Abstract
Background
This study aimed to investigate the association of pan-immune-inflammation value (PIV), PIV/HDL-C (high-density lipoprotein cholesterol), PIV*LDL-C (low-density lipoprotein cholesterol) with the prognosis of patients with acute coronary syndrome (ACS) undergoing percutaneous coronary intervention (PCI).
Methods
A total of 1360 patients with ACS undergoing PCI were consecutively enrolled in this study. They were divided into major adverse cardiovascular events (MACEs) (n = 58) and non-MACEs (n = 1302) groups. The PIV, PIV/HDL-C, and PIV*LDL-C values were measured. The endpoints were MACEs, including cardiogenic mortality, recurrence of myocardial infarction, in-stent restenosis, and rehospitalization for severe heart failure.
Results
The multivariable Cox regression analysis showed that PIV ≥355.79 (hazard ratio [HR]: 2.006, 95% confidence interval [CI]: 1.165-3.455), PIV/HDL-C ≥ 282.86 (HR: 1.987, 95% CI: 1.119-3.527), and PIV*LDL-C ≥ 1431.58 (HR: 2.071, 95% CI: 1.206-3.556) were all independent predictors of MACEs in patients with ACS undergoing PCI (all P < .05). The cumulative survival rates were significantly lower for patients with higher PIV, PIV/HDL-C, and PIV*LDL-C than for patients with lower values of these indices (log-rank tests: all P < .05).
Conclusion
Higher PIV, PIV/HDL-C, and PIV*LDL-C were independent prognostic factors for patients with ACS undergoing PCI and may be novel biomarkers for predicting MACEs.
Introduction
Acute coronary syndromes (ACS), which includes unstable angina, non-ST elevation myocardial infarction (NSTEMI), and ST elevation myocardial infarction (STEMI), 1 is the leading cause of death worldwide, despite the great milestones achieved in its diagnosis and treatment. 2 The rupture of vulnerable atherosclerotic plaques in the coronary artery and systemic vascular inflammation are reportedly the primary causes of ACS.3–7 Previous studies have identified the potential usefulness of immune-inflammatory biomarkers, including neutrophil/lymphocyte ratio (NLR), platelet/lymphocyte ratio (PLR), monocyte/ lymphocyte ratio (MLR), and systemic immune-inflammation index (SII), as prognostic markers in patients with ACS undergoing percutaneous coronary intervention (PCI).8,9
The pan-immune inflammation value (PIV), a novel prognostic biomarker for some malignancies, is calculated as follows: [neutrophil count (
Methods
Study Population
A total of 1360 patients with ACS undergoing PCI were consecutively enrolled in this prospective cohort study from January 2016 to December 2018 at the Affiliated Hospital of Chengde Medical University. The inclusion criteria were patients aged ≥ 65 years; ACS clinical types unstable angina, STEMI, and NSTEMI; 19 coronary arteriography showing stenosis ≥50% in one or more of the left main, left anterior descending, left circumflex, right main, right posterior descending, right marginal, or their main branches; and patients who underwent PCI for the first time. The major exclusion criteria were patients with stable angina, coronary artery spasm, severe heart diseases (eg, aortic dissection and hypertrophic cardiomyopathy), infectious diseases, malignant tumors, systemic inflammatory disorders, hepatic diseases, and renal dysfunction.The definition of typical clinical risk factors for ACS according to our previous study. 9 In our study, all patients are prescribed antiplatelet/anticoagulant therapy and underwent risk assessment and revascularization strategy selection before PCI treatment as suggested by “2014 AHA/ACC guideline for the management of patients with non-ST-elevation acute coronary syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines”. This study was conducted according to the standards of the Declaration of Helsinki and was approved by the Ethics Committee of the Affiliated Hospital of Chengde Medical University (Number: LL076). All participants provided written informed consent.
Baseline Demographics and Clinical Characteristics
The research team collected baseline demographic and clinical data for all the patients, including sex, age, body mass index, dyslipidemia, hypertension, diabetes mellitus, ischemic stroke, history of heart failure, and family history of coronary artery disease, etc Fasting blood samples were collected within the first 24 h of admission before PCI. Data on blood routine indices: white blood cell count, platelet count, total cholesterol, triglycerides, HDL-C, LDL-C, serum uric acid, and serum creatinine were also recorded.
Follow-Up and Study Endpoints
The study endpoint was occurrence of major adverse cardiovascular events (MACEs), including cardiogenic mortality, rehospitalization for severe heart failure, recurrence of myocardial infarction and in-stent restenosis. Cardiogenic mortality was defined as death from heart diseases. Severe heart failure was defined as the New York Heart Association Classification Class IV. Follow-up data were collected via clinic visits at 1, 3, 6, and 12 months after the procedure and once annually thereafter.
Statistical Analyses
Statistical analyses were performed using SPSS 26.0 (SPSS Inc, Chicago, IL, USA), GraphPad Prism 8.0 (GraphPad Software Inc., La Jolla., CA), and R 4.3.3. All continuous variables showed a skewed distribution on Kolmogorov–Smirnov test and are expressed as median and interquartile range, whereas all categorical variables are presented as number (%). The Mann–Whitney U test was used to compare the differences in the continuous variables between the MACEs and non-MACEs groups. Categorical variables were compared using
Results
Baseline Clinical Characteristics
Among the 1579 patients, after excluding patients with malignant tumors (n = 7), infectious diseases (n = 20), stable angina (n = 95), blood system diseases (n = 10), and patients lost to follow-up (n = 87) due to lost of contact details or patients was reluctant to cooperate with our follow-up, a total of 1360 patients were enrolled in this study. The medium follow-up time was 1006 days in the two groups. The clinical characteristics of patients in the MACEs (n = 58) and non-MACEs (n = 1302) groups are outlined in Table 1. Patients in two groups showed significant differences in age ≥65 years, acute myocardial infarction, unstable angina, history of heart failure, cardiogenic shock, white blood cell count, serum creatinine ≥110 μmol/L, left ventricular ejection fraction (LVEF) ≤ 40%, PIV ≥355.79, PIV/ HDL-C ≥ 282.86, and PIV*LDL -C ≥ 1431.58 (all P < .05). Patients in the MACEs group tended to have age ≥65 years, acute myocardial infarction, history of heart failure, and cardiogenic shock.
Baseline Clinical Characteristics of the MACEs and Non-MACEs Groups.

Abbreviations: MACEs, major adverse cardiovascular events; AMI, acute myocardial infarction; UA, unstable angina; HF, heart failure; CAD, coronary artery disease; WBC, white blood cell; TC, total cholesterol; TG, triglyceride; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; SCr, serum creatinine; LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction; ACEI/ARB, angiotensin-converting enzyme inhibitors/angiotensin II receptor blocker; PIV, pan-immune-inflammation value.
Receiver Operating Characteristic Curve Analysis
ROC curve analysis was performed to determine the optimal cutoff values of PIV, PIV/HDL-C, PIV*LDL-C for predicting MACEs (Table 2). The area under the curve (AUC) for PIV was .648 (P < .001, 95% confidence interval [CI]: 0.577-0.719), and optimal diagnostic cutoff point was 355.79, with a sensitivity of 62.10% and specificity of 61.10%. The AUC for PIV/HDL-C was 0.641 (P < .001, 95% CI: 0.571-0.711), and optimal diagnostic cutoff point was 282.86, with a sensitivity of 69.00% and specificity of 52.40%. The AUC for PIC*LDL-C was 0.633 (P = .001, 95% CI: 0.560-0.707), and optimal diagnostic cutoff point was 1431.58, with a sensitivity of 44.80% and a specificity of 76.20%.
Receiver Operating Characteristic (ROC) Curve Analysis for Predictive Factors of MACEs.

Abbreviations: MACEs, major adverse cardiovascular events; AUC, area under curve; CI, confidence interval; Se, sensitivity; Sp, specificity; PIV, pan-immune-inflammation value; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol.
Univariate and Multivariate COX Proportional Hazards Regression Models
Cox proportional hazards model was created to assess the independent predictors of MACEs (Table 3). Significant variables (P < .05) in the comparison between MACEs and non-MACEs groups were included in the univariate Cox regression analysis, and those that retained statistical significance (P < .05) were entered into the multivariate Cox proportional hazard model. After adjusting for history of heart failure, cardiogenic shock, serum creatinine ≥110 μmol/L, age ≥ 65 years, LVEF ≤ 40%, the multivariable Cox regression analysis revealed that PIV ≥355.79 (HR: 2.006, 95% CI: 1.165-3.455, P = .012), PIV/HDL-C ≥ 282.86 (HR: 1.987, 95% CI: 1.119-3.527, P = .019), and PIV*LDL-C ≥ 1431.58 (HR: 2.071, 95% CI: 1.206-3.556, P = 0.008) were independent predictors of MACEs in patients with ACS undergoing PCI.
Cox Proportional Hazard Models for Predictive Factors of MACEs.
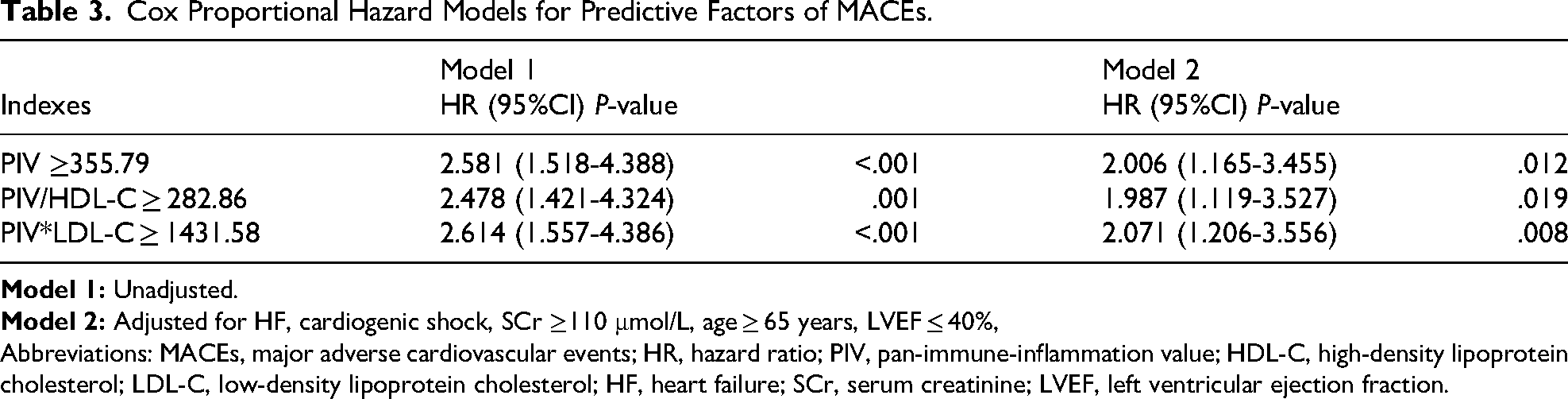
Abbreviations: MACEs, major adverse cardiovascular events; HR, hazard ratio; PIV, pan-immune-inflammation value; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; HF, heart failure; SCr, serum creatinine; LVEF, left ventricular ejection fraction.
Survival Analyses
The Kaplan–Meier curves revealed different survival rates across the three indices. Survival time refers to the event-free survival based on MACEs.The cumulative survival rates for patients with PIV ≥355.79, PIV/HDL-C ≥ 282.86, and PIV*LDL-C ≥ 1431.58 were significantly lower than those for patients with PIV <355.79, PIV/HDL-C < 282.86, and PIV*LDL-C < 1431.58 (log-rank tests: all P < .05 (Figure 1

Kaplan–Meier Curves for Cumulative Survival of PIV (A), PIV/HDL-C (B), and PIV*LDL-C (C) for Patients with ACS Undergoing PCI (Log-Rank P < .001). Survival Time Refers to the Event-Free Survival Based on All MACEs. Abbreviations: PIV, pan-immune-inflammation value; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol.
P for Interaction
The independent association of PIV ≥355.79, PIV/HDL-C ≥ 282.86, and PIV*LDL-C ≥ 1431.58 with prognosis was assessed in various subgroups of heart failure (with or without), LVEF (>40% or ≤40%), age (≥65 or <65 years), and cardiogenic shock (with or without). The three indices showed no significant interaction with heart failure, LVEF, age, and cardiogenic shock (all P for interaction > .05) (Figure 2).

Forest Graphs Based on Subgroup Analysis for Evaluating the Association Between Indices and Prognosis. (A) Subgroup Analysis of HF, (B) Subgroup Analysis of LVEF, (C) Subgroup Analysis of age, (D) Subgroup Analysis of Cardiogenic Shock. Abbreviations: HR, Hazard Ratio; PIV, pan-Immune-Inflammation Value; HDL-C, High-Density Lipoprotein Cholesterol; LDL-C, low-Density Lipoprotein Cholesterol; HF, Heart Failure; LVEF, Left Ventricular Ejection Fraction.
P for Trend and Restricted Cubic Spline (RCS)
The association of the tertiles of PIV, PIV/HDL-C, PIV*LDL-C with MACEs is shown in Table 4. The risk of MACEs increased gradually with increasing PIV, PIV/HDL-C, and PIV*LDL-C tertiles. Regarding PIV, with T1 as reference, the results were as follows: T2 versus T1: HR: 1.743, 95% CI: 0.823–3.691, P = .147; T3 versus T1: HR: 2.810, 95% CI: 1.403–5.628, P = .004; P for trend = .011. For PIV/HDL-C, with T1 as reference, the results were as follows: T2 versus T1: HR: 2.233, 95% CI: 1.051–4.743, P = .037; T3 versus T1: HR: 2.865, 95% CI: 1.387–5.921, P = .004; P for trend = .017. With regard to PIV*LDL-C, taking T1 as reference, the results were as follows: T2 versus T1: HR: 1.411, 95% CI: 0.667–2.983, P = .368; T3 versus T1: HR: 2.685, 95% CI: 1.374–5.247, P = .004; P for trend = .007.
Cox Hazard Proportional Models of MACEs Risk According to Tertiles of Indices.

Abbreviations: MACEs, major adverse cardiovascular events; HR, hazard ratio; CI, confidence interval; PIV, pan-immune-inflammation value; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol.
We used RCS to visualize the relationship between the three indices and MACEs risk (Figure 3). The values of PIV, PIV/HDL-C, and PIV*LDL-C with HR values close to 1 were 289.75, 271.10, and 665.30, respectively. Therefore, PIV >289.75, PIV/HDL-C > 271.10, and PIV*LDL-C > 665.30 were independent risk factors for MACEs. A linear relationship was observed between MACEs and PIV (P overall < .001, P non−linearity = .135), PIV/HDL-C (P overall < .05, P non−linearity = 0.059), and PIV*LDL-C (P overall < .05, P non−linearity = .078).

Restricted Cubic Spline (RCS) of PIV (A), PIV/HDL-C (B), and PIV*LDL-C (C). Abbreviations: MACEs, major adverse cardiovascular events; HR, hazard ratio; PIV, pan-immune-inflammation value; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol.
Discussion
To the best of our knowledge, this study is the first to explore the association of PIV, PIV/HDL-C, and PIV*LDL-C with prognosis in patients with ACS undergoing PCI using different statistical methods. The major findings are as follows: first, PIV ≥355.79, PIV/HDL-C ≥ 282.86, and PIV*LDL-C ≥ 1431.58 were associated with poor prognosis and were independent risk factors for MACEs in patients with ACS undergoing PCI. Second, the three indices showed no significant interaction with heart failure, LVEF, age, and cardiogenic shock. Finally, a linear trend was observed between PIV, PIV/HDL-C, and PIV*LDL-C and MACEs.
Inflammatory processes are involved in the development of atherosclerotic plaques, as well as unstable coronary plaque erosion and rupture.20,21 A high white blood cell count may be considered an independent prognostic factor for patients with ACS. 22 Activated neutrophils promote atherosclerosis, and neutrophil accumulation is an independent predictor of mortality in patients with ACS.23,24 Monocytes play a critical role in the progression of atherosclerosis and plaque rupture. Numerous microparticles derived from monocytes are detected in patients with ACS. 25 Macrophages release tumor necrosis factor that mediates the chemotaxis of B and T cells to the arterial walls, which promotes an inflammatory environment. 26 Platelets are involved in chronic inflammation, which contributes to the progression of atherosclerosis and acute coronary diseases. In particular, activated platelets release potent inflammatory factors and recruit circulating endothelial progenitor cells to the location of vascular injury, thus inducing a prothrombotic state. 27 According to previous studies, NLR, PLR, MLR, and SII can predict MACE in patients with ACS undergoing PCI.8,9 However, these indices comprise only a few peripheral blood cell components, and their predictive ability is relatively limited. As a novel biomarker of systemic immune-inflammatory status, PIV is based on several peripheral blood count parameters, including platelets, neutrophils, monocytes, and lymphocytes. 10 Murat et al reports that PIV seems superior to NLR, PLR, and SII in predicting short and long-term mortality in patients with STEMI. 28 Recently, Akkaya et al reported that higher PIV levels were associated with a higher risk of coronary slow flow phenomenon in patients with normal coronary angiography. 29 Similarly, another study revealed a correlation between high PIV and increased risk of impaired coronary flow in patients with STEMI. 30 Furthermore, in another study, PIV values analyzed at three time points predicted the poor long-term prognosis of patients with STEMI after primary PCI. 31 In the present study, PIV ≥355.79 was associated with poor prognosis and was independently associated with the risk of MACE in patients with ACS undergoing PCI. We did not observe any significant interaction of the three indices with heart failure, LVEF, age, and cardiogenic shock.
HDL-C protects against atherosclerosis through reverse cholesterol transport and antioxidative activity. However, LDL-C is a key pathogenic factor in the development of atherosclerosis. The equilibrium between HDL-C and LDL-C can affect endothelial dysfunction and inflammation.14,15 Therefore, PIV/HDL-C and PIV × LDL-C are comprehensive inflammatory nutrition indices. A previous study reported higher values of PIV/HDL-C and PIV × LDL-C in the severe coronary heart disease group compared to the mild group and had a high diagnostic efficacy for coronary heart disease. 32
In the present study, we combined PIV with HDL-C and LDL-C to predict the prognosis of patients with ACS who underwent PCI. We performed ROC curve analysis to determine the optimal cutoff values of PIV, PIV/HDL-C, and PIV*LDL-C for predicting MACEs; the AUCs indicated that the three indices had good predictive performance. The Kaplan–Meier curves demonstrated differences in the cumulative survival rates across two different groups, and the survival time was significantly lower in the higher index group. Multivariate cox regression models showed that PIV/HDL-C ≥ 282.86 and PIV*LDL-C ≥ 1431.58 were correlated with poor prognosis and were independent risk factors for MACEs in patients with ACS undergoing PCI. We also performed correlation analyses in subgroups of heart failure, LVEF, age, and cardiogenic shock, but no significant interactions were observed between the three indices and heart failure, LVEF, age, and cardiogenic shock, further indicating their independent prediction ability for MACE. Additionally, RCS confirmed the prediction trend of the indices. We found linear trends between PIV, PIV/HDL-C, and PIV*LDL-C and MACEs after a long-term follow-up.
MACEs was designed as a compound endpoint including cardiogenic mortality, rehospitalization for severe heart failure, recurrence of myocardial infarction and in-stent restenosis in the present study.And we found that patients in the MACEs group had higher incidence of age ≥65 years, acute myocardial infarction, history of heart failure, and cardiogenic shock. Additionally,higher PIV, PIV/HDL-C and PIV*LDL-C values were independent risk factors of MACEs. Prior studies also showed that triglyceride-glucose index (TyG index), Geriatric Nutritional Risk Index (GNRI) and SII were valuable factors to predict Major Adverse Cardiovascular Events including all-cause death, non-fatal myocardial infarction and non-fatal stroke or recurrent angina, severe arrhythmia.33,34 Therefore,different definitions of MACEs may have some influence on the prognosis study. Addtionally, the indices only tested on admission without dynamic observations were possible bias factors in the clinical study.
Addtionally,aspirin in Non-MACEs group was significantly higher than that in MACEs group. Dual antiplatelet therapy (DAPT) were administrated for the patients with ACS after PCI at least 12 months as suggested by “2016 ACC/AHA Guideline Focused Update on Duration of Dual Antiplatelet, Therapy in Patients With Coronary Artery Disease” to improve the prognosis of patients. However, there were significantly difference between the two groups due to the poor medication compliance of some patients who were more likely to develop MACEs and worse prognosis. More health education are needed for patients with ACS to improve the compliance in the medical care.
Clinical guidelines and research indicate that TIMI risk index is useful to predict mortality in ACS patients, and reduced LVEF is closely associated with long-term outcomes.35,36 What's more, coronary microvascular dysfunction and obstruction is associated to a much worse outcome in patient undergoing PCI. 37 Therefore, PIV indices preserve their predictive abilities for MACEs independently of post-PCI TIMI, improved LVEF and microvascular obstruction.
This study has some limitations. First, this was a single-center study with a small sample size.So we did not perform Kaplan-Meier analyses for each component of MACEs. Second, our study was based on the clinical features of blood cells and did not investigate any specific mechanism. Third, we only observed indices on admission without dynamic tests, the changes during the follow-up period may cause deviations in the analysis and prediction of MACEs.Therefore, more studies about PIV values at different time points,such as 24 h after PCI, and at discharge, were needed to predict the long-term prognosis of patients with ACS undergoing PCI.
Conclusion
This study showed that higher levels of the inflammatory nutrition indices PIV, PIV/HDL-C, and PIV*LDL-C were independent prognostic factors for patients with ACS undergoing PCI. These novel indices are easily accessible and cost-effective composite biomarkers for predicting long-term MACEs in patients with ACS, which may provide novel insights to optimize post-discharge management and identify high-risk patients earlier.
Footnotes
Acknowledgments
The authors would like to thank the doctors and nurses of the Cardiology Research Team at the Affiliated Hospital of Chengde Medical University and the Hebei Key Laboratory of Panvascular Diseases for their assistance.
ORCID iDs
Ethics Approval
This study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of the Affiliated Hospital of Chengde Medical University (Number: LL076/2015).
Author Contributions
Conceptualization, Yan Liu and Lixian Sun; Data curation, Jingyi Liu, Yuewen Qi and Weichao Shan; Formal analysis, Yan Liu; Methodology, Yan Liu, Xinchen Wang, Ge Song, Chen Wei and Lixian Sun; Resources, Ying Zhang; Software, Yan Liu; Supervision, Lixian Sun; Validation, Lixian Sun; Visualization, Yan Liu; Writing – original draft preparation, Yan Liu and Lixian Sun; Writing – review & editing, Yan Liu and Lixian Sun.
Funding
This study was supported by the Natural Science Foundation of Hebei Province (Grant number H2021406071) to Dr Lixian Sun and the Government funded clinical medicine talent training project (Grant numbers ZF2023252) to Dr Ying Zhang.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Patient Consent Statement
Informed consent for participation was obtained from all subjects involved in the study.
