Abstract
Background
Venous thromboembolism (VTE) is a complex vascular disorder, and emerging research suggests potential neurovascular and metabolic factors in its pathogenesis. However, the specific roles of cortical structure features and cerebrospinal fluid (CSF) metabolites in VTE risk remain underexplored.
Methods
Using Mendelian randomization (MR) analysis, we examined associations between cortical features, CSF metabolites and VTE. Mediation MR and co-localization analyses were employed to explore genetic pathways and potential mediatory effects.
Results
MR analysis initially identified associations between six cortical features and fifteen CSF metabolites with VTE. After false discovery rate (FDR) correction, lingual gyrus thickness remained statistically significant, while isoleucine and methylmalonate showed suggestive associations. Mediation MR analysis revealed no causal relationship between lingual gyrus thickness and the CSF metabolites isoleucine or methylmalonate. Co-localization analysis indicated low posterior probabilities for shared genetic variants, suggesting that these traits influence venous thromboembolism (VTE) through distinct biological mechanisms.
Conclusion
The findings suggest that MRI-based cortical structure features and CSF profiling hold potential as complementary tools for assessing VTE risk. Further research is warranted to investigate the neurovascular and metabolic mechanisms underlying VTE.
Introduction
Venous thromboembolism (VTE) encompasses a complex and clinically significant condition defined by the development of thrombi within the venous system. Its two primary manifestations–deep vein thrombosis (DVT) and pulmonary embolism (PE)—pose distinct and serious health challenges.1,2 DVT predominantly arises in the deep veins of the lower extremities, whereas PE results from embolized thrombi occluding the pulmonary arterial circulation. This condition carries profound health risks, encompassing both the immediate threat of PE and the long-term sequelae of DVT, such as post-thrombotic syndrome. Although considerable progress has been made in the realms of prevention and therapeutic management, VTE remains a leading contributor to global morbidity and mortality, accounting for an estimated 10 million cases worldwide each year.3,4
Diagnosing VTE is challenging due to its varied presentations, which range from localized pain and swelling in DVT to systemic symptoms, such as shortness of breath and hemodynamic instability in PE. DVT diagnosis typically relies on venous ultrasonography, while PE often requires computed tomography pulmonary angiography (CTPA) or ventilation-perfusion (V/Q) scanning. Biomarkers like D-dimer, although useful as screening tools, lack specificity, thus complicating the diagnostic process and potentially delaying treatment. 5
The global epidemiology of VTE highlights a substantial health burden, with an estimated incidence of 1 to 2 cases per 1000 individuals annually. Importantly, VTE is associated with significant morbidity, including long-term complications such as post-thrombotic syndrome and chronic thromboembolic pulmonary hypertension, as well as high mortality rates, particularly in cases of pulmonary embolism.6,7 Well-recognized risk factors include major surgical procedures, malignancies, extended periods of immobility, pregnancy, and the use of oral contraceptives. Furthermore, genetic predispositions such as Factor V Leiden and prothrombin gene mutations significantly elevate the risk of developing VTE. However, these genetic determinants alone fail to fully account for the occurrence of VTE in individuals without identifiable thrombophilic conditions, suggesting the involvement of additional, yet to be elucidated, contributory factors.8,9
The management of VTE primarily revolves around anticoagulation therapy, which aims to prevent both the progression of clots and their recurrence. The mainstays of treatment include low-molecular-weight heparin (LMWH), vitamin K antagonists such as warfarin, and direct oral anticoagulants (DOACs) like rivaroxaban and apixaban, each offering distinct advantages and limitations.10,11 In severe cases of pulmonary embolism (PE) accompanied by hemodynamic instability, more aggressive interventions such as thrombolytic therapy or mechanical thrombectomy may be required. Nevertheless, balancing the antithrombotic benefit with the elevated risk of hemorrhage remains a persistent clinical challenge, especially among older patients or those with comorbid conditions. 12
In recent years, genetic studies have identified a range of novel loci associated with VTE risk, which contribute to improved risk stratification and potentially guide precision medicine approaches.13–15 Among the most clinically relevant are Factor V Leiden (F5, rs6025), the prothrombin G20210A mutation (F2, rs1799963), and ABO variants such as rs817671915.16–18 In parallel, research attention has turned to the potential role of central nervous system (CNS) structures in influencing systemic thrombotic risk. The cerebral cortex, through its involvement in autonomic regulation and neuroimmune signaling, may indirectly affect systemic inflammation and vascular tone. 19 Features such as regional cortical thickness and surface area have been implicated in a range of neurological and systemic disorders, yet their relationship to thromboembolic risk remains poorly understood. Furthermore, studies increasingly highlight the influence of cortical health on autonomic function, neurovascular regulation, and inflammatory responses, all of which are recognized as key contributors to thrombotic events.20,21
Cerebrospinal fluid (CSF) biomarkers represent an additional, yet inadequately investigated, dimension of research concerning VTE. These biomarkers illuminate the essence of central nervous system (CNS) vitality and unveil precious revelations about the dance between neuroinflammation, the robustness of the blood-brain barrier, and the holistic performance of systemic vascular health.22,23 Certain biomarkers within CSF, including inflammatory cytokines, coagulation factors, and neurodegenerative markers, may drive systemic pro-thrombotic states, particularly in the presence of neuroinflammation or CNS injury. 24 The ability of CSF metabolites to reflect both CNS and systemic inflammatory status suggests a plausible mechanistic link to VTE pathogenesis, especially in vulnerable neurological populations.
One of the key challenges in VTE research lies in the heterogeneity of its clinical presentation and the difficulty of identifying high-risk individuals who could benefit from targeted preventive strategies. The wide range of clinical manifestations, from asymptomatic DVT to life-threatening PE, complicates both clinical trial design and risk stratification models. 25 While anticoagulation remains the cornerstone of VTE management, there is ongoing debate regarding the optimal duration of therapy and the selection of patients for extended prophylaxis. Clinical research is further hindered by the difficulty of conducting large-scale, long-term studies in special populations, such as patients with cancer, pregnant women, or those with chronic inflammatory conditions, all of whom have unique thrombotic and bleeding risks.26,27
Mendelian randomization (MR) provides a robust tool to investigate these complex associations. By utilizing genetic variants as instrumental variables, MR enables the estimation of causal relationships between exposures, such as cortical structure and CSF biomarkers, and outcomes like VTE, while effectively reducing confounding and reverse causality. 28 This approach is particularly valuable in scenarios where neuroanatomical and metabolic traits cannot be ethically or practically manipulated in trials. The methodology addresses a critical gap in the literature, where direct mechanistic studies linking brain structures and CSF markers to systemic thromboembolic events are still lacking.
The objective of this study is to assess whether changes in cerebral cortex structure, specifically cortical thickness and surface area, along with alterations in CSF biomarkers, have a causal relationship with the risk of VTE. The central hypothesis posits that genetic variants influencing cortical structure and CSF biomarkers are causally linked to an increased risk of VTE, thus providing a novel perspective on the neurovascular and neuroinflammatory contributions to thromboembolic events.
Materials and Methods
Study Design
Our study utilizes a MR design to investigate potential causal relationships between cerebral cortex structure, CSF biomarkers, and the risk of VTE. The MR framework is particularly suited to minimizing confounding and reverse causality, challenges commonly encountered in observational epidemiology. The study design comprises three stages: an initial MR analysis with cortical structure and CSF biomarkers as exposures and VTE as the outcome, followed by a mediation MR analysis, and concluding with a co-localization analysis (Figure 1).

Overview of the Research and Design of the Study. By
The initial MR analysis investigates direct causal associations between cortical structure features and CSF biomarkers with VTE. Subsequently, a mediation MR analysis explores whether CSF biomarkers mediate the relationship between cortical structure and VTE risk. In this analysis, cortical structure features are treated as the primary exposure, CSF biomarkers as potential mediators, and VTE as the outcome. This mediation step is essential for determining whether changes in CSF biomarkers could represent a pathway through which cortical structures impact VTE risk, thus providing further insight into the neuroinflammatory mechanisms underlying thromboembolic events. 29
Statistically significant results from the initial MR analyses are subjected to co-localization analysis, which assesses whether the same genetic loci influence both cortical structure, CSF biomarkers, and VTE. This step helps to confirm shared genetic etiology, allowing us to pinpoint specific loci that may underlie the observed causal relationships.
Data Sources
Sources of GWAS Data on Cortical Structure
The data on cortical structure, specifically surface area (SA) and thickness (TH), were sourced from the ENIGMA Consortium. This large-scale study utilized magnetic resonance imaging (MRI) to collect brain imaging and genetic data from 51,665 participants, predominantly of European ancestry, across 60 global cohorts. The dataset was carefully adjusted for global genetic effects, ensuring that the resulting genetic correlations (LD score rG) reflect regional specificity, independent of broader genome-wide influences. These thorough adjustments enhance the accuracy of the analysis, allowing for a more precise evaluation of the genetic architecture underlying cortical metrics in humans. 30
CSF Metabolites Data Synopsis
The analysis included 338 cerebrospinal fluid metabolites identified through a comprehensive metabolome-wide association study (MWAS). After rigorous quality control and data processing, 291 independent samples from cognitively healthy individuals of European ancestry were retained. Of the 338 metabolites, 296 were chemically validated and grouped into eight primary metabolic pathways: amino acid metabolism, carbohydrate metabolism, lipid metabolism, nucleotide metabolism, energy metabolism, cofactor and vitamin metabolism, peptide metabolism, and xenobiotic metabolism. The remaining 38 metabolites, although chemically undefined, were included in the analysis due to their potential biological relevance. 31
GWAS Data Sources for VTE
For the outcome, data on VTE were obtained from the FinnGen release, which includes comprehensive genomic and clinical data from Finnish biobank participants. The VTE cases in this dataset were identified based on several ICD codes, including ICD-10 codes I26, I80, O87.1, and O88.2, ensuring consistent diagnostic criteria across participants. These codes encompass a wide range of VTE manifestations, such as pulmonary embolism (I26), phlebitis and thrombophlebitis (I80), deep phlebothrombosis in the puerperium (O87.1), and obstetric blood-clot embolism (O88.2). This dataset consists of a total of 412,181 individuals, with 21,021 VTE cases and 391,160 controls. Genetic analysis was conducted using 19,345,634 single nucleotide polymorphisms (SNPs) to identify genetic variants associated with VTE risk. 32
Assumptions
The Mendelian randomization analysis relies on three core instrumental variable (IV) assumptions:
Relevance: The selected genetic variants must exhibit a strong association with the exposure variables–specifically, cortical structure features like thickness and surface area, and CSF biomarkers indicative of neuroinflammation or neurodegeneration. These associations confirm that the variants serve as reliable proxies for the exposures, providing a robust basis for inferring causality.
33
Independence: It is essential that the genetic variants are independent of confounders that could influence both the exposure (cortical structure or CSF biomarkers) and the outcome (VTE). This principle, akin to randomization in clinical trials, reduces the risk of bias commonly encountered in observational studies, ensuring that observed effects are not confounded by external factors.
34
Exclusion Restriction: This assumption requires that the genetic variants impact the outcome (VTE) solely through the specified exposures–cortical structure or CSF biomarkers–without influencing the outcome via other biological pathways. This condition is fundamental to isolating the causal effect of the exposures on the outcome (Figure 2A and B).
35

Schematic Diagram of Our Mendelian Randomization Assumptions.
Statistical Methods
Main Analysis
The primary analysis employs advanced MR techniques to investigate causal relationships between cortical structure, CSF biomarkers, and VTE, addressing key challenges such as weak instruments and pleiotropy. Single nucleotide polymorphisms (SNPs) are selected based on genome-wide significance and undergo stringent quality control to confirm strong associations with exposures and eliminate confounding. The primary MR method, Inverse Variance Weighted (IVW), aggregates effects from multiple genetic instruments to provide an overall causal estimate, assuming no pleiotropic effects. To address IVW limitations, especially with pleiotropy or weak instruments, additional MR methods are employed. 36
Bayesian Weighted Mendelian Randomization (BWMR) provides a computationally efficient framework for managing uncertainties and correcting pleiotropy through Bayesian inference, enhancing robustness when SNP-exposure associations are weak or pleiotropic effects are present. 37 Constrained Maximum Likelihood (CML) further refines precision by applying constraints on SNP-exposure associations, which is particularly useful in cases involving weaker instruments. 38 The Robust Adjusted Profile Score (RAPS) method further adjusts for both systematic and idiosyncratic pleiotropic effects, ensuring reliable estimates even when genetic instruments deviate from standard MR assumptions. 39 These advanced methods outperform traditional approaches, such as MR-Egger and the Weighted Median, in handling pleiotropy and weak instrument bias. For instance, while MR-Egger effectively detects pleiotropy, it often incurs high estimation variability with weak instruments. Similarly, the Weighted Median method, though reliable when most instruments are valid, does not address pleiotropic bias as comprehensively as BWMR or RAPS. For sensitivity analysis, MR-Egger remains the standard method for detecting directional pleiotropy via its intercept term.
In terms of managing missing data, the study uses summary-level data from genome-wide association studies (GWAS), ensuring that missing data are minimal and imputation is unnecessary. Multiple testing correction is conducted using the false discovery rate (FDR) method, with results considered statistically significant at p_FDR < 0.2. This ensures that significant findings reflect true associations, controlling for the risk of type I errors in the analysis. 40
Mediation Analysis
For the mediation analysis, a two-step approach was used to explore whether CSF biomarkers mediate the causal pathway between cortical structure and VTE. Following the initial MR analysis, which identified significant associations between cortical structure and VTE, the next phase examined if these links were mediated by CSF biomarkers (Figure 2C).
The mediation analysis proceeded in two stages: first, evaluating the causal effects of cortical structure on CSF biomarkers, followed by assessing the causal effects of CSF biomarkers on VTE. Significant associations from both steps were included in the mediation framework to determine if CSF biomarkers mediate the relationship (paths a and b) between cortical structure and VTE (path c). If significant, this result would support the hypothesis that neuroinflammatory or neurodegenerative processes, reflected in CSF biomarkers, contribute to linking cortical changes to thromboembolism risk. 29
Sensitivity Analyses and Additional Analyses
Several sensitivity analyses were carried out to ensure that the causal estimates remained robust and were not skewed by potential pleiotropy or weak instruments. MR-Egger regression was applied to evaluate directional pleiotropy, with a focus on the intercept test. A non-zero intercept would signal that some genetic instruments might influence the outcome through mechanisms unrelated to the primary exposure, potentially biasing the causal inference. To further refine the analysis and correct for any outliers driving pleiotropic effects, MR-PRESSO was utilized, offering an additional layer of adjustment to enhance the reliability of the results.41,42
A leave-one-out analysis was conducted, where each genetic variant was sequentially excluded from the analysis. This step ensures that no single variant disproportionately drives the causal estimate and helps identify variants that may have pleiotropic effects undetected by other methods.
Moreover, Cochran's Q statistic was calculated to assess heterogeneity across the genetic instruments. Significant heterogeneity could indicate violations of MR assumptions, particularly in terms of pleiotropy, thereby providing insight into the reliability of the selected instruments. Lastly, the Steiger test was employed to confirm the correct orientation of the genetic variants, ensuring that the direction of the SNP-exposure and SNP-outcome associations was appropriate for the analysis. 42
Co-Localization Analysis
Beyond the primary MR analysis, co-localization analysis was employed to assess whether the same genetic variants are implicated in both the exposures (cortical structure and CSF biomarkers) and VTE. This approach estimates the posterior probabilities for various hypotheses: H0 (neither trait is associated with the genetic variant), H1 (only cortical structure or CSF is associated), H2 (only VTE is associated), H3 (both traits are linked, but through distinct causal variants at the same locus), and H4 (both traits are associated and share the same causal variant). 43 The primary interest lies in H4, as it suggests that the genetic variants driving both cortical structure or CSF biomarkers and VTE are the same. A high posterior probability for H4 would indicate a shared genetic foundation between these traits, supporting the notion that the same genetic variants may contribute to both changes in cortical structure or CSF biomarkers and an increased risk of VTE.
All statistical analyses were conducted in R-Studio (version 4.4.1), utilizing key packages tailored to specific analytical needs: TwoSampleMR for Mendelian randomization analysis, MR-PRESSO for assessing pleiotropy, BWMR for Bayesian Weighted Mendelian Randomization, MR-RAPS for robust adjusted profile score analysis, and coloc for co-localization analysis.44–46
Results
Exploration of the Causal Effect of Cortical Structure on VTE
A comprehensive MR analysis was conducted to investigate the causal effects of various cortical structure features on VTE, with the inverse-variance weighted (IVW) method designated as the primary analytical approach. Prior to false discovery rate (FDR) correction, several cortical regions were identified as significantly associated with VTE. The lingual gyrus thickness demonstrated a statistically significant protective effect, with an odds ratio (OR) of 0.83 (p = .001). Similarly, the posterior cingulate surface area was associated with a protective effect, showing an OR of 0.85 (p = .021). The validity of the MR assumptions was carefully evaluated to ensure the robustness of these causal inferences on the other hand, the frontal pole thickness was associated with an increased risk of VTE, yielding an OR of 1.16 (p = .030). A similar detrimental effect was observed for the temporal pole thickness, with an OR of 1.20 (p = .030). Lastly, the posterior cingulate thickness showed a positive association with VTE risk, presenting an OR of 1.17 (p = .045). The above results are illustrated in Figures 3 and 4 and Supplementary Table 1A.
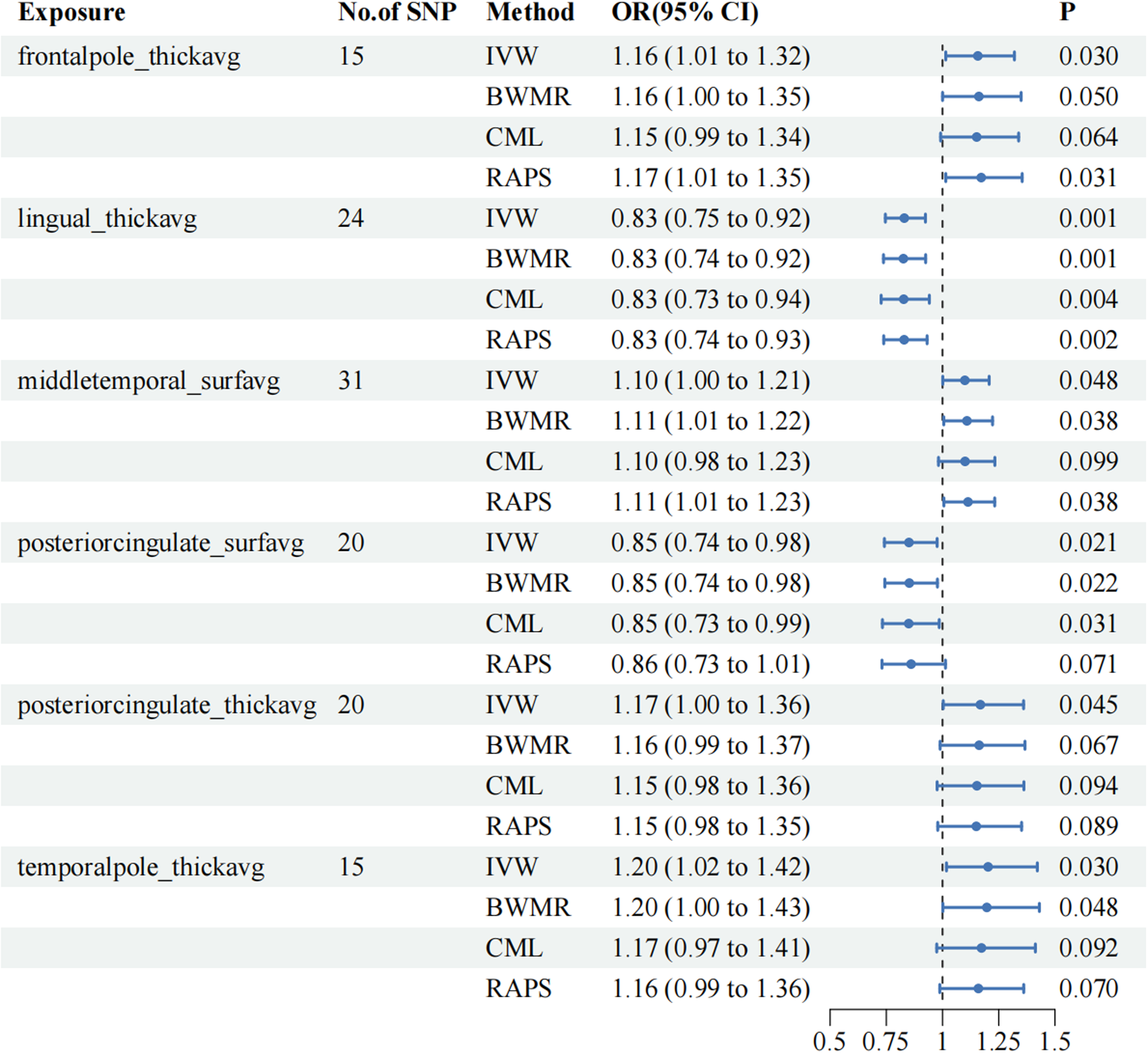
Significant Mendelian Randomization Estimates from Cortical Structure on Genetically Predicted VTE.

Scatter Plot Showed the Causal Associations Between Specific Cortical Structure and VTE.
The validity of the MR assumptions was carefully evaluated to ensure the robustness of these causal inferences. Horizontal pleiotropy, which can bias the estimates by violating the exclusion restriction assumption, was assessed using the MR-Egger intercept and Cochran's Q statistic to test for heterogeneity. For most cortical structure traits, no significant pleiotropy was detected, as indicated by MR-Egger intercepts and Q statistics (p > .05). Specifically, for the lingual gyrus thickness, both MR-Egger and IVW methods demonstrated no evidence of pleiotropy or heterogeneity, with Q values of 20.4375 (p = .5557) and 20.5203 (p = .6104), respectively (Figure 5).

Funnel Plot for the Overall Heterogeneity Between Specific Cortical Structure and VTE : (A) Cortical Thickness (TH) in the Frontal Pole; (B) TH in the Lingual Gyrus; (C) Cortical Surface Area (SA) Within Middle Temporal; (D) SA Within Posterior Cingulate; (E) TH in the Posterior Cingulate; (F) TH in the Temporal Pole.
However, heterogeneity was observed in the posterior cingulate thickness, as shown by Cochran's Q statistic, particularly for the IVW method (Q = 33.3584, p = .0218). This suggests variability in the genetic instruments, which may indicate pleiotropic effects, necessitating careful interpretation of these results. Despite this, the leave-one-out analysis confirmed that no single SNP disproportionately influenced the causal estimates, indicating that the overall findings remained robust across most cortical regions (Figure 6 and Supplementary Table 3A).

Leave One Out Analysis of the MR Results Between Specific Cortical Structure and VTE:.
After adjusting for multiple comparisons using FDR correction, only the association between lingual gyrus thickness and VTE remained statistically significant (p_FDR < 0.05). The IVW method revealed an OR of 0.8301 (p_FDR = 0.0457), reinforcing the protective role of this cortical region in VTE risk. Other regions, while suggestive of associations, did not maintain statistical significance after FDR correction, with p_FDR values exceeding 0.2, indicating that these initial associations were likely false positives due to multiple testing (Supplementary Table 1B).
Lastly, the MR Steiger test was used to confirm the directionality of the causal relationships, ensuring that the genetic variants were more strongly associated with the exposure than with the outcome. This test confirmed the correct directionality for all traits, verifying that the observed associations were causal rather than reversed (Supplementary Table 5).
Investigating the Impact of CSF Metabolites on VTE Through Causal Analysis
In the MR analysis conducted to explore the potential causal relationships between CSF metabolites and VTE, several associations were identified before applying false discovery rate (FDR) correction. The analysis used the inverse variance weighted (IVW) method, which initially revealed significant associations between specific metabolites and VTE risk.
Of particular interest, isoleucine levels were found to have a significant protective effect on VTE, with an odds ratio (OR) of 0.8150 (p = .0006), suggesting that higher levels of isoleucine reduce VTE risk. Similarly, leucine levels showed a protective association with an OR of 0.8146 (p = .0145). In addition, methylmalonate levels also demonstrated a protective association, with an OR of 0.9802 (p = .0003),
Other metabolites also showed initial associations with VTE before FDR correction. Sphingomyelin (d18:1/18:1, d18:2/18:0) levels were positively associated with VTE, with an OR of 1.0526 (95% CI: 1.0055-1.1019, p = .0280). Alpha-ketoglutarate levels displayed a similar positive association (OR = 1.1172, p = .0471). Additionally, metabolites such as 2-piperidone (OR = 0.9746, p = .0105), 4-methyl-2-oxopentanoate (OR = 1.1525, p = .0311), and 5,6-dihydrouridine (OR = 1.1930, p = .0379) also showed initial significant associations. Furthermore, hydroxy-cmpf, glycerate, and palmitoyl sphingomyelin (d18:1/16:0) levels were identified as potentially contributing to VTE risk, with ORs of 1.2939, 1.1747, and 1.0514, respectively (Figure 7 and Supplementary Table 2A).

The Causal Associations Between of CSF and VTE.
However, after applying FDR correction to account for multiple comparisons, most of these associations did not remain statistically significant. Isoleucine showed a suggestive association with VTE, as the p_FDR value was 0.1040, indicating a potential but not definitive relationship. Similarly, methylmalonate levels remained suggestive with a p_FDR value of 0.0937. The association between leucine levels and VTE, although initially significant, did not retain significance after FDR correction, with a p_FDR exceeding 0.2. Other metabolites, including sphingomyelin, alpha-ketoglutarate, and 2-piperidone, also lost their significance after FDR correction (Supplementary Table 2B).
The consistency of the causal associations was further supported by the results of additional MR methods and sensitivity analyses. The MR-Egger intercept and the global test within MR-PRESSO ruled out horizontal pleiotropy (Supplementary Table 3B), reinforcing the validity of the findings. Scatter plots and funnel plots (Figures 8 and 9) also indicated stability in the results, showing no signs of systematic bias. Similarly, leave-one-out analysis confirmed that none of the single nucleotide polymorphisms (SNPs) significantly influenced the causal estimates through pleiotropy (Figure 10). To validate the directionality of the associations, the MR Steiger test demonstrated that all SNPs were more strongly linked to the exposure than to the outcome (Supplementary Table 5), confirming the reliability of these results.
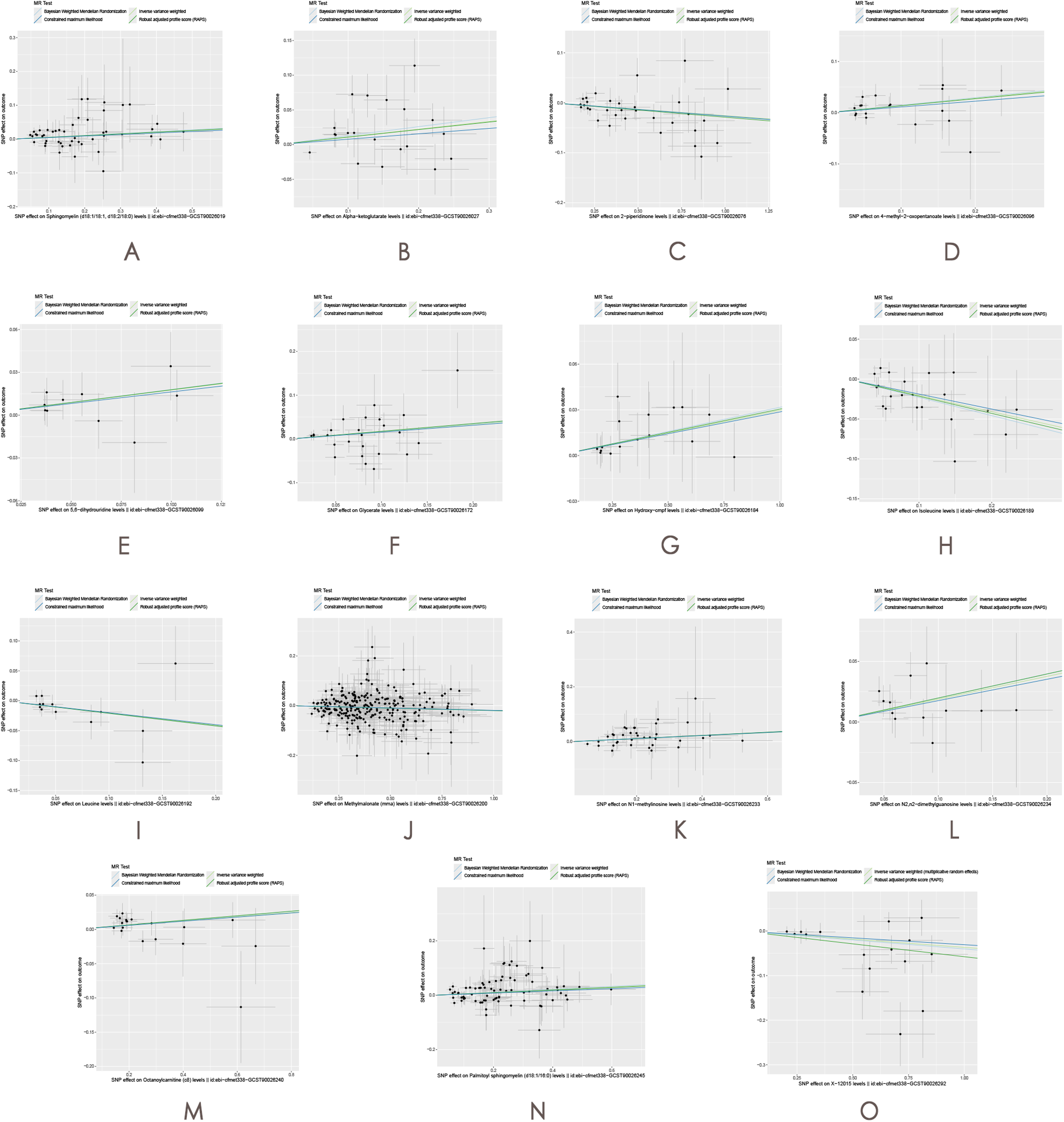
The effect of CSF on VTE. (A) Sphingomyelin(d18:1/18:1,d18:2/18:0) levels; (B) Alpha-ketoglutarate levels; (C) 2-piperidinone levels; (D) 4-methyl-2-oxopentanoate levels; (E) 5,6-dihydrouridine levels; (F) Glycerate levels; (G) Hydroxy-cmpf levels; (H) Isoleucine levels; (I) Leucine levels; (J) Methylmalonate (mma) levels; (K) N1-methylinosine levels; (L) N2,n2-dimethylguanosine levels; (M) Octanoylcarnitine (c8) levels; (N) Palmitoyl sphingomyelin (d18:1/16:0) levels; (O) X-12015 levels.

The Funnel Plot Illustrates the Overall Heterogeneity Between CSF and VTE.

Leave One Out Analysis of the MR Results Between CSF and VTE.
Mediation Analysis of Cortical Structure, CSF Biomarkers, and VTE Risk
In the mediation analysis, cortical structure features and CSF biomarkers, which had initially demonstrated causal relationships with VTE in the primary MR analysis, were further evaluated using a mediation MR approach. The goal was to assess whether CSF biomarkers functioned as mediators in the relationship between cortical structural alterations and VTE risk. For mediation to be established, a significant causal relationship between cortical structure features and CSF biomarkers would be required. However, the mediation MR analysis revealed no evidence supporting such a causal link, indicating that CSF biomarkers do not mediate the relationship between cortical structural changes and VTE risk. These findings ruled out CSF biomarkers as intermediaries in the neurovascular pathways linking cortical features to thromboembolic events (Figure 2C).
A complementary mediation analysis was conducted with reversed roles, evaluating cortical structure as a potential mediator between CSF biomarkers and VTE risk. Similarly, no causal evidence was identified to suggest that changes in CSF biomarkers influence cortical structure. This analysis further supports the conclusion that neither cortical structure features nor CSF biomarkers act as mediators in each other's pathways toward influencing VTE risk (Supplementary Table 4).
Co-Localization Analysis
The co-localization analysis was performed to assess whether cortical structure features and specific CSF metabolites share genetic variants that influence VTE. A key metric in this analysis, the posterior probability for hypothesis 4 (PP.H4), indicates whether both traits–exposure and outcome–are driven by the same genetic variant. For lingual gyrus thickness, the co-localization results showed a low PP.H4 value of 1.40E-02, suggesting that the cortical thickness and VTE are unlikely to share a common genetic variant. Similarly, isoleucine levels and methylmalonate levels also exhibited low PP.H4 values of 5.91E-03 and 7.47E-04, respectively, further indicating that these CSF metabolites do not share a significant genetic basis with VTE (Figure 11 and Supplementary Table 6). The leading SNPs associated with each trait, including rs4971696 for lingual gyrus thickness, rs6504411 for isoleucine, and rs138263676 for methylmalonate, showed no evidence of colocalization with VTE.

Colocalization Analysis Results for the Association Between Specific Cortical Structure Structures/CSF and VTE. (A) TH in the Lingual Gyrus; (B) Isoleucine Levels; (C) Methylmalonate Levels.
Discussion
This study provides novel evidence supporting potential causal relationships between specific cortical structure features, CSF metabolites, and VTE risk. Using Mendelian randomization, we identified six cortical traits and fifteen CSF metabolites that initially showed associations with VTE, but after correcting for multiple testing, only lingual gyrus thickness remained statistically significant. Isoleucine and methylmalonate levels exhibited suggestive associations, while the remaining traits did not maintain significance, emphasizing the importance of controlling for false positives in high-dimensional datasets.
The significant association between lingual gyrus thickness and VTE offers new insights into how cortical morphology might influence systemic vascular risk. Although this region is traditionally associated with visual and cognitive processing, the observed findings suggest it may indirectly modulate vascular health through its influence on autonomic function and inflammatory regulation. Cortical areas involved in sensory integration and neurovascular coupling are known to affect autonomic outflow and cytokine signaling, both of which are critical in thrombus formation. 47 Neuroinflammatory responses regulated by these circuits could contribute to systemic vascular vulnerability, implying that structural alterations in the lingual gyrus may reflect a broader neurovascular mechanism predisposing to VTE.
From a clinical imaging perspective, the detection of cortical thickness changes using MRI is a non-invasive technique already embedded in routine neurological assessment. This makes it a promising candidate for early thromboembolic risk stratification, especially in populations with neurovascular comorbidities.19,47,48 While the lingual gyrus has not previously been directly implicated in VTE, the current findings point to its potential relevance, particularly in patients with neurological disorders who are already at increased thrombotic risk due to immobility or systemic inflammation.
The potential involvement of CSF metabolites adds an additional dimension to our understanding of VTE pathophysiology. Among the metabolites analyzed, isoleucine–a branched-chain amino acid–demonstrated a potential inverse relationship with VTE risk. Although elevated isoleucine has been linked to metabolic conditions such as insulin resistance and cardiovascular disease, it may also exert anti-thrombotic effects through energy metabolism or anti-inflammatory pathways. 49 Dysregulation of the mammalian target of rapamycin (mTOR) pathway, which is influenced by amino acid availability, may also contribute to endothelial dysfunction and a pro-thrombotic state.50,51 Similarly, methylmalonate–a biomarker of vitamin B12 deficiency–was suggestively associated with increased VTE risk. Elevated methylmalonate reflects impaired cellular metabolism and is implicated in systemic inflammation and endothelial dysfunction. In clinical settings, it serves as a sensitive indicator of metabolic stress, particularly in patients with neuropathy or cardiovascular disease. Although direct links between methylmalonate and VTE remain unconfirmed, its role in promoting vascular damage supports a plausible connection to thrombosis. 52 By contributing to systemic metabolic imbalances, these metabolite could facilitate endothelial damage and promote a pro-thrombotic state. 53
Despite these associations, mediation analyses did not reveal any causal pathways linking lingual gyrus thickness with isoleucine or methylmalonate levels, suggesting that cortical structure and CSF metabolites contribute to VTE risk via biologically distinct mechanisms. Cortical traits may influence neurovascular control, whereas metabolites likely act through systemic metabolic and inflammatory routes. Co-localization analyses further supported this conclusion: posterior probability values (PP.H4) were low for all three traits, indicating a lack of shared genetic variants underlying both exposures and VTE. The top associated SNPs–rs4971696 (lingual gyrus thickness), rs6504411 (isoleucine), and rs138263676 (methylmalonate)—did not overlap with known VTE loci, reinforcing the hypothesis of independent genetic influences.
These mechanistic distinctions are crucial for understanding the multifactorial nature of thromboembolic risk. While cortical structural changes may reflect central regulation of vascular tone and inflammation, CSF metabolites likely mark peripheral metabolic states contributing to endothelial dysfunction. Their combined investigation offers a broader systems-level perspective on VTE vulnerability. The implications of this study are significant for understanding VTE risk, particularly concerning cortical structure and metabolic biomarkers. Identifying lingual gyrus thickness and metabolites such as isoleucine and methylmalonate as potential markers for VTE highlights the importance of integrating neurovascular and metabolic pathways in thromboembolic disease research. While no causal mediating effect was observed between cortical structure and CSF metabolites, the associations found suggest that neuroimaging could serve as a non-invasive tool for early risk stratification in clinical settings, particularly for high-risk populations such as those with neurological disorders.
By employing a MR design, this study provides robust causal inferences through genetic variants, reducing confounding and reverse causation biases. The findings also advance our understanding of how cortical structures and metabolic markers contribute to VTE risk through independent biological pathways, as confirmed by co-localization analysis. However, these insights must be interpreted alongside the study's limitations. The reliance on GWAS data from predominantly European populations limits the generalizability of these findings to other ancestries. Additionally, the resolution of MRI may not capture subtle brain structural changes that could influence vascular health. Despite efforts to minimize pleiotropy, it remains a potential confounding factor. Furthermore, the cross-sectional nature of the data limits temporal inferences, and unmeasured environmental factors may have influenced the results.
Moreover, the VTE GWAS data were exclusively derived from the FinnGen cohort, which consists of individuals of Finnish ancestry. While this homogeneous sample helps reduce population stratification bias, it may restrict the external validity of the findings. As such, caution is warranted when extrapolating these results to non-Finnish or more genetically diverse populations.
From a clinical perspective, these results suggest promising applications for non-invasive cortical imaging and metabolic profiling in assessing VTE risk, particularly among neurological patients who face elevated thromboembolic risks. Future research should prioritize diversifying genetic datasets by including populations with broader ancestral backgrounds and conducting longitudinal studies to clarify the temporal dynamics within these relationships.
Conclusion
Our study identifies potential associations between lingual gyrus thickness, isoleucine, and methylmalonate with VTE, highlighting distinct neurovascular and metabolic pathways that may contribute to VTE risk. Although causal mediation was not confirmed, these findings emphasize the importance of exploring both cortical structure and metabolic profiles in VTE research. The results underscore the potential of using non-invasive cortical imaging and metabolic screening for risk stratification, particularly in high-risk neurological patients. Future research should focus on diversifying genetic datasets, employing longitudinal designs, and investigating interactions between neurovascular and metabolic pathways to further elucidate VTE pathophysiology.
Supplemental Material
sj-xlsx-1-cat-10.1177_10760296251360018 - Supplemental material for Linking Cortical Structure and Cerebrospinal Fluid Metabolites with Venous Thromboembolism Risk: A Two-Step Mendelian Randomization and Co-Localization Analysis
Supplemental material, sj-xlsx-1-cat-10.1177_10760296251360018 for Linking Cortical Structure and Cerebrospinal Fluid Metabolites with Venous Thromboembolism Risk: A Two-Step Mendelian Randomization and Co-Localization Analysis by Tianni Liu, Junxian Li, Qiong Liu and Guojun Liang in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgements
We thank all the participants and researchers who contributed to the GWAS and provided the summary data.
Ethics Approval and Consent to Participate
Not applicable. We used summary-level data from publicly available GWAS studies which have received ethical approval from their respective institutional review boards and informed consent from all participants. No administrative permissions were required to access the data.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This work was supported by the Huadu District General Medical Research Special Project [NO.24-HDWS-004].
Data Availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
