper extremity deep venous thrombosis; LEDVT: Lower extremity deep venous thrombosis; ICU: Intensive care unit; PE: Pulmonary embolism; ADL: Activities of daily living.
Comorbidities for HA-UEDVT
The most common comorbidities in both HA-UEDVT groups were hypertension, hematologic neoplasms, diabetes, and breast neoplasms, as shown in Table 2. Compared to the non-CVC-associated HA-UEDVT group, the CVC-associated HA-UEDVT group had a higher incidence of hematologic neoplasms (OR = 2.31, 95% CI: 1.30 to 4.08) and lung neoplasms (OR = 3.28, 95% CI: 1.32 to 8.19), while breast neoplasm was less common (OR = 0.32, 95% CI: 0.15 to 0.68). Compared to the HA-LEDVT group, the non-CVC-associated HA-UEDVT group was significantly more likely to have hematologic neoplasms (OR = 14.99, 95% CI: 8.80 to 25.56), lung neoplasms (OR = 6.41, 95% CI: 2.64 to 15.54), and breast neoplasms (OR = 64.83, 95% CI: 30.80 to 136.42), but less likely to have aortic diseases (OR = 0.43, 95% CI: 0.21 to 0.89) or trauma of upper extremities (OR = 0.08, 95% CI: 0.02 to 0.32).
Comorbidities in Patients Diagnosed with DVT.

NOTE: Data are presented as n (%).
Comparison was between CVC-related and non-CVC-related HA-UEDVT patients. p values are calculated by chi-square test.
Comparison was between non-CVC-related HA-UEDVT and HA-LEDVT patients. p values are calculated by chi-square test.
Kidney disease: including acute kidney injury, chronic kidney disease, nephritis, hydronephrosis, etc.
Hemorrhagic diseases: including gastrointestinal hemorrhage, cerebral hemorrhage, hemorrhage in other organs, etc.
Infectious/contagious diseases: including diseases of various organs caused by bacterial (including special bacteria such as tuberculosis and tetanus), fungal, or viral infections, such as bacterial pneumonia, hepatitis B, hepatitis C, acute/chronic pyelonephritis, viral encephalitis, tuberculosis of various systems, abscesses of various systems, sexually transmitted diseases, etc.
Non-neoplastic gastrointestinal disease: including inflammatory bowel diseases, gastroesophageal reflux disease, peptic ulcer diseases, irritable bowel syndrome, constipation, etc.
Rheumatic immune diseases: including rheumatoid arthritis, systemic lupus erythematosus, Sjögren's syndrome, ankylosing spondylitis, etc.
Aortic diseases: including aortic aneurysm, aortic dissection, aortic stenosis, congenital aortic anomaly, etc.
Trauma of upper extremities: including fractures, dislocations, and soft tissue injuries of the hands, forearms, upper arms, and shoulder joints caused by external violence, such as falling, crashing and cutting.
Abbreviations: DVT, Deep venous thrombosis; CVC, Central venous catheter; HA-UEDVT, Hospital-acquired upper-extremity deep venous thrombosis; HA-LEDVT, Hospital-acquired lower extremity deep venous thrombosis; n, Number of participants; OR, Odds ratio; CI, Confidence interval.
Preventive Treatments
The primary preventive measures before DVT occurrence were subcutaneous or oral anticoagulation therapy (shown in Table 3). There were no statistically significant differences between the two HA-UEDVT groups in prophylactic treatments (35.7% vs 25.2%, p = .077). Compared to the HA-LEDVT group, the prophylactic treatments in HA-UEDVT group were less frequent (30.1% vs 82.4%, p < .001). The number and proportion of patients with different types of neoplasm who developed HA-UEDVT and received prophylactic anticoagulation are shown in Table S4.
Prophylactic Treatments Before DVT Occurrence During Hospitalization.

NOTE: Data are presented as n (%).
Comparison was between CVC-related and non-CVC-related UEDVT patients. p values are calculated by chi-square test.
Comparison was between HA-UEDVT (both CVC and non-CVC) and HA-LEDVT patients. p values are calculated by chi-square test.
Abbreviations: DVT, Deep venous thrombosis; CVC, Central venous catheter; HA-UEDVT, Hospital-acquired upper-extremity deep venous thrombosis; HA-LEDVT, Hospital-acquired lower extremity deep venous thrombosis; LMWH, Low molecular weight heparin; DOACs, Direct oral anticoagulants; n, number of participants; OR, Odds ratio; CI, Confidence interval.
Discussion
Previous research on DVT has predominantly focused on the lower extremities, with less attention given to HA-UEDVT. In this single-center retrospective study, we report for the first time that the incidence trends, epidemiological characteristics, and preventive measures for HA-UEDVT over 13 years in a large medical center in China. This study highlights some important similarities and differences between HA-UEDVT and HA-LEDVT patients and may contribute to a better understanding of HA-UEDVT incidence, aiding in the development of appropriate thrombosis prevention policies and clinical guidelines for future inpatients.
Early reports of HA-UEDVT annual incidence indicated 0.02 cases per 1000 people. 9 A 2012 study estimated the annual incidence at 0.04 - 0.1 cases per 1000 people. 3 The most recent prospective study from France reported an annual incidence of 0.098 cases per 1000 people. 4 Our study identified 239 HA-UEDVT events among 779,848 eligible inpatients over 13 years, with an average annual incidence of 0.019 cases per 1000 people. The annual incidence peaked in 2015 and 2016 at 0.029 cases per 1000 people, consistent with the broader understanding that the widespread use of CVC, amelioration in imaging techniques, and the increase in cancer patients corresponded with an increase in HA-UEDVT incidence.10-13 Examining recent incidence trends, we found the incidence of HA-UEDVT increased in 2020, this phenomenon may be attributed to the epidemic prevention policy of COVID-19 pandemic in China, 14 making the number of hospital admissions for patients with non-urgent treatment of benign diseases decreased significantly in 2020, which was also reflected in the total number of hospitalizations in 2020 (50,972 people) (Table S2). Compared to previous references, our study's annual incidence is relatively low, possibly due to the exclusion of outpatient data and the lack of long-term follow-up of discharged patients. This study provides statistical data on the annual incidence of HA-UEDVT at a tertiary medical center in China. While these data may have certain regional and hospital-specific characteristics, they also offer some value for the development of HA-UEDVT prevention and treatment strategies in other hospitals of the same level within China. At the same time, we also need other higher-quality multicenter studies to formulate Chinese national prevention and treatment guidelines for HA-UEDVT.
After literature searching, no relevant studies regarding the incidence of HA-UEDVT in each department were found, and our data results may play an early warning role for relevant high-risk departments. The overall incidence of HA-UEDVT in medicine departments is 3.13 times higher than those of surgery, mainly because there are a large number of patients in department of respiratory, gastroenterology and hematology, which have a large number of patients with malignant tumors and infectious diseases causing blood hypercoagulability and thrombosis. 15 Secondly, the length of hospitalization in medicine departments is longer, which also increases the risk of HA-UEDVT. Among surgical departments, the highest incidence is breast disease surgery and thoracic surgery, also due to the high number of breast and lung cancer patients. This is consistent with the risk factors, breast cancer is the highest risk of malignant tumors, followed by leukemia and lung cancer, and most of the above high incidence departments need to implant CVC due to chemotherapy and postoperative parenteral nutrition. No HA-UEDVT events were reported in the departments of hepatology, endocrinology, traditional Chinese medicine, pain medicine, osteoarthrosis, spinal surgery, neurosurgery, obstetrics, and otolaryngology, there are following possible reasons: 1), the incidence rate of HA-UEDVT is relatively low; 2), in our center, the chemotherapy for liver cancer patients is not conducted by the hepatology department but is instead managed by the hepatobiliary surgery department. Additionally, there are very few cancer patients receiving treatment in the neurosurgery and otolaryngology departments. The departments with high incidence of HA-UEDVT and HA-LEDVT are significantly different, which also suggests that the related factors of the two are different, and the HA-LEDVT events are mostly caused by advanced age and bed immobility of traumatic diseases. 16 The surgical departments have more invasive operations than those in the medical departments, but the incidences of HA-UEDVT and HA-LEDVT are lower, which seems to be contrary to cognition, this outcome may be related to the emphasis and education of the surgical system on the prevention and treatment of DVT.
This study compared the related factors of HA-UEDVT and non-HA-UEDVT and found that placing a CVC in cancer patients is a strong predictor of HA-UEDVT. This effect may be explained by propensity score matching, which provides more rigorous adjustment for selection bias than standard multivariate analysis. A study from the Registro Informatizado de Enfermedad Trombo Embólica (RIETE) registry reported that 36.3% of HA-UEDVT patients had active malignant tumors at diagnosis. If catheter patients were excluded, 25% of HA-UEDVT patients had active malignant tumors at diagnosis. 6 In our study, 61.5% of HA-UEDVT patients had active malignant tumors at diagnosis, and 52% of non-CVC-related HA-UEDVT patients had active malignant tumors at diagnosis. Compared to previous studies, our results suggest a higher proportion of HA-UEDVT cases with concurrent malignant tumors, which warrants further prospective research. Different malignancies have varying degrees of association with HA-UEDVT. According to a review from the California Cancer Registry, the highest HA-UEDVT risks are associated with leukemia (1.7%), followed by gastric cancer (1.4%), lymphoma (1.3%), and pancreatic cancer (1.1%). 17 In our study, the Lasso regression analysis shows that breast cancer is the strongest related factor, followed by leukemia and lung cancer, with the β values of 4.68, 2.69, and 1.85, respectively. The reasons for this result are considered to be the common use of indwelling intravenous catheters in the departments of breast disease surgery, hematology and thoracic surgery due to the use of chemotherapy drugs, and the need for indwelling catheters in the departments of gastrointestinal surgery and hepatobiliary surgery due to postoperative parenteral nutrition support. However, as shown in Table 2, we found that compared with the non-CVC-related HA-UEDVT group, CVC-associated HA-UEDVT combinations are more common in hematological neoplasia and lung tumors, while less common in breast cancer, suggesting that the higher risk due to breast cancer is less associated with CVC. This strong association between breast cancer and HA-UEDVT, considering the presence of activated specific molecular pathways or the use of chemotherapeutic agents, and the underlying mechanism needs further verification. We also found that several traditional risk factors for HA-LEDVT, such as post-traumatic internal fixation surgery, knee and hip replacement surgery, advanced age, and prior history of PE, did not contribute to HA-UEDVT. In Lasso analysis, patients with HA-LEDVT also had a higher risk of HA-UEDVT. The risk of HA-UEDVT should be evaluated after HA-LEDVT is found in clinic.
In 2016, the American College of Chest Physicians issued ACCP-10 Guidelines for Antithrombotic Therapy and Thromboprophylaxis, which updated the anticoagulant treatment for VTE disease. The guidelines recommended that patients should be stratified for VTE risk and bleeding risk assessment using Caprini or Pauda, IMPROVE VTE Risk Assessment Scale and Bleeding Risk Assessment Scale. For patients with moderate to high VTE risk, drug or mechanical prophylaxis is recommended. 18 In present study, 30.1% of patients received in-hospital thrombosis-related preventive measures, with subcutaneous injection of LMWH being the main method before HA-UEDVT detection, but it was still significantly less than HA-LEDVT (30.1% vs 82.4%, p < .001), this situation can be attributed to the lack of a general consensus on prophylactic anticoagulation treatment for UEDVT, 1 with recommendations for UEDVT prevention only available for certain types of cancer. 19 Previous literature does not support the routine use of thromboprophylaxis for CVC in cancer patients. 20 An open-label randomized trial conducted by Young et al. 21 suggested that prophylactic use of warfarin did not benefit cancer patients with CVCs. In contrast, a randomized, controlled, phase III study by Lavau-Denes et al. 22 indicated that prophylactic anticoagulation significantly reduced the incidence of catheter-related and non-catheter-related DVT in cancer patients, without an increase in serious side effects. Therefore, the use of anticoagulant prophylaxis in cancer patients with CVCs remains controversial. More studies are needed in the future to determine convenient, effective, and safe thrombosis prevention protocols for CVCs. Besides, HA-UEDVT may have some significant complications, including PE and loss of vascular access. In this study, there were 9 cases of PE in the CVC-related HA-UEDVT group, while the non-CVC-related HA-UEDVT group did not have any PE events. Chi-square test was performed to compare the outcome of PE complicated by the CVC-related HA-UEDVT group and the HA-LEDVT group (OR = 1.29; 95% CI: 0.64 to 2.60), showing no statistical significance. These results suggest that the likelihood of PE occurrence in CVC-associated HA-UEDVT group is similar to those in HA-LEDVT group, so we urgently need to raise awareness of anticoagulant therapy in CVC-associated HA-UEDVT group. The duration of anticoagulant therapy depends on the time of catheter removal. A cohort study of 74 CVC-related HA-UEDVT patients observed the results of anticoagulant therapy after 3 months without catheter removal and found no recurrence of VTE in any patient after 3 months. 23 Guidelines from CHEST, ISTH, NCCN, and ESC recommend anticoagulation for at least 3 months, with continuation of treatment as long as the catheter remains in place after 3 months of anticoagulation therapy.24-27 Various anticoagulation options are available, but there have been no randomized controlled trials to observe the efficacy of different anticoagulants for HA-UEDVT. In the past, vitamin K antagonists (VKA), represented by warfarin, and LMWH were the most common outpatient anticoagulation treatments, but the use of warfarin has decreased over time due to newer data showing that LMWH is superior to warfarin in malignancy associated thrombus, while UEDVT has a stronger association with malignancy.28,29 In our study, warfarin accounted for only 1.6% of HA-UEDVT treatments over 13 years. Direct oral anticoagulants (DOACs) including dabigatran, rivaroxaban, apixaban, and edoxaban have become increasingly popular due to their ease of use and expanding indications, but direct data on the efficacy and safety of DOACs in HA-UEDVT are limited to very small studies.30,31 Among our 239 HA-UEDVT patients, 5 patients received DOACs in-hospital, with no recurrence of thrombosis within 1 month. More studies are needed in the future to evaluate their use in this field.
Limitations
This study is a single-center retrospective case-control study, and the explanation of results is affected by data interpretation bias. More prospective multicenter studies are needed to validate the findings. HA-UEDVT is very rare, and even with 13 years of data collection at a single center, the sample size is still small. More original data or high-quality meta-analyses are needed in the future. Due to the longtime span of this study, the type of venous catheter was not distinguished, that is, peripheral implanted central venous catheter (PICC) or CVC. The differences between these two types of venous catheter have been statistically analyzed in a previous study, the incidence of DVT in PICC patients is significantly higher than that in CVC patients, and although PICC catheters have a longer life expectancy, they need to be removed earlier.32,33 In the future, the effects of catheter material, lumen diameter, insertion site and tip position on HA-UEDVT should be considered. Some patients classified as non-CVC-related HA-UEDVT may have had their CVC removed within 30 days before thrombosis was detected, which may have caused some differences between LEDVT and non-CVC-related HA-UEDVT patients.
Conclusion
HA-UEDVT mainly occurs in medicine departments, and its incidence has shown a downward trend in recent years. Having CVC, breast cancer, and HA-LEDVT are highly associated with HA-UEDVT occurrence. There is no statistically significant difference in the incidence of PE between the CVC-related HA-UEDVT group and the HA-LEDVT group, indicating the need for more high-quality prospective studies to guide HA-UEDVT prevention.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251365626 - Supplemental material for An Analysis of Incidence, Related Factors and Prophylactic Treatments of Hospital-Acquired Upper Extremity Deep Vein Thrombosis: A Single-Center 13-Year Retrospective Study
Supplemental material, sj-docx-1-cat-10.1177_10760296251365626 for An Analysis of Incidence, Related Factors and Prophylactic Treatments of Hospital-Acquired Upper Extremity Deep Vein Thrombosis: A Single-Center 13-Year Retrospective Study by Xiao Wang, Yezu Liu, Jing Wang, Chun Bai, Xiaoming Zhang, Xuemin Zhang, Qingle Li, Wei Li, Jingjun Jiang, Yang Jiao and Tao Zhang in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Ethics Approval
Ethical approval to report this case series was obtained from Peking University People's Hospital (2024PHB321).
Informed Consent
Verbal informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
Author Contributions
Conception and design: XW, YZL, JW, CB, XMZ, XMZ, QLL, WL, JJJ, YJ, TZ
Analysis and interpretation: XW, JW, CB, XMZ, XMZ, QLL, WL, JJJ, YJ, TZ
Data collection: XW, XMZ, XMZ, QLL, WL, TZ
Writing the article: XW, YZL, JW, TZ
Critical revision of the article: XW, YZL, JW, XMZ, XMZ, QLL, WL, JJJ, YJ, TZ
Final approval of the article: XW, YZL, JW, XMZ, XMZ, QLL, WL, JJJ, YJ, TZ
Statistical analysis: XW, JW, CB, XMZ, XMZ, TZ
Obtained funding: TZ
Overall responsibility: TZ
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Xiaomi Corporation through an Industry-Academia Collaboration (Grant No. 2025-Z-01), and Peking University People's Hospital Scientific Research Development Fund (Grant No. RDL2024-02).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
