Abstract
Background:
Peripherally inserted central venous catheters (PICCs) are widely used in patients with cancer. Catheter usage is one of the risk factors for venous thromboembolism. We aimed to scrutinize the incidence and risk factors for PICC-related upper extremity venous thrombosis (UEVT) in patients with lung cancer receiving chemotherapy.
Patients and Methods:
We performed a retrospective cohort study of patients with lung cancer with PICC insertion undergoing chemotherapy. Symptomatic PICC-UEVT was diagnosed by ultrasound. The relationship between chemotherapeutic agent exposure and PICC-UEVT was evaluated. Patient-, catheter-, and insertion-related factors were analyzed in univariable and multivariable logistic regression to identify significant independent risk factors for PICC-UEVT in patients with lung cancer.
Results:
A total of 328 patients with lung cancer having PICC undergoing chemotherapy were included, for a total of 34 895 catheter days. Seventeen (5.2%) patients developed PICC-related UEVT, with an incidence of 0.49 per 1000 catheter days. In multivariable logistic analysis, advanced disease was shown to be a significant risk factor for PICC-UEVT (odds ratio [OR]: 4.9; 95% confidence interval [CI]: 1.4-16.7; P = .011). Patients treated with etoposide had a higher risk of PICC-related UEVT (OR: 3.6; 95% CI: 1.1-12.1; P = .042). Patients were followed up after PICC removal for a median duration of 246 days. None of the patients developed pulmonary embolism.
Conclusion:
Patients with lung cancer harboring an advanced disease or treating with etoposide were at higher risk of PICC-UEVT.
Keywords
Introduction
Venous thromboembolism (VTE) is a common complication for patients with cancer. The etiologies of VTE are numerous, and catheter usage is one of the risk factors for VTE. 1,2 Peripherally inserted central venous catheters (PICCs) are widely used in patients with cancer. 3,4 One of the foremost complications related to PICC is upper extremity venous thrombosis (UEVT), 5,6 which may result in pulmonary embolism (PE) and serious clinical outcome. 7 Patients with cancer having PICC tend to develop catheter-related UEVT, 8,9 and the incidence of PICC-related UEVT in patients with cancer was 6% to 15%. 3,4 As the leading cause of cancer death, lung cancer may account up to 14% of cancer-related thrombotic cases, 10 and 3% to 30% of patients with lung cancer may develop vein thrombosis, 11 –13 especially among patients receiving chemotherapy. 14 However, while risk factors and other clinical characteristics of PICC-related UEVT had been evaluated generally, very little work had been focused on patients with lung cancer receiving chemotherapy. The true incidence of PICC-related UEVT in patients with lung cancer remains unclear, and there was a lack of reliable data on the related risk factors. The purpose of this retrospective cohort study was to determine the incidence and risk factors related to symptomatic PICC-related UEVT in patients with lung cancer receiving chemotherapy.
Patients and Methods
Peking Union Medical College Hospital (PUMCH) is a 2000-bed teaching hospital affiliated to Peking Union Medical College and consistently ranks as the top general hospital in China. We conducted a retrospective cohort study of patients with lung cancer having PICC insertion undergoing chemotherapy treatment from January 1, 2008, to December 31, 2013, at PUMCH.
The study was approved by the Ethics Committee of PUMCH (approval number: S-K027). Patients with lung cancer were included if they (1) received an intravenous chemotherapeutic agent with PICC and (2) had a complete PICC data (from insertion to removal). Patients were excluded if they had (1) prophylactic systemic anticoagulation or (2) anticoagulant therapy at the time of the PICC’s insertion. All patients were followed until PICC was removed, and most of the patients were still followed at clinic after PICC was removed.
Insertion and Maintenance of PICC
All PICCs were inserted using sterile technique by PICC nurses of Department of Parenteral and Enteral Nutrition in PUMCH. The insertion site was determined by PICC nurses after a preliminary ultrasound scan of the upper extremity veins. Basilic vein was the first choice. The PICC tip position was confirmed immediately by routine chest radiograph after placement. The PICC placement was considered optimal if the tip of the catheter was located in the middle and distal superior vena cava (SVC) and cavoatrial junction and acceptable if located in proximal SVC. When the tip was located out of SVC, it was defined as malposition.
All PICCs were routinely maintained by PICC nurses using sterile technique weekly. The catheters were flushed with 10 mL normal saline and then sealed with 2 mL heparin saline (50-100 IU) according to the maintenance protocol.
Definition
The PICC-related UEVT was defined as an UEVT within the veins where PICCs were placed or were adjacent to. Upper extremity superficial venous thrombosis (UESVT) was defined as the condition when UEVT occurred only in superficial vein. Upper extremity deep venous thrombosis (UEDVT) was defined as the condition when UEVT occurred in deep vein. In this retrospective study, all UEVT were symptomatic venous thrombosis. Once the patients experienced clinical manifestations such as swelling, pain, or tenderness at the PICC site, venous duplex ultrasonography was performed. The UEVT was diagnosed by duplex ultrasound imaging, including no compressibility of the vein, presence of visible thrombus, and lack of Doppler-detected venous flow. The incidence of PICC-related UEVT was calculated as the total number of PICC-related UEVTs divided by the total number of PICCs placed (%), or divided by 1000 PICC days (/1000 days of use). Length of PICC day was calculated as the number of days between PICC insertion and removal. The time of developing UEVT was calculated as the number of days between PICC insertion and the diagnosis of UEVT. The length of follow-up was calculated as the number of days between PICC removal and the last time of visit to clinic.
Data Collection
All patients were identified from our independent electronic PICC database. Several potential risk factors for catheter-related thrombosis were collected. Patient-related characteristics were collected from medical record, including age, gender, histology (non-small cell lung cancer (NSCLC) or small cell lung cancer), stage (I-IIIA, IIIB-IV), and comorbidities. All chemotherapeutic agents were included, such as platinum-based regimens, taxanes, and antimetabolite-based regimens. Catheter-related and insertion-related characteristics were collected from PICC database, including tip configuration (valve), malposition, length of PICC day, insertion vein, insertion technique, left-sided insertion, and previous PICC insertion. Data were abstracted independently by 2 trained investigators. Disagreements on information were resolved by consensus or retrieving further information from medical records.
Statistical Analysis
Univariable and multivariable logistic regression analyses were conducted to calculate unadjusted and adjusted odds ratio (OR) and their 95% confidence interval (Cis) for the association between all risk factors and PICC-related UEVT. Any clinical characteristics with a value of P < .05 in the univariable analysis was entered into a multivariable analysis. These results were presented as adjusted ORs with 95% CIs. All statistical tests were 2 sided, and P values < .05 were considered statistically significant. Statistical analysis was done with SPSS software (version 19, SPSS Inc, IBM, NY, USA).
Results
Between January 2008 and December 2013, 328 patients with lung cancer having PICC undergoing active chemotherapy were included, for a total of 34 895 catheter days (Table 1). The median age was 59 (range, 22 to 82 years), and 62% (203 of 328) of patients were male. In all, 91% (297 of 328) had NSCLC, 28% (92 of 328) had stage IIIB-IV cancer, and 8% (27 of 328) had a previous PICC insertion. All patients chose 4F catheters, and 66% (297 of 328) chose valve tip. In all, 93% (305 of 328) of the catheters were inserted in basilic vein and 52% (171 of 328) in left upper extremity (Table 1). In all, 62% of patients’ initial chemotherapy regimen was cisplatin-based regimen, followed by carboplatin-based regimen, and docetaxel-based regimen. The chi-square test demonstrated that patients treated with etoposide had a higher risk of PICC-related UEVT. Besides this, all other regimens were not significant (Table 2).
Patient Characteristics.
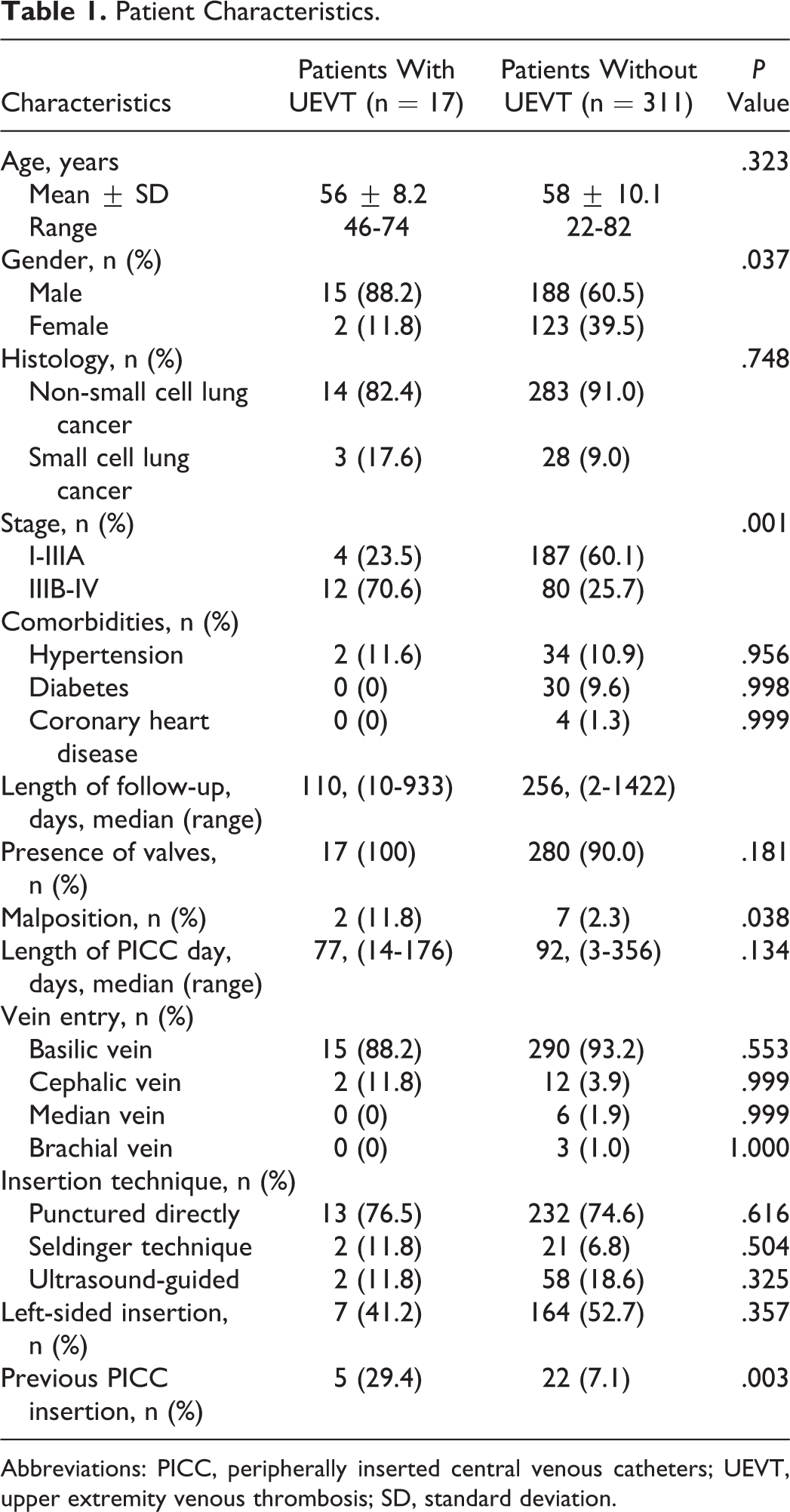
Abbreviations: PICC, peripherally inserted central venous catheters; UEVT, upper extremity venous thrombosis; SD, standard deviation.
Chemical Exposure Characteristics.
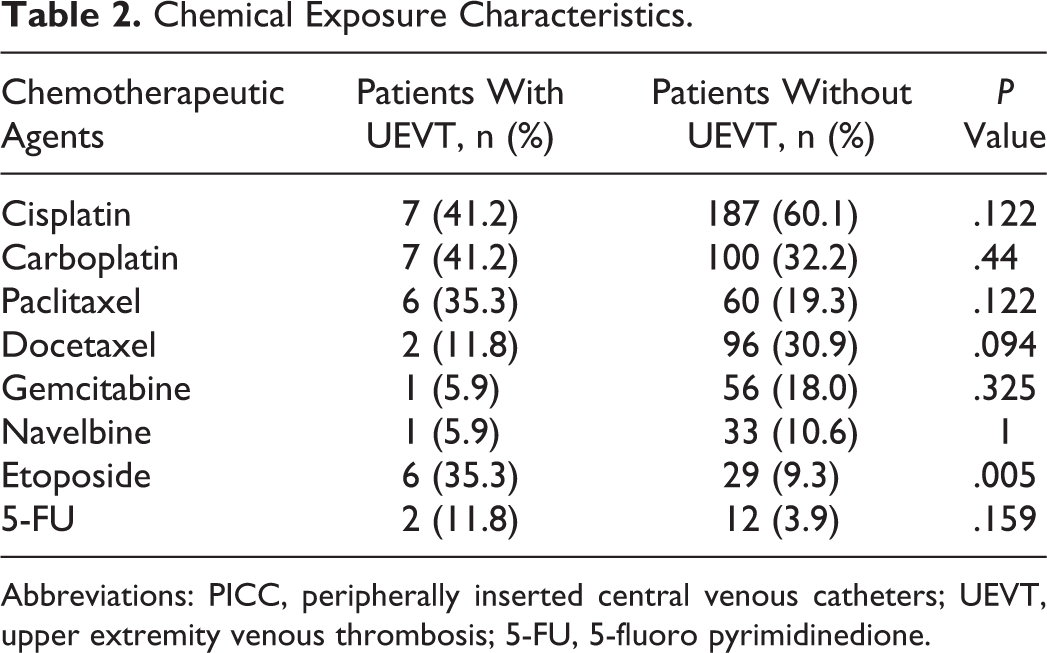
Abbreviations: PICC, peripherally inserted central venous catheters; UEVT, upper extremity venous thrombosis; 5-FU, 5-fluoro pyrimidinedione.
Of all 328 patients, 17 (5.2%) patients developed PICC-related symptomatic UEVT for a total of 1430 PICC days, with an incidence of 0.49 per 1000 catheter days. The median time of developing UEVT was 22 days (range of 3 to 174 days). In all, 29% (5 of 17) of patients developed UESVT and 71% (12 of 17) developed UEDVT. The UEVT occurred in a single vein in 53% (9 of 17) of patients, in more than 1 vein in 48%. The most common vein involved was subclavian vein. Most of the patients with UEVT developed phlebitis and edema of upper extremity. Varicose veins in chest wall were found in 1 patient with jugular venous thrombosis. Seventeen patients who developed PICC-UEVT were treated with low-molecular-weight heparin (LMWH) and followed until PICC was removed.
In all, 277 patients were followed up after PICC removal for a median duration of 246 days. During the follow-up period, 1 patient in control group developed DVT of lower limb, and 1 patient in UEVT group developed UEVT after another PICC insertion. None of the patients developed PE.
To examine the association of baseline characteristics with the development of UEVT, a univariate analysis was conducted. Factors such as age, histology, comorbidities, tip configuration, catheter days, insertion vein, insertion technique, and left-sided insertion were proven not significant predictors of UEVT. Gender, malposition, previous PICC insertion, and advanced disease were all related to a significant increase in risk of UEVT (Table 1). Multivariable analysis was then performed (Table 3). Advanced disease (IIIB-IV; OR: 4.9; 95% CI: 1.4-16.7; P = .011) was shown to be a significant risk factor for PICC-related UEVT in multivariable analysis. Patients treated with etoposide had a higher risk of PICC-related UEVT (OR: 3.6; 95% CI: 1.1-12.1; P = .042).
Multivariable (Adjusted) Analyses.
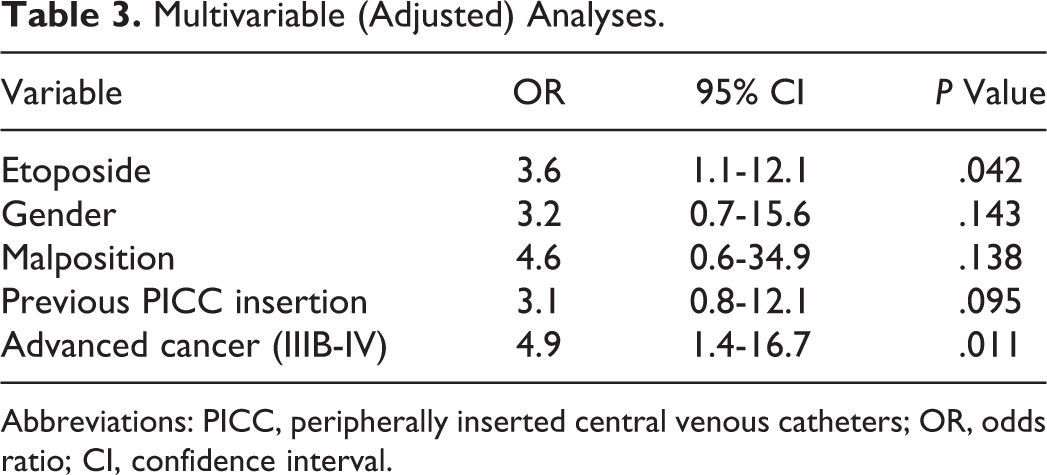
Abbreviations: PICC, peripherally inserted central venous catheters; OR, odds ratio; CI, confidence interval.
Discussion
The UEVT events were characterized in 328 patients with lung cancer using retrospective analysis. Seventeen (5.2%) patients developed PICC-UEVT. Patients with lung cancer harboring an advanced disease or treated with etoposide were at higher risk of PICC-UEVT.
Lung cancer is the cancer with the highest incidence in men and remains the most common cause of cancer-related death among men in China. As a long-term venous catheter for chemotherapy, PICCs were widely used in patients with lung cancer since the first PICC inserted in PUMCH in 1997 in China. This is the largest study to date to examine the incidence and predictive factors of PICC-related symptomatic UEVT in patients with lung cancer undergoing active chemotherapy in China. In our study, the incidence of PICC-related UEVT was 5.2% (UEDVT for 3.7%), a result that is consistent with prior studies. 9,15 Patient-related, catheter-related, and insertion-related risk factors for UEVT were analyzed, as well as the duration of follow-up, since these factors were reported to reflect the incidence of UEVT in patients with cancer. 9 In our study, advanced cancer was a strong risk factor for the development of PICC-related UEVT. This finding is consistent with previously reported literature. 8,16 Patients with lung cancer harboring an advanced disease and/or receiving chemotherapy were at higher risk of VTE. 17,18 Patients with cancer having prothrombotic gene mutations had higher risk of VTE. Arslan et al 11 reported that patients with lung cancer with homozygous MTHFR A1298C mutation significantly tend to develop DVT. In this retrospective analysis, gene mutations were not included and further studies are required. Other patient-related characteristics were not related to the development of VTE.
The association of age and VTE in patients with cancer is controversial. A retrospective study identified advanced age as a risk factor for VTE among patients whose mean age was 45.6. The mean age of patients with VTE was 57.2 while that of control group was 36.8. 19 In our study, the median age was 59, nearly one-third of patients were older than 65, and the result of stratified analysis was not statistically significant. The demographics also may give some inkling of the potential for bias, and other factors including chemotherapy in older patients may confound this risk. 18
Male gender was an independent predictor of implanted port-related VTE in a retrospective cohort study, but there were only 6 patients with lung cancer. 20 Lung cancer has the highest incidence among all cancers in Chinese men, and about two-thirds of patients in our study were male. Gender was only related to UEVT in univariable analysis but not an independent risk factor in multivariable analysis. The incidence of venous thrombosis was higher in patients with NSCLC or adenocarcinoma. In our study, 82% (14 of 17) of UEVT was developed in patients with NSCLC and 47% (8 of 17) in patients with adenocarcinoma. However, univariable analysis showed that NSCLC and adenocarcinoma were not significantly related to UEVT. Studies with larger samples are needed.
Cisplatin-based regimens had been found to be related to additional risk of VTE in patients with lung cancer, 21,22 however, the proportion of catheter-related upper extremity DVT has not been well described. In a cohort study of 243 patients with cancer, no association was found between chemotherapy agents (cisplatin) and catheter-related thrombosis, but there were no patients with lung cancer. 23 In our study, patients treated with etoposide had a higher risk of PICC-related UEVT. Etoposide was usually administered from day 1 to day 5 as a 180-minute infusion every day, which may lead to a limited motion of the upper extremities with PICC and relate to UEVT. In view of the small sample size, we explore the trend of chemotherapeutic regimen related with PICC-UEVT. Studies with larger samples are needed.
Although advanced cancer and chemotherapy increased the risk of UEVT, PICCs are essential to patients with lung cancer receiving chemotherapy, therefore we sought to determine the controllable risk factors such as catheter-related and insertion-related factors. Previously described risk factors for catheter-related and insertion-related thrombosis were not significant determinants in our analysis. Larger diameter of PICC was related to a higher risk of UEVT. 24,25 Large number of prospective studies found that increasing catheter size is significantly related to increased risk of UEVT. 26,27 As 5F and 6F catheters were not used widely in China, all PICCs in our study were 4F catheters. Lumen may interact with diameter, 28 so diameter and lumen were not included in logistic regression analysis model. In our study, previous PICC insertion was related to a significant increased risk of UEVT in univariate analysis. It is mainly because a second PICC insertion was always performed in patients with advanced cancer or metastasis, but the result has not reached statistical significance in multivariable analysis.
In our study, all PICCs were routinely flushed with normal saline and then sealed with heparin saline by PICC nurses using sterile technique every week. It was reported that UEVT rates can be minimized if catheters were maintained appropriately. 29 According to American College of Chest Physicians guidelines, in our study, PICCs were not removed immediately in most patients with PICC-related UEDVT. 30 The treatment was to keep the catheter in place and to add systemic anticoagulants. 31 Considering that pulmonary embolus from superficial venous thrombosis is very rare, PICCs out of use were removed immediately in most patients with PICC-related UESVT. Systemic anticoagulants was added if there was an ongoing need for the catheter in these patients. Anticoagulant therapy could alleviate acute symptoms and prevent PE effectively, 32 and LMWH was safe and efficient. 33 Catheters were removed immediately in patients with contraindications for anticoagulation. 34
There are several limitations in our study. First, this is a retrospective study and patients with PICC-related asymptomatic UEVT were not included, the incidence of which may be 14% to 30%. 9,35,36 Although most of the patients were followed up after PICC removal for a long time and none of the patients developed PE, a prospective study including the asymptomatic UEVT samples is further needed. Second, the number of PICC-related UEVT is relatively small and the demographics may also give some inkling of the potential for bias. A research with larger sample is needed.
Nonetheless, to our knowledge, this is the largest study to date to examine the incidence and predictive factors for PICC-related symptomatic UEVT in patients with lung cancer undergoing active chemotherapy. This study could help clinicians select an appropriate venous chemotherapeutic catheter for patients with lung cancer. It would be of value to be able to predict patients with advanced lung cancer with PICC lines receiving chemotherapy might benefit from VTE prophylaxis. 37
Conclusion
In conclusion, patients with lung cancer harboring an advanced disease or treated with etoposide were at higher risk of PICC-UEVT. Further prospective and large sample studies are needed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
