Abstract
Background
Antiphospholipid Syndrome (APS) is an autoimmune condition that increases thrombosis risk. Although Vitamin K antagonists (VKA) are standard treatment, interest in direct oral anticoagulants (DOAC) has grown. Recent studies evaluated DOAC across all APS subgroups. This review updates our understanding of DOAC's efficacy and safety compared to VKA in preventing thrombotic complications, integrating new findings into the literature.
Methods
A systematic review across PubMed/Medline, Embase, and Cochrane was conducted in accordance with PRISMA guidelines. The quality of randomized controlled trials (RCTs) and cohort studies was assessed using the Cochrane Risk of Bias tool and the Newcastle-Ottawa Scale, respectively. Meta-analysis calculated odds ratios (OR) using the random effect model for arterial and venous thrombotic and bleeding outcomes
Results
Of a total of 2385 records identified, the meta-analysis included 11 studies, comprising 5 RCTs and 7 observational studies, with a combined total of 1813 participants. The analysis revealed no significant difference in the odds of venous thromboembolism (VTE) between DOAC and VKA (OR = 0.80; 95% CI 0.41-1.56). However, a significant increase in thromboembolism recurrence was noted in triple-positive APS patients using DOAC compared to VKA (OR = 3.62; 95% CI 1.10-11.98). The risk of major bleeding was similar between DOAC and VKA (OR = 1.02; 95% CI 0.62-1.68). Additionally, the pooled analysis indicated a higher risk of arterial thrombosis with DOAC (OR = 6.21; 95% CI 2.06-18.76).
Conclusion
Overall, findings suggest DOAC are comparably safe but increase the risk of arterial thrombosis and VTE in triple-positive APS, favoring warfarin for prophylaxis.
Introduction
Antiphospholipid syndrome (APS) is a systemic autoimmune disease that manifests through both thrombotic and non-thrombotic complications in macrovascular and microvascular systems, as well as pregnancy complications that arise from the presence of persistent antiphospholipid antibodies (aPL), which include anticardiolipin antibodies, anti-β2 glycoprotein-I antibodies, and lupus anticoagulant.1,2 The estimated prevalence of APS in the general population ranges from 40 to 50 cases per 100 000 adults. 3
Thrombotic events are mainly due to the ability of aPL to directly induce thrombus formation and platelet activation. 2 After an initial thrombotic event, the risk of recurrence significantly increases by 10%–67%. 4 The likelihood of thrombosis appears to be influenced by the clinical and immunological characteristics of patients, with those who are triple positive for aPL facing the highest risk, estimated at 5.3% per year. 5
To mitigate the risk of thrombotic complications, long-term treatment with oral anticoagulants is essential for APS, particularly as secondary prophylaxis following the first thrombotic event. Vitamin K antagonists (VKA) have been the primary oral anticoagulant option for patients with APS. 6 However, over time, several issues with VKA have become evident, including warfarin resistance and unstable anticoagulation quality.7‐10 In the past decade, direct oral anticoagulants (DOAC) have gained significant attention due to their more predictable anticoagulant response, fixed dosing, and rapid onset and offset of action. The current role of DOAC in treating APS is defined by limited clinical trial data, along with information from manufacturers, case series, cohort studies, and expert consensus.11‐13 The 2020 International Society on Thrombosis and Haemostasis Scientific and Standardization Subcommittee recommended VKA over DOAC for APS patients. 14 The second update of the American College of Chest Physicians (CHEST) guidelines in 2021 further emphasized this recommendation, with panelists agreeing that DOAC should not be used in APS patients, especially those who are triple positive for aPL. 15 Since then, more evidence has emerged not included in these decisions. This systematic review aims to update previous knowledge and combine the most recent evidence evaluating the efficacy and safety of DOAC in preventing thrombotic complications compared to VKA.
Methods
The study was reported in accordance with the recommendations outlined in the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) protocol. 16 PRISMA 2020 Checklist is available in the Supplemental Material (Supplementary Table 1). The study protocol has been registered with PROSPERO under the registration number (CRD42024626338).
Search Strategy and Selection Criteria
We conducted a comprehensive search on PubMed/Medline, Embase, and Cochrane from inception to the first of December 2024 without language restriction. We also searched Google Scholar, the International Clinical Trials Registry Platform (ICTRP), The US Clinical Trials Registry, the Australian New Zealand Clinical Trials Registry (ANZCTR), the Clinical Trial Registry in Japan, International Standard Randomized Controlled Trial Number Registry (ISRCTN registry), EU Clinical Trials, Nederland's Trial Register, and Randomized Controlled Trial Number Registry (ISRCTN registry), EU Clinical Trials, Nederland's Trial Register, and the bibliographies of the included studies and recent review articles to identify any additional relevant studies. The search terms used were “'direct oral anticoagulant”, “novel oral anticoagulant”, “DOAC”, “factor Xa inhibitors”, “antithrombins”, “apixaban”, “rivaroxaban”, “dabigatran etexilate”, “dabigatran “, “edoxaban”, “betrixaban”, “warfarin”, “phenprocoumon”, “acenocoumarol”, “coumadin”, “vitamin K antagonist”, “antivitamin k”, antiphospholipid syndrome”, “anti-phospholipid antibody syndrome”, “antiphospholipid”, More details on search strategies, available in Supplemental Materials
Two independent reviewers (HA, AA) conducted the initial screening for eligible studies in three phases: first, evaluating the titles, followed by a review of the abstracts in the second phase. In the third phase, full texts were assessed according to the study inclusion criteria (Figure 1). Any disagreements between the reviewers were resolved through consensus or by consulting a third reviewer (LA). We included randomized controlled trials (RCTs), cohort studies, and case-control studies that compared the outcomes of the recurrence of arterial and venous thromboembolism, major bleeding, and clinically relevant nonmajor bleeding (CRNMB)

Flow Diagram for Selection of Studies Included in the Systematic Review and the Meta-Analysis.
Data Extraction and Quality Assessment
All required data were extracted using a pre-designed Excel sheet that included the following information: publication year, study design, sample size, and baseline characteristics of included patients, including age, gender, and treatment (type of drug, dose, and duration). Data regarding the subsequent outcomes was gathered from the two treatment groups: thrombotic recurrence (whether venous or arterial), bleeding (including the definition used), and mortality. Two reviewers independently assessed the risk of bias. The quality of the included RCTs was evaluated using the Cochrane Risk of Bias tool, 17 and observational studies were evaluated using the Newcastle-Ottawa Quality Assessment Scale. 18 Duplicate records were excluded using the reference manager Mendeley®. 19 Pre-proof articles, editorials, studies with different interventions, and reviews were also excluded from our study.
Outcomes
The two main efficacy outcomes were (1) arterial thrombotic events and (2) venous thromboembolic events. Additional efficacy outcomes included myocardial infarction, ischemic stroke or transient ischemic attack, pulmonary embolism (PE), deep vein thrombosis (DVT), and a composite of any arterial or venous thromboembolic events of the APS subgroups. The primary safety outcome focused on major bleeding, following the definition provided by the International Society on Thrombosis and Haemostasis. 20 CRNMB, as defined by the International Society on Thrombosis and Haemostasis, was also evaluated. 21 Reviews of definitions used in individual trials were conducted, and a standardized definition was employed across the studies wherever possible.
Statistical Analysis
A meta-analysis was performed utilizing a random-effects model to calculate odds ratios (ORs) and 95% confidence intervals (CIs) for all outcomes. Heterogeneity between studies was assessed using predictive interval and the I² statistic, with values of 25%, 50%, and 75% indicating low, moderate, and substantial heterogeneity, respectively. All statistical analyses were conducted using Comprehensive Meta-analysis Version 4. 22 The Egger's test and a visual assessment of the funnel plot were used for assessing publication bias. Egger's test was conducted only for meta-analyses comprising at least 5 studies. 23
Results
Study Characteristics
Out of 2385 records identified in the literature search, 468 articles remained for full-text evaluation after screening titles and abstracts and removing duplicates. The systematic review included 11 studies.24‐34 Four hundred and fifty-six were excluded because they did not align with the aim of this review regarding the interventions of interest and measured outcomes, or they reviewed articles that had unfitting study methodologies (ie, case series or case-control studies), editorials, study protocols, or clinical guidelines. (Figure 1). Among the included studies, four were randomized controlled trials (RCTs),24‐27 one was post hoc, 28 and the remaining six were observational studies,29‐34], with two being prospective31,34 and four retrospectives.29,31,32,33 The characteristics of the 11 included studies and a summary of their outcomes are illustrated in (Table 1). The quality assessment of the four RCTs was performed using the Cochrane Risk of Bias tool; details are available in the Supplemental Material (Table S5). Additionally, the NOS score range for the six observational studies included falls between 7 and 8, indicating good quality, details are also available in the Supplemental Material (Table S6).
Characteristics of the Included Studies (n = 11).
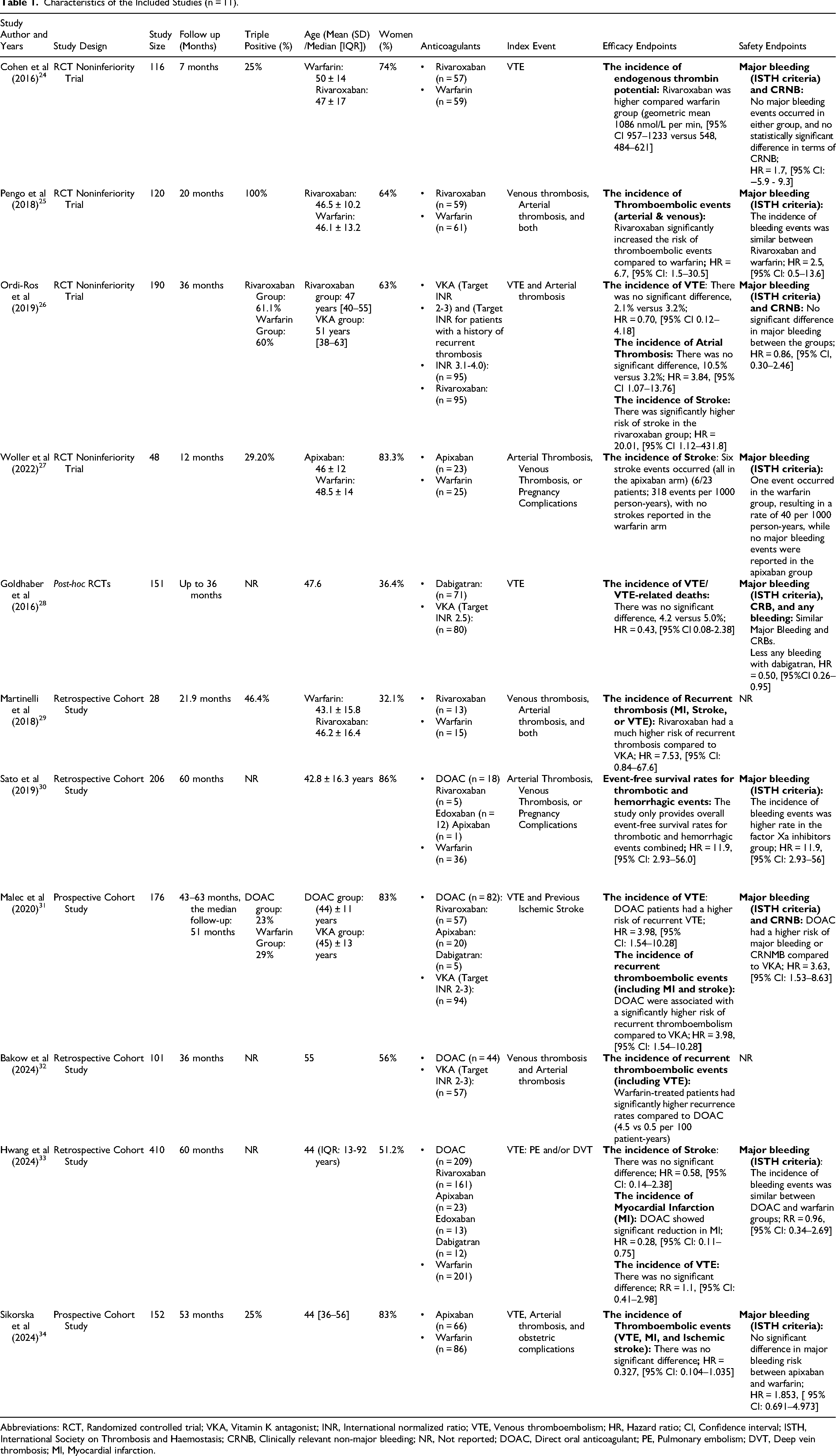
Abbreviations: RCT, Randomized controlled trial; VKA, Vitamin K antagonist; INR, International normalized ratio; VTE, Venous thromboembolism; HR, Hazard ratio; CI, Confidence interval; ISTH, International Society on Thrombosis and Haemostasis; CRNB, Clinically relevant non-major bleeding; NR, Not reported; DOAC, Direct oral anticoagulant; PE, Pulmonary embolism; DVT, Deep vein thrombosis; MI, Myocardial infarction.
Efficacy Outcomes
Arterial Thrombosis
The pooled analysis of arterial thrombosis in observational cohort studies showed no significant difference in the odds of arterial thrombosis between DOAC and VKA (OR = 0.84; 95% CI 0.31-2.31; I² = 49.9%, prediction interval (0.05, 13.43)). However, the pooled analysis of RCTs indicated a significantly increased risk of arterial thrombosis with DOAC compared to VKA (OR = 6.21; 95% CI 2.06-18.76; I² = 0%) (Figure 2).

Risk of Arterial Thrombosis in DOAC Versus VKA.
Myocardial Infarction
The pooled analysis of observational cohort studies revealed no significant difference in the odds of the outcome between DOAC and VKA (OR = 1.24; 95% CI 0.22-6.88; I² = 59.5%, prediction interval (0.00, 1144.42)). Similarly, the pooled analysis of the RCTs demonstrated no significant difference between the groups (OR = 7.62; 95% CI 0.39-150.78; I² = 0%, prediction interval (0.00, 1475.25)) (Figure S1).
Stroke
The combined analysis of observational cohort studies found no significant difference in the odds of stroke between DOAC and VKA (OR = 0.91; 95% CI 0.38-2.18; I² = 0%). The pooled analysis of RCTs demonstrated a significantly higher risk of stroke with DOAC compared to VKA (OR = 5.30; 95% CI 1.74-16.20; I² = 0%) (Figure S2).
VTE
There were no significant differences in the odds of VTE between DOAC and VKA in the observational studies analysis (OR = 0.75; 95% CI 0.33-1.73; I² = 36.7%, prediction interval (0.10, 5.86)), or RCTs analysis (OR = 0.90; 95% CI 0.30-2.69; I² = 0%) (Figure 3).

Risk of VTE in DOAC Versus VKA.
Subgroup Analysis of Recurrent Thromboembolism Based on the Subtype of APS
Triple-Positive Subgroup Analysis
The analysis of two observational cohort studies showed no significant difference in the odds of thromboembolism recurrence between DOAC and VKA (OR = 1.93; 95% CI 0.08-48.98; I² = 75.3%). The analysis of three RCTs showed a statistically significant increase in the odds of thrombosis recurrence with DOAC compared to VKA (OR = 4.00; 95% CI 1.10-14.50; I² = 17.2%, prediction interval (0.00, 203207.9)) (Figure 4).

Analysis of Recurrent Thromboembolism for Triple-Positive APS Subgroup.
Single/Double Positive Subgroup Analysis
The pooled analysis of two RCTs showed no significant difference in the odds of thromboembolism recurrence between DOAC and VKA (OR = 2.28; 95% CI 0.28-18.73; I² = 0%) (Figure S3).
Safety Outcomes
Major Bleeding
Observational cohort studies showed no significant difference in major bleeding odds between DOAC and VKA (OR = 1.06; 95% CI 0.57-1.97; I² = 0%), as did RCTs (OR = 0.94; 95% CI 0.40-2.21; I² = 0%) and the overall pooled analysis (OR = 1.02; 95% CI 0.62-1.68; I² = 0%) (Figure 5). Sensitivity analysis, excluding the Malec study, maintained no significant difference in observational studies (OR = 0.69; 95% CI 0.28-1.70; I² = 0%), and in RCTs (OR = 0.94; 95% CI 0.40-2.21; I² = 0%) (Figure S4).

Risk of Major Bleeding in DOAC Versus VKA.
CRNMB
A single observational cohort study showed no significant difference in the CRNMB odds between DOAC and VKA (OR = 1.55; 95% CI 0.66-3.64; I² = 0%). A similar finding was observed in the analysis of three RCTs (OR = 0.99; 95% CI 0.34-2.84; I² = 51.6%, prediction interval (0.00, 52929.01)) (Figure S5).
Publication Bias
Publication bias, assessed via Egger's regression test for funnel plot asymmetry, was not statistically significant for arterial thrombosis, stroke, and VTE outcome (P = .48, .89, .99 respectively). Analyses were limited to observational cohort studies with at least five included studies. Funnel plots were depicted in Supplementary Figures S6- S15.
Discussion
This study assessed the efficacy and safety of DOAC compared to VKA in preventing thrombotic complications in patients with APS. Our study's key findings revealed no significant difference in the overall risk of VTE, stroke, or major bleeding between DOAC and VKA in patients with APS. However, there was a higher risk of arterial thrombosis and myocardial infarction, but without statistical significance, in APS patients receiving DOAC compared to those receiving VKA. Additionally, our study showed an overall statistically significant increased risk of VTE recurrence in triple-positive APS patients receiving DOAC compared to VKA. Hence, our results do not support the use of DOAC, especially in high-risk APS patients such as those with triple positivity, and for preventing arterial thrombotic and myocardial infarction, in which VKA may offer better protection against thrombotic complications.
Our findings are consistent with the EULAR 2019 recommendations and the 2021 CHEST guidelines, both of which recommend against DOAC use in high-risk APS patients.12,15 Particularly, our meta-analysis confirms the higher risk of arterial thrombosis and myocardial infarction events reported in previous RCTs.24‐27 One RCT conducted by Pengo et al demonstrated increased thrombosis and bleeding events, including myocardial infarction, stroke, and major bleeding in the rivaroxaban group. 25 Similarly, our finding of increased arterial thrombosis in high-risk APS patients aligns with earlier meta-analyses and observational cohort studies, reinforcing the better protection of VKA in APS patients. 35 All of this evidence supports the use of VKA in high-risk APS patients rather than DOAC. High-risk APS patients include those with triple positivity or a history of arterial thrombosis.
In contrast, our study's results differ from some observational cohort studies, in which our study showed no statistically significant differences in VTE, stroke, or major bleeding rates between the two treatment groups.29‐34 The observational studies show more mixed results with moderate to high heterogeneity. These discrepancies in observational studies are likely due to variations in patient characteristics, study design, follow-up duration, and confounders. This variability underscores the importance of stratifying APS patients based on risk profiles when interpreting anticoagulant efficacy and safety data.
Nevertheless, our study demonstrated that bleeding risks provide additional clarity to an area of ongoing debate. In contrast to the usual expectations that DOAC might offer better safety due to their predictable pharmacokinetics and fixed dosing, our study found no significant difference in major bleeding or CRNMB between DOAC and VKA.36,37 The bleeding risk associated with APS patients may be influenced more by the underlying disease and patient comorbidities than by the choice of anticoagulant alone.38,39 Moreover, the international normalized ratio (INR) monitoring of warfarin may not be accurate due to an interaction between LA and the thromboplastin reagent utilized in the INR testing. This could be a barrier for clinicians to identify the risk of both bleeding and thrombotic complications, even in patients who are within the therapeutic INR range. Hence, there is a need for better monitoring strategies for VKA to reduce the risk of thrombosis and bleeding events.40,41
There are several limitations to our study that should be acknowledged. First, the limited number of RCTs and variations in study designs, follow-up durations, and outcome definitions contributed to heterogeneity and may have impacted the precision of our pooled estimates. Second, although our analysis included subgroup comparisons by APS antibody profiles, the number of studies and patients in the single- and double-positive groups was relatively small, which limited the strength of the conclusions for these lower-risk populations. Third, although we included studies that evaluated different DOAC agents, we did not perform a subgroup analysis based on individual DOACs due to limited reporting and small sample sizes for specific agents (eg, apixaban, rivaroxaban, dabigatran). Pooling all DOACs into a single comparator group may have masked clinically meaningful differences. Emerging evidence suggests that certain DOACs, such as apixaban, may have a more favorable safety profile than others. 42 Acknowledging this heterogeneity is crucial for enhancing the clinical applicability of our findings. Fourth, the included observational studies may be subject to treatment selection bias and residual confounding. Additionally, adherence data, time in the therapeutic range for VKA users, and dosing consistency were not uniformly reported across studies, which may influence the interpretation of outcomes. Despite these limitations, our findings provide updated insights that reinforce guideline recommendations and highlight areas where future, more targeted research is needed to inform the selection of anticoagulants in APS.
Conclusion
In conclusion, although DOACs may offer equivalence in efficacy and safety compared to VKA in the APS population, their use in high-risk APS patients, especially those who are triple aPL positive, is associated with a substantially increased risk of recurrence of thrombosis events. Our findings reinforce current guidelines and emphasize the need for risk stratification in selecting anticoagulation options in APS. Further studies are needed to optimize personalized patient treatment in this population.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251364269 - Supplemental material for Evaluating the Efficacy and Safety of Direct Oral Anticoagulants Compared to Vitamin K Antagonists in Patients with Antiphospholipid Syndrome: Updated Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-cat-10.1177_10760296251364269 for Evaluating the Efficacy and Safety of Direct Oral Anticoagulants Compared to Vitamin K Antagonists in Patients with Antiphospholipid Syndrome: Updated Systematic Review and Meta-Analysis by Lama Alfehaid, Nada Alsuhebany, Hussah Albahlal, Albatoul Alomran, Abdulmajeed M Alshehri, Mohammed AlSheef and Abdulaali Almutairi in Clinical and Applied Thrombosis/Hemostasis
Footnotes
ORCID iDs
Author Contribution
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
