Abstract
The aim of this single-centre 10-year retrospective observational study was to evaluate the profile of Chinese cancerous patients complicated with venous thromboembolism (VTE) based on demographic features, clinical characteristics, and medication exposure. Consecutive 1482 patients with solid tumor complicated with VTE at a tertiary center between 2012 and 2021 were retrospectively enrolled. Data were collected on demographics, comorbidities, discharge diagnoses, laboratory examination data, treatment details, and imaging description of the lesion. The overall incidence of clinical VTE was 1.35% in hospitalized patients with cancer in our center. Lung cancer was the most frequent tumor subtype for developing VTE events, accounting for 24.83% of all cases. Over half of the patients (66.60%) were observed to have an increased risk of VTE within the first 6 months of cancer diagnosis. Close to half of the patients (46.49%) had received chemotherapy within 6 months prior to the diagnosis of VTE. The frequency of massive ascites group (>2000 mL) in gynecological patients with VTE was significantly larger than that of nonmassive ascites group (≤2000 mL) (P < .001). Patients with ovarian, vulvar, lung cancers were considered at high risk for VTE. The assessment and monitoring of VTE in patients with cancer within the first 6 months of cancer diagnosis should be strengthened. VTE occurrence was closely related to advanced age and stage, adenocarcinoma, obesity and noval anticancer therapies in patients with cancer. Early detection of VTE-related examination may lead to earlier intervention for patients with gynecological tumors with preoperative massive ascites.
Keywords
Introduction
Venous thromboembolism (VTE), which comprises deep vein thrombosis (DVT) and pulmonary thromboembolism (PE), is a major global burden of disease. It has been established that there is a strong association between cancer and thromboembolic events. 1 Patients with malignancy are at a high risk of VTE, account for approximately 20% of all patients complicated with vein thrombosis, and have a 4-fold to 9-fold increased risk of developing VTE compared to the general population.2,3 Furthermore, cancer-associated thrombosis is commonly associated with higher morbidity and mortality, increased hospital admission, reduced quality of life and poor survival.4–6 As a result, VTE is responsible for 9% of death in patients with cancer, making it the second leading cause of death in patients with cancer. 7
Despite the well-established association of thrombosis in patients with cancer, it is equally important to note that the risk of VTE appears to increase with the use of newer anticancer therapies, such as targeted therapy, radiotherapy, and recently immunotherapy.8,9 In recent years, as a result of improvements in recognition and diagnosis of the disease, increasing attention is being paid to the importance of thrombosis in patients with cancer under the in-hospital VTE prevention and control system. Hence, the characteristics of patients with cancer presenting with VTE are evolving, but there are currently few studies related to the epidemiology and the main etiology of VTE specific to the Chinese cancerous population. 10 Therefore, in this single-centre, retrospective analysis, records of patients with solid tumor complicated with VTE based on 1482 cases were analyzed to estimate the rates of VTE in patients with cancer by different tumor subtype and therapeutic treatments. This study focuses on the rates, risk factors, and treatments medication for cancer-associated thrombosis to reflect the regularity of VTE among the Chinese cancerous population, as well as to arouse the highlight of oncologist on the VTE risk in routine clinical practice.
Materials and Methods
Study Design
In this cross-sectional retrospective study, we enrolled 109,769 hospitalized patients who were confirmed diagnosis of solid malignancies from The Affiliated Tumor Hospital of Xinjiang Medical University, which was a tertiary center in Northwest China, during the period of January 1, 2012 to December 31, 2021. The study was approved by the Ethics Committee of The Affiliated Tumor Hospital of Xinjiang Medical University (IRB file no.2019BC007) and was conducted in accordance with the ethical standards laid down in the 2013 Declaration of Helsinki, its later amendments and Chinese Good Clinical Practice guidelines. However, the requirement to obtain informed consent for any research utilizing patients’ medical information was waived owing to the retrospective design of the study. The patients’ records and information were anonymized and de-identified prior to the analysis.
Patients and Eligibility
Patients were eligible if they met the following criteria: (a) 18 years or older; (b) inpatients who were histopathologically proven malignant tumors; (c) with DVT and/or PE events confirmed by objective imaging techniques (compression venous ultrasonography [CUS] or computed tomography pulmonary angiography [CTPA]); and (d) complete case data. The exclusion criteria included the following: (a) inpatients hospitalized for<24 h; (b) second malignancy or other concomitant malignant diseases; (c) patients who had confirmed the presence of VTE at the time of admission; (d) patients receiving long-term therapeutic anticoagulation before hospitalization; (e) superficial venous thrombosis or thrombophlebitis; (f) previous hematological diseases, rheumatoid arthritis, limited liver and kidney function and/or damage; and (g) incomplete medical records.
In this study, both symptomatic VTE and asymptomatic VTE detected incidentally during routine imaging were included in this analysis. Thus, a total of 1482 patients with solid tumor complicated with VTE were included in the final analysis. Of those, 735 patients were diagnosed as PE by CTPA or contrast-enhanced CT, with single/bilateral/multilobar pulmonary artery embolism and its branches being the main type. And 974 patients were diagnosed as DVT by CUS (for upper extremity, lower extremity and neck or thoracic thrombus) or contrast-enhanced CT (for splanchnic thrombus), with venous blood stasis in the lower limbs or venous valve insufficiency.
Data Collection
To prevent bias, data from eligible patients’ medical charts were collected using an electronic medical record (EMR) by both trained data management personnel and a member of the statistical team. The diagnosis information including VTE events (DVT and/or PE) and comorbidities were abstracted from EMR through International Statistical Classification of Diseases, 10th Clinical Modification (ICD-10 CM) code (I80, 82, 26 + subindices). The abstracted data included demographic information, comorbidities, discharge diagnoses, laboratory examination data, treatment details, and imaging description of the lesion.
Statistical Analysis
Statistical analysis was performed with SPSS 25.0 software (IBM Corp, Armonk, New York, USA). Data with skewed distribution were expressed as median and interquartile range [M (P25, P75)] and the categorical variables were presented as whole numbers and proportions. Multiple imputation with chained equations was used to replace missing data for body mass index (BMI) values. To present the anatomic distributions of VTE, Venn diagram analysis was plotted using the R package VennDiagram (https://cran.r-project.org/web/packages/VennDiagram/). The distribution of variables was assessed using the Kolgomorov-Sminorv test. Group comparisons for categorical variables were based on chi-squared test or Fisher's exact test, while comparisons for continuous variables with skewed distribution were based on Mann-Whitney U test and Wilcoxon signed-rank test. Two-sided P < .05 was regarded as statistically significant.
Results
Annual Incidence of Cancer-Associated VTE
Over the 10-year period, there were among 109,769 individual patients with cancers in the electronic database upon discharge diagnosis. Of them, 1482 (1.35%) patients experienced clinically apparent VTE events during their hospitalization period. Among these patients, 974 cases (0.89%) were diagnosed with DVT and 735 cases (0.67%) were diagnosed with PE (including 227 cases complicated with DVT and 508 cases without).
The annual incidence of cancer-associated thrombosis (CAT) ranged from 0.59% in 2014 to 1.92% in 2021, with an average annual incidence rate of 1.25% over the research decade. The incidence rate of VTE displayed a general ascending trend, but showed a descending trend from 2017 (1.77%) to 2019 (1.11%). Correspondingly, the incidence of DVT showed a similar trend to that of VTE (Figure 1A). The reported annual rate of PE initially showed a general ascending trend but eventually rapidly descending again, with the rate of PE complicated with DVT increasing progressively from 0.01% in 2012 to 0.55% in 2020 (Figure 1A). For the incidence rate of PE, DVT, PE&DVT, and VTE, a gradually ascending trend was shown with statistical significance between the 2 intervals (χ² = 39.03,15.97, 91.84,106.12, all P < .001, respectively) (Figure 1B).
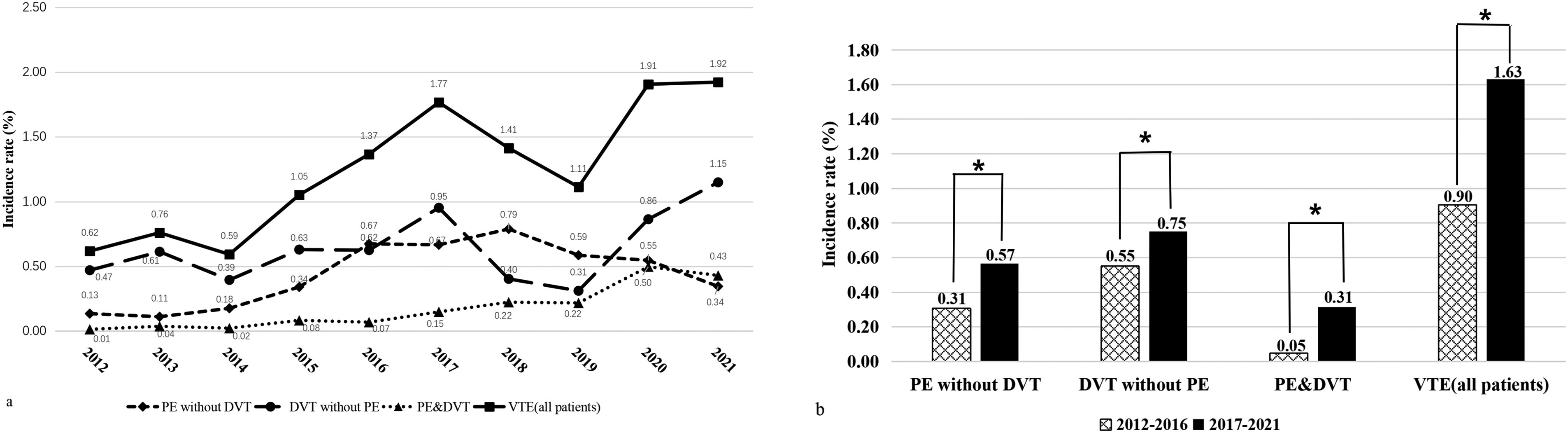
Annual incidence of VTE in patients with cancer and the temporal trends in disease frequencies. (A) The annual incidence of VTE. (B) The changing incidence of VTE according to periods. *P < 0.001. Abbreviations: DVT, deep venous thrombosis; PE, pulmonary embolism; VTE, venous thromboembolism.
Incidence of VTE by Cancer Subtypes
Lung cancer was the most frequent tumor subtype for developing VTE events, accounting for 24.83% of all cases, followed by ovarian cancer (9.72%), gastric cancer (8.77%), breast cancer (8.23%), cervical cancer (7.69%), and colorectal carcinoma (7.56%) (Figure 2).

Cancer subtypes among hospitalized VTE patients with malignancy, 2012-2021. Abbreviations: VTE, venous thromboembolism.
The incidence rates of VTE in several common types of solid cancer are reported in Table 1. The patients were divided into extremely high-risk, high-risk, middle-risk and low-risk groups based on the VTE incidence rate. Patients with ovarian and vulvar cancers were considered at extremely high risk (4.53%) for VTE. Patients with lung, pancreas, endometrial cancers and lymphoma were considered at relatively high risk (2.66%) for VTE, which was associated with 2-fold higher risk of VTE as compared to the moderate risk group (1.39%). Meanwhile, low risk (0.68%) was observed in patients with breast and brain cancer. Significant differences were observed among different risk groups (χ2 = 419.58, P<.001), while there was no significant difference within each risk group (χ² = 0.08, 1.37, 6.26, all P > .05, respectively) (Table 1).
Incidence Rates of VTE in Several Common Types of Solid Cancer.

P value has been calculated on the known components of the variables within each risk group.
P value has been calculated on the known components of the variables among 3 different risk groups.
Abbreviation: VTE, venous thromboembolism.
The Anatomic Distributions of Cancer-Associated VTE
Of the 1482 patients with VTE, 747 patients (50.40%) developed DVT alone, 508 patients (35.28%) developed PE alone, and 227 patients (15.32%) developed both DVT and PE. Among 974 patients with DVT, the most commonly involved area was the lower extremities (73.51%), followed by the neck or thorax (14.58%), and upper extremities (12.83%) (Figure 3). In the lower extremity, distal DVT was more common than proximal DVT (42.30% vs 21.87%), while DVT in upper extremities were more often observed on the right (43.20%) than the left side (39.20%) (Table 2).

The Wayne diagram of overlapping and anatomic distributions of VTE. The red, blue, yellow, and green ellipse represented UEDVT, LEDVT, SPDVT, and N&TDVT, respectively. The overlap regions represented the intersection between PE and any anatomic parts of DVT. Abbreviations: DVT, deep venous thrombosis; LEDVT, lower extremities DVT; N&TDVT, neck or thorax DVT; PE, pulmonary embolism; UEDVT, upper extremities DVT; SPDVT, splanchnic veins DVT; VTE, venous thromboembolism.
The Incidence of PE and CVC of Patients With Cancer With DVT Diagnosis According to Anatomic Site.

Proximal DVT included thrombosis involving the popliteal vein and above.
Abbreviations: CVC, central venous catheter; DVT, deep vein thrombosis; LEDVT, lower extremity DVT; N&TDVT, neck or thoracic DVT; PE, pulmonary thromboembolism; SPDVT, splanchnic DVT; UEDVT, upper extremity DVT.
Data on PE and central venous catheter (CVC) of patients with cancer with DVT diagnosis are presented in Table 2. The incidences of patients with DVT in the lower extremities developing PE were 21.15%, which were higher than the risk of PE developing with DVT in other sites (2.98%). In further analysis, catheter-related thrombosis (CRT) occurred in 125 patients (88.03%) with DVT in the neck or thorax and 92 patients (73.60%) in the upper extremities. Of these, 91.4% (111 of 125) patients in the internal jugular vein and 89.53% (77of 92) patients in the subclavian vein had been inserted with central venous catheters. However, only 194 of 716 patients (27.09%) with DVT in the lower extremities experienced CRT events. This implied that DVT development in the upper extremities and neck or thorax was strongly associated with CVC indwelling.
Demographics and Epidemiology of Cancer-Associated VTE
Among the total of 1482 patients with cancer-associated VTE, there was no significant difference in the incidence of VTE between sexes, with 40.2% being male and 59.8% female (χ² = 3.48, P = .062). The average age at onset of VTE was 60 years old, with patients aged between 50 and 70 years accounting for more than three-fourth of the cohort (76.57%)(Figure 4A). In addition, elderly patients (≥60 years) were more likely to develop PE complications than in the younger patients (χ² = 14.19, P<.001) (Figure 4A). The tumour node metastases (TNM) classification distribution of cancer-associated VTE was 26.45% for early stage (stage I-II), and 70.55% for advanced stage (stage III-IV), respectively. Meanwhile, patients with advanced stage were more likely to cause PE complications than those with early stage (χ² = 14.58, P<.001) (Figure 4C). Among 1482 patients with VTE, the most commonly histological type was adenocarcinoma (45.82%), followed by squamous cell carcinoma (16.60%) and invasive ductal carcinoma (7.96%) (Figure 4D).

The distribution of cancer-associated VTE based on different demographic and epidemiological characteristics. (A,a) The percentage of VTE in different age groups and the distribution of VTE based on different age groups. (B,b) The percentage of VTE in different ethnic groups and the distribution of VTE based on different ethnic groups. (C,c) The percentage of VTE in different AJCC staging groups and the distribution of VTE based on different AJCC staging groups. (D,d) The percentage of VTE in different pathological types and the distribution of VTE based on different pathological type groups. Abbreviations: AJCC, American Joint Committee on Cancer; DVT, deep vein thrombosis; PE, pulmonary thromboembolism; VTE, venous thromboembolism.
The average BMI of the patients was 24.8 ± 4.2 kg/m2. The majority of participants (48.92%) had a baseline BMI within the normal range according to World Health Organization (WHO) cut-off points (18.5-24.9 kg/m2) and only 10.26% of the patients were obese with BMI values over 30 kg/m2. However, 37.85% of patients had a BMI within the normal range of 18.5 to 23.9 kg/m2, and 18.76% of patients were classified as obese according to the Chinese population standards.
The majority of patients (89.34%) had good physical condition with an Eastern Cooperative Oncology Group (ECOG) performance status (PS) score of 0 or 1, and only 158 of them (10.66%) had an ECOG PS of 2 or higher. The blood types distribution of patients with cancer with VTE was 29.08%, 22.67%, 20.24%, and 15.59% for A, B, O, and AB blood types, respectively (Table 3). The most common comorbidities in patients with VTE were hyperlipidemia (43.25%), followed by hypertension (24.16%). The most common symptom among patients diagnosed with VTE was limb/neck swelling with pain, which was seen in 215 (14.51%) of the patients.
The Demographic and Clinical Characteristics of Patients With Cancer With VTE by Ethnicities.
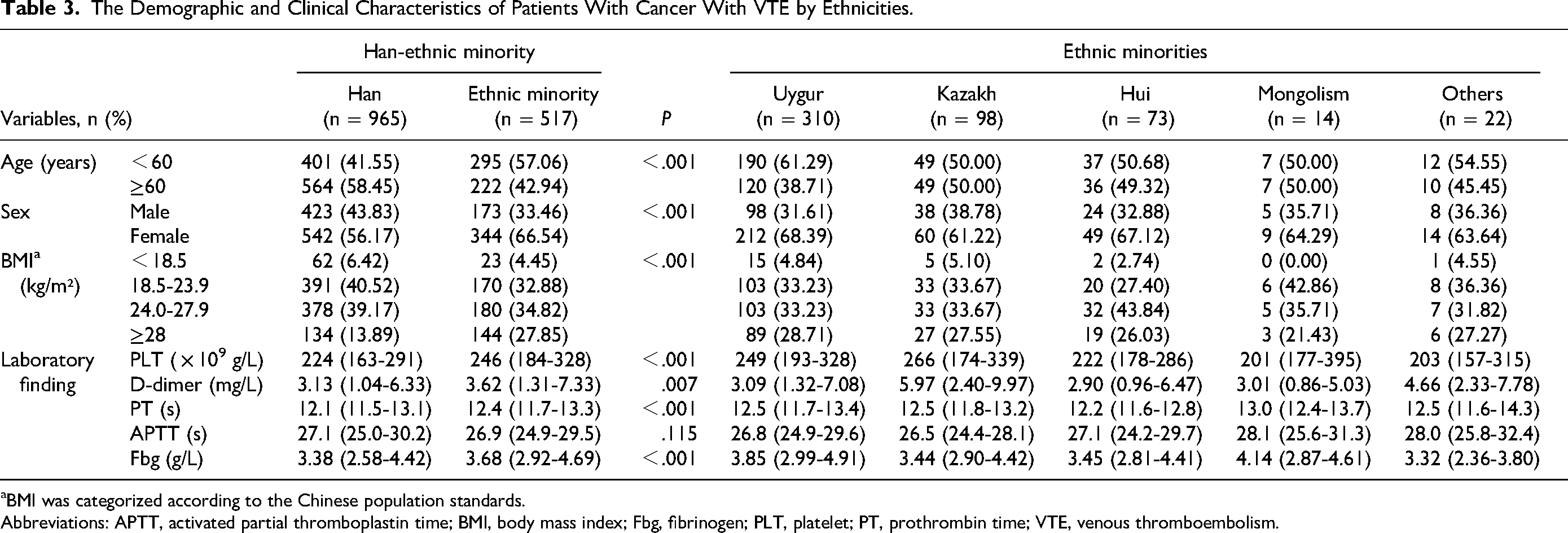
BMI was categorized according to the Chinese population standards.
Abbreviations: APTT, activated partial thromboplastin time; BMI, body mass index; Fbg, fibrinogen; PLT, platelet; PT, prothrombin time; VTE, venous thromboembolism.
Regarding ethnicities, 965 cases (65.11%) were Han ethnic and 517 cases (34.89%) were minority ethnics and the majority (59.96%) (310 of 517) of the participants were Uygur ethnic minorities (Figure 4B). There was a statistically significant difference in age between Han and Ethnic minority groups (χ² = 32.494, P < .001). There was a significant difference in the proportion of males and females across different ethnicities groups (χ² = 15.063, P < .001). Moreover, significant differences for the values of platelet, D-dimer and fibrinogen between Han and Ethnic minority groups were observed (P = .000-.007) (Table 3).
The Length of Time From Diagnosis of Cancers to VTE Diagnosis
Time-to-event analysis of VTE is shown in Figure 5. And 216 patients (14.57%) were diagnosed with VTE before their diagnosis of malignancy. For the majority of patients with VTE (n = 760, 51.28%), VTE events occurred within 3 months of cancer diagnosis (Figure 5). There were 227 (15.32%), 94 (6.34%),71 (4.79%), and 70 (7.42%) patients diagnosed with VTE between 3 to 6 months, 6 months to 1 year, 1 to 2 years, 2 to 5 years of cancer diagnosis, respectively. Only 44 patients (2.97%) developed VTE events 5 years after their cancer diagnosis.

Histogram of the frequency of VTE occurrence during the first 5 years after the diagnosis. (Time before and after cancer diagnosis is calculated in months. Time 0 corresponds to the diagnosis time point.) Abbreviation: VTE, venous thromboembolism.
Therapeutic Treatments of Patients With Cancer With VTE
Close to half of the patients (n = 689, 46.49%) had received chemotherapy within 6 months prior to the diagnosis of VTE. Of these, 485 patients (32.73%) had previously received platinum-containing chemotherapy regimens and 293 patients (19.77%) had received taxane-based regimens. Additionally, chemotherapy cycle number ≥3 was associated with an increased risk of VTE compared with cycle number 1 and 2 (31.92% vs 14.57%) (Table 4).
The Therapeutic Treatments of Patients With Cancer by VTE Status.

All the anticancer therapies were within 6 month before the VTE diagnosis.
The immunotherapy were within 3 month before the VTE diagnosis.
The supportive treatment were within 1 month before the VTE diagnosis.
*The percentage means total PE/total VTE.
Abbreviations: 3D-CRT, 3-dimensional conformal radiotherapy; CD20, lymphocyte antigen CD20; DVT, deep vein thrombosis; EGFR-TKI, epidermal growth factor receptor-factor tyrosine kinase inhibitors; EPO, erythropoietin; G-CSF, granulocyte-colony stimulating factor; HER2, human epidermal receptor 2; IMRT, intensity-modulated radiotherapy; PD1, programed cell death 1; PDL1, programed death ligand 1; PE, pulmonary thromboembolism; VEGF, vascular endothelial growth; VTE, venous thromboembolism.
Postoperative patients were at a high risk of VTE 6 months after surgery, accounting for nearly one-third of the participants (32.05%). The number of patients undergoing open surgery was much larger (282 of 475, 59.37%) compared to those undergoing minimally invasive surgery (169 of 475, 35.58%). As for the site of surgery, gynecological surgery was dominant (163 of 475, 34.32%) (Table 4).
Among patients with cancer with VTE, 207 patients (13.97%) were under radiotherapy within 6 months before the VTE diagnosis, of which 183 (88.41%) received intensity-modulated radiotherapy (IMRT) and 24 (11.59%) received 3-dimensional conformal radiotherapy (3D-CRT). And 236 patients (15.92%) were being treated with or had received target therapy within half year before the VTE confirmation. One hundred and six (7.15%) patients were treated with antivascular endothelial growth (VEGF) agents and 76 (5.13%) patients were treated with epidermal growth factor receptor-factor tyrosine kinase inhibitors (EGFR-TKI). Only 2.56% of the patients (n = 38) were under immunotherapy within 3 months before the VTE diagnosis. Among them, the majority of the patients (84.21%) received antiprogramed cell death 1 (PD1) monotherapy; nevertheless, 6 patients (15.79%) were treated with antiprogramed death ligand 1 (PDL1) immunotherapy (Table 4).
Analysis of Gynecological Tumor-Related VTE
Baseline characteristics among patients with gynecological tumors with VTE stratified by whether or not patients underwent recent operation are presented in Table 5. The morbidity of gynecological tumor-related VTE was 2.44% and 2.05% in the nonoperative and postoperative groups, respectively (P = .094). The nonoperative group had a significantly lower likelihood of developing early DVT/PE symptoms, but had a significantly higher likelihood of developing PE complications compared with the postoperative group (P = .020 and .027, respectively).The frequency of massive ascites group (>2000 mL) in gynecological patients with VTE was significantly larger than that of nonmassive ascites group (≤2000 mL; P < .001). Significant differences were observed in neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), albumin, and cancer antigen 125 (CA125) levels were observed between the nonoperative and postoperative groups (P = .000 - .027). While there were no significant differences in hemoglobin, platelet, and D-dimer between the 2 groups (P = .167 - .868).
The Demographic and Clinical Characteristics of Gynecological Tumor-Related VTE.
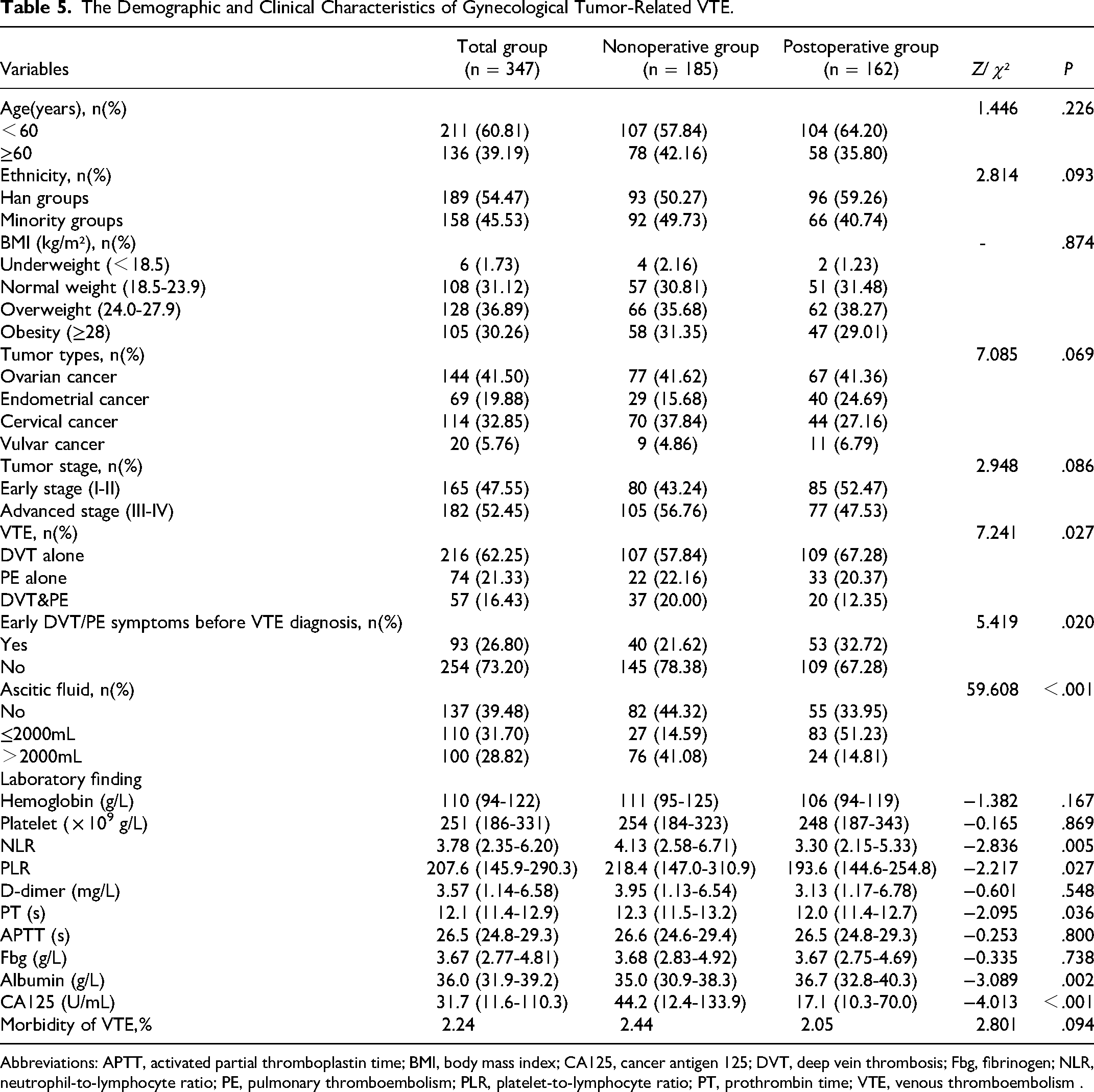
Abbreviations: APTT, activated partial thromboplastin time; BMI, body mass index; CA125, cancer antigen 125; DVT, deep vein thrombosis; Fbg, fibrinogen; NLR, neutrophil-to-lymphocyte ratio; PE, pulmonary thromboembolism; PLR, platelet-to-lymphocyte ratio; PT, prothrombin time; VTE, venous thromboembolism .
Discussion
Cancer is well recognized as a risk factor for development of VTE, and VTE is a common and potentially life-threatening complication in patients with cancer.1–7 Of note, it has been established that there is a strong association between cancer and thromboembolic events. Hence, early detection of high-risk factors for patients with cancer combined with VTE should be paid for particular attention. Currently, there are few studies related to the epidemiology and the main etiology of VTE specific to the Chinese cancerous population.10,11 In particular, there are no contemporary large-scale studies addressing cancer-associated VTE epidemiology for ethnic minority populations residing in Xinjiang, Northwest China. Therefore, in this single-centre retrospective observational study, with a specific focus on ethnic minority populations and noval anticancer therapies, we evaluated the profile of Chinese cancerous patients complicated with VTE based on demographic features, clinical characteristics, and medication exposure. To our knowledge, this study provides for the first time a profile of the current spectrum of cancer-associated VTE disease in this region, which can serve as a useful reference for the development of VTE risk stratification tools and clinical decision making on thromboprophylaxis.
In the present study, we reported a 1.35% incidence of clinical VTE, a 0.89% incidence of DVT and a 0.67% incidence of PE among hospitalized patients with cancer between 2012 and 2021 in our institution. The overall rate of cancer-associated VTE in our center (1.35%) was similar to the incidence reported in other single-center study (1.79%) by Peng et al. 10 Previous studies have reported that Asian population had a swing incidence of cancer-associated VTE ranging from 0.47% to 13.2%.10–14 Despite potential fluctuations in incidence, the incidence of VTE was significantly lower among Asian patients with comorbid cancer with VTE than the reported that of patients from Western populations.3,15,16 The reasons for this discrepancy might be due to the ethnic and population differences in genetic predisposition, the uneven distribution of medical resources, under-recognition of thrombotic disease, and possibly less symptomatic VTE in Asian patients.17,18 Further, our study showed a gradually ascending trend in VTE incidence for the study duration, with 0.62% in 2012 and 1.92% in 2021. The annual rate of PE exhibited an upward tendency before 2017 while showed a downward trend during the later period. Under the in-hospital VTE prevention and control system, improved awareness and standardizing the diagnosis and management of VTE might be one of the reasons for such trends. Besides, emerging treatment modalities such as targeted therapy and immunotherapy may also be a reason for such upward trends.
Nowadays, it is well known that type of malignancy strongly influences the incidence of VTE.10,15,16,19 Based on our analysis, the results showed that lung cancer was the most frequent tumor subtype for developing VTE events, accounting for 24.83% of all cases, followed by ovarian, gastric, breast, cervical and colorectal cancers. This result is consistent with the findings of several recent studies.10,20 Despite differences in VTE rate according to type of malignancy, the patients with cancer with the highest proportion for developing VTE events do not necessarily mean that their incidence is indeed highest. When patients were grouped based on the VTE incidence rate, our current data showed that patients with ovarian and vulvar cancers were considered at extremely high risk (4.53%) while patients with breast and brain cancers were considered at low risk (0.68%) for VTE. Similar to our findings, previous studies also found that patients with tumors of pancreas, ovarian, lung and gastric cancers are generally reported to be greater risk of VTE than other types of malignancies.10,11,16,19,21,22 However, it needs to be noted that the incidence of VTE in brain tumors especially glioblastoma (GBM) remains highly controversial. Given the data from our center, only 0.40% GBM patients were diagnosed with VTE, which was consistent with domestic study results, 10 but inconsistent with other previous reports.21,23 This is likely owing to racial difference, genetic variation, admission bias, and anticancer therapies.24,25
CVCs especially peripheral insertion central catheter is increasingly used in the treatment of patients with cancer for administration of continuous infusional chemotherapy, and to avoid frequent venipuncture. Despite these strengths, moreover most studies noted that the use of long-term CVC was strongly associated with a substantial risk of upper extremity DVT (UEDVT).26,27 Consistent with these previous studies, the present study also found the same results that CRT occurred in 88.03% of patients with DVT in the neck or thorax and 73.60% of patients in the upper extremities. Of these, nearly 91.4% of patients in the internal jugular vein and 89.53% of patients in the subclavian vein had been inserted with CVCs. Such results suggested that CVCs indwelling might be the main cause of UEDVT and neck or thorax DVT among patients with cancer, consistent with findings of the current studies.10,26,27 This phenomenon might be correlated with vascular endothelial injury and slow blood flow caused by the deep venous indwelling catheter, or compression of the cervical lymph nodes metastasis. Moreover, as mentioned earlier, the incidences of patients with DVT in the lower extremities developing PE were 21.15%, which were higher than the risk of PE developing with DVT in other sites (2.98%). Further analyses of the 716 patients with lower extremity DVT (LEDVT) showed higher incidence of higher risk PE in patients with isolated distal DVT than proximal DVT (42.30% vs 21.87%). The results of the present study are consistent with previous literatures.10,28 Thereby, it is concluded that high rate of catheter-related DVT and the severe consequence of LEDVT should be paid for particular attention for front-line clinicians and nurses.
In addition, we evaluated the effect of patient-related factors on cancer-associated VTE, including age, gender, ethnicity, BMI, comorbid diseases, and so on. It is widely accepted that patients with cancer with advanced age, female sex, and obesity have an increased risk of developing VTE.10,19,20 Our findings show that females had higher VTE rates than males, with 40.2% being male and 59.8% female, but there was no significant difference in the incidence of VTE between sexes. Our results also demonstrated that elderly patients (≥60 years) were more likely to develop PE complications than in the younger patients, indicating the effect of advanced age on severe embolic events, which agrees with the results of previous study. 29 Moreover, our current data showed that the blood types distribution of patients with cancer with VTE was 67.34% and 20.24% for non-O and O blood types, which is consistent with the findings of recent study. 30 Furthermore, it should be noted that ethnic and regional differences are non-negligible factors in the study of risk factors for VTE. 19 Thus, with a specific focus on ethnic minority populations, 65.11% were Han ethnic and 34.89% were minority ethnics and the majority (59.96%) of the participants were Uygur ethnic minorities in our study. Our study further found that participants in Uygur group reported onset at a younger age than those in Han group. These discrepancies may also be caused by Uygurs’ unique ethnic origin, diet, lifestyles, living habits, and living environments.
According to remarkable evidence from systematic review as well as cohort and case-control studies, obesity is an independent factor for VTE and might predispose to thromboembolic events.10,31–33 With respect to BMI, it was shown that the majority of participants had a baseline BMI within the normal range according to WHO cut-off points (18.5-24.9 kg/m2) and only 10.26% of the patients were obese with BMI values over 30 kg/m2. Nevertheless, 18.76% of patients were classified as obese according to the Chinese population standards. Plausible explanations for this could be attributed to body structure, dietary habits and ethnic differences. Moreover most studies noted that the modified version of Khorana scale setting the cut-off points of BMI at 24 kg/m2 for Chinese cancer population could improve the predictive value in identifying and communicating risks. 34 This suggests that we should pay attention to the construction of VTE risk assessment model specific to Chinese patients with cancer by modifying BMI cut-off value.
Several studies have reported that cancer-related factors (such as clinical stage and histological type) may be essential for risk stratification in patients with cancer with VTE.35–37 Regarding histological types of tumors, our results showed that the most commonly histological type was adenocarcinoma (45.82%) among all patients with VTE. Similar to our findings, previous studies also found patients with adenocarcinoma was associated with a markedly higher risk of VTE compared with other pathological types.10,35 Another recent study by Li et al 36 has demonstrated that adenocarcinoma was one of the most powerful predictors for VTE development in the predictive scoring system. This is consistent with widespread belief that adenocarcinoma plays a critical role in activating procoagulants factors by secreting mucin, resulting in thrombus formation. Additionally, our studies revealed that the TNM classification distribution of cancer-associated VTE was 26.45% for early stage (stage I-II), and 70.55% for advanced stage (stage III-IV), respectively. Our study results are also consistent with the findings of Gade et al, 37 who showed that advanced and metastatic cancer has been linked to an increased risk of VTE as compared to tumors that are localized. The observed difference in incidence of VTE by stage likely correlate well with biological and clinical aggressiveness of tumors. 38
Further, we have also conducted time-to-event analysis of VTE to explore the length of time from diagnosis of cancers to VTE diagnosis. Over half of the patients (66.60%) were observed to have an increased risk of VTE within the first 6 months of cancer diagnosis, specifically after the first 3 months, in consistent with Wang's conclusion that most VTE events occurred within the first half years after the cancer diagnosis. 39 Otherwise, the increase in VTE risk gradually slows down with time elapse. There were 6.34%, 4.79%, and 7.42% patients diagnosed with VTE between 6 months to 1 year, 1 to 2 years, 2 to 5 years of cancer diagnosis, respectively. Only 44 patients (2.97%) developed VTE events 5 years after their cancer diagnosis. This phenomenon may be related to hypercoagulability caused by malignancy, vascular endothelial damage by anticancer therapy and prolonged bed time after surgery. Therefore, the assessment and monitoring of VTE in patients with cancer within the first 6 months of cancer diagnosis should be strengthened.
Taking into consideration the anticancer therapy half year before VTE diagnosis, chemotherapy, widely used in close to half of the patients (46.49%), has been reported to be associated with a 2-fold to 6-fold increased VTE risk. 40 We further found that platinum-based and taxane-based chemotherapy may have the greater relationship with the increased risk of VTE compared with other chemotherapies, which was consistent with findings reported in the literature. 10 Moreover, given the data from our study, chemotherapy cycle number ≥3 was associated with an increased risk of VTE compared with cycle number 1 and 2. There are many different theories explaining the possible mechanism, including vascular endothelium damage, reduced endogenous anticoagulant factors (antithrombin, protein C, protein S) and platelet activation. 41 However, there is less evidence available on the influence or mechanism of radiotherapy (RT) in patients with cancer with VTE. The present study showed 13.97% patients were under radiotherapy within 6 months before the VTE diagnosis, of which 183 (88.41%) received IMRT and 24 (11.59%) received 3D-CRT. which is similar to other reports.10,42 In particular, 15.92% patients were being treated with or had received target therapy and 7.15% were anti-VEGF and 5.13% were EGFR-TKI, which is slightly higher than results of previous study. 10 This implies that the application of targeted drugs (especially anti-VEGF and EGFR-TKI agents) needs to be particularly focused on patients in hypercoagulant state.
Additionally, it is worth noting that VTE have been increasingly reported as immune-related adverse events (irAEs) in recent studies.43–45 The current study showed only 2.56% patients were under immunotherapy, among them, the majority of the patients (84.21%) received anti-PD1 monotherapy; nevertheless, 6 patients (15.79%) were treated with anti-PDL1 immunotherapy. One of the reasons that the incidence of immune-related VTE is not higher could be due to the lack of anti-PD1/PDL1 therapy in earlier years. Interestingly, several possible mechanisms may underlie these findings. One possible mechanism is that activated T cells induce increased production of tissue factor (TF) to promote procoagulant activity. 44 Second, inflammatory reactions by the expressions of interleukin-8 (IL-8) and many other inflammatory cytokines may be another mechanism. 45 Therefore, the development of irAE reinforces the importance of continuous and close monitoring of patients receiving immunotherapy. Currently, it is warranted to develop and validate an immunotherapy-specific VTE risk assessment model to guide prophylaxis decisions for patients on immune checkpoint inhibitor (ICIs).
According to our statistics, postoperative patients were at a high risk of VTE 6 months after surgery, accounting for nearly one-third of the participants (32.05%). As for the site of surgery, gynecological surgery was dominant. Therefore, we focused on exploring the baseline characteristics among patients with gynecological tumors with VTE stratified by whether or not patients underwent recent operation. Our results also revealed that significant differences were observed in NLR, PLR, albumin and CA125 levels were observed between the nonoperative and postoperative groups, indicating the effect of pre-operative poor nutritional status, infections, and large tumor burden on VTE risk, which is highly consistent with those of some previous reports on the risk factors for thrombosis in gynecological malignancies.11,46 Meanwhile, the frequency of massive ascites group (>2000 mL) in gynecological patients with VTE was significantly larger than that of nonmassive ascites group (≤2000 mL). More importantly, our findings indicate that the morbidity of gynecological tumor-related VTE in the nonoperative groups (2.44%) was higher in postoperative groups (2.05%), although the difference is not statistically significant. Therefore, given these results, for patients with gynecological tumors with preoperative massive ascites, early detection of VTE-related examination may lead to earlier intervention to avoid VTE-related high morbidity and mortality.
Some limitations of the current study must be considered. The retrospective data collection from patient records inevitably leads to incomplete data acquisition resulting in selection and information bias. Thus, we only included patients with imaging evidence of VTE by CTPA or CUS and cases of asymptomatic CA-VTE might have been missed, which may result in a bias underestimating of the incidence of VTE. Additionally, due to the limitation of data, we only conducted a cross-sectional descriptive analysis of demographics and epidemiology of cancer-associated VTE, and the prognosis of disease was not assessed. A further limitation is the lack of a control group. Thus, it is difficult to quantitatively validate the current prediction models and to predict VTE in clinical practice in our study. Concerning ethnic minority populations and noval anticancer therapies, development of immunotherapy-specific VTE risk assessment model to guide prophylaxis decisions for Chinese patients with cancer on ICIs is needed priority attention in future research.
Conclusions
The overall incidence of clinical VTE was 1.35% in hospitalized patients with cancer in our center. The annual incidence rates showed a fluctuating and upward trend over the research decade. Patients with ovarian, vulvar, lung, pancreas, endometrial cancers, and lymphoma were considered at high risk for VTE. DVT development in the upper extremities and neck or thorax was strongly associated with CVC indwelling. The assessment and monitoring of VTE in patients with cancer within the first 6 months of cancer diagnosis should be strengthened. VTE occurrence was closely related to advanced age, Uygur ethnic minorities, tumor advanced stage, adenocarcinoma, obesity, non-O blood types, and noval anticancer therapies in patients with cancer. Especially, the application of targeted drugs (especially anti-VEGF and EGFR-TKI agents) and immunotherapy (especially anti-PD1 monotherapy) needs to be particularly focused on patients in hypercoagulant state. Early detection of VTE-related examination may lead to earlier intervention for patients with gynecological tumors with preoperative massive ascites.
Footnotes
Author Contributions
HML contributed to the study design, the data statistics and analysis, and writing the manuscript. YT, HWN, LLH, GLC, CXZ, and KK performed data collection. HML and YT analyzed and interpreted the data. QL designed and supervised the research and revised the manuscript. All authors read and approved the final manuscript.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This study was approved by the Ethics Committee of The Affiliated Tumor Hospital of Xinjiang Medical University (Approval ID: 2019BC007), which waived the requirement for patient signed informed consent owing to the retrospective nature of the study. And all procedures performed in this study involving human participants were in accordance with the ethical standards of the Ethics Committee of The Affiliated Tumor Hospital of Xinjiang Medical University and with the declaration of Helsinki.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (grant number 81760014) and Natural Science Foundation of Xinjiang Uygur Autonomous Region (2022D01D74)
