Abstract
Lung cancer is frequently complicated by coagulation abnormalities, primarily presenting as venous thromboembolism (VTE) but also including bleeding events in select patients. This hypercoagulable state arises from a multifaceted interplay between tumor-derived procoagulants, systemic inflammation, endothelial dysfunction, autoimmune mechanisms, and treatment-associated effects. These disruptions not only elevate thrombotic risk but also influence prognosis and therapeutic decisions. Furthermore, the presence of antiphospholipid antibodies or acquired coagulation inhibitors, such as lupus anticoagulant and anti-Factor VIII antibodies, poses a unique clinical challenge by increasing the risk of both thrombosis and hemorrhage. Recent data highlight the expanding role of tumor-derived extracellular vesicles, immune-mediated coagulopathies, and prothrombotic effects of immune checkpoint inhibitors in lung cancer–associated thrombosis. Novel biomarkers-such as prothrombin fragment 1 + 2, integrin β2– positive extracellular vesicles, and endothelial activation markers-are being investigated for risk stratification and personalized treatment decisions. Diagnostic approaches-including coagulation assays, biomarker profiling, and imaging-are discussed alongside emerging tools for individualized anticoagulation and management of acquired inhibitors. Early recognition and tailored management of cancer-associated coagulopathy are essential to improve patient outcomes. This review aims to provide practical insights for clinicians navigating the thrombohemorrhagic spectrum in lung cancer care.
Keywords
Introduction
Lung cancer remains the leading cause of cancer-related mortality worldwide, accounting for more than 1.8 million deaths annually. 1 Despite advances in early detection and targeted therapies, the prognosis for many patients remains poor, largely due to systemic complications beyond tumor burden itself. Among these, coagulation disorders represent a significant clinical challenge, with venous thromboembolism (VTE) being one of the most frequent and consequential events.2,3
The intricate association between cancer and thrombosis, first observed by Armand Trousseau in the nineteenth century, is now recognized as a distinct clinical entity termed cancer-associated thrombosis (CAT). 4 Lung cancer, particularly non-small cell lung cancer (NSCLC) and adenocarcinoma, exhibits one of the highest VTE incidences among all malignancies, with reported rates approaching 15% to 20% depending on disease stage and treatment modalities.5,6
Multiple factors contribute to the hypercoagulable state observed in lung cancer patients. Tumor-derived procoagulants, such as tissue factor (TF) and cancer cell- derived extracellular vesicles, initiate and propagate the coagulation cascade. 7 Additionally, systemic inflammation characterized by elevated interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α) levels promotes a prothrombotic milieu through endothelial activation and platelet aggregation. 8 Emerging evidence also implicates neutrophil extracellular traps (NETs) in facilitating thrombus formation in cancer patients, including those treated with immune checkpoint inhibitors. 9
Importantly, the presence of coagulation abnormalities in lung cancer patients carries independent prognostic implications. Studies have consistently demonstrated that VTE occurrence is associated with decreased overall survival, increased hospitalization rates, and treatment interruptions. 10 Moreover, a subset of patients develops acquired coagulation inhibitors, such as lupus anticoagulant or antibodies against clotting factors, which paradoxically predispose them to both thrombotic and hemorrhagic complications, further complicating management. 11
Early identification of patients at high thrombotic risk remains a cornerstone of optimal lung cancer management. Traditional clinical risk scores, such as the Khorana score, offer some predictive value but are limited in their applicability to lung cancer populations, necessitating the integration of novel biomarkers like soluble P-selectin, D-dimer, and TF-positive extracellular vesicles into stratification models. 12 Recent efforts are also focusing on molecular and genetic predictors of VTE risk, such as ROS1 rearrangements, which appear to confer heightened thrombotic susceptibility. 13 Recent efforts are increasingly focusing on molecular and genetic predictors of venous thromboembolism (VTE) risk in lung cancer. Among these, rearrangements involving the ROS1 gene have garnered particular attention. ROS1 fusions, which occur in approximately 1%–2% of non-small cell lung cancers (NSCLC), especially in younger, non-smoking patients with adenocarcinoma histology, are associated with a significantly elevated incidence of thromboembolic events. This prothrombotic predisposition may stem from oncogenic signaling cascades that promote tissue factor expression and the release of procoagulant extracellular vesicles. The association between ROS1 rearrangements and heightened thrombotic risk has been substantiated in both clinical trials and recent comprehensive reviews, underscoring the potential utility of ROS1 status as a biomarker for VTE risk stratification in NSCLC patients.13,14
This review aims to provide an updated, clinically relevant overview of coagulation abnormalities in lung cancer, emphasizing pathophysiological mechanisms, diagnostic approaches, prognostic implications, and emerging therapeutic considerations.
Mechanisms of Hypercoagulability in Lung Cancer
Lung cancer is associated with a complex hypercoagulable state driven by multifactorial processes involving tumor biology, systemic inflammation, endothelial dysfunction, immune responses, and treatment-related effects.5,15,16 These overlapping mechanisms promote thrombin generation, suppress fibrinolysis, and enhance platelet activation, contributing to the high incidence of venous thromboembolism (VTE) in this patient population.2,3
Tumor-Derived Procoagulants
Lung cancer cells, particularly adenocarcinomas, frequently overexpress tissue factor (TF), a key initiator of the extrinsic coagulation cascade. 6 TF forms a complex with activated factor VII (FVIIa), leading to downstream activation of factor X and thrombin generation. Moreover, TF-bearing microparticles released by tumor cells amplify coagulation by activating endothelial cells and platelets. Recent studies have highlighted the role of tumor-derived extracellular vesicles rich in TF as pivotal mediators of cancer-associated thrombosis. 7 TF expression also correlates with tumor invasiveness and metastatic potential, linking procoagulant phenotype with tumor progression. 17
Recent data by Lucotti et al 18 have expanded our understanding of the role of extracellular vesicles (EVs) in cancer-associated thrombosis and metastasis. The study identified a lung-specific pro-thrombotic niche enriched in reprogrammed macrophages that secrete small EVs (sEVs) containing integrin β2. These vesicles interact with platelet glycoprotein Ib (GPIb), enhancing platelet aggregation and facilitating thrombus formation. Importantly, integrin β2 on sEVs forms heterodimers with integrin αX and promotes both thrombosis and pulmonary metastasis. The chemokine CXCL13 was shown to drive the reprogramming of macrophages into a pro-thrombotic phenotype, highlighting its central role in shaping the local thromboinflammatory microenvironment. Inhibition of integrin β2 significantly reduced thrombotic burden and metastatic spread in vivo. Furthermore, elevated circulating levels of sEV–β2 were detected in patients with pancreatic cancer prior to VTE onset, suggesting a potential role as a predictive biomarker. These findings provide new insight into the mechanisms linking inflammation, EV-mediated coagulation, and metastatic dissemination in lung cancer.
Inflammatory Cytokine Activation
The tumor microenvironment in lung cancer is enriched with inflammatory cytokines, including interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), and interleukin-1 beta (IL-Ιβ), which upregulate TF and suppress endogenous anticoagulants such as antithrombin and protein C.19,20 IL-6 further enhances thrombopoiesis via stimulation of thrombopoietin, contributing to elevated platelet turnover. These cytokines also promote the expression of plasminogen activator inhibitor-1 (PAI-1), thereby impairing fibrinolysis. Recent clinical evidence suggests that IL-6 inhibitors may have a potential role in modulating thrombosis risk in cancer patients. 8
Endothelial Dysfunction and Platelet Activation
Endothelial dysfunction is a hallmark of the prothrombotic state in lung cancer. 5 Factors such as oxidative stress, cytokine-induced injury, and tumor invasion reduce the expression of thrombomodulin and tissue plasminogen activator (tPA), while upregulating endothelial adhesion molecules like E-selectin and ICAM-1. Tumor platelet interactions exacerbate coagulation, as platelets not only facilitate clot formation but also shield circulating tumor cells from immune surveillance, promoting metastasis. 6 Emerging data suggest that platelet-derived growth factors may actively promote a metastatic niche formation in lung cancer. 21
Autoimmunity and Acquired Inhibitors
Autoimmune manifestations, including the presence of antiphospholipid antibodies and lupus anticoagulant, have been reported in lung cancer and are associated with paradoxical thrombosis despite laboratory signs of hypocoagulability. 4 Rarely, lung cancer patients may develop acquired coagulation inhibitors, such as antibodies to factor VIII or IX, resulting in spontaneous, life-threatening bleeding episodes. Recent case series highlight the diagnostic challenge of acquired hemophilia in malignancy- associated settings, requiring specialized assays such as mixing studies and Bethesda titers for timely intervention. 11
Treatment-Related Risks
Antineoplastic therapies, including platinum-based chemotherapy, anti-angiogenic agents (eg, bevacizumab), and immune checkpoint inhibitors (ICIs), contribute significantly to thrombotic risk in lung cancer. 6 ICIs, in particular, have been associated with increased thromboinflammatory responses. 22 Surgical interventions—especially in elderly or immobilized patients—further exacerbate the risk of both thrombosis and bleeding. Balancing disease control with the potential for thrombohemorrhagic complications remains crucial in oncologic decision-making.2,15 Balancing disease control with the potential for thrombohemorrhagic complications remains crucial in oncologic decision-making. These overlapping prothrombotic and hemorrhagic pathways are summarized below (Figure 1, Table 1).
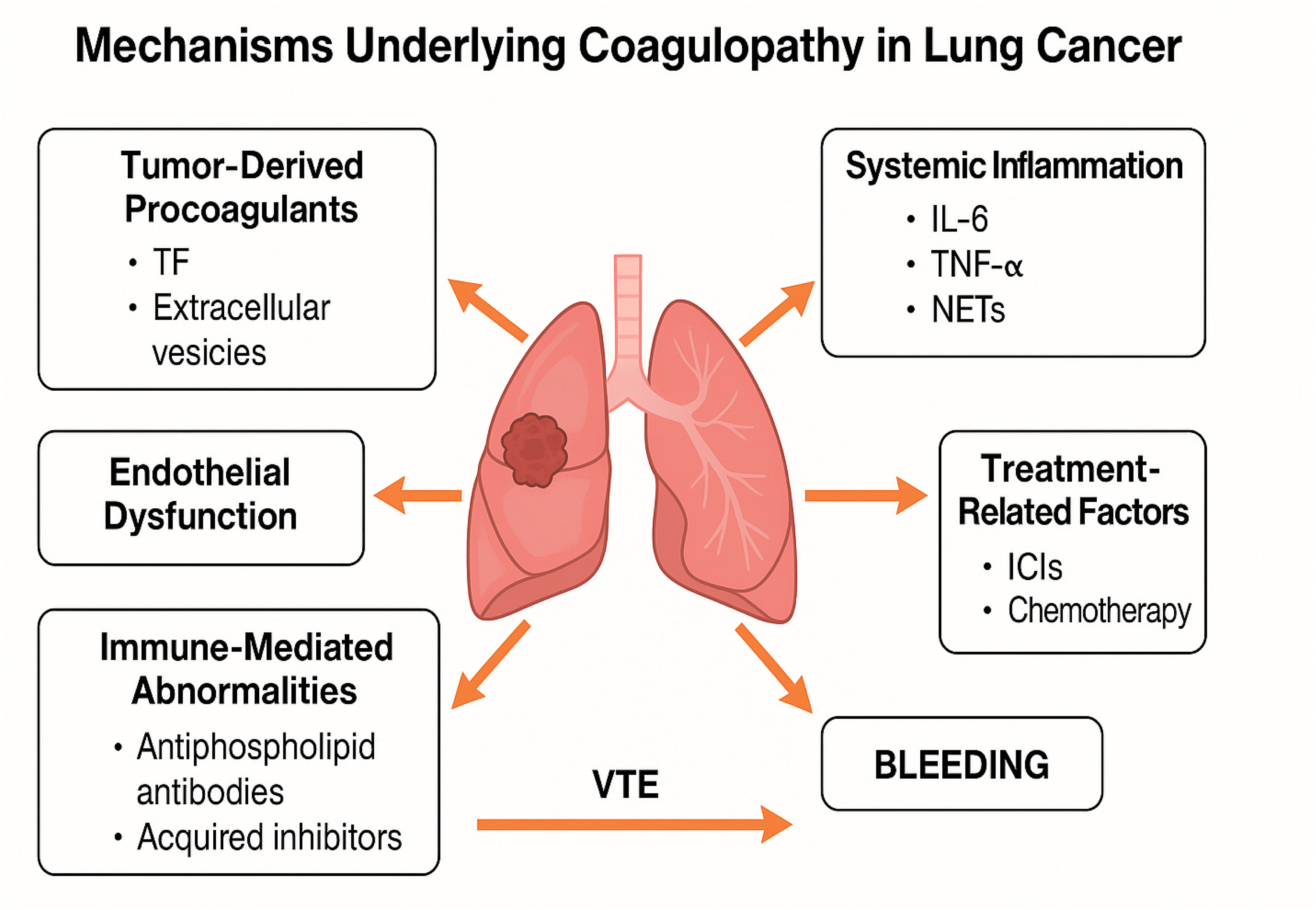
Mechanisms Underlying Coagulopathy in Lung Cancer. The diagram illustrates the multifactorial mechanisms contributing to coagulation abnormalities in lung cancer. Tumor-derived procoagulants such as tissue factor (TF) and extracellular vesicles initiate thrombogenesis. Systemic inflammation involving IL-6, TNF-α, and neutrophil extracellular traps (NETs) enhances the prothrombotic state. Endothelial dysfunction and immune-mediated abnormalities, including antiphospholipid antibodies and acquired inhibitors, further exacerbate coagulation disorders. Treatment- related factors, particularly immune checkpoint inhibitors (ICIs) and chemotherapy, also increase thrombotic and bleeding risks. These overlapping pathways culminate in venous thromboembolism (VTE) and, in select cases, hemorrhagic complications.
Mechanisms, Biomarkers, and Clinical Implications of Coagulopathy in Lung Cancer.

A Summary of the Main Pathogenic Mechanisms Driving Coagulopathy in Lung Cancer, Along with Associated Biomarkers and Their Clinical Relevance for Diagnosis, Prognosis, and Therapeutic Decision-Making.
*EVs = extracellular vesicles, VTE = venous thromboembolism, CRP = C-reactive protein, NETs = Neutrophil Extracellular Traps, PAI-1 = plasminogen activator inhibitor-1, MPO = myeloperoxidase, ICI = immune checkpoint inhibitor, VWF = von Willebrand factor.
Diagnostic Strategies in Cancer-Associated Coagulopathy
The diagnosis of cancer-associated coagulopathy (CAC) in lung cancer patients necessitates a multifaceted approach, integrating clinical assessment, laboratory biomarkers, and imaging modalities. Early identification of patients at heightened risk for venous thromboembolism (VTE) is paramount, as it enables timely prophylactic interventions and tailored therapeutic strategies.
Clinical Risk Assessment Tools
Several risk assessment models (RAMs) have been developed to stratify VTE risk in cancer patients. The Khorana score, which incorporates factors such as cancer type, platelet count, hemoglobin level, leukocyte count, and body mass index, is widely utilized. However, its predictive accuracy in lung cancer patients is moderate, prompting the development of lung cancer-specific models. The COMPASS-CAT model, for instance, includes variables like metastatic disease, history of VTE, and use of erythropoiesis-stimulating agents, offering improved risk stratification in this cohort. 23
Laboratory Biomarkers
Notably, a prospective observational study by Tsubata et al 24 introduced a novel risk assessment model for VTE in advanced lung cancer. Their findings highlighted that low platelet counts and elevated diastolic blood pressure (DBP) were independent risk factors for thromboembolism, deviating from conventional assumptions. Interestingly, they also reported that elevated D-dimer levels did not correlate with VTE risk in their cohort, suggesting that D-dimer may lack specificity in this population. Instead, prothrombin fragment 1 + 2 (PT F1 + 2), a marker of thrombin generation, was identified as a more suitable biomarker for VTE risk stratification.
These insights emphasize the need for refined, context-specific biomarker validation in lung cancer patients, as laboratory biomarkers remain a cornerstone in evaluating the hypercoagulable state. Among the most studied markers, elevated D-dimer levels are frequently observed and have traditionally been associated with increased VTE risk. Additionally, soluble P-selectin, a marker of platelet activation, and thrombin generation markers, such as PT F1 + 2 and thrombin–antithrombin complexes, have demonstrated predictive value for VTE in cancer patients. 25
Building upon these foundations, recent diagnostic approaches have incorporated advanced assays and technologies that enhance sensitivity and specificity for cancer-associated coagulopathy. Thrombin generation assays (TGAs), including calibrated automated thrombography, provide a global assessment of coagulation potential and have shown promise in identifying hypercoagulable profiles in cancer patients. 26 Moreover, high-sensitivity ELISA platforms and flow cytometry-based detection of tissue factor– bearing extracellular vesicles (TF-EVs) are increasingly used in research settings to quantify procoagulant activity. 7 Novel assays targeting neutrophil extracellular trap (NET) markers such as MPO-DNA complexes and cell-free DNA also offer emerging diagnostic potential, particularly in the context of immunotherapy-associated thrombosis. 9 Integration of these tools with traditional markers like D- dimer and PT F1 + 2 may improve diagnostic accuracy and risk stratification in lung cancer–associated thrombosis.
Imaging Modalities
Imaging studies are integral to the diagnosis of VTE. Compression ultrasonography is the first-line modality for detecting deep vein thrombosis, while computed tomography pulmonary angiography (CTPA) is the gold standard for diagnosing pulmonary embolism. In certain cases, ventilation-perfusion (V/Q) scanning may be employed, particularly when CTPA is contraindicated.
Molecular and Genetic Markers
Emerging evidence suggests that molecular and genetic factors may influence thrombotic risk in lung cancer patients. For example, patients harboring ROS1 rearrangements have been found to have a higher incidence of VTE compared to those without such genetic alterations. Further research is warranted to elucidate the role of genetic profiling in VTE risk assessment. 27
Integrative Approaches
An integrative diagnostic approach, combining clinical risk models, biomarker assessment, and imaging studies, offers the most comprehensive strategy for evaluating CAC in lung cancer patients. Such a multifaceted assessment enables personalized risk stratification and informs clinical decision-making regarding prophylactic and therapeutic interventions.
Discussion
The interplay between lung cancer and coagulopathy represents a clinically significant and complex phenomenon with profound implications for patient management and prognosis. Recent advances in our understanding of cancer biology have underscored the dynamic role of the hemostatic system in promoting tumor progression, immune evasion, and metastatic dissemination. 28 Hypercoagulability is not merely a complication of malignancy but an integral component of the tumor microenvironment.
Tumor-derived procoagulant activity, primarily mediated through tissue factor (TF) and TF-positive extracellular vesicles, continues to emerge as a key driver of cancer- associated thrombosis.7,29 These microparticles facilitate cross-talk between cancer cells, endothelial cells, and platelets, creating a systemic prothrombotic state. Importantly, circulating levels of TF-bearing vesicles have been correlated with VTE risk and worse overall survival in lung cancer patients, suggesting their potential role as prognostic biomarkers. 10
Inflammatory mediators also significantly modulate the thrombotic milieu. Interleukin- 6 (IL-6), tumor necrosis factor-alpha (TNF-α), and neutrophil extracellular traps (NETs) promote thrombin generation and impair endogenous anticoagulant pathways.8,9 The emerging role of NETs as scaffolds for thrombus formation, especially in the context of immune checkpoint inhibitor therapy, introduces new complexities into the pathophysiology of cancer-associated thrombosis. 30
Endothelial dysfunction, characterized by downregulation of thrombomodulin and upregulation of adhesion molecules such as ICAM-1 and VCAM-1, further amplifies the procoagulant state.5,21 In lung cancer patients, endothelial activation markers have been linked not only to VTE but also to poor treatment outcomes, highlighting the need for endothelial-targeted therapeutic strategies.
The autoimmune dimension of coagulopathy in lung cancer is increasingly recognized. Acquired antiphospholipid syndrome and inhibitors to clotting factors, such as factor VIII, present substantial diagnostic and therapeutic challenges. A growing body of evidence suggests that paraneoplastic immune dysregulation contributes to the development of such abnormalities, necessitating heightened clinical vigilance and access to specialized coagulation testing.11,31
Beyond acquired hemophilia and antiphospholipid syndrome, bleeding in lung cancer patients may also manifest in the form of massive hemoptysis, particularly in advanced or centrally located tumors. A recent prospective study by Salvayre et al 32 involving ICU patients reported that severe hemoptysis in lung cancer is not only life-threatening but also prone to recurrence, with significant implications for in-hospital mortality and long-term outcomes. These findings underscore the importance of early identification of bleeding sources, multidisciplinary management involving interventional radiology, and consideration of palliative bronchial artery embolization in selected cases. As such, hemorrhagic complications should be considered alongside thrombotic events when stratifying risk and planning anticoagulation strategies in this population.
Treatment-related risks must also be carefully navigated. Platinum-based chemotherapy, antiangiogenic agents like bevacizumab, and immune checkpoint inhibitors have all been implicated in enhancing thrombotic risk.6,22 Particularly, ICIs have been associated with a heightened incidence of thromboinflammatory events, possibly through NETosis and cytokine storms, underscoring the need for proactive thromboembolic risk monitoring in these patients.
Despite these insights, substantial challenges remain in optimizing the management of cancer-associated thrombosis (CAT) in lung cancer. Current clinical risk models, such as the Khorana score, show limited predictive accuracy in this population, prompting calls for lung cancer-specific stratification tools integrating biomarkers such as D- dimer, soluble P-selectin, and TF-bearing extracellular vesicles. 33
In this context, recent evidence from a prospective observational study by Tsubata et al 24 offers a fresh perspective on VTE risk prediction in advanced lung cancer. Their findings challenge existing paradigms by demonstrating that low platelet count and elevated diastolic blood pressure (DBP) were independently associated with increased VTE risk-parameters not typically emphasized in standard models. Moreover, the study revealed that elevated D-dimer levels, though widely used, did not significantly correlate with thrombotic events in this cohort. Instead, prothrombin fragment 1 + 2 (PT F1 + 2), a marker of thrombin generation, emerged as a more reliable biomarker of VTE risk stratification. These findings advocate for a revision of current biomarker-driven risk assessment tools and support the integration of alternative physiological and hemostatic parameters tailored to lung cancer patients. These insights not only inform future diagnostic strategies but also have therapeutic implications, particularly regarding anticoagulant selection and monitoring.
Therapeutically, low molecular weight heparin (LMWH) remains the cornerstone of anticoagulation. However, direct oral anticoagulants (DOACs) have gained ground due to ease of administration and favorable safety profiles, although concerns about bleeding, particularly in patients with gastrointestinal involvement or coagulopathy, persist. 34 Individualized risk-benefit assessments remain critical, especially in patients with concurrent bleeding diatheses.
Looking forward, research should focus on validating novel biomarkers for thrombotic risk stratification and exploring the efficacy of targeted therapies that modulate the tumor–coagulation interface. Inhibitors of TF, NETs, and platelet–tumor interactions represent promising avenues.18,30 Furthermore, integrating omics technologies to characterize the thromboinflammatory signatures of lung cancer may offer personalized approaches to thrombosis prevention.
Ultimately, a multidisciplinary model of care, incorporating oncology, hematology, radiology, and supportive services, is essential to effectively address the thrombohemorrhagic challenges faced by lung cancer patients. Tailored strategies based on evolving risk profiles, therapeutic interventions guided by biomarker-driven models, and early intervention will be pivotal in improving both survival and quality of life.
Conclusion and Future Directions
Coagulation disorders in lung cancer result from a complex interplay of tumor-derived procoagulants, chronic inflammation, immune dysregulation, endothelial injury, and treatment-related effects. These mechanisms predominantly manifest as venous thromboembolism (VTE), but may also include bleeding complications, particularly in patients with acquired coagulation inhibitors or thrombocytopenia.
These abnormalities extend beyond acute clinical events. They may reflect underlying tumor biology and influence prognosis, making early recognition and proactive management critical in oncologic care. Advances in diagnostic tools-including targeted biomarker panels, functional coagulation assays, and refined imaging-are enhancing our ability to stratify risk and tailor therapy.
Future research should aim to integrate molecular, inflammatory, and coagulation data into unified risk assessment models. Prospective studies assessing anticoagulation strategies in patients with immune-mediated coagulopathies or specific biomarker profiles are urgently needed. Additionally, therapies targeting tissue factor expression, neutrophil extracellular traps (NETs), or platelet–tumor interactions may open new avenues for intervention.
Ultimately, a multidisciplinary, patient-centered approach remains essential for managing this complex landscape. With improved tools for risk prediction and an emphasis on personalized care, clinicians can better mitigate thrombohemorrhagic complications and improve outcomes in lung cancer patients.
Clinical Integration: Key Recommendations for Managing Cancer-Associated Coagulopathy
Effective management of cancer-associated coagulopathy requires a structured, multidisciplinary approach integrating established guidelines with emerging biomarker-based strategies:
All patients with malignancy-particularly those with lung cancer-should undergo thromboembolic risk assessment at diagnosis using validated tools such as the Khorana score and D-dimer levels. Risk reassessment is recommended at key treatment milestones.
28
In patients with unexplained bleeding, prolonged aPTT, or thrombotic complications, advanced testing including lupus anticoagulant assays, mixing studies, and inhibitor screens is critical to differentiate acquired versus inherited defects.
35
Anticoagulant therapy must be individualized based on bleeding risk, tumor type, and comorbidities. Low molecular weight heparin (LMWH) remains the standard first-line option, while direct oral anticoagulants (DOACs) may be appropriate in select patients with lower bleeding risk.
36
Suspected acquired factor inhibitors warrant urgent hematology consultation and access to bypassing agents (eg, recombinant factor VIIa or activated prothrombin complex concentrates) alongside immunosuppressive therapy.
37
Optimal outcomes depend on coordinated care involving oncologists, hematologists, pharmacists, and supportive care teams, particularly in high-risk or unstable patients.
Structured patient education regarding signs of venous thromboembolism (VTE) and bleeding must be an integral component of every treatment plan.
Measurement of prothrombin fragment 1 + 2 (F1 + 2) may offer added diagnostic value in VTE risk stratification, especially in cases with non-predictive D-dimer levels.
26
Evidence suggests that elevated diastolic blood pressure and thrombocytopenia represent independent VTE predictors in patients with advanced lung cancer and should be incorporated into individualized models where applicable.
24
In the presence of severe hemoptysis, especially with centrally located tumors or cavitating lesions, anticoagulation must be withheld or deferred. Management should include prompt evaluation by interventional radiology, thoracic oncology, and intensive care specialists.
32
Recent research highlights the predictive utility of small extracellular vesicles (sEVs) expressing integrin β2 (sEV–β2) in cancer-associated thrombosis and metastatic progression. Additionally, CXCL13-mediated macrophage activation may serve as an upstream marker of prothrombotic remodeling within the tumor microenvironment.
18
These findings may contribute to future biomarker-driven risk stratification frameworks.
Footnotes
Ethics Considerations
The present research complies with the guidelines for human studies, and the research was conducted ethically in accordance with the World Medical Association Declaration of Helsinki. This retrospective review of patient data did not require ethical approval in accordance with local/national guidelines.
Authors’ Contributions
Α.Μ. and F.G. were responsible for the conception and design of the study. All authors were involved in data acquisition, interpretation, drafting and review of the article as well as its final approvement.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated in preparing this review of the literature.
