Abstract
Introduction
This study investigated the roles of d-dimers, fibrinogen, activated partial thromboplastin time (aPTT), and Factor XIII (FXIII) in critically ill COVID-19 patients.
Methods
68 patients were included into the study based on the inclusion criteria. Severe illness was defined as COVID-19 pneumonia leading to acute respiratory failure requiring mechanical ventilation and ICU (Intensive Care Unit) care. Patients who received Extracorporeal Membrane Oxygenation therapy were excluded due to its effects on the coagulation system. Blood samples were collected on day one and days five to seven, to measure coagulation parameters (aPTT, d-dimer, fibrinogen, and FXIII).
Results
In total, 59% of the patients died during their hospital stay. Coagulation parameters showed elevated d-dimer levels in 95.6% of patients, while FXIII activity significantly decreased during the first week in ICU. A drop in FXIII activity over the first week (FXIII Δ) emerged as a significant predictor of in-hospital mortality (HR = 2.174, P = .031), while d-dimers did not. No significant changes in aPTT, fibrinogen, or d-dimer levels were observed between days one and five to seven.
Conclusion
The change in Factor XIII activity during the first week in ICU (FXIII Δ) emerged as an independent predictor of in-hospital mortality. D-dimer levels were elevated in nearly all patients, but did not serve as an independent predictor of in-hospital mortality, possibly due to the homogeneity of the cohort. Overall, the results emphasize the importance of monitoring d-dimers and Factor XIII in predicting disease severity and outcomes in COVID-19 patients.
Introduction
Since its beginning in late 2019, the COVID-19 pandemic has posed a significant challenge to the global health system. Over the last years, extensive research has been conducted on the pathophysiology of severe disease courses and risk factors for mortality. COVID-19 was initially considered to be a primarily respiratory disease due to early involvement of the lungs and acute respiratory failure observed in critically ill patients. However, research has shown that coagulation abnormalities play a crucial role in COVID-19 associated mortality. The mechanisms leading to this coagulopathy are complex and multifaceted, involving platelet hyperactivation, a hyper-active innate immune response, endothelial damage and an imbalance of fibrinolysis.1,2
Today, coagulation abnormalities are regarded as one of the central pathophysiological features in severe COVID-19 disease, leading to a prothrombotic state with high risk of venous and arterial thrombosis, multiorgan failure and overall increased mortality,1,3 even under prophylactic treatment with low molecular weight heparin (LMWH). 4 To provide an overview of coagulation abnormalities, inflammation, clot formation and stability, this study focused on d-dimers, fibrinogen, aPTT, and FXIII as prognostic outcome markers for COVID-19 disease.
During acute infection, proinflammatory cytokines are upregulated, which leads to an activation of procoagulant pathways and ultimately causes microvascular thrombosis. 3 To monitor fibrin formation and degradation, d-dimers emerged as an important marker early on during the pandemic. 5 Regarding COVID-19, they were found to be elevated in almost all hospitalized patients and can be associated with disease severity, the occurrence of venous thromboembolisms (VTE) and mortality.6–8 Another important parameter for monitoring inflammation and coagulation is fibrinogen. It is one of the acute-phase proteins and can be elevated in the context of inflammation and infection. In their review, Kangro et al state that elevated fibrinogen levels not only indicate a higher risk of thrombosis but also actively contribute to clot formation by promoting denser, more stable fibrin networks that resist breakdown, worsening the hypercoagulable state in COVID-19 and leading to poor outcomes. 9 At the same time, low fibrinogen levels can reflect advanced stages of the disease due to consumptive coagulopathy, also being associated with higher mortality rates. 10
In this context, aPTT is an important parameter to assess the intrinsic and common pathway of coagulation, providing insight into the overall function of clotting factors and the balance between procoagulatory and anticoagulatory pathways. Several studies have reported increased aPTT in COVID-19 patients compared to healthy controls, 11 although its prognostic value for in-hospital mortality remains questionable. 12
While there has been extensive research on coagulopathy in COVID-19, only a few studies have reported impaired Factor XIII-activity in severely ill COVID-19 patients.13,14 These investigations included patients with a wide range of disease severities, which makes it challenging to draw clear conclusions about the specific role of FXIII in disease progression and death in critically ill COVID-19 patients.
The primary objective of this study was to verify existing research about the roles of d-dimers, fibrinogen, and aPTT, and to investigate the impact of Factor XIII activity on the survival of critically ill COVID-19 patients at the time of admission to the Intensive Care Unit (ICU), as well as the impact of dynamic changes in these coagulation parameters during the first week of treatment on overall in-hospital mortality. To achieve a more homogenous patient cohort, this study only included patients with severe disease progression in need of mechanical ventilation or high-flow oxygen therapy, while excluding the potential confounding effects of Extracorporeal Membrane Oxygenation (ECMO)-therapy on coagulation parameters. Our goal is to contribute to the growing knowledge on the prognostic significance of coagulation biomarkers for COVID-19 as well as the potential future waves of the disease.
Methods
Study Design and Patient Population
This prospective single-center observational study was conducted between March 01, 2020, and December 31, 2021, to assess the influence of coagulation parameters on the survival outcomes of critically ill COVID-19 patients upon admission to the ICU.
This study is part of the CAPNETZ-Umbrella study and the sub-study “Coronavirus Disease 2019 (COVID-19)” under the supervision of Prof. Dr Gernot Rohde, Medical Clinic I at Frankfurt University Hospital. Ethical approval was granted by the Ethics Committee of the Faculty of Medicine at Goethe University Frankfurt for this substudy (ref. 11/17) and for additional biomaterial analyses (ref. 20-627). Furthermore, prospective data collection and analysis to improve the understanding of COVID-19 in critically ill patients was approved under reference number 20-643. No ethical or professional objections were raised, and all protocols were positively evaluated.
A total of 424 patients treated at the University Hospital Frankfurt were initially considered for the study. We then focused on patients who had a complete follow-up after five to seven days after ICU admission. Patients were included if they were at least 18 years of age and tested positive for COVID-19. The diagnosis was confirmed using Real-Time-polymerase chain reaction (PCR) testing on admission. Only patients who met both criteria, and for whom COVID-19 was the primary reason for ICU admission, were eligible for the study. To minimize potential confounding effects on coagulation parameters, patients who required ECMO-therapy during the study period were excluded. Ultimately, 68 patients were included for the assessment of dynamic changes in coagulation parameters, all requiring invasive mechanical ventilation or high-flow oxygen therapy due to COVID-19 related respiratory failure.
Among the 424 patients, 281 were excluded from the analysis due to missing essential data. In 266 of these cases, complete coagulation profiles were unavailable five to seven days following ICU admission, making it impossible to evaluate changes over time. An additional 15 patients lacked information necessary for evaluating disease severity at admission, namely SAPS II and PaO₂/FiO₂ ratio, owing to incomplete documentation in the electronic medical records. Consequently, these patients could not be reliably stratified according to illness severity at presentation.
Data Collection
Relevant clinical data were extracted from the electronic medical records using a patient data management system (PDMS; Metavision 5.4, iMDsoft, Tel Aviv, Israel), including clinical characteristics, age, sex, body mass index, relevant comorbidities, therapeutic interventions, and clinical outcomes.
Scores
Simplified Acute Physiology Score II (SAPS II)
To evaluate and compare the overall health status of the patients, we measured the SAPS II score on day one of ICU admission. The SAPS II is a dedicated scoring system designed for use in ICUs, aimed at quantifying the severity of illness in patients aged ≥ 18 years. It encompasses 12 physiological variables, including vital signs, such as heart rate, respiratory rate, temperature, and blood pressure, along with age, chronic health conditions, and the type and reason for admission. A higher SAPS II score indicates a more severe illness and a potentially higher risk of mortality. 15
PaO2/FiO2-Ratio
To evaluate lung function, we calculated the ratios of the partial pressure of arterial oxygen to the fractional concentration of oxygen in the inspired air for each patient upon their ICU admission. 16
Blood Sample Collection
Peripheral venous blood was drawn from all patients on day one and day five to seven after ICU admission. The follow-up interval was chosen to capture early dynamic changes in coagulation parameters during the acute phase of critical illness. Additionally, logistical factors such as limited laboratory capacities and the workload on staff, especially over the weekends, influenced the timing of blood sampling, which led to a clustering of follow-up samples around weekdays.
A total of 9 mL of blood were collected into tubes containing 1 mL of Tri-Sodium-Citrate (9NC/10 mL Tri-Sodium Citrate Monovette® from Sarstedt). Citrate plasma samples were centrifuged at 1500×g for 15 min at room temperature and then heated to 37 °C for testing. The chromogenic determination of Factor XIII activity was performed using the Berichrom® F XIII assay on Siemens budd-chiari-syndrome (BCS) XP analysis system according to the manufacturer's instructions. aPTT, d-dimers, and fibrinogen levels were measured on Werfens anti-cardiolipin (ACL)-TOP analysis system using the HemosIL D-Dimer HS 500 assay, HemosIL Fibrinogen-C XL reagent, and HemosIL SynthASil APTT Reagent.
Statistical Analysis
Statistical analyses were performed using IBM SPSS Statistics version 28.0.1.1. Continuous variables are presented as median and interquartile range (IQR), and normality of data distribution was assessed using the Kolmogorov-Smirnov Test. Categorical variables were presented as absolute frequencies and percentages.
Group comparisons for continuous variables were performed using the Wilcoxon signed-rank test (eg, coagulation parameters between ICU admission and follow-up) or Student's t-test (eg FXIII between tocilizumab and control group) for paired measurements, and the Mann-Whitney U test for independent samples (eg, comparison between survivors and non-survivors). Categorical variables were compared using Fisher's exact test. To assess associations between coagulation parameters, Spearman's rank correlation coefficient (ρ) was used. Univariate logistic regression was performed to identify potential predictors of in-hospital mortality, reporting odds ratios with 95% confidence intervals. Variables with a P-value < .050 in univariate analysis were considered for further multivariable analysis.
For survival analysis, Cox proportional hazards regression was applied to determine the independent prognostic value of coagulation markers, adjusting for potential confounders. Hazard ratios with 95% CIs were reported. The Kaplan-Meier method was used to visualize survival distributions.
All tests were conducted at a two-sided significance level of P < .050. Given the exploratory nature of this study, no adjustments for significance were made.
Results
A total of 68 patients treated at the ICU for respiratory failure caused by COVID-19 infection, were included for detailed coagulation factor analysis. The median age was 66 years (IQR: 56–75), with the youngest patient being 35 and the oldest 89 years old. The median length of ICU stay was 14 days (IQR: 9-23), and 59% of the patients died during their hospital stay. Among these, 24 patients died from multiple organ failure, 11 patients experienced respiratory failure due to SARS-CoV-2 infection, three patients died from sepsis, and two patients from intracerebral haemorrhage. (Figure 1)

Patient Inclusion.
At study inclusion, all patients were administered anticoagulation therapy, with 62 patients receiving low-molecular-weight heparin and six patients receiving unfractionated heparin (UFH). Additionally, 46 patients received antiplatelet therapy in the form of aspirin. During the first week of ICU treatment, 53 patients were treated with antibiotics, and 18 patients received antiviral treatment, including tocilizumab in 11 patients and remdesivir in seven patients. Baseline characteristics of all 68 patients are shown in Table 1.
Baseline Characteristics of the Patient Cohort.

Abbreviation: PAOD, peripheral arterial occlusive disease.
Table 2 presents the coagulation parameters measured on ICU admission (day one) and after five to seven days of ICU stay.
Coagulation Parameters at ICU Admission and After Days Five to Seven.

N = 68 patients. Data represent median (interquartile range).
Abbreviations: aPTT, activated partial thrombine time; FXIII, Factor XIII; ICU, Intensive Care Unit.
*indicates statistical significance (P < .050).
Upon ICU admission, aPTT was within the normal range (27-35 s) in 64.7% of the patients (44 out of 68). APTT was reduced in 10 patients (14.7%), and 14 (20.6%) had elevated aPTT levels. At the same time, fibrinogen levels were elevated above 498 mg/dL in 33.8% (23 of 68) of patients. The median FXIII activity level was 89.4%, with 20.6% (14 out of 68) of the patients falling below the normal range of 70%. D-dimer levels were elevated in almost all patients, with 95.6% of patients (65 of 68) above the normal range of 500 µg/L.
In follow-up measurements, no significant changes were observed between day one and days five to seven regarding aPTT, fibrinogen- and d-dimer levels. However, there was a significant drop in FXIII activity in 51.5% of patients below the low normal range (Figure 2). Overall, FXIII activity decreased by 22.4% (IQR: 6.5-35.1) during the first week in the ICU. Importantly, no FXIII substitutions or FXIII-containing substitutes like fresh frozen plasma were administered to any patient between the two blood collection points. Four patients received substitution therapy after day seven; all these patients died, including one who suffered a fatal intracranial haemorrhage. Spearman's rank correlation analysis showed a significant negative correlation between d-dimers and FXIII levels on day one (ρ = −.301, P = .013), as well as on days five to seven (ρ = −.358, P = .003).
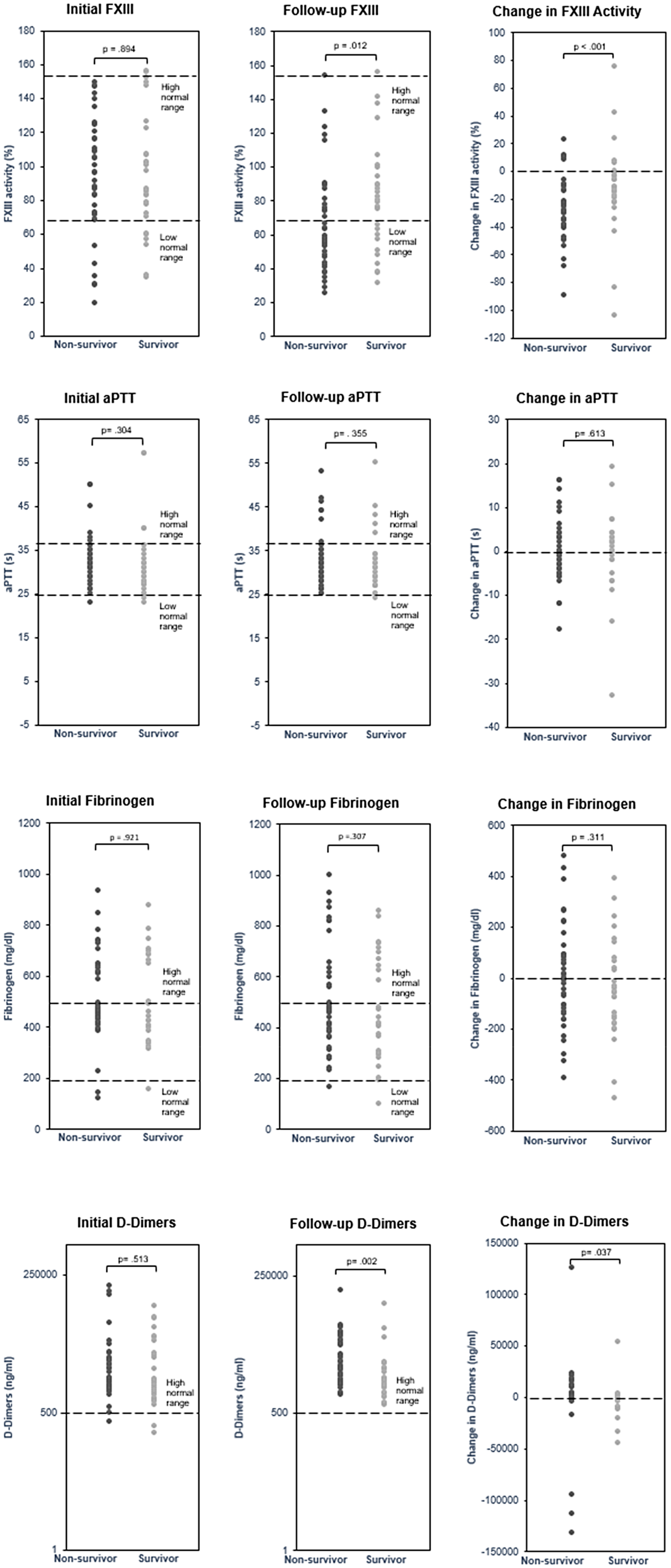
FXIII, aPTT, Fibrinogen and d-Dimers in Non-Survivors and Survivors.
To assess whether tocilizumab administration influenced FXIII levels, as reported for rheumatoid arthritis, we analyzed its potential confounding effects. While there was a significant decrease in FXIII activity from day one to days five to seven in the group without tocilizumab administration (W = 1.34 × 10−6; P < .001), as well as in the tocilizumab group (W = .005; P = .005), an independent-samples t-test comparing the FXIII changes (FXIII Δ) between the tocilizumab and control groups indicated no significant difference (t[66] = .084; P = .934).
In-Hospital Mortality
Upon admission to the ICU, there was no significant difference between survivors and non-survivors in the levels of fibrinogen (P = .921), d-dimers (P = .513), FXIII activity (P = .894) and aPTT (P = .304) as shown in Figure 2. On days five to seven however, non-survivors showed significantly lower median FXIII activity levels compared to survivors (P = .012). Non-survivors had a median FXIII activity of 57.5% (IQR: 46.8-80.0) with 63% falling below the normal range. In contrast, survivors had a median FXIII activity of 79.7% (IQR: 57.7-98.2), with only 36% below the normal range. At this time point, d-dimer levels were elevated in all patients, and there was a significant difference between survivors and non-survivors (P = .002). No differences were observed for aPTT (reduced in 11.8% and elevated in 19.1% of patients) (P = .355) and fibrinogen (elevated in 35.3% of patients) (P = .307) (Figure 2).
We conducted median splits for aPTT, d-dimer levels, fibrinogen levels and FXIII activity. Patients were divided into “low” and “high” groups for each parameter based on levels on day one and days five to seven. Changes in these levels (Δ) were also split by median, separating patients with substantial changes from those with smaller changes. The separating values for each parameter are listed in Table 2.
In univariate logistic regression analysis (Table 3), d-dimers on days five to seven and FXIII Δ were predictive for in-hospital mortality. A notable trend was observed for FXIII activity on days five to seven. We then performed cox regression analysis to assess the discriminatory ability of FXIII Δ and d-dimers on days five to seven by adjusting for group differences, as listed in Tables 4 and 5.
Univariate Logistic Regression Analysis.

N = 68 patients.
Abbreviations: aPTT, activated partial thrombine time; FXIII, Factor XIII
*indicates statistical significance (P < .050).
Baseline and Clinical Characteristics of the Cohort Separated by Change in d-Dimer Levels.

N = 68 patients. Continuous data are expressed as median (Interquartile range), categorial data as number (%).
Abbreviation: ASS, Acetylsalicylic acid; SAPS, Simplified Acute Physiology Score.
*indicates statistical significance (P < .050).
Baseline and Clinical Characteristics of the Cohort Separated by Change in FXIII Activity.
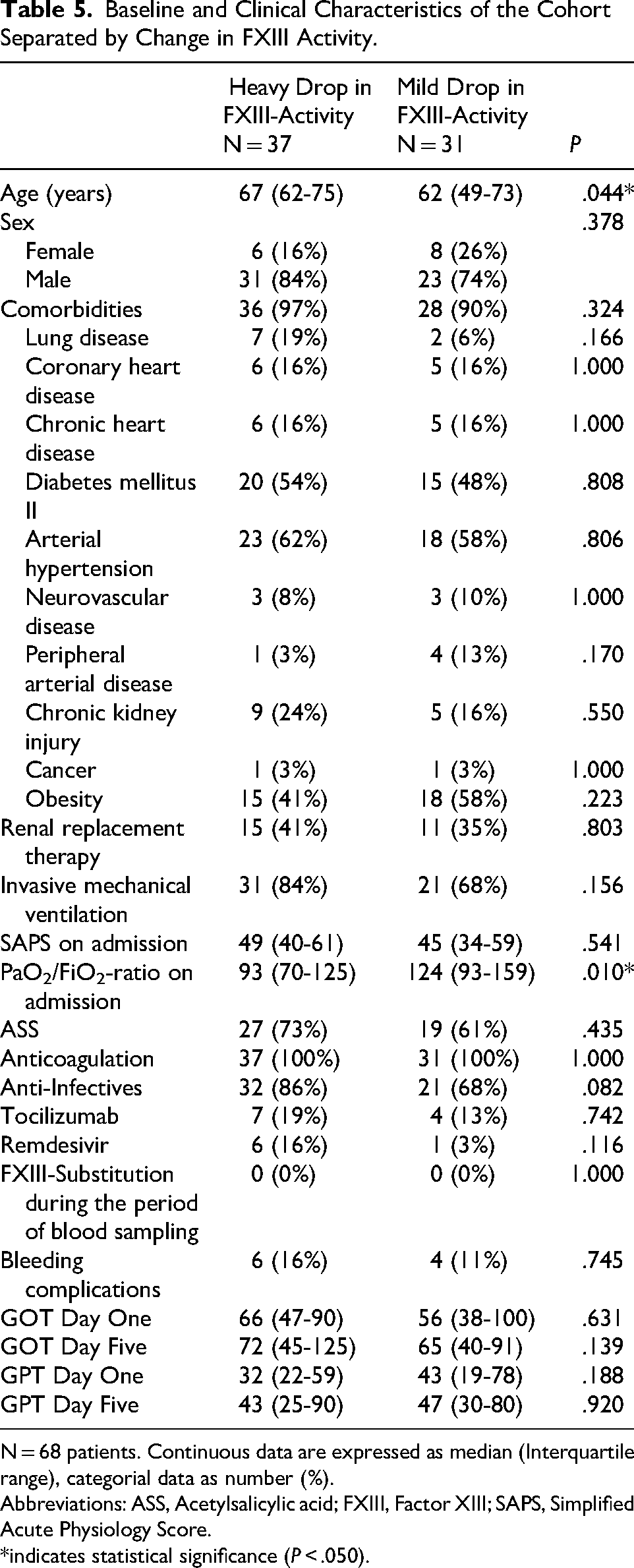
N = 68 patients. Continuous data are expressed as median (Interquartile range), categorial data as number (%).
Abbreviations: ASS, Acetylsalicylic acid; FXIII, Factor XIII; SAPS, Simplified Acute Physiology Score.
*indicates statistical significance (P < .050).
For follow-up d-dimers, significant differences were observed between the groups in terms of sex (P = .033) and PaO2/FiO2-ratio on admission (P = .004) (Table 4). The distribution of comorbidities, therapeutic interventions, and overall disease severity, as measured by the SAPS II, was similar in both groups.
Regarding FXIII Δ, there were significant group differences in age (P = .044) and PaO2/FiO2-ratio on admission (P = .010) (Table 5). To explore a potential link between FXIII activity and liver function, we measured glutamat-pyruvate transaminase (GPT) and GOT levels, as the B subunit of FXIII is exclusively synthesized by hepatocytes. GPT levels were elevated in 28% and GOT levels in 47% of patients on day one and 31% (GPT) and 35% (GOT) on days five to seven. However, no significant differences were observed between the FXIII Δ groups in these measurements.
Interestingly, the rate of bleeding complications during the first week of hospitalization did not differ significantly between the groups. Among the six patients (five male and one female) with a substantial decrease in FXIII-activity who experienced bleeding, one had nasal bleeding, two had rectal bleeding with melena, two had upper gastrointestinal bleeding and one was bleeding next to the outlet of a thoracic drainage. In the other group, two male patients experienced gastrointestinal bleeding with melena, and one suffered from intracerebral haemorrhage.
Group Differences in Demographic and Clinical Characteristics of the Study Cohort
We then performed Cox regression analysis to assess the survival distribution between the “low” and “high” groups for FXIII Δ and follow-up d-dimer levels.
FXIII Δ emerged as an independent predictor of in-hospital mortality (HR = 2.174; 95%CI [1.075, 4.386]; P = .031). In contrast, follow-up d-dimer levels were not predictive of in-hospital mortality after adjusting for sex and PaO2/FiO2-ratio (HR = 1.608; 95%CI [.798, 3.239]; P = .184).
Figure 3 shows the adjusted survival curve illustrating the estimated probability of survival for both FXIII Δ groups over time.
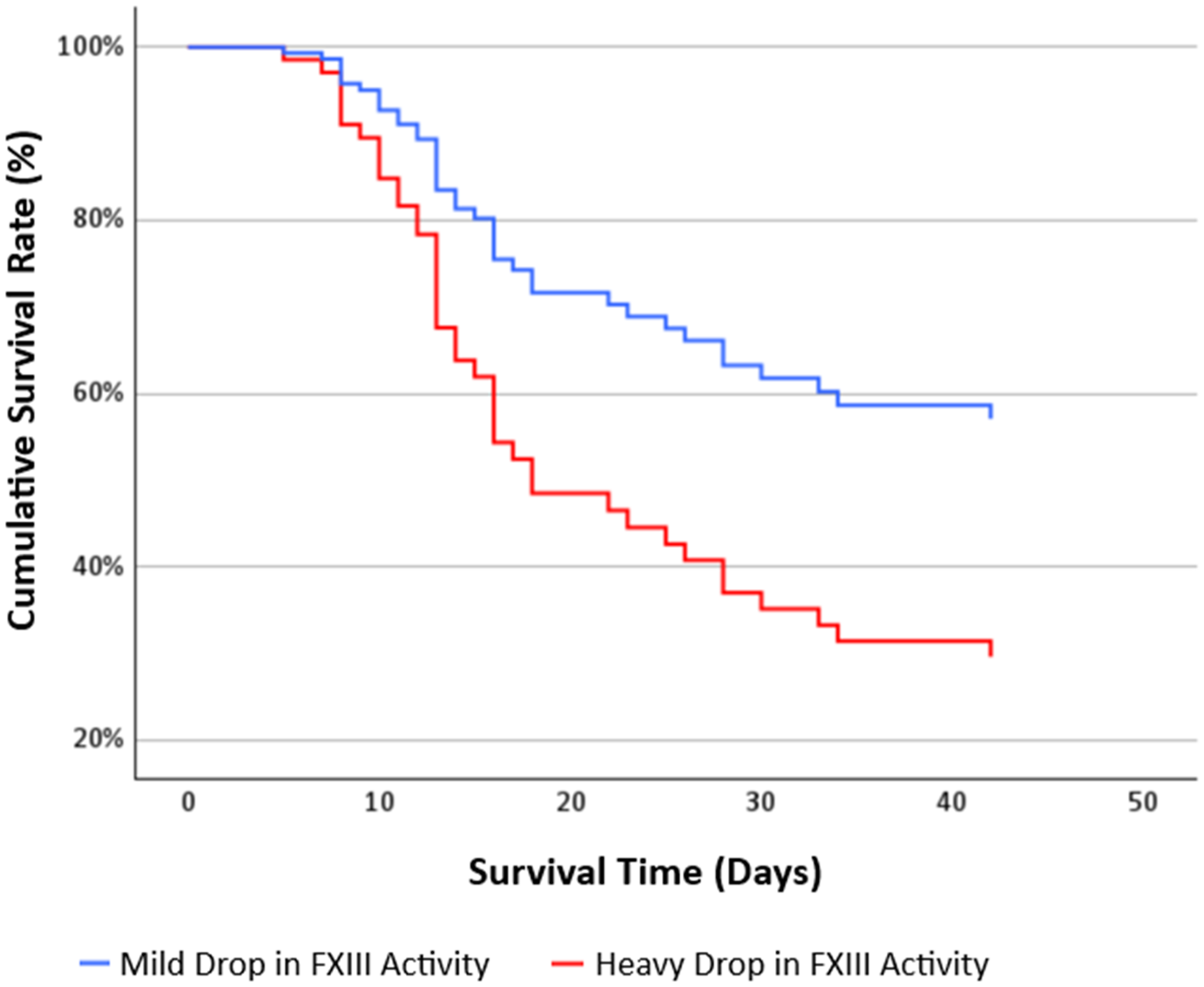
Kaplan- Meier Survival Analysis Stratified by Change in FXIII Activity and Adjusted for Group Differences.
Discussion
COVID-19 can severely impair organ function and coagulation, leading to a high mortality rate. Therefore, factors that can predict the outcome of patients with severe disease progression are warranted. This study evaluated the coagulation profiles of patients admitted to the ICU with severe COVID-19 disease, focusing on d-dimers, fibrinogen, aPTT, and FXIII levels.
To the best of our knowledge, this study is the first to investigate dynamic changes in these parameters within a homogenous cohort of critically ill COVID-19 patients who required respiratory support but did not undergo ECMO-therapy during hospitalization. Coagulation parameters were measured on the first day at ICU and follow up measurements were always carried out on the fifth to seventh day. This minimized confounding factors such as the effect of ECMO therapy on coagulation and the time-dependent change in coagulation factors during the course of the disease.
In the ICU, coagulopathy is common due to various changes such as disseminated intravascular coagulation, impaired synthesis of coagulation factors, sepsis or drug induced thrombocytopenia. 17 For critically ill COVID-19 patients in specific, numerous research findings have shown a high prevalence of both venous and arterial thrombotic events as well as bleeding complications. The incidence of venous thromboembolism is reported between 10% and 31% and the rate of arterial thrombotic events about 5%.18–20 Bleeding complications were observed in 0.9% up to 4.9%, depending on whether patients received therapeutic or prophylactic anticoagulation.20,21
Regarding the prevalence of thrombotic and thromboembolic events, the observed increase in d-dimer levels in our cohort is consistent with previous publications. Berger et al analyzed d-dimer levels in 2782 hospitalized COVID-patients and found elevated levels in most patients (86%) during their hospital stay. Elevated d-dimers were associated with higher odds of critical illness and higher mortality. 6 Similarly, Zhou et al found that an elevation in d-dimers above 1 μg/mL was associated with an 18-fold increase in death. 8 Since d-dimers are fibrin degradation products, their increase suggests increased fibrinolysis as a result of increased clot formation, which is commonly seen in severe COVID-19 disease. 1 In our cohort however, d-dimers did not emerge as an independent predictor of mortality. This may be the case due to several factors. A possible explanation for this might be the homogeneity of our patient population. All included patients were critically ill and required oxygenation therapy. This selection could have led to generally elevated d-dimer levels across the cohort, causing d-dimers to lose their discriminatory power between survivors and non-survivors. Second, the timing of d-dimer measurement could have influenced the results. Since d-dimers were measured on day one and days five to seven, it is possible that peak d-dimer levels occurred outside of these time points.
Interestingly, aPTT remained within the normal range in most patients. This could be attributed to the complex alterations in coagulation parameters in COVID-19 patients. One possible explanation is the use of anticoagulant therapy with LMWH or UFH in all patients, which can prolong aPTT, combined with an acute phase response characterized by an increase in FVIII,22,23 which can shorten the aPTT. This balance may result in normal aPTT values, which could mask the underlying coagulopathy.
At ICU admission, elevated fibrinogen levels were observed in one-third of the patients. This finding further supports the presence of a hypercoagulable state. Fibrinogen is part of the acute phase response and its elevation is an indication of the underlying inflammation and coagulation activation. 24
Our analysis showed that the change in FXIII activity from day one to days five to seven was predictive of in-hospital mortality. However, it is important to mention that the follow-up FXIII activity alone also differed significantly between survivors and non-survivors (Figure 2). This suggests that it may serve as a practical, though less robust, alternative marker when baseline data is unavailable.
We also observed a significant correlation between increased d-dimer levels and decreased FXIII activity. This may reflect a compensatory mechanism or consumptive depletion of FXIII due to increased clot formation.
While d-dimer levels are now a well-documented marker of high mortality in COVID-19 patients,6,25 the change in FXIII activity presents a novel finding.
Factor XIII deficiency can either be congenital or acquired. Acquired FXIII deficiency appears to be relatively common in ICU patients. In a study by Lichter et al, FXIII activity was measured in 34 COVID-19 patients hospitalized in the ICU. In this study, 74% of the patients had activity levels below 79%. However, six of the patients were undergoing ECMO-therapy and the time of measurement was not reported by the authors. 26 Another publication by Lawrie et al reported FXIII activity below the normal range of 70% in 21% out of 1004 hospitalized COVID-19 patients. 27 The prevalence of congenital Factor XIII deficiency on the other hand has been estimated at 1:2.000.000 and typically manifests with bleeding of the umbilical stump in the first days after birth as well as intracranial haemorrhage. 28
In clinical practice, mild deficiencies often go undetected, 29 probably because FXIII-deficiency cannot be identified using global coagulation tests such as aPTT or prothrombin time, which are routinely used to screen for coagulation abnormalities.
To account for potential confounding factors, we investigated the use of tocilizumab in our study population and its potential impact on FXIII levels. Previous studies have suggested a potential correlation between tocilizumab use and FXIII decline in patients suffering from rheumatoid arthritis (RA).30,31 Watanabe et al demonstrated that FXIII-A expression was upregulated in fibroblast-like synoviocytes under IL-6 stimulation and blocking the IL-6 receptor with tocilizumab led to a significant reduction in FXIII expression. 30 However, our findings did not show a significant correlation between tocilizumab use and lower FXIII activity in critically ill COVID-19 patients, as well as no significant difference in FXIII activity between patients who received tocilizumab and those who did not. This discrepancy could be attributed to the distinct pathophysiological mechanisms underlying COVID-19. While the literature supports the role of IL-6 receptor inhibition in reducing FXIII levels in RA, our findings suggest that the broader pathophysiology of COVID-19 disease, namely the overall dysregulated coagulation system with an imbalance of procoagulant and anticoagulant forces may overshadow any potential effects of tocilizumab.
There are three possible mechanisms that may explain the observed decrease in FXIII-activity: decreased synthesis due to liver failure, formation of antibodies against FXIII and/or consumption of coagulation factors.
While the FXIII-A subunit is primarily synthesized by cells of bone marrow origin, FXIII-B is exclusively secreted by hepatocytes. 32 Its purpose is to protect FXIII-A from premature degradation and also supporting the accelerated activation of plasmatic FXIII by fibrin-polymers. 32 Severe deficiency of FXIII-B can therefore result in increased clearance of FXIII-A and thus decreased FXIII activity. However, FXIII-B is present in plasma in excess with about 50% circulating unbound under physiological conditions. 32 Several studies have found that SARS-CoV-2 infection is related to liver dysfunction and elevated transaminases. angiotensin-converting enzyme (ACE)-2 receptors get expressed on liver cells, especially cholangiocytes, but also on hepatocytes, enabling a direct infection and damaging of host cells by SARS-CoV-2. 33 Guan et al reported elevated levels of GOT in 39% and elevated levels of GPT in 20% of critically ill COVID-19 patients, 34 which is consistent with our findings. In our study, we found elevated liver enzyme levels in some patients, but no direct link between increased transaminases and decreased FXIII activity, making a synthesis disorder unlikely.
Another potential mechanism may involve the formation of antibodies against FXIII. Viruses such as the EBV (Epstein-Barr virus) and CMV (Cytomegalovirus) are well recognized contributors to the development of autoimmune reactions and the production of autoantibodies. In COVID-19, studies showed a marked extent of autoantibody formation against extracellular antigens. 35
This formation of autoantibodies can be attributed to a hyperstimulation of the immune system by SARS-CoV-2, resulting in tissue damage with an increased release of endogenous antigens, molecular mimicry between viral and human proteins as well as neutrophil activation and NETosis. 36
To date, only a few cases of patients with FXIII antibodies have been reported worldwide, leading to life-threatening bleeding with no standard treatment available. 29 In a review of 93 reported cases of autoimmune acquired FXIII deficiency (AH13), the median residual FXIII activity was 6.0%. This is considerably lower than FXIII levels observed in our patients, where the median activity was 89.4% (71.4-119.5) on day one and 66.2% (49.9-89.1) at the time of follow-up. In addition, 86% of patients with AH13 developed severe bleeding complications, mostly in intramuscular and subcutaneous tissues, which did not occur in our patient cohort. Hence, it seems unlikely that the formation of autoantibodies against FXIII is the primary cause of the decline in FXIII activity observed in COVID-19 patients. However, since there is no standard screening test to assess abnormal FXIII activity, milder forms of AH13 may go unnoticed in clinical practice. 37
As COVID-19 is primarily thought to be a procoagulatory condition, the combination of elevated d-dimers and FXIII consumption likely reflects microthromboembolism formation in the pulmonary microvasculature, consistent with findings in previous studies, which link such thrombosis to impaired gas exchange.38,39 This aligns with the observed impact of FXIII decline on worsening oxygenation and ventilation disorders in our patients. The latter assumption is compatible with the observed impact of FXIII decline on worsening of oxygenation and ventilation disorders in our patients.
SARS-CoV-2 directly damages the vascular endothelium and causes platelet hyperactivation, excessive immune response with complement activation and formation of neutrophil extracellular traps (NETs) as well as loss of the antithrombotic endothelial surface and release of procoagulant substances such as tissue factor and von Willebrand factor. This leads to a continuous deposition of fibrin in the lungs and the development of venous thromboembolism with consumption of coagulation factors. 1 Since FXIII activity declined during the first week at ICU, and considering that activity levels were only slightly below the normal range in most patients, it appears that a consumption based mechanism is the most probable cause for our observations.
Previous investigations often included patients with a wide range of disease severity and clinical interventions which makes it difficult to draw clear conclusions about the role of FXIII in disease progression and death of critically ill COVID-19 patients. Therefore, we focused on a specific subgroup, including only patients who required mechanical ventilation but did not undergo ECMO-therapy, to exclude its potential confounding effects on coagulation parameters.
Our study has some limitations. With 68 patients, our sample size is relatively small, and larger studies are needed to verify our conclusions. Some participants were lost to follow up because they were transferred to other medical facilities during ongoing therapy. In a few cases, data relevant to the study, but not immediately necessary for patient care, were incompletely collected due to the urgent nature of pandemic containment efforts, leading to an exclusion of those patients, and possibly contributing to selection bias.
Additionally, it was often not possible to record clinical manifestations, such as pulmonary embolisms, in all patients. As a result, we focused on overall survival as the primary endpoint and used standardized scores like the SAPS II and the PaO2/FiO2-ratio to assess disease severity.
We also did not measure the time between onset of illness and ICU admission. The hypercoagulable state in COVID-19 patients is observed early during infection and can progress to a hypocoagulable state due to the excessive consumption of clotting factors. 40 Thus, the difference in timing of testing may contribute to confounding of results.
Lastly, our study focuses on COVID-19 patients infected in a time when earlier variants (alpha, beta and delta) were predominant, whereas today Omicron remains the dominant variant. Recent studies indicate that Omicron can still lead to a hypercoagulable state, although with a lower overall coagulopathy risk compared to Beta- and Delta-variants. 41 Despite the general reduction in disease severity with Omicron, coagulopathy remains a concern in immunocompromised and severely pre-diseased patients. Studies highlight that d-dimers remain an important prognostic marker for severe illness, including ICU admission and mortality. 42 Given these findings, FXIII monitoring may still hold clinical value in high-risk patients. The integration of FXIII activity in a broader panel of coagulation markers could help refine risk stratification and improve patient outcomes.
In conclusion, this study provides valuable insights into coagulopathy in critically ill COVID-19 patients, highlighting a drop in FXIII activity as a new potential predictor of in-hospital mortality. Due to the limited sample size, larger scale studies are needed to verify our findings, establish cut-off values and explore potential causal relationships between FXIII activity and disease progression, especially in the context of the Omicron variant.
The Role and Responsibility of Each Author
CC wrote the first draft, that has consecutively improved by all authors. WM, SR and SR developed the study concept together with European Hematology Association (EHA). FG contributed to the evaluation of the data.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
